Biomarker-based Immunoassays Market
The Biomarker-based Immunoassays Market is segmented by Sample Type (Blood, Tissue, Urine, Others), Product (Reagents & Kits, Instruments/Analyzers, Services), Biomarker Type (Efficacy & Pharmacodynamic, Predictive/Prognostic, Others), Disease (Cancer, Cardiovascular, Neurological, Others), End Use (Hospitals & Clinics, Diagnostic Labs, Research), and Region. Forecast for 2026 to 2036.
Biomarker-based Immunoassays Market Size, Market Forecast and Outlook By FMI
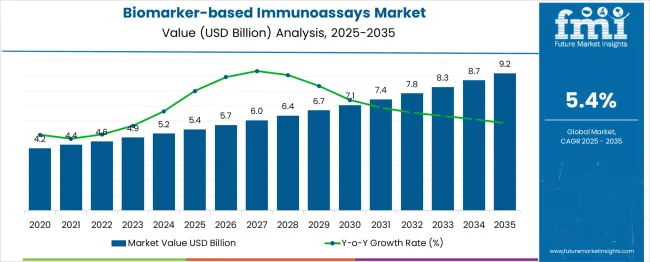
In 2025, the biomarker‑based immunoassays market was valued at USD 5.4 billion and is projected to grow to USD 5.69 billion in 2026 and USD 9.63 billion by 2036, reflecting a CAGR of 5.4%. Demand trends reflect a pivot from generalized screening to precision biomarker quantitation, driven by the urgency to reduce mortality in oncology and neurology through earlier intervention.
Growth is anchored in the "compliance gate" mechanism, where new regulatory approvals for high-sensitivity assays force healthcare providers to upgrade installed bases to meet stricter diagnostic standards. As per FMI's projection, hospitals and labs are prioritizing platforms that deliver femtogram-level sensitivity to satisfy FDA and EMA mandates for earlier disease detection, effectively rendering older, low-sensitivity photometric systems obsolete in premium care settings. This retrofit cycle is distinct from organic volume growth, as it compels capital expenditure on next-generation digital immunoassays and multiplexing equipment even during budget tightening.
Summary of Biomarker-based Immunoassays Market
- Biomarker-based Immunoassays Market Definition
- Industry covers antibody-based diagnostic tools for quantifying disease-specific biological markers.
- Demand Drivers in the Market
- Rising cancer and neurodegenerative disease prevalence requiring early detection.
- Shift toward minimally invasive blood-based testing.
- Regulatory approval of high-sensitivity companion diagnostics.
- Key Segments Analyzed in the FMI Report
- Sample Type: Blood dominates with 60.2% share due to non-invasive preference.
- Product: Reagents & Kits hold 42.8% share driven by recurring test volumes.
- Geography: High-growth Asia Pacific versus innovation-led North America.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare at Future Market Insights, opines, "In the updated version of the Biomarker-based Immunoassays Market Report for 2026 to 2036, CXOs will find that value is migrating from hardware to proprietary biomarker content. My findings point at a future where the ability to detect femtogram-level markers like pTau217 will determine platform viability, as 'good enough' sensitivity is no longer sufficient for new therapies."
- Strategic Implications/Executive Takeaways
- Prioritize antibody profiling partnerships with pharma to lock in companion diagnostic revenue.
- Decentralize testing via point-of-care formats to capture urgent care volume.
- Expand manufacturing in APAC to hedge against supply chain rigidities.
- Methodology
- Validated through first-hand corporate production and capacity data.
- Zero reliance on speculative third-party market research reports.
- Based on verifiable industrial benchmarks and verified sources.
Industry leaders are explicitly linking these technological shifts to patient outcomes. As noted by Professor Yu Jintai, Deputy Director of Neurology at Huashan Hospital, concluded, “Blood testing, with its minimally invasive and convenient nature, holds great promise as a diagnostic tool. Despite challenges in identifying biomarkers, we are excited to work with Danaher to make significant strides in early diagnosis, ultimately benefiting Alzheimer’s disease patients through more accessible and effective diagnostics.[1]" This development signals a shift where supplier success depends on integrating complex research biomarkers into routine clinical workflows.
Global demand dispersion highlights distinct regional maturity curves. China (7.3% CAGR) and India (6.8% CAGR) are scaling rapid screening infrastructure to address massive patient volumes, while Germany (6.2%), France (5.7%), and the UK (5.1%) focus on integrating advanced at-home biomarker testing and precision oncology into national health systems. The USA (4.6% CAGR) and Brazil (4.1% CAGR) remain volume-heavy markets transitioning toward value-based diagnostic reimbursement models.
Biomarker-based Immunoassays Market Definition
The biomarker-based immunoassay market comprises the global engineering, production, and deployment of biochemical tests that use antibody-antigen reactions to measure specific protein or genetic markers indicative of disease states. These assays quantify biological indicators such as cardiac troponin, prostate-specific antigen (PSA), or emerging neuro-markers like GFAP to guide clinical decision-making.
Biomarker-based Immunoassays Market Inclusions
Scope includes the full spectrum of diagnostic formats, ranging from high-throughput central lab analyzers to rapid lateral flow tests and next-generation digital platforms. It covers reagents, calibrators, and dedicated instrument hardware used across oncology, cardiology, and infectious disease applications. FMI analysts opine that the inclusion of associated immunoassay CDMO services is critical, as pharmaceutical partners increasingly outsource companion diagnostic development.
Biomarker-based Immunoassays Market Exclusions
The market excludes general chemistry testing and non-immunological molecular diagnostics (like PCR or NGS) unless they are part of a hybrid immunoassay platform. It also omits research-only antibodies sold without clinical validation for diagnostic use, as well as general lab consumables not specific to clinical immunodiagnostics workflows.
Biomarker-based Immunoassays Market Research Methodology
- Primary Research: Interviews with lab directors, regulatory consultants at FDA/EMA, and R&D heads at major diagnostic firms.
- Desk Research: Analysis of approved 510(k) clearances, WHO disease burden reports, and corporate annual filings.
- Market-Sizing and Forecasting: Bottom-up modeling based on test volume data from national health services validated against instrument install bases.
- Data Validation and Update Cycle: Cross-referencing reagent shipment volumes with reported disease incidence rates to ensure alignment.
Segmental Analysis
Biomarker-based Immunoassays Market Analysis by Sample Type
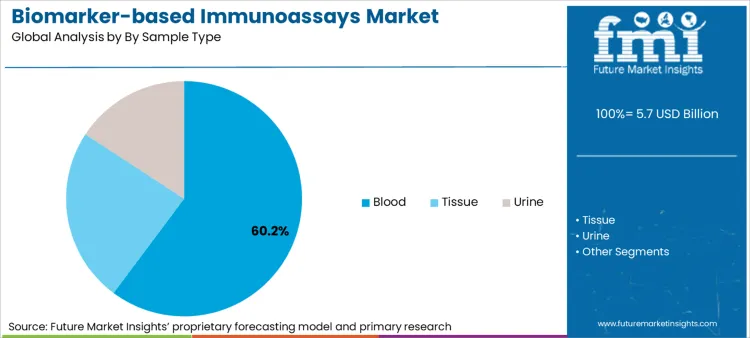
Blood samples command the largest share of the market, driven by the universal clinical preference for "liquid biopsy" workflows that reduce patient trauma and enable serial monitoring. According to FMI's estimates, blood-based testing accounts for 60.2% of revenue, as it allows for the simultaneous detection of multiplexed markers like PSA and cTn without surgical intervention.
- Diagnostic trigger: Demand is reinforced by the need for non-invasive monitoring, where blood assays replace tissue biopsies to track disease progression, shortening patient recovery times and enabling more frequent testing intervals.
- Sensitivity shift: Adoption accelerates where new digital platforms can detect low-abundance markers in plasma, allowing labs to identify neurological damage or early-stage cancer that was previously undetectable in blood samples.
- Volume consolidation: Procurement shifts because centralized labs can automate high-volume blood panel processing, reducing the cost-per-test and consolidating revenue around high-throughput hematology and immunoassay lines.
Biomarker-based Immunoassays Market Analysis by Product
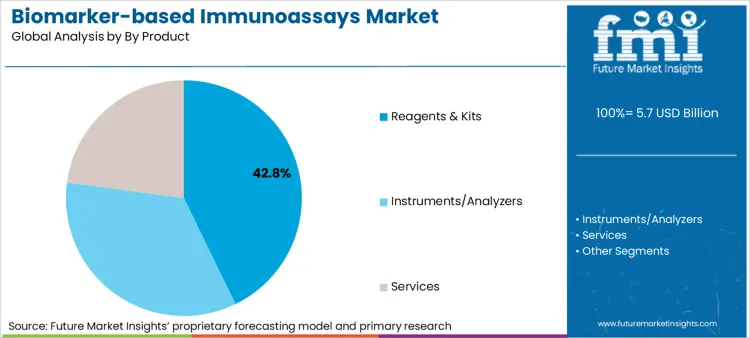
Reagents and kits lead the sector with a share of 42.8%, owing to their ability to serve as the recurring revenue engine for the industry, generating consistent cash flow that outpaces instrument capital expenditure. Manufacturers prioritize locked-in reagent contracts to secure long-term value from installed analyzers.
- Recurring revenue model: Growth is anchored in the "razor-and-blade" mechanism, where the installation of an analyzer guarantees a 5-to-7 year stream of proprietary reagent purchases, insulating suppliers from capex down-cycles. [4]
- Menu expansion: Value capture improves as vendors launch new disease-specific kits for existing platforms, allowing labs to expand their test menu without incurring new hardware costs or retraining staff.
- Supply chain resilience: Buyer priority shifts toward suppliers with regional reagent manufacturing hubs, ensuring that critical cytokine autoantigen biomarker test kits remain available during logistics disruptions or import freezes.
Biomarker-based Immunoassays Market Analysis by Disease
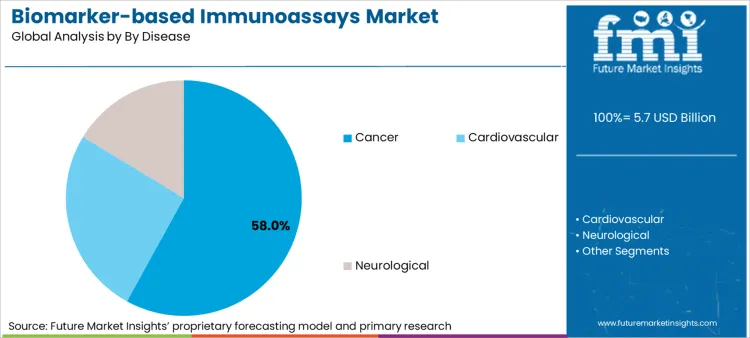
Oncology remains the dominant disease segment, representing 58% of application volume due to the critical nature of tumor markers in staging and therapy selection. The shift toward personalized medicine has made biomarker quantification a prerequisite for prescribing modern biological therapies.
- Therapeutic linkage: Uptake is driven by the mandatory pairing of drugs with companion diagnostics, where a positive biomarker result is the regulatory gatekeeper for accessing advanced cancer treatments.
- Screening intensity: Expansion tracks the rising global cancer burden, estimated to increase at a rapid pace, compelling health systems to implement population-scale screening programs using standardized immunoassays.
- Neurological emergence: Investment flows toward neuro immunoassay development as breakthrough therapies for Alzheimer's create a sudden, massive requirement for blood-based pTau and amyloid detection to qualify patients for treatment.
Biomarker-based Immunoassays Market Drivers, Restraints, and Opportunities
Drivers and Restraints
The primary driver propelling market expansion is the escalating global burden of chronic diseases, particularly cancer, which saves millions of lives annually. This mortality crisis forces health ministries and insurers to pivot from reactive treatment to proactive screening, fundamentally altering procurement behavior. Hospitals are aggressively adopting high-throughput immuno-oncology assay platforms to stratify patients earlier, a shift validated by Roche's projection that cancer deaths could rise to 18.5 million by 2050 without better diagnostic intervention. [11] Diagnostic volume is decoupling from general hospital admissions and realigning with preventive health mandates, driving sustained demand for predictive biomarker kits.
Industry faces a significant restraint in the form of sensitivity limitations and the high cost of next-generation platforms. While standard assays work well for established markers, they often fail to detect ultra-low concentrations required for early neurodegenerative diagnosis. For instance, optimizing detection limits for kidney damage biomarkers often bottoms out at 0.12 ng/mL, which may still be insufficient for presymptomatic screening in primary care. [3] This technical ceiling forces labs to invest in expensive digital or mass-spec hybrid systems, creating a capital barrier that slows adoption in cost-sensitive emerging markets and smaller community clinics.
Market Opportunities
- Digital sensitivity leap: Suppliers can capture premium tier pricing by launching digital immunoassays capable of femtogram-level detection, as demonstrated by startups securing government grants to commercialize ultra-sensitive devices for Alzheimer's markers.
- Point-of-care decentralization: Growth opportunities exist in porting complex central lab tests to bedside formats, evidenced by FDA clearances for rapid TBI blood tests that deliver lab-quality brain injury results in 15 minutes.
- Companion diagnostic partnerships: Diagnostic firms can secure long-term volume by co-developing assays with pharma, leveraging the trend where companies work with hundreds of partners to deliver PD-L1 biomarker testing for novel cancer drugs.
Regional Analysis
Based on the regional analysis, the Biomarker-based Immunoassays market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
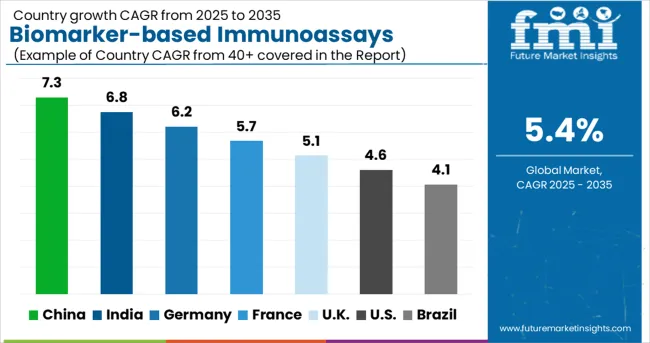
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 7.3% |
| India | 6.8% |
| Germany | 6.2% |
| France | 5.7% |
| UK | 5.1% |
| USA | 4.6% |
| Brazil | 4.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific Biomarker-based Immunoassays Market Analysis
Asia Pacific is rapidly transforming from a volume-importer to a technology-incubator, driven by heavy government investment in deep-tech diagnostics. FMI analysts opine that the region's focus on localized manufacturing and "health sovereignty" is creating a new competitive tier of suppliers capable of serving both domestic mass markets and export channels.
- China: Demand for biomarker-based immunoassays in China is set to grow at 7.3% CAGR through 2036, supported by strategic research collaborations that localize advanced marker discovery before procurement cycles translate into higher deployment volumes. This trajectory is supported by the recent Danaher-Huashan Hospital partnership, which aims to accelerate early Alzheimer's identification through domestic clinical trials. [6]
- India: Growth tracks a CAGR of 6.8% as stringent quality control mandates for infectious disease screening force blood banks to upgrade from manual methods to automated ELISA and CLIA systems. This shift is reflected in the National Institute of Biologicals' enforcement of approved screening methods for HIV and Hepatitis, ensuring that only validated commercial kits are procured for national safety.
FMI’s report includes a comprehensive evaluation of the Asia Pacific landscape. Beyond China and India, the analysis highlights South Korea and Japan as critical markets where aging populations are driving demand for radioimmunoassay replacements and neuro-diagnostic innovations. In South Korea, government-backed funding for startups like Bredis Healthcare signals a strong push toward commercializing home-grown digital immunoassay technologies.
Europe Biomarker-based Immunoassays Market Analysis
Europe remains the global center for "value-based diagnostics," where reimbursement is increasingly tied to the clinical utility of the biomarker rather than the test volume. The region prioritizes automation and connectivity to manage workforce shortages while maintaining high standards for oncology and cardiac care.
- Germany: The Germany biomarker-based immunoassays sector is poised to expand at 6.2% CAGR during 2026 to 2036 because rising cancer incidence rates require robust, high-throughput screening infrastructure which lifts utilisation rates and justifies technology refresh decisions. This demand is underscored by RKI data showing over half a million new cancer cases annually, necessitating efficient diagnostic workflows. [7]
- France: France is likely to see biomarker-based immunoassays revenues increase at 5.7% CAGR during 2026 to 2036, given the national push for early detection programs and the resulting push for capacity and workflow upgrades.
- UK: The outlook for biomarker-based immunoassays in the UK indicates a 5.1% CAGR from 2026 to 2036 as GP referral pathways for cancer symptoms tighten, strengthening demand from both public programmes and private operators. The pressure to diagnose 90% of cancer patients presenting with symptoms drives the adoption of rapid exclusion tests in primary care.
FMI’s report includes a detailed breakdown of the European market. The study also identifies Switzerland and the Netherlands as key hubs for high-value coronavirus immunoassay research and headquarters for major diagnostic conglomerates. These markets are critical for early adoption of novel biomarkers before they scale across the wider EU bloc.
North America Biomarker-based Immunoassays Market Analysis
North America is characterized by a "speed-to-answer" culture, where regulatory clearances for rapid point-of-care devices are driving market value. The integration of complex biomarker panels into emergency medicine is reshaping the traditional central lab model.
- USA: The USA is expected to record 4.6% CAGR in biomarker-based immunoassays demand over 2026 to 2036 as FDA clearances for rapid brain injury tests bridge the gap between ER triage and lab confirmation, tightening the link between operational constraints and adoption-led investment. This is evidenced by the recent clearance of point-of-care TBI assays that provide results in 15 minutes, allowing immediate clinical intervention.
FMI’s report includes an in-depth assessment of North America. Canada is also analyzed as a growing market where provincial health systems are harmonizing testing standards. The region offers significant opportunities for united states immunoassay cdmo players to support the burgeoning biotech sector in developing proprietary companion diagnostics.
Latin America Biomarker-based Immunoassays Market Analysis
Latin America is emerging as a volume-growth engine, where expanding public health coverage is increasing access to basic diagnostic services. The focus is on cost-effective screening for high-mortality conditions like cancer and infectious diseases.
- Brazil: Brazil is projected to post 4.1% CAGR for biomarker-based immunoassays between 2026 and 2036 as survival rates for conditions like cholangiocarcinoma remain low due to late diagnosis, improving the conversion rate from pilots to enterprise rollouts. Government data indicates 5-year survival rates as low as 9% for late-stage cases, driving a frantic policy push for earlier biomarker screening. [10]
FMI’s report includes continued coverage of Latin America. Mexico and Chile are identified as opportunistic markets where private hospital chains are investing in modern diagnostic equipment. These countries are increasingly adopting international quality standards, opening the door for global suppliers to introduce mid-tier automated platforms.
Competitive Aligners for Market Players
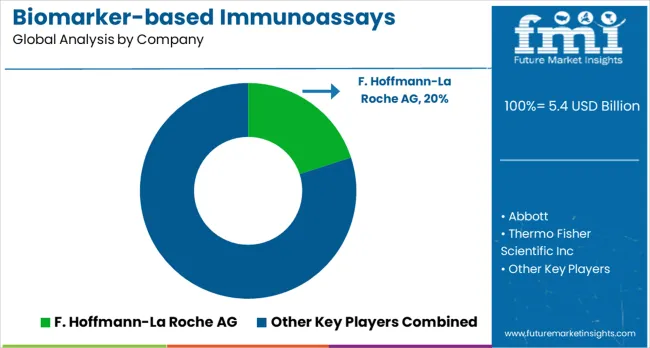
The competitive landscape is defined by a "menu wars" dynamic, where the primary differentiator is no longer just hardware speed, but the breadth of the exclusive biomarker menu available on the platform. Players like Roche and Abbott are locking in market share by securing FDA approvals for proprietary companion diagnostics that competitors cannot offer, effectively creating a monopoly on testing for specific new drug therapies.
Consolidation of the "care continuum" is another key strategy, with companies moving to bridge the gap between central labs and near-patient testing. By offering the same high-sensitivity biomarker assays on both massive track systems and portable handheld devices, manufacturers ensure they capture revenue regardless of where the patient interacts with the health system. This is exemplified by the push to bring traumatic brain injury markers from the core lab directly to the ER bedside.
Regulatory expertise has become a critical competitive moat. The ability to navigate increasingly complex dual-track approvals, simultaneously satisfying FDA breakthrough designations and EU IVDR compliance, separates tier-1 integrators from smaller reagent suppliers. Companies that can deliver fully validated, compliant "lab-ready" solutions with guaranteed supply chains are winning long-term contracts over lower-cost but riskier alternatives. [5]
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
- In April 2025, Labcorp launched the nationwide pTau‑217/Beta Amyloid 42 Ratio blood-based biomarker immunoassay to aid Alzheimer’s disease diagnosis (measures pTau‑217 and Beta Amyloid 42 in ratio) [8].
- In June 2025, Fujirebio expanded its neuro testing portfolio by launching the fully automated Lumipulse® G sTREM2 RUO immunoassay for quantitative measurement of sTREM2 (a microglial activation biomarker) on LUMIPULSE® G analyzers [9].
- In June 2025, Quanterix announced its HD‑X Simoa Immunoassay Analyzer received Class 1 Medical Device registration in South Korea (MFDS) via distributor HS Biosystems, supporting expanded reach into IVD and large reference labs [12].
- In September 2025, Beckman Coulter Diagnostics launched the industry’s first fully automated, high-throughput Access BD‑Tau RUO immunoassay for use on the DxI 9000 and Access 2 analyzers, and also announced development of an Aβ‑42 RUO immunoassay [2].
Key Players in Biomarker-based Immunoassays Market
- F. Hoffmann-La Roche
- Abbott
- Thermo Fisher Scientific
- Eurofins Scientific
- QIAGEN
- Bio-Rad Laboratories
- Siemens Healthineers
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 5.69 billion (2026) to USD 9.63 billion (2036), at a CAGR of 5.4% |
| Market Definition | The biomarker-based immunoassay market comprises the global engineering, production, and deployment of biochemical tests that use antibody-antigen reactions to measure specific protein or genetic markers indicative of disease states. |
| Sample Type Segmentation | Blood, Tissue, Urine, Others |
| Product Segmentation | Reagents & Kits, Instruments/Analyzers, Services |
| Application Coverage | Cancer, Cardiovascular, Neurological, Others |
| Regions Covered | North America, Latin America, Europe, Asia Pacific, Middle East & Africa |
| Countries Covered | USA, Canada, Brazil, Mexico, Germany, UK, France, China, India, Japan, South Korea |
| Key Companies Profiled | F. Hoffmann-La Roche, Abbott, Thermo Fisher Scientific, Eurofins Scientific, QIAGEN, Bio-Rad Laboratories, Siemens Healthineers |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with lab directors and regulatory consultants |
Biomarker-based Immunoassays Market Analysis by Segments
Sample Type
- Blood
- Tissue
- Urine
- Others
Product
- Reagents & Kits
- Instruments/Analyzers
- Services
Biomarker Type
- Efficacy & Pharmacodynamic
- Predictive/Prognostic
- Others
Disease
- Cancer
- Cardiovascular
- Neurological
- Others
End Use
- Hospitals & Clinics
- Diagnostic Labs
- Research
Region
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- [1] G-MedTech. (2025, January 19). Danaher and Huashan Hospital collaborate on early Alzheimer’s disease diagnosis.
- [2] BD-Quanterix Corporation. (2025, June 23). Quanterix expands reach with IVD and large reference labs.
- [3] National Institutes of Health (NIH). (2024). Optimization of immunoassay LODs for kidney damage biomarkers.
- [4] Roche Diagnostics. (2024). Companion diagnostics scale and pharmaceutical partnerships.
- [5] National Institute of Biologicals (NIB). (2025). Regulatory approval for infectious disease biomarkers in India.
- [6] Danaher Corp & Huashan Hospital. (2025). Strategic partnership for Alzheimer's biomarker research.
- [7] Robert Koch-Institut (RKI). (2025). Cancer incidence in Germany.
- [8] Labcorp. (2025). Labcorp launches first-of-its-kind immunoassay to accelerate path to diagnosis of Alzheimer’s disease.
- [9] Beckman Coulter Diagnostics. (2025, September 10). Beckman Coulter Diagnostics launches industry first fully automated, high throughput.
- [10] Instituto Nacional de Câncer (INCA). (2026). Cholangiocarcinoma survival rates and recurrence in Brazil.
- [11] Roche Diagnostics. (2025). Early cancer detection progress and global mortality projections.
- [12] Fujirebio Diagnostics. (2025). FDA endorses first-of-its-kind blood test for Alzheimer’s disease [Press release].
This Report Addresses
- Market intelligence to enable structured strategic decision making across mature and emerging diagnostic economies
- Market size estimation and 10 year revenue forecasts from 2026 to 2036, supported by validated volume and install base benchmarks
- Growth opportunity mapping across blood, tissue, and urine sample types with emphasis on non-invasive transitions
- Segment and regional revenue forecasts covering oncology, neurology, and cardiology applications
- Competition strategy assessment including menu expansion models, reagent lock-in positioning, and regulatory capability benchmarking
- Product innovation and sensitivity tracking including digital immunoassays and point-of-care cleared grades
- Regulatory impact analysis covering FDA breakthrough designations, IVDR mandates, and export driven quality upgrades
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is the demand for biomarker-based immunoassays in the global market in 2026?
Demand for biomarker-based immunoassays in the global market is estimated to be valued at USD 5.69 billion in 2026.
What will be the market size of biomarker-based immunoassays in the global market by 2036?
Market size for biomarker-based immunoassays is projected to reach USD 9.63 billion by 2036.
What is the expected demand growth for biomarker-based immunoassays in the global market between 2026 and 2036?
Demand for biomarker-based immunoassays in the global market is expected to grow at a CAGR of 5.4% between 2026 and 2036.
Which sample type is poised to lead global sales by 2026?
Blood samples are expected to be the dominant form, capturing approximately 60.2% of global market share in 2026 due to the preference for non-invasive testing.
How significant is the role of Hospitals & Clinics in driving biomarker-based immunoassays adoption in 2026?
Hospitals & clinics represent a critical segment, holding a substantial share as acute care settings prioritize rapid diagnostic turnaround.
What is driving demand in China?
Growth is driven by government-backed research collaborations and massive infrastructure scaling for early disease screening.
What compliance standards or regulations are referenced for China?
National Medical Products Administration (NMPA) standards and partnership-driven clinical trial validations are referenced.
What is the China growth outlook in this report?
China is projected to grow at a CAGR of 7.3% during 2026 to 2036.
Why is Europe described as a priority region in this report?
Europe is a hub for value-based diagnostics, prioritizing high-sensitivity assays to improve long-term patient outcomes and reduce hospitalization costs.
What type of demand dominates in Europe?
Replacement driven demand for premium grade, automated platforms with extensive biomarker menus dominates regional consumption.
What is Germany growth outlook in this report?
Germany is projected to expand at a CAGR of 6.2% during 2026 to 2036.
Does the report cover France in its regional analysis?
Yes, France is included within Europe under the regional scope of analysis.
What are the sources referred to for analyzing France?
National health data and cancer incidence reports are used to benchmark demand in France.
What is the main demand theme linked to France in its region coverage?
Demand is associated with national screening programs aimed at early oncology detection.
Does the report cover Brazil in its regional analysis?
Yes, Brazil is included within Latin America under the regional coverage framework.
What is the main Brazil related demand theme in its region coverage?
Demand is driven by urgent requirements to improve survival rates for late-stage cancers through better screening.
Which product formats or configurations are strategically important for North America supply chains?
Rapid point-of-care cartridges and cartridges for decentralized testing are prioritized to speed up time-to-result.
What is biomarker-based immunoassays and what is it mainly used for?
It is a diagnostic technique using antibodies to detect specific biological markers for diseases like cancer and heart conditions.
What does biomarker-based immunoassays market mean in this report?
It refers to the global trade and consumption of reagents, kits, and instruments for biomarker quantification.
What is included in the scope of this biomarker-based immunoassays report?
Scope covers reagents, instruments, and services across blood, tissue, and urine samples for various disease indications.
What is excluded from the scope of this report?
General chemistry tests, pure molecular diagnostics without immunoassay components, and research-only antibodies are excluded.
What does market forecast mean on this page?
Market forecast represents a model based projection built on defined assumptions for strategic planning purposes.
How does FMI build and validate the biomarker-based immunoassays forecast?
Forecast is developed using hybrid top down and bottom up modeling validated through primary interviews and install base data.
What does zero reliance on speculative third party market research mean here?
Primary interviews and verifiable public datasets are used instead of unverified syndicated market estimates.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By By Sample Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By By Sample Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By By Sample Type , 2026 to 2036
- Blood
- Tissue
- Urine
- Blood
- Y to o to Y Growth Trend Analysis By By Sample Type , 2021 to 2025
- Absolute $ Opportunity Analysis By By Sample Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By By Product, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By By Product, 2026 to 2036
- Reagents & Kits
- Instruments/Analyzers
- Services
- Reagents & Kits
- Y to o to Y Growth Trend Analysis By By Product, 2021 to 2025
- Absolute $ Opportunity Analysis By By Product, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By By Biomarker Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By By Biomarker Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By By Biomarker Type, 2026 to 2036
- Efficacy & Pharmacodynamic
- Predictive/Prognostic
- Efficacy & Pharmacodynamic
- Y to o to Y Growth Trend Analysis By By Biomarker Type, 2021 to 2025
- Absolute $ Opportunity Analysis By By Biomarker Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By By Disease
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By By Disease, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By By Disease, 2026 to 2036
- Cancer
- Cardiovascular
- Neurological
- Cancer
- Y to o to Y Growth Trend Analysis By By Disease, 2021 to 2025
- Absolute $ Opportunity Analysis By By Disease, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By By End Use, 2026 to 2036
- Hospitals & Clinics
- Diagnostic Labs
- Research
- Hospitals & Clinics
- Y to o to Y Growth Trend Analysis By By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By By Sample Type
- By By Product
- By By Biomarker Type
- By By Disease
- By By End Use
- Competition Analysis
- Competition Deep Dive
- F. Hoffmann-La Roche
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Abbott
- Thermo Fisher Scientific
- Eurofins Scientific
- QIAGEN
- Bio-Rad Laboratories
- Siemens Healthineers
- F. Hoffmann-La Roche
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by By Sample Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by By Product, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by By Biomarker Type, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by By Disease, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by By End Use, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by By Sample Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by By Product, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by By Biomarker Type, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by By Disease, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by By End Use, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by By Sample Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by By Product, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by By Biomarker Type, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by By Disease, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by By End Use, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by By Sample Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by By Product, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by By Biomarker Type, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by By Disease, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by By End Use, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by By Sample Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by By Product, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by By Biomarker Type, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by By Disease, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by By End Use, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by By Sample Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by By Product, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by By Biomarker Type, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by By Disease, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by By End Use, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by By Sample Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by By Product, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by By Biomarker Type, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by By Disease, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by By End Use, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by By Sample Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by By Product, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by By Biomarker Type, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by By Disease, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by By End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by By Sample Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by By Sample Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by By Sample Type
- Figure 6: Global Market Value Share and BPS Analysis by By Product, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by By Product, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by By Product
- Figure 9: Global Market Value Share and BPS Analysis by By Biomarker Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by By Biomarker Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by By Biomarker Type
- Figure 12: Global Market Value Share and BPS Analysis by By Disease, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by By Disease, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by By Disease
- Figure 15: Global Market Value Share and BPS Analysis by By End Use, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by By End Use, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by By End Use
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by By Sample Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by By Sample Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by By Sample Type
- Figure 32: North America Market Value Share and BPS Analysis by By Product, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by By Product, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by By Product
- Figure 35: North America Market Value Share and BPS Analysis by By Biomarker Type, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by By Biomarker Type, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by By Biomarker Type
- Figure 38: North America Market Value Share and BPS Analysis by By Disease, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by By Disease, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by By Disease
- Figure 41: North America Market Value Share and BPS Analysis by By End Use, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by By End Use, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by By End Use
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by By Sample Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by By Sample Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by By Sample Type
- Figure 48: Latin America Market Value Share and BPS Analysis by By Product, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by By Product, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by By Product
- Figure 51: Latin America Market Value Share and BPS Analysis by By Biomarker Type, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by By Biomarker Type, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by By Biomarker Type
- Figure 54: Latin America Market Value Share and BPS Analysis by By Disease, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by By Disease, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by By Disease
- Figure 57: Latin America Market Value Share and BPS Analysis by By End Use, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by By End Use, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by By End Use
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by By Sample Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by By Sample Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by By Sample Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by By Product, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by By Product, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by By Product
- Figure 67: Western Europe Market Value Share and BPS Analysis by By Biomarker Type, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by By Biomarker Type, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by By Biomarker Type
- Figure 70: Western Europe Market Value Share and BPS Analysis by By Disease, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by By Disease, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by By Disease
- Figure 73: Western Europe Market Value Share and BPS Analysis by By End Use, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by By End Use, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by By End Use
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by By Sample Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by By Sample Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by By Sample Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by By Product, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by By Product, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by By Product
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by By Biomarker Type, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by By Biomarker Type, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by By Biomarker Type
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by By Disease, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by By Disease, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by By Disease
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by By End Use, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by By End Use, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by By End Use
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by By Sample Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by By Sample Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by By Sample Type
- Figure 96: East Asia Market Value Share and BPS Analysis by By Product, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by By Product, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by By Product
- Figure 99: East Asia Market Value Share and BPS Analysis by By Biomarker Type, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by By Biomarker Type, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by By Biomarker Type
- Figure 102: East Asia Market Value Share and BPS Analysis by By Disease, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by By Disease, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by By Disease
- Figure 105: East Asia Market Value Share and BPS Analysis by By End Use, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by By End Use, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by By End Use
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by By Sample Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by By Sample Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by By Sample Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by By Product, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by By Product, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by By Product
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by By Biomarker Type, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by By Biomarker Type, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by By Biomarker Type
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by By Disease, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by By Disease, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by By Disease
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by By End Use, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by By End Use, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by By End Use
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by By Sample Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by By Sample Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by By Sample Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by By Product, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by By Product, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by By Product
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by By Biomarker Type, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by By Biomarker Type, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by By Biomarker Type
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by By Disease, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by By Disease, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by By Disease
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by By End Use, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by By End Use, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by By End Use
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

