Fibrin Glue Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
The Fibrin Glue Market is segmented by Product (Viral Inactivation, Autologous Donation, Recombinant Production, and Others), Type (Hemostat, Adhesive, and Sealant), Application (Cardiac Surgery, Pulmonary Surgery, Burn Bleeding, Vascular Surgeries, Orthopedic Surgeries, Lacerations of Liver and Spleen, Neurosurgery, Plastic Surgery, General Surgery, and Wound Management), and Region, by FMI.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Fibrin Glue Market Size, Market Forecast and Outlook By FMI
In 2025, the fibrin glue market was valued at USD 46.3 billion. Based on Future Market Insights' analysis, demand for fibrin glue is estimated to grow to USD 48.7 billion in 2026 and USD 76.9 billion by 2036. FMI projects a CAGR of 5.2% during the forecast period. Sector is expected to expand by an absolute dollar increase of USD 30.6 billion between 2026 and 2036. This represents steady, incremental expansion rather than a structural step-change, reflecting the essential but procedure-linked nature of fibrin sealants in surgery. Growth remains supported by rising surgical volumes and trauma care utilization, while pricing sensitivity in public healthcare systems and competition from synthetic hemostatic agents moderate the pace of expansion.
Industry is underpinned by a sustained increase in surgical volumes and a clinical preference for biologically derived agents that facilitate rapid hemostasis. As hospitals continue to work through backlogs of elective procedures and expand the use of minimally invasive techniques, the demand for versatile sealing solutions, ranging from liquid formulations to specialized patches, has become a key growth driver for major medical device manufacturers.
Summary of Fibrin Glue Market
- Market definition
- Fibrin glue is a biologic surgical sealant derived from clotting proteins that surgeons use to control bleeding and bond tissue during operative procedures.
- Demand drivers
- Rising cardiovascular and orthopedic surgical volumes are increasing hospital procurement of advanced hemostats that reduce intraoperative blood loss and shorten operating time.
- Expansion of minimally invasive and robotic surgeries is prompting surgeons to adopt precise tissue sealants that function in confined operative fields.
- Growth in trauma and emergency care infrastructure in emerging economies is raising demand for fast-acting fibrin-based hemostatic products in tertiary hospitals.
- Key segments analysed
- By product, recombinant fibrin glue holds approximately 55% share in 2026 due to lower pathogen transmission risk compared to plasma-derived variants.
- By type, hemostats account for nearly 60% of total demand, reflecting the product’s primary role in bleeding control.
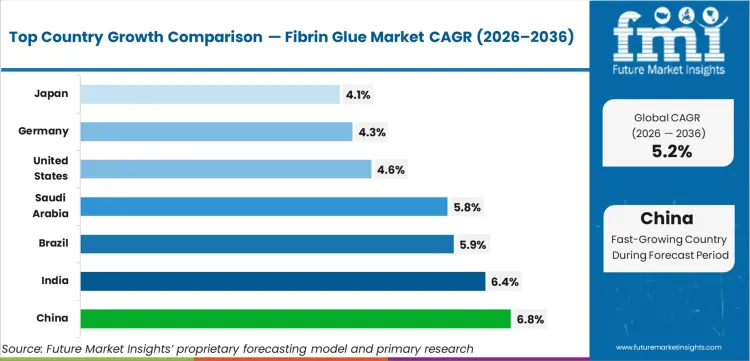
- By geography, China and India represent the fastest-growing national markets, while the United States remains the largest revenue contributor.
- Analyst opinion at FMI
- Nikhil Kaitwade, Principal Consultant for Chemicals & Materials at Future Market Insights, opines, ‘Fibrin glue demand is increasingly tied to surgical complexity and hospital protocol standardization rather than basic volume expansion, making product differentiation in safety and handling characteristics central to competitive positioning.’
- Strategic Implications/Executive Takeaways
- Manufacturers should prioritize recombinant production capacity to align with hospital infection-control policies and plasma safety concerns.
- Companies need to strengthen hospital tender participation strategies in emerging markets where public procurement drives bulk purchasing.
- Portfolio expansion into procedure-specific applicator systems can improve margins and deepen integration into surgical workflows.
- Methodology
- Market sizing integrates company-level revenue disclosures with national surgical procedure statistics for volume validation.
- Forecast assumptions incorporate regulatory approvals, reimbursement changes, and capacity expansion announcements.
- Estimates are triangulated across supply-side revenues, hospital demand indicators, and trade data to maintain internal consistency.
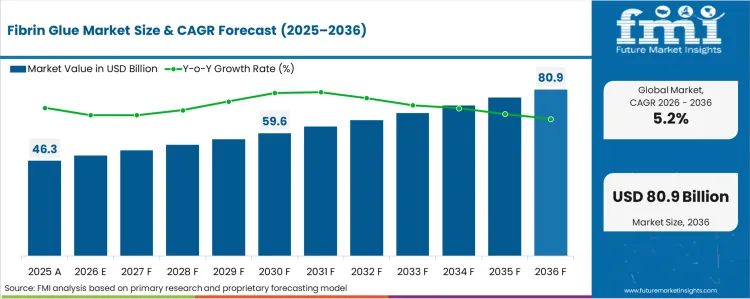
| Metric | Value |
|---|---|
| Estimated Value (2026E) | USD 48.7 billion |
| Forecast Value (2036F) | USD 76.9 billion |
| CAGR (2026 to 2036) | 5.2% |
Source: FMI analysis based on primary research and proprietary forecasting model
Companies are focusing on the operational efficiency of these products, positioning them as essential tools for improving patient outcomes and reducing surgical theater time in high-acuity environments such as cardiovascular and orthopedic surgery. As Andrew Hider, President and Chief Executive Officer of Baxter International, noted regarding the company’s surgical segment performance, “the Advanced Surgery business showed robust growth, increasing by 11% with contributions both across the portfolio and around the globe” [1].
China is projected to record a CAGR of 6.8% through 2036, supported by expanding tertiary hospital capacity and rising volumes of cardiovascular and orthopedic procedures in urban centers. India follows with a CAGR of 6.4%, driven by rapid growth in private hospital chains and wider adoption of advanced surgical sealants in Tier 1 and Tier 2 cities. Brazil is expected to grow at 5.9% CAGR, reflecting higher trauma surgery volumes and gradual modernization of public hospital procurement standards. The United States and Germany, representing mature markets, are projected to expand at 4.6% and 4.3% CAGR respectively. These countries generate largely replacement and procedure-linked demand rather than new market creation, with cost-containment frameworks in public reimbursement systems acting as a structural constraint on pricing expansion.
Fibrin Glue Market Definition
Fibrin glue is a biological tissue adhesive made from fibrinogen and thrombin, two proteins involved in natural blood clotting. When combined at the surgical site, these components form a stable fibrin clot that seals tissue, controls bleeding, and supports wound healing. The fibrin glue market covers the production and sale of these sealants for clinical use across hospitals and ambulatory surgical centers. Its primary function is hemostasis and tissue sealing during surgical procedures such as cardiovascular surgery, orthopedic repair, neurosurgery, and trauma care, where rapid bleeding control and secure tissue approximation are required.
Market Inclusions
This report covers global and regional market sizes for fibrin glue from 2026 to 2036, along with historical reference data. It includes segment breakdowns by product type such as recombinant and plasma-derived variants, by application including hemostats and surgical sealants, and by end-use setting. The analysis also incorporates pricing trends, procurement models in public and private hospitals, and selected trade flow observations for biologic sealants.
Market Exclusions
The scope excludes synthetic cyanoacrylate adhesives, collagen-based hemostatic sponges, oxidized regenerated cellulose products, and gelatin matrices unless fibrin components are incorporated. It also omits downstream finished surgical kits where fibrin glue is bundled but not separately priced. Veterinary sealants, research-grade laboratory fibrin products, and autologous platelet-rich plasma preparations are not included, as the focus remains on regulated, commercially manufactured fibrin sealants for human clinical use.
Research Methodology
- Primary Research: FMI analysts conducted structured interviews with hospital procurement managers, surgical specialists, biologics manufacturers, and regional distributors to assess demand patterns, pricing benchmarks, and product preferences.
- Desk research: Public sources reviewed include company annual reports, investor presentations, regulatory filings, transplant and surgical statistics from government health agencies, and disclosures from national reimbursement authorities.
- Market-sizing and forecasting: A hybrid approach was applied, combining bottom-up aggregation of company revenues and procedure volumes with top-down validation against national surgical case data and healthcare expenditure benchmarks.
- Data validation and update cycle: All estimates were cross-checked through triangulation of supply-side revenues, demand-side procedure counts, and import-export records where available. Forecast assumptions are revisited periodically as new regulatory approvals, capacity expansions, or pricing changes are disclosed.
Segmental Analysis
Fibrin Glue Market Analysis by Production Method
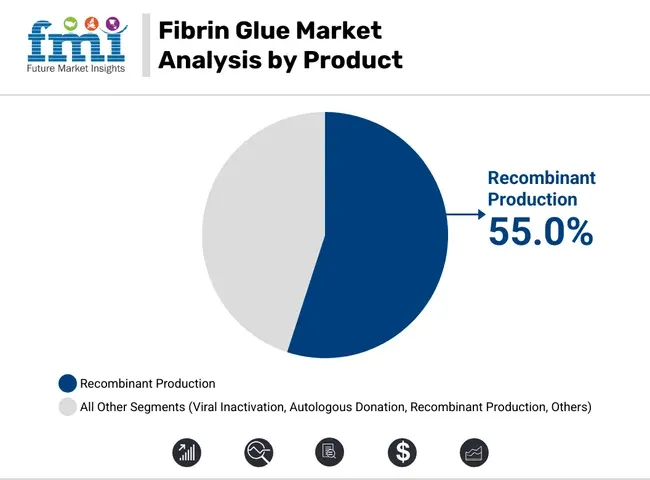
Recombinant production is estimated to hold a 55% share in 2026, as per FMI’s fibrin glue market assessment. Hospitals and regulators tend to favor manufacturing routes that reduce plasma-borne exposure concerns and simplify supplier qualification for high-risk surgical use. This preference strengthens uptake in facilities with strict infection-control and pharmacovigilance requirements.
- Manufacturing shift: Corza Medical closed the acquisition of TachoSil’s manufacturing operations in Linz (July 2024), highlighting how suppliers are restructuring production footprints around quality systems and continuity of supply for fibrin-based products. [2]
- Recombinant R&D: Patent filings disclose development of recombinant fibrinogen variants designed for improved stability in fibrin sealant applications, indicating sustained innovation aimed at non-plasma inputs. [3]
- Handling constraints: Product labeling for plasma-derived fibrin products continues to flag infectious-agent transmission risk, reinforcing buyer scrutiny of plasma dependence in tender evaluations. [4]
Fibrin Glue Market Analysis by Type
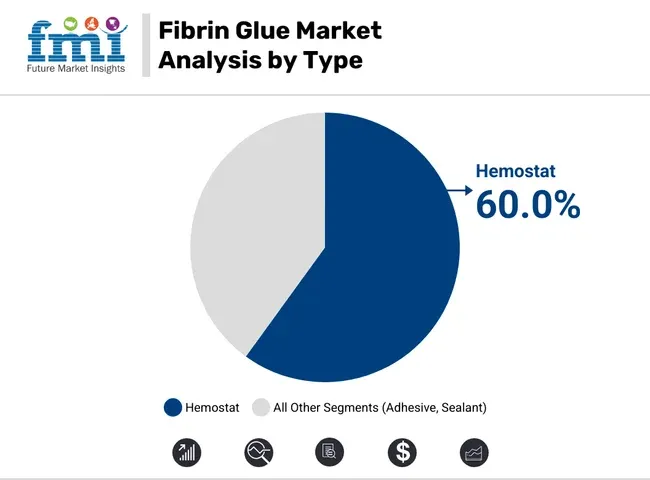
Hemostats are estimated to hold 60% share in 2026, reflecting fibrin glue’s core role in controlling surgical bleeding when standard techniques are insufficient. Surgeons value rapid clot formation at the point of care, especially in soft tissue procedures where suturing or cautery can be impractical. This keeps the category anchored to operating-room utilization rather than discretionary use.
- Asset consolidation: Corza’s July 2024 completion of the TachoSil manufacturing acquisition strengthens dedicated capacity for fibrin-based hemostatic patches, supporting availability for hospital purchasing cycles. [2]
- Indication expansion: Grifols reported FDA approval (October 2024) to use its fibrin sealant in pediatric surgical bleeding control, widening the addressable hemostasis pool in regulated settings. [5]
- Cold-chain reality: India’s CDSCO published Baxter’s September 2024 voluntary recall notice for TISSEEL kits tied to stability findings, underscoring storage-condition sensitivity that influences distributor and hospital inventory practices. [6]
Fibrin Glue Market Drivers, Restraints, and Opportunities
The fibrin glue market has historically expanded in line with surgical procedure volumes rather than population growth alone. Its valuation reflects the institutionalization of biologic hemostasis in operating-room protocols, particularly in cardiovascular, transplant, and complex orthopedic surgeries where bleeding control carries direct outcome implications. The market is neither early-stage nor saturated; it operates as a procedure-dependent specialty biologics segment anchored in hospital formularies and governed by stringent biologics regulation under agencies such as the USA Food and Drug Administration and the European Medicines Agency.
The central shift underway is a move from plasma-derived sealants toward recombinant and pathogen-reduced formulations. While recombinant variants often carry higher unit prices, hospitals weigh these premiums against infection-control priorities and medico-legal risk. At the same time, certain low-complexity procedures are reverting to mechanical or synthetic hemostatic options when cost pressure intensifies. The result is a rebalancing effect: premium products lift average selling prices, but tighter protocol-driven utilization tempers overall volume acceleration.
- Biologics regulation tightening: Oversight under frameworks such as the USA FDA’s Biologics License Application pathway increases compliance costs and post-marketing surveillance obligations. Manufacturers must invest in pharmacovigilance and validated plasma sourcing, which raises entry barriers but also stabilizes long-term supplier positions.
- Procedure complexity escalation: Growth in organ transplantation and minimally invasive cardiac interventions is expanding the addressable need for rapid, conformable tissue sealing. These higher-acuity cases require reliable clot formation in confined operative fields, supporting specialized fibrin formats over generic topical agents.
- Hospital budget compression: In publicly funded health systems, bundled reimbursement models and diagnosis-related group payment structures constrain discretionary biologic use. Procurement committees increasingly require head-to-head clinical justification before approving higher-priced recombinant products, limiting unchecked premiumization.
Regional Analysis
The fibrin glue market is assessed across North America, Latin America, Europe, Asia Pacific, and the Middle East and Africa, reflecting how hospital purchasing, payer reforms, and clinical preferences shape consumption patterns. Geographical segmentation is presented at regional and country levels, and the full report includes market attractiveness analysis by region and key surgical end-use clusters.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 6.8% |
| India | 6.4% |
| Brazil | 5.9% |
| Saudi Arabia | 5.8% |
| United States | 4.6% |
| Germany | 4.3% |
| Japan | 4.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific Market Analysis
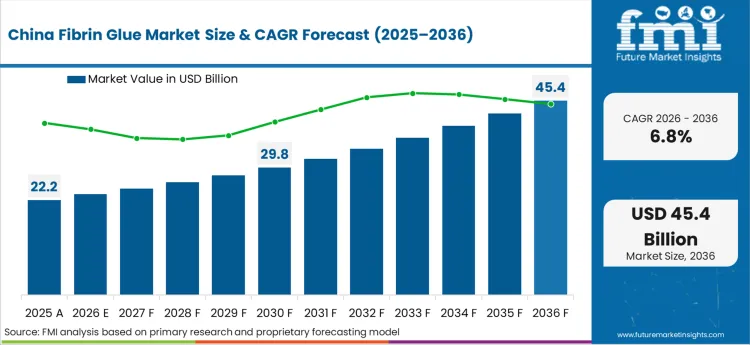
Asia Pacific is the cost-control testbed, where hospital purchasing is shaped as much by payer reform as by clinical preference. Domestic and multinational suppliers compete on supply continuity, usability in high-throughput surgical settings, and compliance with evolving reimbursement rules. Demand tends to concentrate in top-tier hospitals, with buyers prioritizing products that can justify utilization under tighter case-based payment.
- China: The market for fibrin glue in China is forecast to register a CAGR of 6.8% through 2036. Growth is shaped by how hospitals allocate spend under expanding case-based payment rules. In September 2024, an official briefing from China’s State Council Information Office cited wide adoption of DRG and DIP payment reform, reinforcing the direction of travel toward stricter utilization control for adjunct products used in surgery. [8] Domestic manufacturers are rapidly pivoting their R&D toward advanced, ready-to-use liquid formulations that eliminate thawing time, directly catering to the high-throughput efficiency demands of top-tier urban surgical centers.
- India: Sales of fibrin glue products in India are on track to record a CAGR of 6.4% through 2036. Demand builds through institutional purchasing rather than consumer pull, with large public hospitals and government-linked procurement channels shaping access. In April 2024, AIIMS Rishikesh floated procurement documentation for human fibrin sealant under a defined purchasing process, showing that fibrin sealants are treated as planned surgical inputs rather than ad hoc emergency items. [10] Procurement has also broadened beyond single-institution buying. To capitalize on the expanding network of specialized surgical hubs, tier-one suppliers are aggressively scaling up their localized cold-chain distribution networks to guarantee product integrity across diverse climatic zones.
- Japan: Japan's fibrin glue industry is projected to witness growth at a CAGR of 4.1% through 2036. The nation's hyper-aging demographic drives consistently high volumes of complex cardiovascular and orthopedic surgeries, inherently increasing the clinical demand for advanced hemostatic agents. Rapid integration of robotic-assisted minimally invasive surgeries is prompting suppliers to engineer highly specialized, extended-length applicator tips specifically tailored for precise robotic articulation in the operating room.
The FMI report gets in-depth into the Asia Pacific landscape by analyzing hyper-local reimbursement coding shifts, domestic biologic manufacturing subsidies, and the pricing elasticity of hemostatic agents. While China, India, and Japan drive the primary volume, other nations like South Korea and Australia are emerging as highly lucrative hubs, experiencing a notable rise in demand tied to their rapidly expanding medical tourism sectors and advanced cardiovascular centers of excellence.
North America Market Analysis
North America is the protocol-driven adoption engine, where fibrin sealants are embedded into surgical pathways and evaluated through formulary governance. Key players such as Grifols and Baxter compete on labeled indications, product consistency, and distribution reliability for time-sensitive biologics. Risk management committees that weigh clinical outcomes, adverse event history, and storage requirements influence purchases.
- United States: The United States fibrin glue sector is poised to expand at a CAGR of 4.6% through 2036. Labeled indications, hospital protocol governance, and a strong compliance culture around biologics anchor the market. In October 2024, Grifols announced FDA approval for its fibrin sealant solution to control surgical bleeding in pediatric patients, expanding the eligible treated population and supporting broader protocol adoption in children’s hospitals. [16] Regulatory labeling updates also influence hospital practice and contracting. The FDA’s biologics product page for TISSEEL, updated in October 2025, specifies use as an adjunct to hemostasis for adult and pediatric patients when conventional techniques are ineffective or impractical, which supports standardized inclusion in surgical pathways. [17] With a growing emphasis on value-based care, leading biologic manufacturers are heavily investing in comprehensive health economic models to conclusively demonstrate how premium sealants drastically reduce postoperative bleeding complications and subsequent readmission costs to hospital value analysis committees.
The FMI report provides an in-depth analysis of North American group purchasing organization (GPO) contracting, off-label usage protocols, and the financial impact of outpatient ambulatory surgical center (ASC) expansion. Beyond the United States, Canada represents a critical and increasingly lucrative hub, seeing a steady pull from centralized provincial health authorities prioritizing advanced surgical biologics to minimize hospital length-of-stay.
Europe Market Analysis
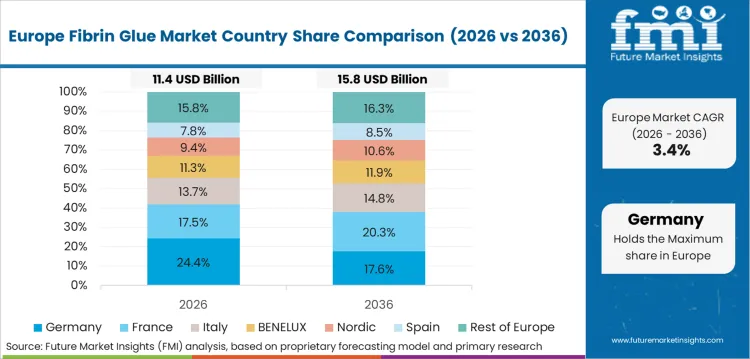
Europe is the biologics compliance stronghold, where national regulators and EU-wide pharmacovigilance influence how fibrin sealants are stocked and applied. Companies compete on safety documentation, controlled spray delivery, and training support for operating room staff. Hospital buyers also scrutinize traceability and labeling discipline, which shapes vendor qualification and contract renewals.
- Germany: Fibrin glue revenue in Germany is anticipated to increase by a CAGR of 4.3% through 2036. Germany’s demand is steady and replacement-led, shaped by strict biologics oversight and tightly managed hospital purchasing. The Paul-Ehrlich-Institut maintains a table of fibrin sealants with valid marketing authorisation, which underlines the emphasis on regulated product status and formal oversight for blood-derived and biologic medicines used in surgery. [18] Application practice is also influenced by safety controls around delivery methods. The European Medicines Agency completed a review of fibrin sealants administered by spray application, concluding that benefits outweighed risks provided measures are implemented to minimize gas embolism risk. [19] In response to Europe's aggressive push for sustainable healthcare, forward-looking suppliers are innovating their packaging solutions, transitioning toward fully recyclable, temperature-stable casing to perfectly align with stringent hospital green-procurement mandates.
Through the FMI report, stakeholders gain an in-depth understanding of Europe's strict EMA pharmacovigilance tracking, cross-border blood plasma sourcing dynamics, and the clinical shift toward synthetic alternative evaluations. United Kingdom and France, remain highly lucrative hubs, characterized by a steady rise in demand for specialized adjuncts utilized in complex neurosurgical and cardiovascular procedures within national health trusts.
Latin America Market Analysis
Latin America is the public-procurement anchor market, where tender specifications and registration status define access. Suppliers win share by meeting documentation requirements, ensuring cold-chain delivery, and supporting clinicians with product handling training. Demand is concentrated in high-volume public hospitals, and purchasing cycles follow budget calendars more than discretionary clinical preference.
- Brazil: FMI estimates the fibrin glue market in Brazil to expand at an annual growth rate of 5.9% through 2036. Public tendering and price-reference discipline are major determinants of utilization. A federal procurement notice published through Compras.gov.br lists the acquisition of fibrin sealant to meet hospital complex demand, indicating that access is frequently mediated by structured tenders rather than decentralized clinician preference. [13] Sub-national procurement reinforces this pattern. A September 2024 procurement document from the Federal District health authority includes fibrin sealant kit specifications, which highlights the importance of meeting technical requirements and documentation standards to remain eligible for bids. [14] To successfully navigate the complexities of these public tender cycles, multinational suppliers are increasingly forging strategic alliances with local distributors, specifically to guarantee uninterrupted cold-chain integrity and streamline import logistics across the vast Brazilian geography.
The FMI report delivers an in-depth assessment of the Latin American supply chain, breaking down regional tender forecasting, localized pricing indices, and the impact of fluctuating currencies on imported biologics. Mexico and Colombia are surfacing as highly lucrative hubs, showing a steady rise in demand for advanced surgical sealants driven by heavy investments in modernizing private healthcare infrastructure.
Middle East and Africa Market Analysis
The Middle East and Africa (MEA) region is rapidly evolving, driven by massive state investments in world-class medical cities and a growing focus on localized pharmaceutical manufacturing. The market places an incredibly high premium on uninterrupted supply chains and rigorous product tracking for human-derived biologic products.
- Saudi Arabia: The fibrin glue landscape in Saudi Arabia is set to achieve a CAGR of 5.8% through 2036. Driven by the ambitious Vision 2030 healthcare sector transformation, the Kingdom is aggressively expanding its tertiary care capabilities, particularly in oncology, neurosurgery, and trauma care. The Saudi Food and Drug Authority (SFDA) enforces strict serialization and tracking requirements for all blood-derived products to ensure maximum patient safety and ethical compliance. To secure long-term contracts within the booming network of state-of-the-art medical cities, global biologic giants are establishing localized secondary packaging and training hubs, directly aligning with the Kingdom's localization mandates.
The FMI report gets in-depth on the MEA market by mapping out giga-project hospital procurement pipelines, shifting localized regulatory frameworks, and the establishment of local biopharma industrial parks. UAE is seeing a surge in demand driven by premium medical tourism, while South Africa remains a pivotal advanced surgical distribution gateway for the broader Sub-Saharan continent.
Competitive Aligners for Market Players
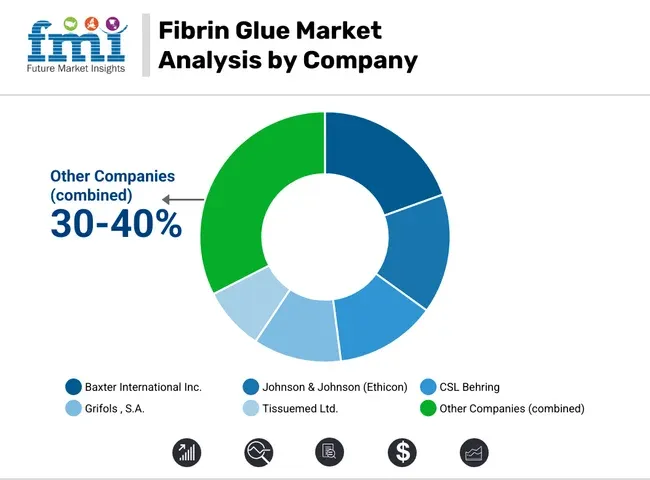
The competitive landscape for fibrin glue is moderately concentrated, led by a small set of global biologics and surgical adjunct suppliers with established hospital access. While share positions vary by region and indication mix, the market is not a long tail of small brands because regulatory approvals, plasma supply requirements, and operating-room protocol adoption create high barriers. The primary competitive variable is not list price alone, it is clinical acceptability under hospital protocols, covering label breadth, handling consistency, and reliable cold-chain supply.
Structural advantages sit with companies that control more of the biologics chain and the execution layer inside hospitals. Players with plasma collection or fractionation scale, validated quality systems, and long-running regulatory dossiers can protect continuity of supply and maintain buyer confidence during tender renewals. Separately, firms with strong applicator and packaging know-how gain an edge because preparation time, spray control, and waste reduction influence whether surgeons and operating-room teams standardize a product across service lines.
Buyer behavior is procurement-led and risk-managed. Large hospital systems and group purchasing bodies reduce dependency by qualifying multiple suppliers, splitting awards across framework contracts, and embedding service-level requirements such as delivery performance, training support, and batch traceability. This approach limits supplier pricing power in routine use cases, while specialty indications and products that reduce rework, shorten time-to-hemostasis, or lower wastage retain better pricing resilience.
Recent developments
- Kedrion Biopharma announced that medicines produced by fractionating Portuguese plasma would be distributed to hospitals by IPST, following the publication of Ordinance 173/2024/1 in July 2024. [19]
- Octapharma reported the completion of a Vienna site expansion that increased production volumes by 50% and expanded packaging and logistics capacity, February 2025. [20]
- Grifols announced plans to build a new manufacturing site in Spain to double plasma fractionation capacity in Europe in July 2025. [21]
Key Players in Fibrin Glue Market
- Baxter International Inc.
- Johnson & Johnson (Ethicon)
- CSL Behring
- Grifols, S.A.
- Takeda Pharmaceutical Company Limited
- STB Lifesaving Technologies
- Zimmer Biomet Holdings, Inc.
- Mallinckrodt Plc
- Shanghai RAAS Blood Products Co. Ltd.
- Hualan Biological Engineering Inc.
- Kaketsuken (Kaketsuken Co., Ltd.)
- Tissuemed Ltd.
- Vivostat A/S
- Haemostasis LLC
- CryoLife, Inc.
- Medtronic PLC
- Artivion, Inc. (formerly CryoLife)
- ProFibrix BV
- Sealantis Ltd.
- BioGlue (CryoLife)
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 48.7 billion (2026) to USD 76.9 billion (2036), at a CAGR of 5.2% |
| Market Definition | The fibrin glue market comprises the global production, distribution, and clinical utilization of biologically derived hemostatic agents that mimic natural blood coagulation, used in surgical procedures to control bleeding, seal tissues, and promote wound healing. |
| Product Segmentation | Viral Inactivation, Autologous Donation, Recombinant Production (55%), Others |
| Type Segmentation | Hemostat (60%), Adhesive, Sealant |
| Application Segmentation | Cardiac Surgery (38%), Pulmonary Surgery, Burn Bleeding, Vascular Surgeries, Orthopedic Surgeries, Lacerations of Liver and Spleen, Neurosurgery, Plastic Surgery, General Surgery, Wound Management |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia Pacific, Middle East and Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia, and 40 plus countries |
| Key Companies Profiled | Baxter International Inc., Johnson & Johnson (Ethicon), CSL Behring, Grifols, S.A., Takeda Pharmaceutical Company Limited, STB Lifesaving Technologies, Zimmer Biomet Holdings, Inc., Mallinckrodt Plc, Shanghai RAAS Blood Products Co. Ltd., Hualan Biological Engineering Inc., Kaketsuken, Tissuemed Ltd., Vivostat A/S, Haemostasis LLC, CryoLife, Inc., Medtronic PLC, Artivion, Inc., ProFibrix BV, Sealantis Ltd., BioGlue (CryoLife) |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling validated through primary interviews with fibrin glue manufacturers, surgeons, and hospital procurement specialists, supported by surgical procedure data and regulatory approval records |
Fibrin Glue Market Analysis by Segments
Product
- Viral Inactivation
- Autologous Donation
- Recombinant Production
- Others
Type
- Hemostat
- Adhesive
- Sealant
Application
- Cardiac Surgery
- Pulmonary Surgery
- Burn Bleeding
- Vascular Surgeries
- Orthopedic Surgeries
- Lacerations of Liver and Spleen
- Neurosurgery
- Plastic Surgery
- General Surgery
- Wound Management
Region
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia Pacific
- Middle East and Africa
Bibliography
- [1] Baxter International Inc. (2026, February 12). Baxter (BAX) Q4 2025 earnings call transcript. The Motley Fool.
- [2] Corza Medical. (July 2024). Corza Medical completes acquisition of TachoSil manufacturing operations. Corza Medical.
- [3] World Intellectual Property Organization. (2023). WO2023222770A1: Novel recombinant fibrinogen variants for fibrin sealants. Patentscope.
- [4] Baxter Healthcare Corporation. (2025). ARTISS fibrin sealant: Prescribing information (PDF). DailyMed, USA National Library of Medicine.
- [5] Grifols. (October 2024). Grifols receives FDA approval to treat surgical bleeding in pediatric patients with its fibrin sealant solution. Grifols.
- [6] Central Drugs Standard Control Organization. (September 2024). Voluntary recall of fibrin sealant TISSEEL kits (PDF). Government of India.
- [7] National Healthcare Security Administration of China. (November 2021). Notice on issuing the Three-Year Action Plan for DRG/DIP payment reform.
- [8] State Council Information Office of the People’s Republic of China. (September 2024). National Healthcare Security Administration briefing on payment reform progress.
- [9] Zhang, X., et al. (2024). Hospital response to a new case-based payment system in China. Health Policy and Planning.
- [10] AIIMS Rishikesh. (April 2024). Human fibrin sealant procurement document.
- [11] Chhattisgarh Medical Services Corporation. (February 2026). Tender document listing fibrin glue and related specifications.
- [12] Central Drugs Standard Control Organization. (September 2024). Voluntary recall notice for TISSEEL fibrin sealant kits, Baxter. [13] Compras.gov.br. (February 2025). Electronic tender for acquisition of fibrin sealant for hospital complex demand.
- [14] Secretaria de Saúde do Distrito Federal. (September 2024). Electronic tender edital listing fibrin sealant kit specifications.
- [15] Grifols. (October 2024). Grifols receives FDA approval to treat surgical bleeding in pediatric patients with its fibrin sealant solution.
- [16] USA Food and Drug Administration. (October 2025). TISSEEL, approved blood product information.
- [17] Paul-Ehrlich-Institut. (2026). Table of fibrin sealant with a valid marketing authorisation.
- [18] European Medicines Agency. (November 2012). Fibrinogen-containing solutions for sealant authorised for administration by spray application, EMA referral outcome.
- [19] Kedrion Biopharma. (July 2024). Medicines produced by Kedrion from Portuguese plasma now distributed to hospitals by IPST.
- [20] Octapharma. (February 2025). Octapharma expands production for the global market at its manufacturing site in Vienna.
- [21] Grifols. (July 2025). Grifols will build new manufacturing site in Spain to double its plasma fractionation capacity in Europe.
This Report Addresses
- Market intelligence to enable structured strategic decision-making across mature and emerging fibrin glue markets.
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, supported by validated surgical procedure data and manufacturer sales benchmarks.
- Growth opportunity mapping across product types, types, and applications, with emphasis on recombinant production technologies and cardiac surgery applications.
- Segment and regional revenue forecasts covering cardiac, orthopedic, general surgery, and wound management applications.
- Competition strategy assessment including recombinant technology positioning, clinical evidence generation, and regulatory approval strategies.
- Technology roadmap tracking including recombinant manufacturing advances, novel formulation development, and shelf-life improvements.
- Regulatory impact analysis covering FDA approvals, EMA oversight, PMDA requirements, and global biologic product standards.
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use.
Frequently Asked Questions
Where does the fibrin glue market stand today in value terms?
In 2025, the fibrin glue market was valued at USD 46.3 billion, forming the base for the current forecast cycle.
What revenue level should we plan for in the near term?
Demand for fibrin glue is estimated to reach USD 48.7 billion in 2026, reflecting steady procedure-linked expansion.
How large is the opportunity by the end of the forecast horizon?
The market is projected to grow to USD 76.9 billion by 2036, as per FMI’s forecast.
What pace of growth is expected across the decade?
FMI projects a CAGR of 5.2% during the forecast period from 2026 to 2036.
What is the absolute revenue expansion implied in the forecast?
The market expands from USD 48.7 billion in 2026 to USD 76.9 billion by 2036, adding USD 28.2 billion over the forecast window.
How meaningful is the jump from 2025 to 2026?
The increase from USD 46.3 billion in 2025 to USD 48.7 billion in 2026 represents measured year-on-year expansion rather than a demand spike.
Is this a high-growth or mid-growth biologics segment?
At a 5.2% CAGR through 2036, fibrin glue reflects mid-single-digit, protocol-driven expansion rather than rapid double-digit scaling.
What level of compounding should financial planning assume?
Strategic models should align with a 5.2% CAGR leading to USD 76.9 billion by 2036.
How much incremental headroom exists beyond the 2026 baseline?
From USD 48.7 billion in 2026 to USD 76.9 billion in 2036, the market creates USD 28.2 billion in additional addressable revenue.
Does the long-term outlook materially exceed the base-year valuation?
Yes, the market rises from USD 46.3 billion in 2025 to USD 76.9 billion by 2036 under the 5.2% CAGR scenario.
What structural forces keep growth at 5.2% rather than accelerating faster?
Expansion is anchored in surgical volumes and hospital protocol adoption, which support steady growth but moderate acceleration beyond the 5.2% CAGR forecast.
How secure is demand if procedure volumes fluctuate?
Even with procedure variability, the forecast still points to USD 76.9 billion by 2036, indicating durable institutional demand.
Is pricing power strong enough to shift the CAGR meaningfully?
Pricing improvements may support margins, yet overall growth remains aligned with the projected 5.2% CAGR through 2036.
Does the 2036 valuation suggest market maturity or transition?
Reaching USD 76.9 billion by 2036 indicates continued transition toward advanced sealants, not stagnation at the 2025 level of USD 46.3 billion.
How resilient is this segment compared to broader surgical adjunct markets?
The rise from USD 48.7 billion in 2026 to USD 76.9 billion by 2036 under a 5.2% CAGR reflects consistent, procedure-backed resilience.
Would you classify the opportunity as incremental or transformative?
The move from USD 46.3 billion in 2025 to USD 76.9 billion in 2036 reflects incremental scaling supported by a 5.2% CAGR.
How sensitive is the outlook to reimbursement discipline?
Despite reimbursement controls, the market is still expected to reach USD 76.9 billion by 2036 at a 5.2% CAGR.
Does the near-term step to USD 48.7 billion signal structural acceleration?
The 2026 value of USD 48.7 billion builds steadily on the 2025 base of USD 46.3 billion without indicating abrupt acceleration beyond the 5.2% CAGR path.
How does long-term growth compare with base-year stability?
Growth from USD 46.3 billion in 2025 to USD 76.9 billion in 2036 reflects sustained expansion over the decade.
Should capital allocation decisions align with aggressive or measured expansion?
With a projected 5.2% CAGR leading to USD 76.9 billion by 2036, the outlook supports disciplined, mid-growth capital planning rather than speculative expansion.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Recombinant Production
- Autologous Donation
- Viral Inactivation
- Others
- Recombinant Production
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type, 2026 to 2036
- Hemostat
- Adhesive
- Sealant
- Hemostat
- Y to o to Y Growth Trend Analysis By Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Type
- Competition Analysis
- Competition Deep Dive
- Johnson & Johnson (Ethicon)
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- CSL Behring
- Grifols, S.A.
- Takeda Pharmaceutical Company Limited
- STB Lifesaving Technologies
- Zimmer Biomet Holdings, Inc.
- Mallinckrodt Plc
- Johnson & Johnson (Ethicon)
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Type, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Type
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product
- Figure 23: North America Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Type, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Type
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product
- Figure 30: Latin America Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Type, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Type
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product
- Figure 37: Western Europe Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Type, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Type
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Type, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Type
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product
- Figure 51: East Asia Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Type, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Type
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Type, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Type
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Type, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Type
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

