Liposomal Doxorubicin Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
The liposomal doxorubicin market is segmented by Type (Branded; Generic), Application (Breast Cancer; Ovarian Cancer; AIDS-related Kaposi's Sarcoma; Multiple Myeloma; Other Solid Tumors) and Distribution Channel (Hospital Pharmacies; Retail Pharmacies; Mail-Order Pharmacies). Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Liposomal Doxorubicin Market Size, Market Forecast and Outlook By FMI
The liposomal doxorubicin market is expected to expand from USD 1.4 billion in 2026 to USD 2.4 billion by 2036. The market is anticipated to register a 6.3% CAGR during the forecast period. Branded formulations are likely to lead with an 87% share. Breast cancer applications are projected to account for 35% of application demand in 2026.
Summary of the Liposomal Doxorubicin Market
- Demand and Growth Drivers
- Oncology centers are expected to use liposomal doxorubicin for patients needing anthracycline therapy with lower cardiac risk.
- Combination therapy adoption is likely to increase use across breast cancer and ovarian cancer treatment programs.
- Cold-chain discipline is anticipated to influence supplier selection as injectable nanomedicines require strict storage control.
- Product and Segment View
- Branded formulations are expected to lead the type segment as physician trust favors established clinical safety records.
- Breast cancer is likely to stay the leading application as metastatic treatment plans often require tolerable chemotherapy options.
- Hospital pharmacies are anticipated to dominate distribution as oncology injectables need controlled handling and reliable product access.
- Geography and Competitive Outlook
- France is expected to lead country-level expansion as public oncology programs improve access to innovative therapies.
- Japan is likely to record strong uptake as aging patients require chemotherapy options with better tolerability.
- Companies with stable sterile injectable capacity are anticipated to strengthen standing across hospital procurement systems.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant for Healthcare at FMI, suggests, “The liposomal doxorubicin market is expected to favor manufacturers with proven formulation control and dependable oncology supply. Branded suppliers are likely to protect their position as hospitals prioritize safety records and consistent availability.”
- Liposomal Doxorubicin Market Value Analysis
- The liposomal doxorubicin market is shifting from standard chemotherapy use toward safer delivery-led oncology treatment.
- Demand is likely to rise as physicians seek anthracycline options with reduced cardiotoxicity for vulnerable patients.
- Adoption is expected to gain traction through hospital pharmacies managing sterile injectable storage and treatment scheduling.
- Spending is anticipated to be backed by cancer incidence pressure and continued need for advanced chemotherapy formats.
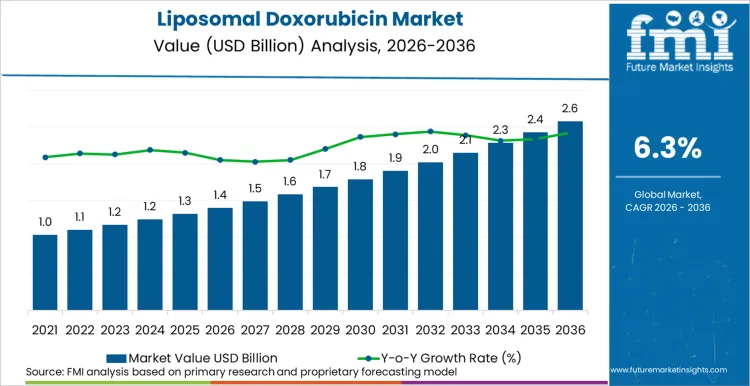
Liposomal Doxorubicin Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 1.4 Billion |
| Industry Value (2036) | USD 2.4 Billion |
| CAGR (2026-2036) | 6.3% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
What Primary Drivers Are Currently Accelerating Adoption of Lipid-Encapsulated Chemo?
The imperative for improved therapeutic indices is acting as a primary catalyst for the integration of drug delivery technology that targets tumor tissues passively via the Enhanced Permeability and Retention (EPR) effect, as per FMI's estimates. Recent advancements in pharmaceutical lipids have enabled the engineering of "stealth" liposomes that evade immune detection, significantly prolonging circulation time compared to free drugs. Efficacy appeals to oncologists seeking to treat elderly patients or those with pre-existing heart conditions who cannot tolerate standard anthracyclines. Manufacturers are capitalizing on trends by developing lipid nanoparticles that serve as versatile platforms for co-delivering doxorubicin with other synergistic agents.
Clinical validation of reduced side effects has expanded the scope of solution selection beyond palliative care. Formulations emphasizing safety are increasingly relying on phospholipid purity to ensure stable vesicle formation. FMI analysts opine that converters are introducing small molecule injectable variations that offer simplified reconstitution protocols for nurses. Integrating oncology adjuvants into treatment regimens is becoming considered standard, ensuring that liposomal doxorubicin is used effectively in adjuvant settings, meeting the rigorous survival benchmarks of modern oncology protocols.
How is Categorical Structure of Liposomal Doxorubicin Market Divided Across Key Segments?
The global landscape is intricately segmented to address the distinct technical requirements of solid tumor treatment, categorizing the industry by type, application, and distribution channel. Structural division allows stakeholders to identify specific capital flows, such as surging demand for chemotherapy drugs in ovarian cancer maintenance versus investment in lung cancer therapeutics, as per FMI's projection. By analyzing segments, manufacturers can tailor production lines to meet distinct handling needs of high-volume branded supply versus the cost-efficiency of emerging generics. Segmentation highlights the shift from broad-spectrum chemotherapy to targeted breast cancer drug protocols favoring liposomal formulations.
Why Does Branded Type Maintain Dominance?
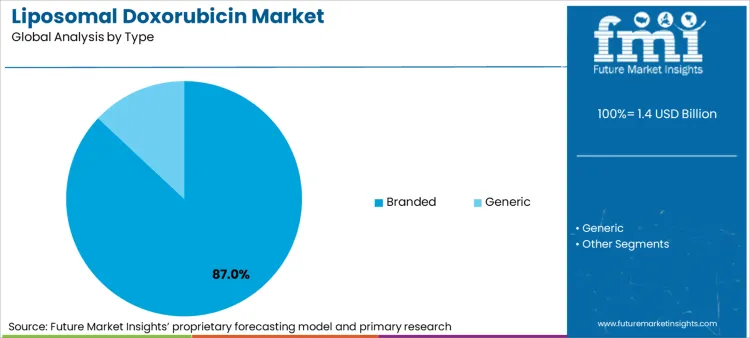
Branded formulations account for an 87% share in 2026, driven by the strong clinical legacy and physician trust associated with the pioneer product, Doxil/Caelyx. Leadership position is underpinned by the complex manufacturing process of "stealth" liposomes, which creates a high technical barrier for generic competitors to prove true bioequivalence. FMI is of the opinion that the segment benefits from the ubiquity of oncology based in vivo cro data supporting the branded version's safety profile over decades. Research focused on long-term survival suggests that oncologists prefer the established brand for curative-intent settings. Critical role in combination trials further solidifies standing, as most new parp inhibitors are tested in conjunction with the branded reference drug.
Why Does Breast Cancer Application Lead Market?
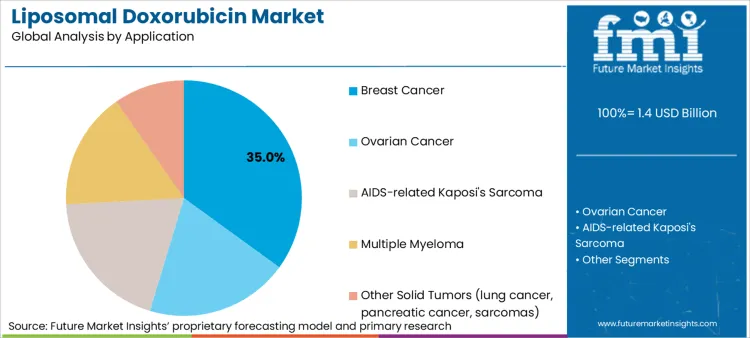
Breast Cancer commands 35% of the sector in 2026, reflecting the high incidence of metastatic cases where anthracyclines remain a potent option. Dominance is inextricably linked to the drug's ability to reduce the risk of congestive heart failure, a major concern for breast cancer survivors living longer due to improved therapies. Manufacturers rely on specialized monoclonal antibodies data to position liposomal doxorubicin as a viable partner for HER2-targeted agents. FMI analysts estimate that settings are essential for managing metastatic disease, driving priority status in procurement budgets. Relentless pursuit of better quality of life ensures that liposomal formulations remain top priority for cancer immunotherapy combination regimens.
Which Technical Dynamics Are Reshaping Trajectory of Nanomedicine R&D?
How Will Active Targeting Redefine Efficacy?
Ligand-targeted liposome adoption is set to reshape drug delivery priorities by demanding precise tumor homing. As passive targeting limits become apparent, researchers access pharmaceutical drug delivery market innovations to attach antibodies to liposome surfaces, as per FMI's estimates. Shift toward active targeting creates demand for sophisticated conjugation chemistry.
Manufacturers failing to explore targeted alternatives risk losing visibility among precision medicine advocates. Availability of peripheral t-cell lymphoma treatment options suggests potential utility in hematological malignancies. Rise of microfluidic manufacturing stimulates innovation, with companies differentiating offerings by providing liposomes with uniform size distribution for consistent biodistribution.
What Role Does Combination Therapy Play in Market Expansion?
Synergistic regimen adoption is set to reshape prescription strategies by demanding compatible formulations. As monotherapy resistance proliferates, sponsors access endometrial cancer treatment protocols combining liposomal doxorubicin with carboplatin or immunotherapy. FMI is of the opinion that the shift toward multi-modal care creates demand for drug stability compatibility studies.
Manufacturers failing to validate co-administration safety risk losing visibility among clinical trial designers. Availability of papillary thyroid cancer research drives complimentary consumption in rare cancer sectors. Rise of biomarker-driven prescriptions stimulates innovation, with companies differentiating offerings by aligning with specific genetic profiles.
How Will Liposomal Doxorubicin Expansion Unfold Across Key Global Regions?
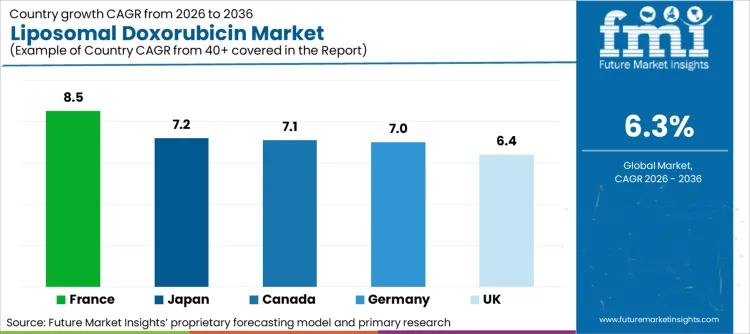
The global landscape for liposomal doxorubicin is characterized by divergent reimbursement policies and cancer care infrastructure. Established markets prioritize branded efficacy and safety profiles, whereas emerging economies rely on cost-effective generics and biosimilars. Western Europe is emerging as a rapid growth hub due to favorable reimbursement for high-cost oncology drugs. North American sectors pivot towards combination therapy trials and expanding indications. As per FMI's projection, government initiatives in developing nations supporting cancer care access ensure sustained demand for essential chemotherapies across all geographies.
| Country | CAGR (2026 to 2036) |
|---|---|
| France | 8.5% |
| Japan | 7.2% |
| Canada | 7.1% |
| Germany | 7.0% |
| UK | 6.4% |
Source: Future Market Insights' proprietary forecasting model and primary research
What Is Driving Explosive Growth in France's Oncology Sector?
The Liposomal Doxorubicin industry in France is poised to expand at 8.5% CAGR. Expansion is driven by the robust "Ten-Year Cancer Control Strategy" which emphasizes equitable access to innovative therapies.
According to the French National Cancer Institute (INCa), the number of new cancer cases in metropolitan France was estimated at 433,136 in 2023, necessitating a steady supply of core chemotherapeutics. FMI analysts opine that this high incidence rate drives revenue generation for established branded drugs in public hospitals. The government's focus on supportive care encourages facilities to opt for liposomal formulations to minimize cardiac side effects, aligning with long-term survivorship goals.
How is Aging Demographics Reshaping Japan's Market?
Sales of liposomal doxorubicin in Japan are set to rise at 7.2% CAGR. Growth is underpinned by the "super-aged" society, where the prevalence of ovarian and breast cancer increases with age, requiring tolerable chemotherapy options.
As per statistics from the National Cancer Center Japan, the projected number of cancer incidence in 2023 was approximately 1,028,500, highlighting the massive demand for oncological drugs. FMI analysts estimate that this demographic reality drives demand for treatments that preserve quality of life in elderly patients. Regulatory pressure from the PMDA for post-marketing safety surveillance validates the need for high-quality branded formulations, maintaining Japan's position as a premium market.
Why Is Canada Prioritizing Generic Access?
Demand for liposomal doxorubicin in Canada is projected to grow at 7.1% CAGR. The single-payer healthcare system is driving the integration of cost-effective generic alternatives to manage the rising oncology drug budget. According to Statistics Canada, cancer remains the leading cause of death, with an estimated 247,100 new cancer cases diagnosed in 2024. FMI is of the opinion that this burden highlights a massive market opportunity for companies providing bioequivalent generics. Government focus on value-based healthcare encourages facilities to opt for lower-cost therapeutics once patents expire, ensuring sustainable access.
What Role Does DKG Certification Play in Germany's Market?
The Liposomal Doxorubicin market in Germany is forecast to register 7.0% CAGR. The certification of cancer centers by the German Cancer Society (DKG) drives the standardization of treatment protocols, favoring evidence-based therapies like liposomal doxorubicin. According to the Robert Koch Institute (RKI), there are approximately 500,000 new cancer cases annually in Germany. FMI analysts estimate that this clinical volume drives demand for reliable supply chains of essential cancer drugs. High standards for pharmacy compounding attract significant investment in closed-system transfer devices to ensure safe handling.
How is NHS Funding Influencing UK's Market?
The Liposomal Doxorubicin industry in the UK is projected to expand at 6.4% CAGR. The NHS's centralized procurement and NICE guidelines dictate market access, favoring treatments with proven cost-effectiveness. As per NHS Digital statistics, there were 325,167 cancer registrations in England in 2022, a number that continues to rise. FMI's projection suggests that the shift necessitates systems capable of delivering high-volume chemotherapy efficiently. Government focus on reducing cancer waiting times encourages consumers (NHS Trusts) to opt for readily available formulations to clear treatment backlogs.
What Strategic Moves Are Defining the Competitive Landscape?
The Liposomal Doxorubicin market is consolidated at the top, dominated by the originator Johnson & Johnson (Janssen), but faces increasing pressure from a fragmented tier of generic manufacturers. FMI analysts opine that key players like Johnson & Johnson (20% share) maintain their lead by leveraging the strong brand equity of Doxil/Caelyx and ensuring robust supply chain reliability in a market prone to shortages.
The landscape features a dichotomy between this dominant innovator and generic challengers like Sun Pharma, Cipla, and Dr. Reddy's, who compete on price and availability. This structure fosters intense competition on bioequivalence data and tender-based procurement contracts. A critical strategic divergence exists between the "Brand-Loyalty" maintenance of the originator and the "Market-Penetration" strategy of generic firms. Johnson & Johnson differentiates itself through continued investment in post-market surveillance and combination trials, reinforcing the safety profile of Doxil in new indications like multiple myeloma combinations.
Cadila Pharmaceuticals and Cipla leverage their agile manufacturing capabilities to fill supply gaps during global shortages, offering a cost-effective alternative to healthcare systems strained by oncology budgets. Companies actively engage in strategic collaborations with hospital group purchasing organizations (GPOs) to secure long-term volume commitments, stabilizing revenue in a volatile generic market.
Key Players in Liposomal Doxorubicin Market
- Johnson & Johnson Services Inc.
- Merck & Co. Inc.
- Cadila Pharmaceuticals Ltd.
- Cipla Limited
- GlaxoSmithKline plc.
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy’s Laboratories Ltd.
- Baxter International Inc.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Type | Branded (Doxil/Caelyx), Generic |
| Application | Breast Cancer, Ovarian Cancer, AIDS-related Kaposi's Sarcoma, Multiple Myeloma, Other Solid Tumors (lung cancer, pancreatic cancer, sarcomas) |
| Distribution Channel | Hospital Pharmacies, Retail Pharmacies, Mail-Order Pharmacies |
| Regions | North America, Europe, East Asia, South Asia, Latin America, MEA |
Source: Future Market Insights – analysis driven by proprietary forecasting models and primary research
Key Segments
Type:
- Branded
- Generic
Application:
- Breast Cancer
- Ovarian Cancer
- AIDS-related Kaposi's Sarcoma
- Multiple Myeloma
- Other Solid Tumors
Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Mail-Order Pharmacies
Bibliography
- French National Cancer Institute. (2024). Key figures on cancer in France.
- National Cancer Center Japan. (2024). Cancer statistics in Japan.
- Statistics Canada. (2024). Cancer statistics at a glance. Government of Canada.
- Robert Koch Institute. (2024). Cancer in Germany report.
- NHS England. (2024). Cancer registration statistics, England. Government of the United Kingdom.
Frequently Asked Questions
How big is the global liposomal doxorubicin market?
The global liposomal doxorubicin market is estimated to be valued at USD 1.4 Billion in 2026.
What is the growth outlook for the liposomal doxorubicin market over the next 10 years?
The market is projected to reach USD 2.4 Billion by 2036, growing at a steady CAGR of 6.3%.
Which type drives adoption for liposomal doxorubicin?
Branded formulations lead adoption with an 87% share, driven by the complex manufacturing and regulatory barriers for generics.
How does adoption differ by region?
France leads growth at 8.5% CAGR driven by strong public health oncology initiatives, while Japan focuses on geriatric cancer care.
Who are the leading players in the liposomal doxorubicin market?
Johnson & Johnson Services Inc. leads with a 20% share, defined by the enduring market presence of its flagship product Doxil/Caelyx.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Roots Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended A9oach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Offering
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Offering , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Offering , 2026 to 2036
- Solution
- Services
- Solution
- Y to o to Y Growth Trend Analysis By Offering , 2021 to 2025
- Absolute $ Opportunity Analysis By Offering , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment Model
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment Model, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment Model, 2026 to 2036
- Cloud-based
- On-premises
- Cloud-based
- Y to o to Y Growth Trend Analysis By Deployment Model, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment Model, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- AI & ML
- Blockchain
- Big Data Analytics
- IoT
- Robotic Process Automation (RPA)
- Others
- AI & ML
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Insurance Companies
- Insurance Intermediaries
- Insurtech Firms
- Others
- Insurance Companies
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Offering
- By Deployment Model
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Offering
- By Deployment Model
- By Technology
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Offering
- By Deployment Model
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Offering
- By Deployment Model
- By Technology
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Offering
- By Deployment Model
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Offering
- By Deployment Model
- By Technology
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Offering
- By Deployment Model
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Offering
- By Deployment Model
- By Technology
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Offering
- By Deployment Model
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Offering
- By Deployment Model
- By Technology
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Offering
- By Deployment Model
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Offering
- By Deployment Model
- By Technology
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Offering
- By Deployment Model
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Offering
- By Deployment Model
- By Technology
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Offering
- By Deployment Model
- By Technology
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Offering
- By Deployment Model
- By Technology
- By End User
- Competition Analysis
- Competition Deep Dive
- Lemonade
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- ZhongAn Online
- Ping An OneConnect
- Oscar Health
- Root Insurance
- Acko
- Digit Insurance
- PolicyBazaar
- Alan
- Next Insurance
- Hippo
- Qover
- BIMA
- Trov
- Lemonade
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Offering , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Offering , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Offering , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Offering , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Offering , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Offering , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Offering , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Offering , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Offering , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Offering , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Offering
- Figure 6: Global Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Deployment Model
- Figure 9: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Technology
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Offering , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Offering , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Offering
- Figure 29: North America Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Deployment Model
- Figure 32: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Technology
- Figure 35: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by End User
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Offering , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Offering , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Offering
- Figure 42: Latin America Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Deployment Model
- Figure 45: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Technology
- Figure 48: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by End User
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Offering , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Offering , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by Offering
- Figure 55: Western Europe Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Deployment Model
- Figure 58: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Technology
- Figure 61: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by End User
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Offering , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Offering , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Offering
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Deployment Model
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by End User
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Offering , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Offering , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by Offering
- Figure 81: East Asia Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Deployment Model
- Figure 84: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Technology
- Figure 87: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by End User
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Offering , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Offering , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Offering
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Deployment Model
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Offering , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Offering , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Offering
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Deployment Model
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis

