Auto-Injectors Market
The Auto-Injectors Market is segmented by Product Type (Prefilled Auto-Injectors and Fillable Auto-Injectors); Indication (Anaphylaxis; Multiple Sclerosis; Rheumatoid Arthritis; and Other Uses); Distribution Channel (Online Pharmacies; Retail Pharmacies; Hospital Pharmacies; and Drug Stores); Usage Type (Disposable Devices and Reusable Devices); Activation Mechanism (Spring-Based Devices; Motor-Driven Devices; and Manual-Assist Devices); and End User (Home Care; Hospitals; Clinics; and Emergency Settings). Forecast for 2026 to 2036.
Auto-Injectors Market Size, Market Forecast and Outlook By FMI
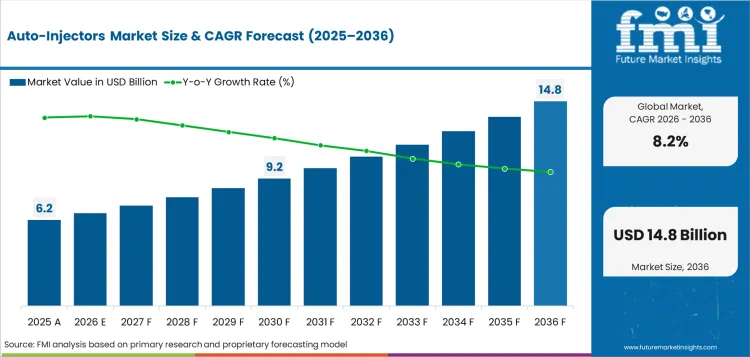
The auto-injectors sector was valued at USD 5.73 billion in 2025. FMI analysis projects the market at USD 6.20 billion in 2026 and USD 13.60 billion by 2036 at 8.2% CAGR through 2036. Disposable devices are estimated to hold 68.0% usage type share in 2026, while, prefilled auto-injectors are projected to account for 64.0% product type share.
Summary of the Auto-Injectors Market
- Demand and Growth Drivers
- Biologic therapy use supports demand for auto injectors as patients shift routine dosing into home care.
- Anaphylaxis care supports growth because epinephrine devices are central to emergency allergy response.
- Home based treatment supports adoption as patients and caregivers look for simple self use delivery formats.
- Product and Segment View
- Prefilled auto injectors lead product demand because fixed dose formats reduce handling steps.
- Disposable devices lead because safety needs and limited cleaning requirements support use after each dose.
- Spring based devices hold demand as manufacturers favor simple activation and reliable dose delivery.
- Geography and Competitive Outlook
- India offers high growth potential because specialty care access and biologic therapy use are expanding.
- The United Kingdom sustains demand as self injection support connects with organized care pathways.
- Companies combining dose accuracy and patient training support can improve access through drug device partnerships.
- Analyst Opinion
- Sabyasachi Ghosh as Associate Vice President at FMI says, "I see auto-injectors moving from emergency allergy use into a daily care tool for biologic drugs and long-term therapy. The next value pool is not only the device. It is the ability to pair the device with training and refill access. Patient support will decide repeat use after the first dose. Suppliers with proven human-factor testing and reliable fill-finish links will have an edge. Cost pressure will stay high as payers compare device ease with drug price. Clients should watch prefilled formats and pharmacy channels as the main signals for volume gain."
- Auto-Injectors Market Value Analysis
- The auto-injectors market is moving from emergency allergy use into a broader self-administration device category.
- Adoption is being driven by biologic therapies that require simple subcutaneous dosing outside clinical settings.
- The market is also benefiting from rising use of prefilled formats that reduce handling steps for patients.
- Demand is further supported by home care treatment, pharmacy refill access, and the need for safer long-term therapy delivery.
Auto-Injectors Market Definition
The market covers mechanical and electromechanical devices that deliver a preset injectable dose through a hidden needle. It includes prefilled and fillable devices used for anaphylaxis and autoimmune care. It also covers neurology therapy; diabetes support; and hormone treatment. The scope excludes standard syringes and infusion pumps. It also excludes IV sets and non-injectable drug formats.
Auto-Injectors Market Inclusions
Market scope includes prefilled devices; fillable devices; disposable formats; reusable formats; spring-based systems; motor-driven systems; hospital channels; retail channels; online channels; home care use; clinic use; and emergency use across all major regions.
Auto-Injectors Market Exclusions
Market scope excludes standard needles; standard syringes; insulin pumps; infusion pumps; inhaled therapies; oral drugs; topical patches; and needle-free platforms sold without auto-injector functionality.
Auto-Injectors Market Research Methodology
- Primary Research: FMI analysts reviewed input from device makers and pharmaceutical partners. Hospital pharmacists; retail pharmacists; and specialty care teams added views on device use. The review focused on dosing needs; training gaps; refill flow; and channel choice.
- Desk Research: FMI used regulatory guidance and device standards to define the addressable product pool. Clinical references and company product information were checked against combination product rules.
- Market Sizing and Forecasting: The model combined device revenue and therapy use rates. Country-level channel access and estimated replacement patterns were used to test segment shares.
- Data Validation: Forecasts were checked against biologic drug use and anaphylaxis device demand. Pharmacy access and leading supplier portfolios were used as validation checks.
Why is the Auto-Injectors Market Growing?
- Biologic drugs support device use as repeat subcutaneous dosing needs simple and safe patient handling.
- Anaphylaxis care supports emergency device placement as schools and public venues keep epinephrine access in focus.
- Home care use rises as patients seek fewer clinic visits and clearer dosing support for chronic therapy.
- Pharmacy access improves repeat supply as online ordering supports refill discipline and private delivery.
- Device makers invest in training aids as user errors can reduce confidence during the first dose.
Chronic therapy is shifting toward self-use formats that reduce the need for repeated clinic visits. The shift supports suppliers that can pair device design with patient training and refill support. Buyers will favor platforms that lower first-dose anxiety and reduce support calls.
Market Segmentation Analysis
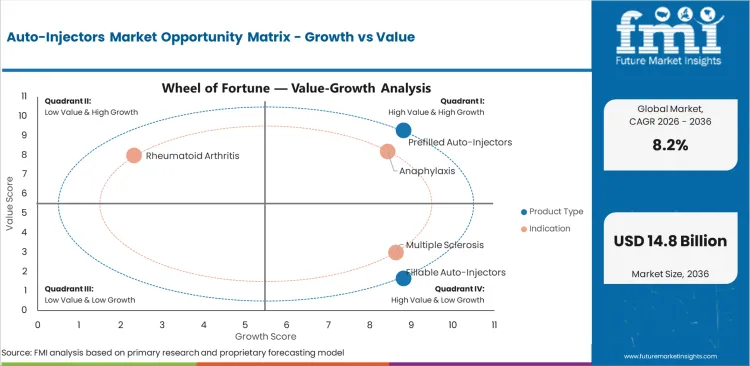
- Prefilled auto-injectors are projected to account for 64.0% product type demand in 2026 as fixed-dose design reduces user error.
- Anaphylaxis is expected to represent 39.0% indication demand in 2026 as epinephrine devices support urgent response.
- Online pharmacies are estimated to hold 34.0% channel share in 2026 with refill ordering and private delivery.
- Disposable devices are anticipated to hold 68.0% usage type share in 2026 as single-use handling reduces cleaning needs.
The market for auto-injectors is divided into groups based on product type, indication, distribution channel, usage type, activation mechanism, and end user. There are prefilled auto-injectors and fillable auto-injectors by product type. By indication, the market includes anaphylaxis, multiple sclerosis, rheumatoid arthritis, and other uses. By distribution channel, the market includes online pharmacies, retail pharmacies, hospital pharmacies, and drug stores. By usage type, the market includes disposable devices and reusable devices. By activation mechanism, the market includes spring-based devices, motor-driven devices, and manual-assist devices. By end user, the market includes home care, hospitals, clinics, and emergency settings.
Auto-Injectors Market Analysis By Product Type
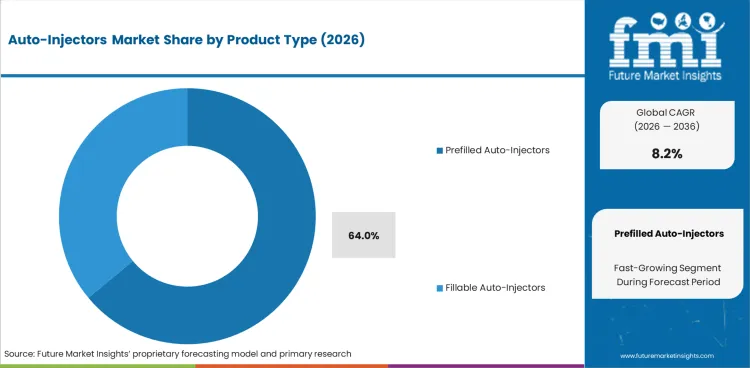
- Prefilled auto-injectors are projected to account for 64.0% of Product Type demand in 2026 as ready-to-use dosing reduces user steps and limits incorrect dose handling.
- Fillable devices keep value in specialist care as dose changes and trial use can still need staff preparation before patient use.
Auto-Injectors Market Analysis By Indication
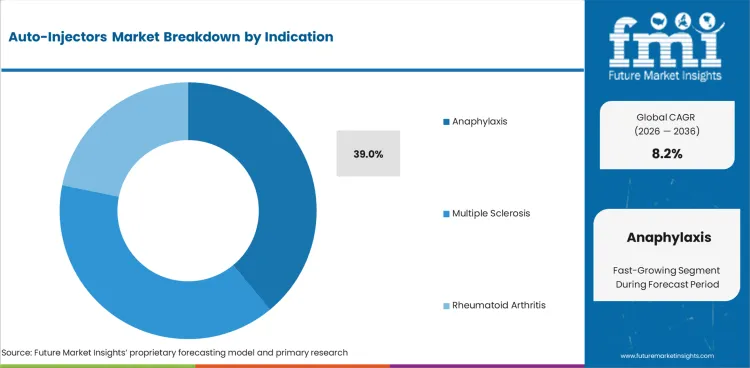
- Anaphylaxis is expected to represent 39.0% of Indication demand in 2026 as emergency epinephrine use needs fast action by non-clinical users.
- Multiple sclerosis and rheumatoid arthritis add steady repeat use as long-term biologic therapy favors simple home dosing over clinic-based injection.
Auto-Injectors Market Analysis By Distribution Channel
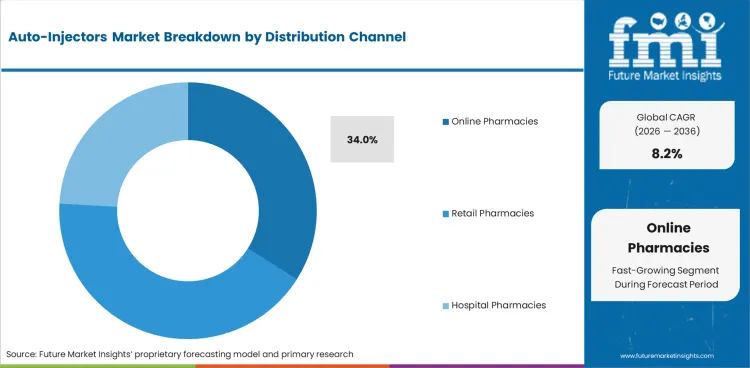
- Online pharmacies are estimated to hold 34.0% of Distribution Channel demand in 2026 as refill ordering suits repeat therapy and privacy needs.
- Hospital pharmacies keep an important role during therapy start as first-dose checks and cold-chain handling need professional oversight.
Auto-Injectors Market Analysis By Usage Type
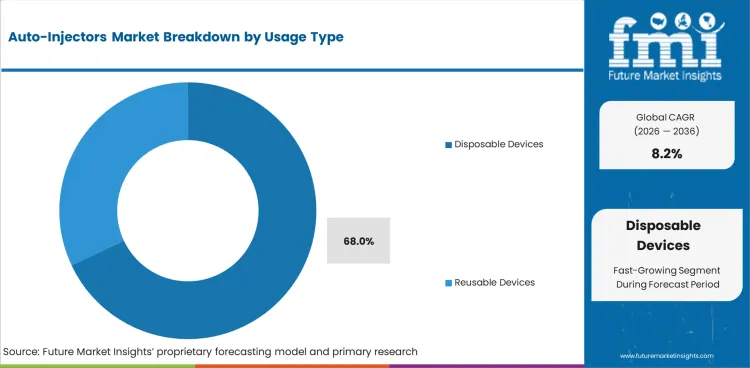
- With 68.0% share in 2026, disposable devices are anticipated to lead Usage Type demand as single-use designs reduce cleaning and cross-use concerns.
- Reusable devices work best in monitored chronic care programs that can justify higher initial cost through repeated cartridge use.
Auto-Injectors Market Analysis By Activation Mechanism
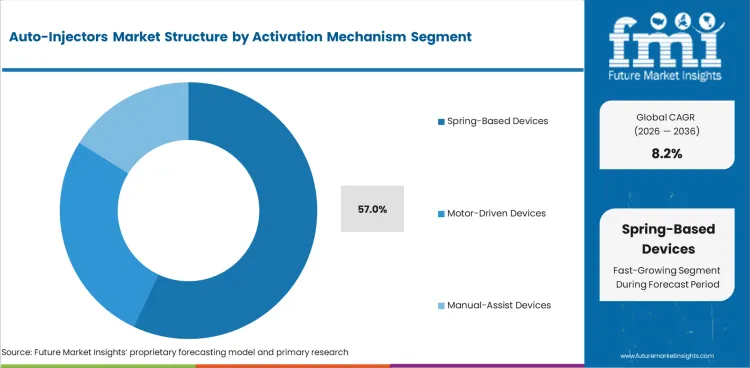
- Spring-based devices are projected to account for 57.0% of Activation Mechanism demand in 2026 as the design offers simple activation and reliable dose push.
- Motor-driven systems appeal to high-viscosity biologics as controlled force can help deliver thicker drug formulations with less user effort.
Insights into the Home Care End User Segment
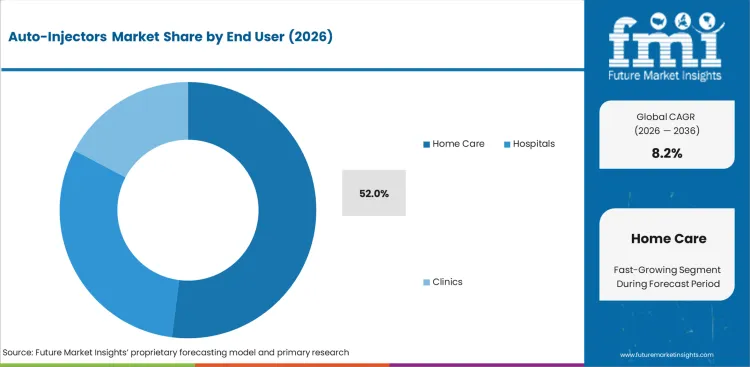
- Home care is expected to account for 52.0% of End User demand in 2026 as trained patients use auto-injectors for repeat therapy outside hospitals.
- Emergency settings keep share in allergy care as epinephrine devices must support fast action by family members and staff.
Auto-Injectors Market Drivers, Restraints and Opportunities
- Biologic delivery drives device use as many therapies need repeat subcutaneous dosing that patients can perform at home.
- Human-factor testing raises supplier costs as drug-device products need proof of safe use by intended users.
- Online refill channels create opportunity as chronic therapy patients need steady access and fewer missed doses.
- High drug-device prices restrain use in cash-pay settings and delay access for lower-income patients.
- Connected dose support offers opportunity for smart devices that record use and help care teams track adherence.
The market for auto-injectors keeps growing because biologic medicines need simple self-use devices, allergy care needs fast emergency dosing, and patients want fewer clinic visits. Even though there are problems like high device costs and strict usability testing, there are still chances for growth in home care treatment and connected dose-support devices.
Training error control
Ease of use is now a buying criterion rather than a design extra. The FDA focus on human-factor evaluation has pushed suppliers to test how patients read instructions and activate devices. That review burden raises development cost and slows weak platforms. It also rewards designs with clear feedback and hidden needles. Fewer steps improve user confidence. Buyers will press for evidence that new users can complete dosing with low error risk.
Analysis of Auto-Injectors Market By Key Countries
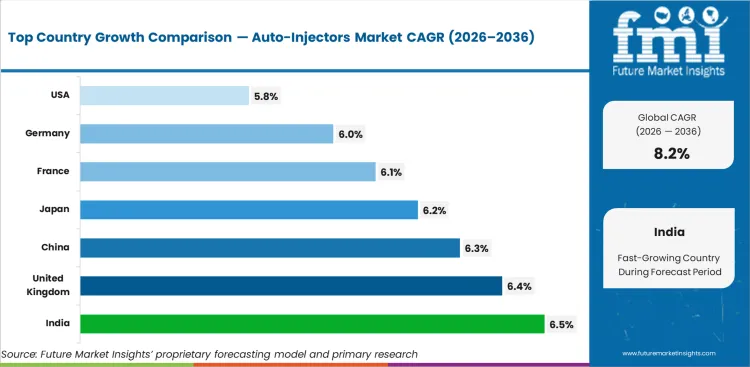
| Country | CAGR |
|---|---|
| India | 6.5% |
| United Kingdom | 6.4% |
| China | 6.3% |
| Japan | 6.2% |
| France | 6.1% |
| Germany | 6.0% |
| United States | 5.8% |
Source: FMI analysis based on published auto-injectors country forecasts and 2026 therapy access evidence.
Auto-Injectors Market CAGR Analysis By Country
- India is projected to record 6.5% CAGR through 2036 as specialty clinics expand access to biologic therapy in large cities.
- The United Kingdom is expected to expand at 6.4% CAGR through 2036 as NHS pathways support patient training for self-use therapy.
- China is forecast to grow at 6.3% CAGR through 2036 as local biologic use and digital pharmacy access expand together.
- Japan is expected to advance at 6.2% CAGR through 2036 as older patients favor compact devices with simple activation.
- France is projected to rise at 6.1% CAGR through 2036 as reimbursement support improves access to self-use biologics.
- Germany is forecast at 6.0% CAGR through 2036 as outpatient care uses auto-injectors for autoimmune therapy.
- The United States is expected to post 5.8% CAGR through 2036 as high baseline use makes added share gains steadier.
Country growth varies by therapy access and pharmacy reach. Device training depth explains the spread. India and the United Kingdom track above the global country set due to stronger specialty care. China follows due to digital pharmacy access. The United States grows at a lower rate since baseline use is already high. The following country sections explain how each care system shapes product volumes.
Demand Outlook for Auto-Injectors Market in India
India has a fast-expanding specialty care base that supports new demand for self-injection devices. Urban hospitals in Delhi and Mumbai are improving access to biologics for autoimmune and endocrine care. Bengaluru adds a strong private specialty base. India is projected to record 6.5% CAGR through 2036 as therapy access expands beyond top private hospitals. This pace reflects lower starting use and larger room for first-time patient training. Suppliers need low-step devices and pharmacy education to improve repeat use.
- Apollo Hospitals and Fortis networks support specialty drug access across major metros through trained nursing support.
- Jan Aushadhi outlets improve medicine reach in smaller districts and can support refill learning for chronic care patients.
- Maharashtra and Karnataka have larger private specialty clusters that can support early device training programs.
Sales Analysis of Auto-Injectors Market in the United Kingdom
The United Kingdom benefits from organized care pathways and clear patient education through the NHS. Auto-injector use is tied to allergy care and long-term therapy for rheumatoid arthritis or multiple sclerosis. The United Kingdom is expected to expand at 6.4% CAGR through 2036 as NHS self-use training reduces user hesitation. Pharmacy supply and clinician guidance support repeat use after the first prescription. Suppliers should focus on clear instructions and reliable refill access.
- NHS England care pathways support patient teaching for long-term injectable therapy in community settings.
- Medicines and Healthcare products Regulatory Agency oversight keeps drug-device safety expectations clear for suppliers.
- School allergy readiness in England supports steady epinephrine device familiarity among caregivers.
Demand Analysis of Auto-Injectors Market in China
China is adding more biologic therapy use through hospital systems and online health platforms. Local device makers and global suppliers compete on price and training support. Compatibility with specialty drugs adds another filter. China is forecast to grow at 6.3% CAGR through 2036 as biologic access and e-pharmacy ordering expand together. Larger cities will create the first volume layer. Regional hospitals will need lower-cost devices and simple patient teaching to extend use.
- Beijing and Shanghai tertiary hospitals form early use clusters for specialty biologic therapy.
- National Medical Products Administration review shapes drug-device launch timelines and device safety files.
- JD Health and Alibaba Health support digital pharmacy habits that can improve refill discipline.
Opportunity Analysis of Auto-Injectors Market in Japan
Japan favors compact and reliable devices that reduce handling stress for older patients. Auto-injectors gain interest in arthritis and endocrine care as home care tools improve. Allergy management adds a separate use base. Japan is expected to advance at 6.2% CAGR through 2036 as older patients favor simple activation and clear feedback. Domestic design strength supports refined device formats. Suppliers should match local comfort needs and language-light training steps.
- Tokyo and Osaka hospitals lead specialty care uptake through high physician access and advanced pharmacy support.
- Pharmaceuticals and Medical Devices Agency review keeps focus on device usability and patient safety evidence.
- Japan's aging care system favors compact packaging and simple activation for patients with reduced hand strength.
Future Outlook for Auto-Injectors Market in France
France has a strong reimbursement base and growing use of biologics in autoimmune care. Auto-injector demand benefits from care models that support patient autonomy after physician guidance. France is projected to rise at 6.1% CAGR through 2036 as self-use biologics gain access through community pharmacy networks. Allergy preparedness adds another device base in schools and public venues. Suppliers should focus on training materials suited to pharmacy counseling.
- Paris and Lyon specialty centers support early use of biologic delivery devices in autoimmune care.
- Haute Autorité de Santé review influences reimbursement access for drug-device products.
- French community pharmacies help patient counseling after hospital specialists start therapy.
Demand Outlook for Auto-Injectors Market in Germany
Germany has strong outpatient care and a large pharmaceutical base that supports self-use drug delivery. Auto-injectors are used in allergy care and chronic therapy as patients seek fewer clinic visits. Germany is forecast at 6.0% CAGR through 2036 as outpatient specialists use devices for autoimmune therapy. The growth pace is steady due to mature access and strict quality expectations. Suppliers must show dose accuracy and strong device support.
- North Rhine-Westphalia and Bavaria hold dense care networks that support specialty pharmacy access.
- Federal Institute for Drugs and Medical Devices oversight shapes safety evidence for drug-device products.
- German sickness funds keep price pressure high and favor devices with clear therapy value.
In-depth Analysis of Auto-Injectors Market in the United States
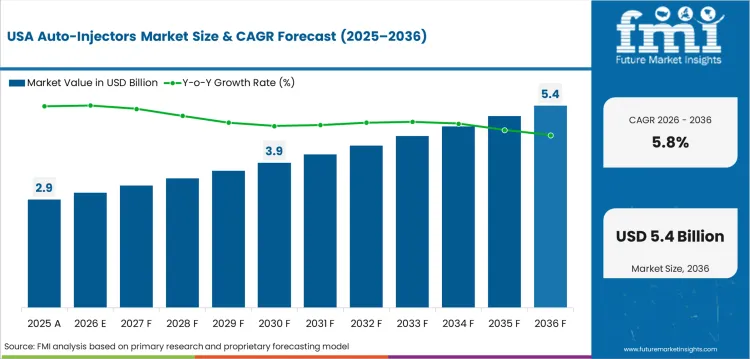
The United States has high baseline use across epinephrine devices and biologic therapy. That maturity keeps the growth rate lower than faster-expanding countries. The United States is expected to post 5.8% CAGR through 2036 as high current use leaves smaller room for share gains. Demand stays tied to specialty pharmacy access and payer coverage. Suppliers need strong patient services and reliable device supply to defend accounts.
- FDA guidance on combination products raises the evidence bar for auto-injector usability and safety.
- CVS Specialty and other specialty pharmacy channels support repeat biologic supply for chronic care patients.
- State school allergy policies support ongoing familiarity with epinephrine auto-injector use.
Competitive Landscape and Strategic Positioning
- • Becton Dickinson is a key player in auto-injectors because it has strong device engineering capability and works closely with pharmaceutical companies on drug-device delivery systems.
- • Sanofi, Pfizer, Viatris, Amgen, Eli Lilly, Novartis, and Bayer link auto-injector use with therapy portfolios in allergy care, autoimmune disease, and chronic treatment.
- • Newer device-focused players are working on easier activation, better patient feedback, connected dose support, and systems suited for high-viscosity biologics.
Becton Dickinson has a strong position because it supports pharmaceutical companies with prefilled delivery platforms and device development know-how. Sanofi has a strong link with allergy and chronic therapy portfolios. Pfizer and Viatris bring broad pharmaceutical access and established patient channels. Amgen, Eli Lilly, Novartis, and Bayer connect auto-injector use with biologic therapies and long-term treatment needs.
Barriers include high device development costs, strict human-factor testing, payer pressure, and the need to prove safe use by patients outside clinical settings. Strategic priorities include reducing user steps, improving dose accuracy, supporting home care treatment, and building stronger drug-device partnerships. Adjacent formats such as injection pens shape expectations for familiar handling. Wearable injectors influence comfort expectations during longer dosing cycles. Needle free injectors add pressure around needle anxiety and patient preference.
Key Companies in the Auto-Injectors Market
Key global companies leading the auto-injectors market include:
- • Becton, Dickinson and Company, Sanofi, Pfizer Inc., and Viatris Inc. have strong device or pharmaceutical infrastructure, established therapy access, and broad experience with injectable drug delivery systems.
- • Amgen Inc., Eli Lilly and Company, Novartis AG, and Bayer AG have strong positions because biologic therapy portfolios support repeat auto-injector use across chronic care settings.
- • Janssen Global Services LLC and other device-focused partners are gaining relevance by supporting patient training, dose reliability, and platform development for home-based treatment.
Competitive Benchmarking: Auto-Injectors Market
| Company | Device Platform Breadth | Human-Factor Testing Depth | Pharma Partner Access | Geographic Footprint |
|---|---|---|---|---|
| Becton, Dickinson and Company | High | High | Strong | Global |
| Sanofi | High | High | Strong | Global |
| Pfizer Inc. | Medium | High | Strong | Global |
| Viatris Inc. | Medium | Medium | Strong | Global |
| Amgen Inc. | Medium | High | Strong | Global |
| Eli Lilly and Company | Medium | High | Strong | Global |
| Novartis AG | Medium | High | Strong | Global |
| Bayer AG | Low | Medium | Moderate | Global |
| Janssen Global Services LLC | Medium | High | Strong | Global |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Auto-Injectors Market
- • In March 2026 embecta agreed to acquire Owen Mumford for up to £150 million, expanding drug-delivery device capabilities for generic and branded therapy segments globally.
- • In January 2026 Ypsomed and BD expanded their self-injection partnership with a 5.5 mL prefillable syringe compatible with YpsoMate 5.5 autoinjectors for large-volume biologics delivery.
Key Players in the Auto-Injectors Market
Major Global Players:
- • Becton, Dickinson and Company
- • Sanofi
- • Pfizer Inc.
- • Viatris Inc.
- • Amgen Inc.
- • Eli Lilly and Company
- • Novartis AG
- • Bayer AG
Emerging Players/Device-Focused Partners
- • Janssen Global Services LLC
- • Human-factor testing partners
- • Drug-device platform developers
- • Connected dose-support technology providers
- • Specialty pharmacy support partners
Report Scope and Coverage
| Parameter | Details |
|---|---|
| Quantitative Units | USD 5.73 billion in 2025 to USD 13.60 billion by 2036, at a CAGR of 8.2% |
| Market Definition | The auto-injectors market covers preset-dose injectable devices used for emergency care and chronic self-administration across allergy, autoimmune, neurology, and other therapy areas. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, UK, France, Germany, Japan, China, India, and other major healthcare markets |
| Key Companies Profiled | Becton, Dickinson and Company, Sanofi, Pfizer Inc., Viatris Inc., Amgen Inc., Eli Lilly and Company, Novartis AG, Bayer AG, and Janssen Global Services LLC |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up methodology using therapy mix, device revenue, channel access, and replacement-use assumptions. |
Auto-Injectors Market by Segments
Auto-Injectors Market Segmented by Product Type:
- Prefilled Auto-Injectors
- Fillable Auto-Injectors
Auto-Injectors Market Segmented by Indication:
- Anaphylaxis
- Multiple Sclerosis
- Rheumatoid Arthritis
- Other Uses
Auto-Injectors Market Segmented by Distribution Channel:
- Online Pharmacies
- Retail Pharmacies
- Hospital Pharmacies
- Drug Stores
Auto-Injectors Market Segmented by Usage Type:
- Disposable Devices
- Reusable Devices
Auto-Injectors Market Segmented by Activation Mechanism:
- Spring-Based Devices
- Motor-Driven Devices
- Manual-Assist Devices
Auto-Injectors Market Segmented by End User:
- Home Care
- Hospitals
- Clinics
- Emergency Settings
Auto-Injectors Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Western Europe
- Germany
- UK
- France
- Italy
- Spain
- Nordics
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Rest of Middle East & Africa
Research Sources and Bibliography
- Embecta Corp. (2024, November 21). Embecta announces definitive agreement to acquire Owen Mumford.
- Ypsomed. (2025, January 9). Ypsomed and BD expand partnership to advance self-injection systems for large-volume biologics.
- Food and Drug Administration. (2024, June). Essential drug delivery outputs for devices intended to deliver drugs and biological products: Draft guidance for industry.
- National Institute for Health and Care Excellence. (2024). Ongoing training in adrenaline auto-injector use.
- Guo, J., Weng, J., Zhu, Q., Zhou, F., Chen, Q., Gu, X., & Zhou, W. (2024). A review of recent FDA-approved biologic-device combination products. (4), 866-879.
This Report Answers
- What is the current and future size of the Auto-Injectors Market?
- How fast is the Auto-Injectors Market expected to expand between 2026 and 2036?
- Which product type is likely to lead the Auto-Injectors Market by 2026?
- Which indication is expected to account for the highest demand by 2026?
- What factors are driving demand for auto-injectors globally?
- How is biologic therapy use influencing auto-injector adoption?
- Why are disposable devices expected to remain the leading usage type?
- How are online pharmacies supporting repeat access to auto-injectors?
- Which countries are projected to show the fastest expansion through 2036?
- What is driving market expansion in India and the United Kingdom?
- Who are the key companies active in the Auto-Injectors Market?
- How does FMI estimate and validate the Auto-Injectors Market forecast?
Frequently Asked Questions
What is the expected size of the Auto-Injectors Market by 2036?
The Auto-Injectors Market is projected to reach USD 13.60 billion by 2036 from USD 6.20 billion in 2026.
What is the expected CAGR for the Auto-Injectors Market?
The Auto-Injectors Market is forecast to advance at 8.2% CAGR during the 2026 to 2036 forecast period.
Which product type leads the Auto-Injectors Market?
Prefilled auto-injectors are expected to lead product type demand with 64.0% share in 2026 as dosing steps are lower.
Which country grows fastest in the Auto-Injectors Market?
India is projected to record 6.5% CAGR through 2036 as specialty care access expands across large urban centers.
Which channel leads the Auto-Injectors Market?
Online pharmacies are estimated to hold 34.0% share in 2026 as refill ordering supports patients using chronic therapy.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Prefilled Auto-Injectors
- Fillable Auto-Injectors
- Prefilled Auto-Injectors
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication, 2026 to 2036
- Anaphylaxis
- Multiple Sclerosis
- Rheumatoid Arthritis
- Anaphylaxis
- Y to o to Y Growth Trend Analysis By Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Online Pharmacies
- Retail Pharmacies
- Hospital Pharmacies
- Online Pharmacies
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Usage Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Usage Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Usage Type, 2026 to 2036
- Disposable Devices
- Reusable Devices
- Disposable Devices
- Y to o to Y Growth Trend Analysis By Usage Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Usage Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Activation Mechanism
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Activation Mechanism, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Activation Mechanism, 2026 to 2036
- Spring-Based Devices
- Motor-Driven Devices
- Manual-Assist Devices
- Spring-Based Devices
- Y to o to Y Growth Trend Analysis By Activation Mechanism, 2021 to 2025
- Absolute $ Opportunity Analysis By Activation Mechanism, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Home Care
- Hospitals
- Clinics
- Home Care
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Indication
- By Distribution Channel
- By Usage Type
- By Activation Mechanism
- By End User
- Competition Analysis
- Competition Deep Dive
- Becton, Dickinson and Company
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Sanofi
- Pfizer Inc.
- Viatris Inc.
- Amgen Inc.
- Eli Lilly and Company
- Novartis AG
- Bayer AG
- Becton, Dickinson and Company
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Usage Type, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Activation Mechanism, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Usage Type, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Activation Mechanism, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Usage Type, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Activation Mechanism, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Usage Type, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Activation Mechanism, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 31: Eastern Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 32: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Usage Type, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Activation Mechanism, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 38: East Asia Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 39: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 40: East Asia Market Value (USD Million) Forecast by Usage Type, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Activation Mechanism, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 43: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 45: South Asia and Pacific Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 46: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 47: South Asia and Pacific Market Value (USD Million) Forecast by Usage Type, 2021 to 2036
- Table 48: South Asia and Pacific Market Value (USD Million) Forecast by Activation Mechanism, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 50: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 51: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 52: Middle East & Africa Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 53: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 54: Middle East & Africa Market Value (USD Million) Forecast by Usage Type, 2021 to 2036
- Table 55: Middle East & Africa Market Value (USD Million) Forecast by Activation Mechanism, 2021 to 2036
- Table 56: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Indication
- Figure 9: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Distribution Channel
- Figure 12: Global Market Value Share and BPS Analysis by Usage Type, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Usage Type, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Usage Type
- Figure 15: Global Market Value Share and BPS Analysis by Activation Mechanism, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Activation Mechanism, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Activation Mechanism
- Figure 18: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by End User
- Figure 21: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 23: Global Market Attractiveness Analysis by Region
- Figure 24: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 29: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 30: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 31: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 32: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Product Type
- Figure 35: North America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Indication
- Figure 38: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Distribution Channel
- Figure 41: North America Market Value Share and BPS Analysis by Usage Type, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Usage Type, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Usage Type
- Figure 44: North America Market Value Share and BPS Analysis by Activation Mechanism, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Activation Mechanism, 2026-2036
- Figure 46: North America Market Attractiveness Analysis by Activation Mechanism
- Figure 47: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 49: North America Market Attractiveness Analysis by End User
- Figure 50: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 51: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Product Type
- Figure 54: Latin America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Indication
- Figure 57: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 60: Latin America Market Value Share and BPS Analysis by Usage Type, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Usage Type, 2026-2036
- Figure 62: Latin America Market Attractiveness Analysis by Usage Type
- Figure 63: Latin America Market Value Share and BPS Analysis by Activation Mechanism, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Activation Mechanism, 2026-2036
- Figure 65: Latin America Market Attractiveness Analysis by Activation Mechanism
- Figure 66: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 68: Latin America Market Attractiveness Analysis by End User
- Figure 69: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 70: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Product Type
- Figure 73: Western Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Indication
- Figure 76: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 77: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 78: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 79: Western Europe Market Value Share and BPS Analysis by Usage Type, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Usage Type, 2026-2036
- Figure 81: Western Europe Market Attractiveness Analysis by Usage Type
- Figure 82: Western Europe Market Value Share and BPS Analysis by Activation Mechanism, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Activation Mechanism, 2026-2036
- Figure 84: Western Europe Market Attractiveness Analysis by Activation Mechanism
- Figure 85: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 87: Western Europe Market Attractiveness Analysis by End User
- Figure 88: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 94: Eastern Europe Market Attractiveness Analysis by Indication
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 97: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by Usage Type, 2026 and 2036
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by Usage Type, 2026-2036
- Figure 100: Eastern Europe Market Attractiveness Analysis by Usage Type
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Activation Mechanism, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Activation Mechanism, 2026-2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Activation Mechanism
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by End User
- Figure 107: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 108: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 109: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 110: East Asia Market Attractiveness Analysis by Product Type
- Figure 111: East Asia Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 112: East Asia Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 113: East Asia Market Attractiveness Analysis by Indication
- Figure 114: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 115: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 116: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 117: East Asia Market Value Share and BPS Analysis by Usage Type, 2026 and 2036
- Figure 118: East Asia Market Y-o-Y Growth Comparison by Usage Type, 2026-2036
- Figure 119: East Asia Market Attractiveness Analysis by Usage Type
- Figure 120: East Asia Market Value Share and BPS Analysis by Activation Mechanism, 2026 and 2036
- Figure 121: East Asia Market Y-o-Y Growth Comparison by Activation Mechanism, 2026-2036
- Figure 122: East Asia Market Attractiveness Analysis by Activation Mechanism
- Figure 123: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 125: East Asia Market Attractiveness Analysis by End User
- Figure 126: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 128: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 129: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 130: South Asia and Pacific Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 131: South Asia and Pacific Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 132: South Asia and Pacific Market Attractiveness Analysis by Indication
- Figure 133: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 134: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 135: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 136: South Asia and Pacific Market Value Share and BPS Analysis by Usage Type, 2026 and 2036
- Figure 137: South Asia and Pacific Market Y-o-Y Growth Comparison by Usage Type, 2026-2036
- Figure 138: South Asia and Pacific Market Attractiveness Analysis by Usage Type
- Figure 139: South Asia and Pacific Market Value Share and BPS Analysis by Activation Mechanism, 2026 and 2036
- Figure 140: South Asia and Pacific Market Y-o-Y Growth Comparison by Activation Mechanism, 2026-2036
- Figure 141: South Asia and Pacific Market Attractiveness Analysis by Activation Mechanism
- Figure 142: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 143: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 144: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 145: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 147: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 148: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 149: Middle East & Africa Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 150: Middle East & Africa Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 151: Middle East & Africa Market Attractiveness Analysis by Indication
- Figure 152: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 153: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 154: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 155: Middle East & Africa Market Value Share and BPS Analysis by Usage Type, 2026 and 2036
- Figure 156: Middle East & Africa Market Y-o-Y Growth Comparison by Usage Type, 2026-2036
- Figure 157: Middle East & Africa Market Attractiveness Analysis by Usage Type
- Figure 158: Middle East & Africa Market Value Share and BPS Analysis by Activation Mechanism, 2026 and 2036
- Figure 159: Middle East & Africa Market Y-o-Y Growth Comparison by Activation Mechanism, 2026-2036
- Figure 160: Middle East & Africa Market Attractiveness Analysis by Activation Mechanism
- Figure 161: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 162: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 163: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 164: Global Market - Tier Structure Analysis
- Figure 165: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

