Branded Generics Market
Branded Generics Market Size and Share Forecast Outlook 2026 to 2036
Branded Generics Market Forecast and Outlook 2026 to 2036
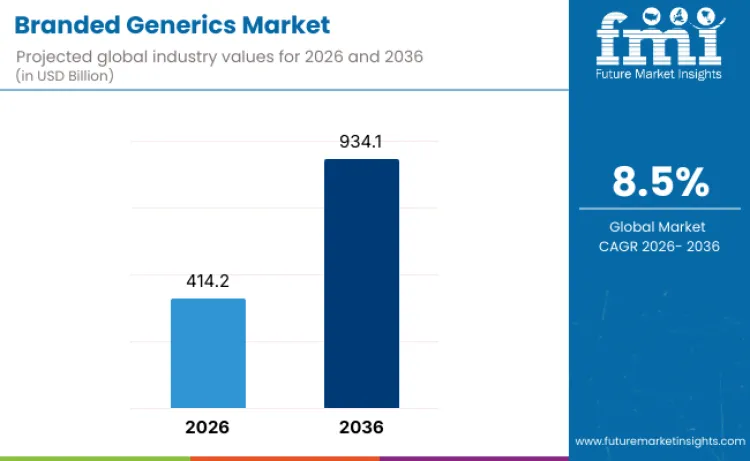
Branded generics revenue is forecast to reach USD 414.2 billion in 2026 and expand at an 8.5% CAGR through 2036, taking the market to USD 934.1 billion by the end of the period. Growth is being reinforced by rising demand for lower-cost, branded alternatives to originator medicines, especially in emerging economies where access and affordability shape prescribing and procurement decisions. The ongoing patent-expiry cycle is widening the addressable opportunity set, while improving manufacturing depth and regulatory readiness among generic producers is supporting faster product rollouts and broader therapeutic coverage.
Competition is shifting toward value-added branded formulations, including differentiated dosing formats and patient-friendly presentations, rather than commodity generics alone, as companies seek stronger positioning with prescribers, pharmacies, and payers. Expansion is also supported by stricter bioequivalence and quality-validation requirements that raise confidence in branded generics and strengthen brand-led substitution in high-volume categories. Demand remains anchored in chronic disease therapy, where long treatment durations and large patient pools sustain repeat purchasing across retail and institutional channels. In parallel, public and private cost-containment measures are accelerating the use of branded generics within formulary-driven pathways, particularly in developing markets where healthcare infrastructure and insurance coverage are expanding alongside the need for affordable branded medicines.
Quick Stats for Branded Generics Market
- Branded Generics Industry Value (2026): USD 414.2 Billion
- Branded Generics Industry Forecast Value (2036): USD 934.1 Billion
- Branded Generics Industry Forecast CAGR: 8.5%
- Branded Generics Industry Leading Application Segment: Anti-diabetic drugs (31.7%)
- Branded Generics Industry Leading Therapy Area: Cardiovascular diseases (20.8%)
- Branded Generics Industry Key Growth Countries: India, China, Brazil, South Korea, UK, USA
- Branded Generics Industry Key Players: Mylan (Viatris), Novartis AG, Teva Pharmaceuticals, Pfizer Inc., Sun Pharmaceutical, Aspen Pharmacare, Abbott Laboratories, Bausch Health, GSK Pharmaceuticals, Zydus Lifesciences
Branded Generics Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 414.2 Billion |
| Industry Value (2036) | USD 934.1 Billion |
| CAGR (2026-2036) | 8.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
What Drivers Are Accelerating Biosimilar Integration?
Momentum toward complex generics is speeding up the adoption of biosimilar branded alternatives, as manufacturers improve process control and scale in biologics production. These advances are helping biosimilars meet comparability expectations while expanding availability and supporting broader market access in high-cost specialty categories. Strong uptake of monoclonal antibody biosimilars, especially within oncology, has reinforced confidence in biosimilars and encouraged expansion into additional therapy areas. At the same time, education on interchangeability, switching protocols, and specialty-pharmacy handling is helping formulary committees move more decisively from evaluation to inclusion. Health systems are responding with structured procurement and contracting strategies that prioritize predictable supply and measurable cost reduction. Alongside this, routine pharmacovigilance and real-world monitoring are increasingly built into biosimilar rollout plans, strengthening ongoing safety oversight and sustaining prescriber and payer confidence.
How Is the Branded Generics Market Segmented Across Key Categories?
Global landscape is intricately segmented to address diverse requirements of the pharmaceutical industry, categorizing sector by drug classification, therapeutic indication, formulation type, and distribution channel. Structural division allows stakeholders to identify specific treatment targets, such as rising demand for anti-diabetic formulations in metabolic disorders or preference for extended-release preparations in chronic pain management. By analyzing these segments, manufacturers can tailor development strategies to meet distinct needs of hospital formularies versus retail pharmacy requirements. Segmentation also highlights shift from basic generic substitution approaches to comprehensive branded therapeutic interventions. As per FMI's estimates, industry sophistication leads to granular segmentation becoming essential for understanding prescription patterns and reimbursement frameworks, ensuring product development aligns with evolving healthcare policies and therapeutic guidelines.
Why Do Anti-Diabetic Drugs Maintain Dominance?
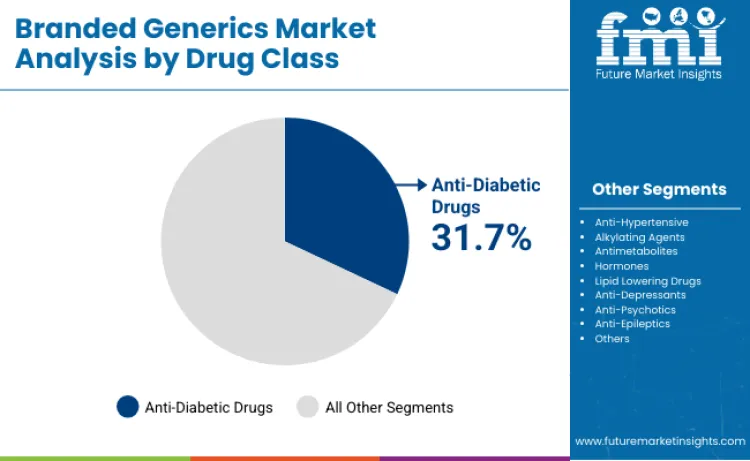
Anti-diabetic drugs are projected to hold 31.7% of drug class share in 2026, supported by the scale of diabetes treatment demand and the need for continuous therapy in long-duration care pathways. Their leadership is reinforced by standardized chronic disease management protocols and the routine use of insulin and non-insulin regimens across major healthcare systems. The segment also benefits from steady product upgrades, particularly combination therapies that reduce pill burden and support tighter glycemic control in real-world settings.
- Large, recurring patient base tied to long-term therapy cycles
- Combination regimens improving treatment convenience and outcomes
- Broader positioning linked to complication management pathways, including renal and cardio-metabolic risk considerations
- Guidelines emphasizing early intervention sustaining volume across primary and specialist care
Why Are Oral Formulations the Preferred Delivery Method?
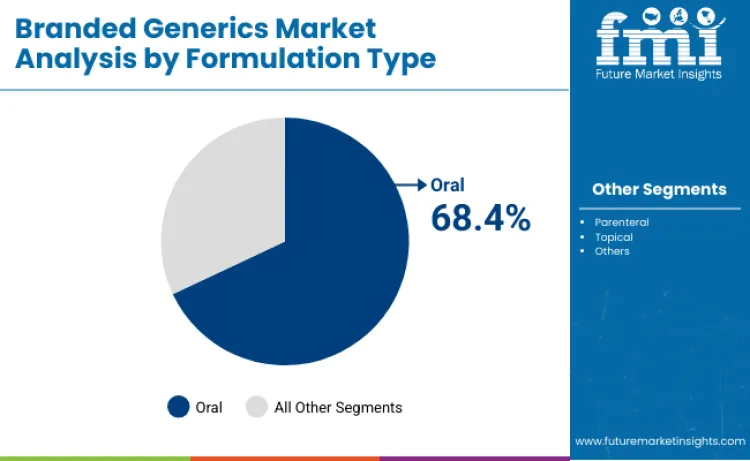
Oral formulations are expected to command 68.4% share in 2026, mainly because they align with patient preference for convenient administration and fit routine outpatient care models. Their dominance is closely linked to better adherence versus injectable routes and the ability to scale high-volume supply at competitive cost points. Manufacturers continue to optimize release profiles and patient experience to protect share in chronic-use categories.
- Convenient dosing supports higher persistence in long-term regimens
- Strong fit with retail pharmacy and outpatient prescribing workflows
- Formulation improvements (release control, palatability) expanding use across age groups
- Cost and manufacturing scalability supporting broad accessibility
Why Does Cardiovascular Disease Lead Therapy Applications?
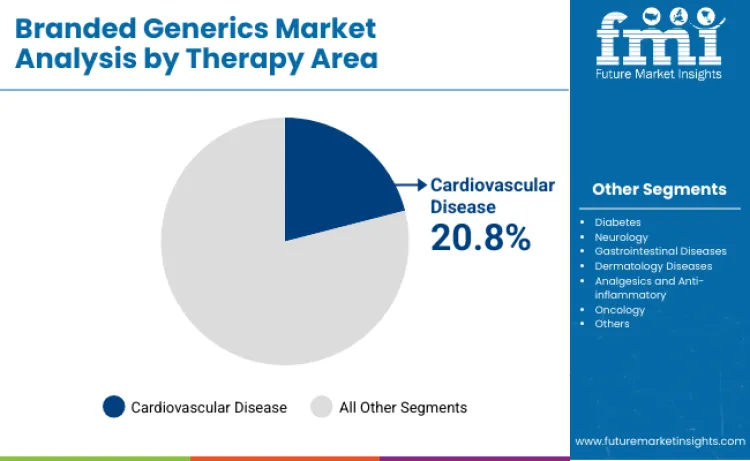
Cardiovascular disease is projected to account for 20.8% of therapy-area share in 2026, reflecting the high treatment volumes associated with hypertension, coronary artery disease, and prevention-focused care. This category remains a major demand center because clinical practice often relies on multi-drug regimens and stepwise intensification, sustaining repeat prescribing and stable consumption patterns. Manufacturers that expand fixed-dose combinations and maintain broad portfolio coverage tend to capture outsized share in these high-prevalence indications.
- High prevalence conditions generating consistent prescription volume
- Guideline-driven use of combination therapy sustaining demand
- Strong clinical validation supporting wide adoption across care settings
- Fixed-dose combinations improving adherence and strengthening product pull-through
Market Dynamics
How Will Patent Cliff Scenarios Impact Market Opportunities?
Patent expires in high-revenue drug classes are expected to expand opportunities by opening large, established demand pools to generic entry and increasing the need for clear differentiation among branded alternatives. As exclusivity ends, companies with faster development and launch execution can capture share early, especially in categories where substitution happens quickly through payer and hospital procurement decisions. The opportunity mix is also shifting toward complex generics, where specialized manufacturing and formulation capabilities enable differentiation and support stronger positioning versus standard commodity generics. At the same time, products that do not communicate therapeutic equivalence and consistent quality risk losing traction with cost-focused buyers. Authorized generics are also likely to play a larger role as originators and partners use them for lifecycle management, pricing control, and channel defense.
How Will Regulatory Harmonization Transform Global Access?
Greater alignment in regulatory expectations is improving multi-country access by reducing repetitive submission work and enabling more coordinated launch strategies. As quality standards and review approaches converge across markets, manufacturers can apply unified development and manufacturing frameworks that support faster entry and lower compliance overhead than country-by-country pathways. Wider use of digital submission systems and regulatory tracking tools also strengthens transparency, improves compliance control, and helps companies manage approvals across multiple jurisdictions more efficiently, supporting broader global rollout plans.
How Will Healthcare Cost Pressures Affect Market Growth?
Rising cost pressures across healthcare systems are reinforcing demand for branded generics that deliver affordability while meeting consistent quality requirements. Government and payer cost-containment actions are accelerating substitution in high-burden therapy areas, which supports volume growth for trusted branded alternatives and strengthens price-based competition. At the same time, purchasers are increasingly requiring clearer cost-effectiveness justification, pushing manufacturers to invest in health-economics evidence that supports formulary access, tender wins, and sustained uptake across public and private channels.
How Will the Branded Generics Industry Evolve in Major Nations?
Global landscape for branded generics is characterized by diverse regulatory frameworks, influenced by healthcare reimbursement systems and pharmaceutical policy standards. Established markets prioritize quality-assured generic alternatives and comprehensive therapeutic substitution programs, whereas emerging economies focus on affordable medication access and basic healthcare coverage. Asia-Pacific is emerging as rapid growth hub due to expanding pharmaceutical manufacturing and increasing chronic disease management requirements. Conversely, North American sectors pivot towards biosimilar integration and comprehensive specialty generic protocols. Government initiatives in developing nations supporting pharmaceutical industry development ensure sustained demand for branded generic solutions across all therapeutic segments.
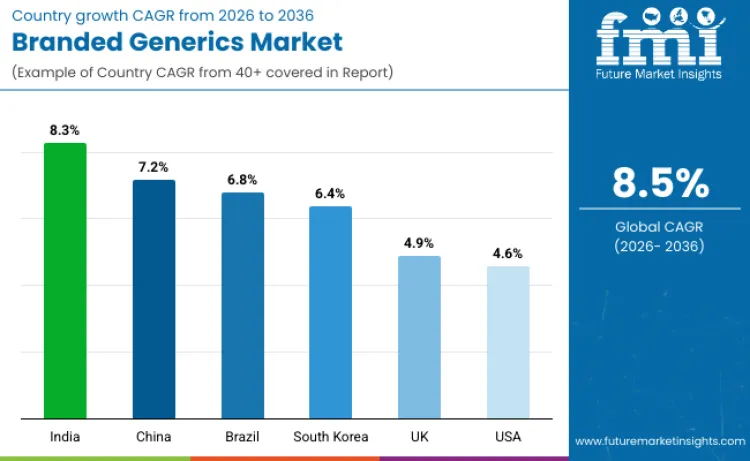
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 8.3% |
| China | 7.2% |
| Brazil | 6.8% |
| South Korea | 6.4% |
| UK | 4.9% |
| USA | 4.6% |
Source: Future Market Insights analysis, supported by a proprietary forecasting model and primary research
What is supporting growth in India’s branded generics sales?
Sales of branded generics in India are expected to expand at an 8.3% CAGR through 2036, supported by large-scale pharmaceutical manufacturing capacity and policy momentum for generic substitution. Access-led programs focused on affordable medicines and broader insurance coverage are widening the treated population across public and private channels. Rising chronic disease therapy volumes are also increasing repeat purchasing, strengthening demand for trusted branded alternatives that balance price and perceived quality. Major manufacturers expanding branded-generic portfolios are improving launch speed, strengthening distribution, and reducing market-entry friction. Continued emphasis on quality-assured generics, along with export-driven scale benefits, reinforces India’s role as a major growth engine for branded generic adoption.
What factors are driving expansion of branded generics in China?
Branded generics in China are projected to grow at a 7.2% CAGR, largely due to procurement reforms that prioritize volume, affordability, and measurable savings. Investments in domestic pharmaceutical capacity are expanding supply depth and improving availability across high-burden therapy areas. Rising healthcare spending, paired with payer pressure to control costs, is supporting structured purchasing decisions that favor established branded generic options. Policies encouraging local production and stronger rollout execution are helping adoption scale across tier-1 and tier-2 cities through hospital procurement and retail pharmacy channels.
What is reinforcing Brazil’s branded generics growth potential?
Brazil’s branded generics industry is forecast to rise at a 6.8% CAGR, supported by healthcare system modernization and sustained demand for affordable therapies across both urban and broader populations. Universal care priorities and affordability goals are accelerating uptake of cost-effective branded alternatives, especially in long-term chronic treatments. Collaboration between international companies and domestic producers is improving product availability and supply consistency, while expanding insurance coverage strengthens purchasing power and prescription volumes. Clear bioequivalence and safety expectations under a structured regulatory environment also enhance provider confidence, supporting wider branded generic use.
What is fueling branded generics growth in South Korea?
Sales of branded generics in South Korea are expected to grow at a 6.4% CAGR, driven by prescription behavior that values brand familiarity and consistent therapeutic performance alongside cost control. Rising healthcare expenditures are increasing pressure to use lower-cost options, supporting branded generics where quality assurance and predictable supply are important. A well-developed pharmaceutical ecosystem and high regulatory standards encourage compliant product development, sustaining trust among prescribers and payers. Ongoing focus on prescription affordability and system efficiency continues to strengthen adoption across reimbursed care settings.
What is behind the UK’s branded generics expansion?
Revenue from branded generics in the UK is set to increase at a 4.9% CAGR, shaped by NHS cost-management priorities and procurement practices that reward reliable, quality-assured alternatives. Greater familiarity with therapeutic equivalence and standardized substitution supports continued use of branded generics in routine prescribing. Growth in biosimilar uptake is also influencing the broader value-driven purchasing environment, reinforcing preference for proven, lower-cost options in specialty segments. Strong compliance expectations and established suppliers support consistent quality, while persistent budget optimization keeps branded generics relevant in formularies.
What is driving branded generics sales growth in the USA?
Branded generics sales in the USA are projected to grow at a 4.6% CAGR, supported by a mature substitution environment and ongoing cost pressure across payers and providers. Formulary management and pharmacy benefit structures increasingly favor lower-cost branded options where supply reliability and patient continuity can be maintained. Strong regulatory oversight and quality expectations support confidence in branded generic utilization, while large manufacturers’ portfolios improve availability across high-volume therapeutic categories. Continued focus on affordability and coverage efficiency sustains demand across retail, mail-order, and institutional channels.
How is competition evolving in the Branded Generics Market?
Competition in the Branded Generics Market is increasingly shaped by efforts to move beyond price-led positioning and build stronger therapeutic differentiation versus commodity generics. Large players such as Teva Pharmaceuticals and Novartis are allocating capital toward biosimilar-linked partnerships and complex-generic development programs that strengthen portfolio depth and support higher-value segments. A clear market direction is the creation of broader therapeutic platforms, where branded offerings are aligned to specific disease-management needs and designed to retain prescriber and payer preference through consistency, availability, and performance expectations.
Companies are also strengthening market access through collaborations with healthcare systems, focusing on formulary placement, substitution pathways, and procurement alignment that can secure repeat volume. Innovation is becoming a key lever, particularly through drug-delivery upgrades that improve convenience, adherence, or clinical performance and help justify differentiated positioning. At the same time, consolidation remains active, with specialized generic firms acquiring assets and capabilities to expand pipelines, improve manufacturing control, and defend margins in a scale-driven market. Teva continues to hold a strong position, supported by its global production footprint and specialization across major therapy areas.
Key Players in Branded Generics Market
- Mylan Pharmaceuticals Inc. (Viatris Inc.)
- Novartis AG
- Teva Pharmaceuticals
- Pfizer Inc.
- Sun Pharmaceutical
- Aspen Pharmacare Holding Ltd.
- Abbott Laboratories
- Bausch Health Companies Inc.
- GlaxoSmithKline Pharmaceuticals Ltd.
- Zydus Lifesciences Ltd.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Drug Class Segments | Anti-Diabetic; Anti-Hypertensive; Alkylating Agents; Antimetabolites; Hormones; Lipid Lowering Drugs; Anti-Depressants; Anti-Psychotics; Anti-Epileptics; Others |
| Therapy Area Categories | Oncology; Cardiovascular Diseases; Diabetes; Neurology; Gastrointestinal Diseases; Dermatology Diseases; Analgesics and Anti-inflammatory; Others |
| Formulation Type Categories | Oral; Parenteral; Topical; Others |
| Distribution Channel Types | Hospital Pharmacies; Retail Pharmacies; Online Pharmacies; Drug Stores |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Key Countries | India, China, Brazil, South Korea, UK, USA |
| Key Companies Profiled | Mylan Pharmaceuticals Inc.; Novartis AG; Teva Pharmaceuticals; Pfizer Inc.; Sun Pharmaceutical; Aspen Pharmacare Holding Ltd.; Abbott Laboratories; Bausch Health Companies Inc.; GlaxoSmithKline Pharmaceuticals Ltd.; Zydus Lifesciences Ltd. |
| Additional Attributes | Dollar sales measured for branded generics used in pharmaceutical therapeutic applications, specified by drug classification (anti-diabetic, cardiovascular agents, oncology drugs), formulation methodology, distribution channel focus (hospital, retail, online), therapeutic indication, and compliance alignment with evolving pharmaceutical regulations and bioequivalence standards. |
Branded Generics Market by Segments
Drug Class:
- Anti-Diabetic
- Anti-Hypertensive
- Alkylating Agents
- Antimetabolites
- Hormones
- Lipid Lowering Drugs
- Anti-Depressants
- Anti-Psychotics
- Anti-Epileptics
- Others
Therapy Area:
- Oncology
- Cardiovascular Diseases
- Diabetes
- Neurology
- Gastrointestinal Diseases
- Dermatology Diseases
- Analgesics and Anti-inflammatory
- Others
Formulation Type:
- Oral
- Parenteral
- Topical
- Others
Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Drug Stores
Bibliography
- USA Food and Drug Administration. (2024). Abbreviated New Drug Application (ANDA): Generic drug development and bioequivalence requirements. USA Department of Health & Human Services, Silver Spring, MD, United States.
- European Medicines Agency. (2024). Guideline on the investigation of bioequivalence for medicinal products in the EU. EMA, Amsterdam, Netherlands.
- European Directorate for the Quality of Medicines & HealthCare. (2024). European Pharmacopoeia (Ph. Eur.): Quality standards for medicines and active substances. EDQM, Strasbourg, France.
Frequently Asked Questions
How big is the global branded generics market?
The global branded generics market is valued at USD 414.2 billion in 2026.
What is the growth outlook over the next 10 years?
The branded generics market is projected to grow at an 8.5% CAGR from 2026 to 2036, reaching USD 934.1 billion by 2036.
Which therapeutic areas or applications drive demand?
Demand is driven by chronic disease therapies, led by anti-diabetic drugs with a 31.7% share and cardiovascular diseases with a 20.8% share.
What are the key risks, regulatory, or development constraints?
Key constraints include strict bioequivalence requirements, pricing pressure from procurement systems, and complexity in advanced branded generic development.
How do approvals, patents, and regulations impact this market?
Patent expirations expand market opportunity, while regulatory approvals and compliance standards determine launch timing and substitution adoption.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand Side Trends
- Supply Side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Roots Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter's Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Billion) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Y-o-Y Growth Trend Analysis 2021 to 2025
- Absolute $ Opportunity Analysis 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Drug Class
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Drug Class, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Drug Class, 2026 to 2036
- Anti-Diabetic
- Anti-Hypertensive
- Alkylating Agents
- Antimetabolites
- Hormones
- Lipid Lowering Drugs
- Anti-Depressants
- Anti-Psychotics
- Anti-Epileptics
- Others
- Y-o-Y Growth Trend Analysis By Drug Class, 2021 to 2025
- Absolute $ Opportunity Analysis By Drug Class, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Therapy Area
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Therapy Area, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Therapy Area, 2026 to 2036
- Oncology
- Cardiovascular Diseases
- Diabetes
- Neurology
- Gastrointestinal Diseases
- Dermatology Diseases
- Analgesics and Anti-inflammatory
- Others
- Y-o-Y Growth Trend Analysis By Therapy Area, 2021 to 2025
- Absolute $ Opportunity Analysis By Therapy Area, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Formulation Type
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Formulation Type, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Formulation Type, 2026 to 2036
- Oral
- Parenteral
- Topical
- Others
- Y-o-Y Growth Trend Analysis By Formulation Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Formulation Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Billion) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Billion) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Drug Class
- By Therapy Area
- By Formulation Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Therapy Area
- By Formulation Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Drug Class
- By Therapy Area
- By Formulation Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Therapy Area
- By Formulation Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Drug Class
- By Therapy Area
- By Formulation Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Therapy Area
- By Formulation Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Drug Class
- By Therapy Area
- By Formulation Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Therapy Area
- By Formulation Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Drug Class
- By Therapy Area
- By Formulation Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Therapy Area
- By Formulation Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Drug Class
- By Therapy Area
- By Formulation Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Therapy Area
- By Formulation Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Drug Class
- By Therapy Area
- By Formulation Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Therapy Area
- By Formulation Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Therapy Area
- By Formulation Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Drug Class
- By Therapy Area
- By Formulation Type
- Competition Analysis
- Competition Deep Dive
- Mylan Pharmaceuticals Inc. (Viatris Inc.)
- Novartis AG
- Teva Pharmaceuticals
- Pfizer Inc.
- Sun Pharmaceutical
- Aspen Pharmacare Holding Ltd.
- Abbott Laboratories
- Bausch Health Companies Inc.
- GlaxoSmithKline Pharmaceuticals Ltd.
- Zydus Lifesciences Ltd.
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Branded Generics Market Key Takeaways
- Global Branded Generics Market Size (USD Billion), 2021-2036
- Global Branded Generics Market Y-o-Y Growth (%), 2021-2036
- Global Branded Generics Market Absolute $ Opportunity Analysis (USD Billion), 2026-2036
- Global Branded Generics Market Pricing Analysis (USD/Unit), 2021-2036
- Global Branded Generics Market Size (USD Billion) By Drug Class, 2021-2036
- Global Branded Generics Market Size (USD Billion) By Therapy Area, 2021-2036
- Global Branded Generics Market Size (USD Billion) By Formulation Type, 2021-2036
- Global Branded Generics Market Size (USD Billion) By Region, 2021-2036
- North America Branded Generics Market Size (USD Billion) By Country, 2021-2036
- North America Branded Generics Market Size (USD Billion) By Drug Class, 2021-2036
- North America Branded Generics Market Size (USD Billion) By Therapy Area, 2021-2036
- North America Branded Generics Market Size (USD Billion) By Formulation Type, 2021-2036
- North America Branded Generics Market Attractiveness Analysis By Country
- North America Branded Generics Market Attractiveness Analysis By Drug Class
- North America Branded Generics Market Attractiveness Analysis By Therapy Area
- North America Branded Generics Market Attractiveness Analysis By Formulation Type
- Latin America Branded Generics Market Size (USD Billion) By Country, 2021-2036
- Latin America Branded Generics Market Size (USD Billion) By Drug Class, 2021-2036
- Latin America Branded Generics Market Size (USD Billion) By Therapy Area, 2021-2036
- Latin America Branded Generics Market Size (USD Billion) By Formulation Type, 2021-2036
- Latin America Branded Generics Market Attractiveness Analysis By Country
- Latin America Branded Generics Market Attractiveness Analysis By Drug Class
- Latin America Branded Generics Market Attractiveness Analysis By Therapy Area
- Latin America Branded Generics Market Attractiveness Analysis By Formulation Type
- Western Europe Branded Generics Market Size (USD Billion) By Country, 2021-2036
- Western Europe Branded Generics Market Size (USD Billion) By Drug Class, 2021-2036
- Western Europe Branded Generics Market Size (USD Billion) By Therapy Area, 2021-2036
- Western Europe Branded Generics Market Size (USD Billion) By Formulation Type, 2021-2036
- Western Europe Branded Generics Market Attractiveness Analysis By Country
- Western Europe Branded Generics Market Attractiveness Analysis By Drug Class
- Western Europe Branded Generics Market Attractiveness Analysis By Therapy Area
- Western Europe Branded Generics Market Attractiveness Analysis By Formulation Type
- Eastern Europe Branded Generics Market Size (USD Billion) By Country, 2021-2036
- Eastern Europe Branded Generics Market Size (USD Billion) By Drug Class, 2021-2036
- Eastern Europe Branded Generics Market Size (USD Billion) By Therapy Area, 2021-2036
- Eastern Europe Branded Generics Market Size (USD Billion) By Formulation Type, 2021-2036
- Eastern Europe Branded Generics Market Attractiveness Analysis By Country
- Eastern Europe Branded Generics Market Attractiveness Analysis By Drug Class
- Eastern Europe Branded Generics Market Attractiveness Analysis By Therapy Area
- Eastern Europe Branded Generics Market Attractiveness Analysis By Formulation Type
- East Asia Branded Generics Market Size (USD Billion) By Country, 2021-2036
- East Asia Branded Generics Market Size (USD Billion) By Drug Class, 2021-2036
- East Asia Branded Generics Market Size (USD Billion) By Therapy Area, 2021-2036
- East Asia Branded Generics Market Size (USD Billion) By Formulation Type, 2021-2036
- East Asia Branded Generics Market Attractiveness Analysis By Country
- East Asia Branded Generics Market Attractiveness Analysis By Drug Class
- East Asia Branded Generics Market Attractiveness Analysis By Therapy Area
- East Asia Branded Generics Market Attractiveness Analysis By Formulation Type
- South Asia and Pacific Branded Generics Market Size (USD Billion) By Country, 2021-2036
- South Asia and Pacific Branded Generics Market Size (USD Billion) By Drug Class, 2021-2036
- South Asia and Pacific Branded Generics Market Size (USD Billion) By Therapy Area, 2021-2036
- South Asia and Pacific Branded Generics Market Size (USD Billion) By Formulation Type, 2021-2036
- South Asia and Pacific Branded Generics Market Attractiveness Analysis By Country
- South Asia and Pacific Branded Generics Market Attractiveness Analysis By Drug Class
- South Asia and Pacific Branded Generics Market Attractiveness Analysis By Therapy Area
- South Asia and Pacific Branded Generics Market Attractiveness Analysis By Formulation Type
- Middle East & Africa Branded Generics Market Size (USD Billion) By Country, 2021-2036
- Middle East & Africa Branded Generics Market Size (USD Billion) By Drug Class, 2021-2036
- Middle East & Africa Branded Generics Market Size (USD Billion) By Therapy Area, 2021-2036
- Middle East & Africa Branded Generics Market Size (USD Billion) By Formulation Type, 2021-2036
- Middle East & Africa Branded Generics Market Attractiveness Analysis By Country
- Middle East & Africa Branded Generics Market Attractiveness Analysis By Drug Class
- Middle East & Africa Branded Generics Market Attractiveness Analysis By Therapy Area
- Middle East & Africa Branded Generics Market Attractiveness Analysis By Formulation Type
- USA Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- USA Branded Generics Market Share Analysis (%) By Drug Class, 2025
- USA Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- USA Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- Canada Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- Canada Branded Generics Market Share Analysis (%) By Drug Class, 2025
- Canada Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- Canada Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- Mexico Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- Mexico Branded Generics Market Share Analysis (%) By Drug Class, 2025
- Mexico Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- Mexico Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- Brazil Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- Brazil Branded Generics Market Share Analysis (%) By Drug Class, 2025
- Brazil Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- Brazil Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- Chile Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- Chile Branded Generics Market Share Analysis (%) By Drug Class, 2025
- Chile Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- Chile Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- Germany Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- Germany Branded Generics Market Share Analysis (%) By Drug Class, 2025
- Germany Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- Germany Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- UK Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- UK Branded Generics Market Share Analysis (%) By Drug Class, 2025
- UK Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- UK Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- Italy Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- Italy Branded Generics Market Share Analysis (%) By Drug Class, 2025
- Italy Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- Italy Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- Spain Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- Spain Branded Generics Market Share Analysis (%) By Drug Class, 2025
- Spain Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- Spain Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- France Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- France Branded Generics Market Share Analysis (%) By Drug Class, 2025
- France Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- France Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- India Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- India Branded Generics Market Share Analysis (%) By Drug Class, 2025
- India Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- India Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- ASEAN Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- ASEAN Branded Generics Market Share Analysis (%) By Drug Class, 2025
- ASEAN Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- ASEAN Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- Australia & New Zealand Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- Australia & New Zealand Branded Generics Market Share Analysis (%) By Drug Class, 2025
- Australia & New Zealand Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- Australia & New Zealand Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- China Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- China Branded Generics Market Share Analysis (%) By Drug Class, 2025
- China Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- China Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- Japan Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- Japan Branded Generics Market Share Analysis (%) By Drug Class, 2025
- Japan Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- Japan Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- South Korea Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- South Korea Branded Generics Market Share Analysis (%) By Drug Class, 2025
- South Korea Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- South Korea Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- Russia Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- Russia Branded Generics Market Share Analysis (%) By Drug Class, 2025
- Russia Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- Russia Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- Poland Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- Poland Branded Generics Market Share Analysis (%) By Drug Class, 2025
- Poland Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- Poland Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- Hungary Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- Hungary Branded Generics Market Share Analysis (%) By Drug Class, 2025
- Hungary Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- Hungary Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- Kingdom of Saudi Arabia Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- Kingdom of Saudi Arabia Branded Generics Market Share Analysis (%) By Drug Class, 2025
- Kingdom of Saudi Arabia Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- Kingdom of Saudi Arabia Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- Turkiye Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- Turkiye Branded Generics Market Share Analysis (%) By Drug Class, 2025
- Turkiye Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- Turkiye Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- South Africa Branded Generics Pricing Analysis (USD/Unit), 2021-2036
- South Africa Branded Generics Market Share Analysis (%) By Drug Class, 2025
- South Africa Branded Generics Market Share Analysis (%) By Therapy Area, 2025
- South Africa Branded Generics Market Share Analysis (%) By Formulation Type, 2025
- Branded Generics Market Competition Dashboard
- Branded Generics Market Competition Benchmarking Analysis
- Branded Generics Market Share Analysis of Top Players By Regional, 2025
- Branded Generics Market Share Analysis of Top Players By Drug Class, 2025
- Branded Generics Market Share Analysis of Top Players By Therapy Area, 2025
- Branded Generics Market Share Analysis of Top Players By Formulation Type, 2025
- Mylan Pharmaceuticals Inc. (Viatris Inc.) - Company Profile
- Novartis AG - Company Profile
- Teva Pharmaceuticals - Company Profile
- Pfizer Inc. - Company Profile
- Sun Pharmaceutical - Company Profile
- Aspen Pharmacare Holding Ltd. - Company Profile
- Abbott Laboratories - Company Profile
- Bausch Health Companies Inc. - Company Profile
- GlaxoSmithKline Pharmaceuticals Ltd. - Company Profile
- Zydus Lifesciences Ltd. - Company Profile
- Assumptions & Acronyms Used in Branded Generics Market Report
List of Figures
- Branded Generics Market Size (USD Billion), 2021-2036
- Branded Generics Market Y-o-Y Growth (%), 2021-2036
- Branded Generics Market Absolute $ Opportunity Analysis (USD Billion), 2026-2036
- Branded Generics Market Value Chain Analysis
- Branded Generics Market Supply Chain Analysis
- Branded Generics Market Investment Feasibility Matrix
- Branded Generics Market PESTLE Analysis
- Branded Generics Market Porter's Five Forces Analysis
- Branded Generics Market Product Life Cycle Analysis
- Branded Generics Market Opportunity Map Analysis
- Branded Generics Market Scenario Forecast Analysis
- Branded Generics Market Production and Consumption Statistics
- Branded Generics Market Import and Export Statistics
- Global Branded Generics Market Share (%) By Drug Class, 2025
- Global Branded Generics Market Share (%) By Drug Class, 2036
- Global Branded Generics Market Share (%) By Therapy Area, 2025
- Global Branded Generics Market Share (%) By Therapy Area, 2036
- Global Branded Generics Market Share (%) By Formulation Type, 2025
- Global Branded Generics Market Share (%) By Formulation Type, 2036
- Global Branded Generics Market Share (%) By Region, 2025
- Global Branded Generics Market Share (%) By Region, 2036
- Global Branded Generics Market Attractiveness Analysis By Region
- North America Branded Generics Market Size (USD Billion), 2021-2036
- North America Branded Generics Market Share (%) By Country, 2025
- North America Branded Generics Market Share (%) By Country, 2036
- North America Branded Generics Market Share (%) By Drug Class, 2025
- North America Branded Generics Market Share (%) By Drug Class, 2036
- North America Branded Generics Market Share (%) By Therapy Area, 2025
- North America Branded Generics Market Share (%) By Therapy Area, 2036
- North America Branded Generics Market Share (%) By Formulation Type, 2025
- North America Branded Generics Market Share (%) By Formulation Type, 2036
- North America Branded Generics Market Attractiveness Analysis By Country
- North America Branded Generics Market Attractiveness Analysis By Drug Class
- North America Branded Generics Market Attractiveness Analysis By Therapy Area
- North America Branded Generics Market Attractiveness Analysis By Formulation Type
- Latin America Branded Generics Market Size (USD Billion), 2021-2036
- Latin America Branded Generics Market Share (%) By Country, 2025
- Latin America Branded Generics Market Share (%) By Country, 2036
- Latin America Branded Generics Market Share (%) By Drug Class, 2025
- Latin America Branded Generics Market Share (%) By Drug Class, 2036
- Latin America Branded Generics Market Share (%) By Therapy Area, 2025
- Latin America Branded Generics Market Share (%) By Therapy Area, 2036
- Latin America Branded Generics Market Share (%) By Formulation Type, 2025
- Latin America Branded Generics Market Share (%) By Formulation Type, 2036
- Latin America Branded Generics Market Attractiveness Analysis By Country
- Latin America Branded Generics Market Attractiveness Analysis By Drug Class
- Latin America Branded Generics Market Attractiveness Analysis By Therapy Area
- Latin America Branded Generics Market Attractiveness Analysis By Formulation Type
- Western Europe Branded Generics Market Size (USD Billion), 2021-2036
- Western Europe Branded Generics Market Share (%) By Country, 2025
- Western Europe Branded Generics Market Share (%) By Country, 2036
- Western Europe Branded Generics Market Share (%) By Drug Class, 2025
- Western Europe Branded Generics Market Share (%) By Drug Class, 2036
- Western Europe Branded Generics Market Share (%) By Therapy Area, 2025
- Western Europe Branded Generics Market Share (%) By Therapy Area, 2036
- Western Europe Branded Generics Market Share (%) By Formulation Type, 2025
- Western Europe Branded Generics Market Share (%) By Formulation Type, 2036
- Western Europe Branded Generics Market Attractiveness Analysis By Country
- Western Europe Branded Generics Market Attractiveness Analysis By Drug Class
- Western Europe Branded Generics Market Attractiveness Analysis By Therapy Area
- Western Europe Branded Generics Market Attractiveness Analysis By Formulation Type
- Eastern Europe Branded Generics Market Size (USD Billion), 2021-2036
- Eastern Europe Branded Generics Market Share (%) By Country, 2025
- Eastern Europe Branded Generics Market Share (%) By Country, 2036
- Eastern Europe Branded Generics Market Share (%) By Drug Class, 2025
- Eastern Europe Branded Generics Market Share (%) By Drug Class, 2036
- Eastern Europe Branded Generics Market Share (%) By Therapy Area, 2025
- Eastern Europe Branded Generics Market Share (%) By Therapy Area, 2036
- Eastern Europe Branded Generics Market Share (%) By Formulation Type, 2025
- Eastern Europe Branded Generics Market Share (%) By Formulation Type, 2036
- Eastern Europe Branded Generics Market Attractiveness Analysis By Country
- Eastern Europe Branded Generics Market Attractiveness Analysis By Drug Class
- Eastern Europe Branded Generics Market Attractiveness Analysis By Therapy Area
- Eastern Europe Branded Generics Market Attractiveness Analysis By Formulation Type
- East Asia Branded Generics Market Size (USD Billion), 2021-2036
- East Asia Branded Generics Market Share (%) By Country, 2025
- East Asia Branded Generics Market Share (%) By Country, 2036
- East Asia Branded Generics Market Share (%) By Drug Class, 2025
- East Asia Branded Generics Market Share (%) By Drug Class, 2036
- East Asia Branded Generics Market Share (%) By Therapy Area, 2025
- East Asia Branded Generics Market Share (%) By Therapy Area, 2036
- East Asia Branded Generics Market Share (%) By Formulation Type, 2025
- East Asia Branded Generics Market Share (%) By Formulation Type, 2036
- East Asia Branded Generics Market Attractiveness Analysis By Country
- East Asia Branded Generics Market Attractiveness Analysis By Drug Class
- East Asia Branded Generics Market Attractiveness Analysis By Therapy Area
- East Asia Branded Generics Market Attractiveness Analysis By Formulation Type
- South Asia and Pacific Branded Generics Market Size (USD Billion), 2021-2036
- South Asia and Pacific Branded Generics Market Share (%) By Country, 2025
- South Asia and Pacific Branded Generics Market Share (%) By Country, 2036
- South Asia and Pacific Branded Generics Market Share (%) By Drug Class, 2025
- South Asia and Pacific Branded Generics Market Share (%) By Drug Class, 2036
- South Asia and Pacific Branded Generics Market Share (%) By Therapy Area, 2025
- South Asia and Pacific Branded Generics Market Share (%) By Therapy Area, 2036
- South Asia and Pacific Branded Generics Market Share (%) By Formulation Type, 2025
- South Asia and Pacific Branded Generics Market Share (%) By Formulation Type, 2036
- South Asia and Pacific Branded Generics Market Attractiveness Analysis By Country
- South Asia and Pacific Branded Generics Market Attractiveness Analysis By Drug Class
- South Asia and Pacific Branded Generics Market Attractiveness Analysis By Therapy Area
- South Asia and Pacific Branded Generics Market Attractiveness Analysis By Formulation Type
- Middle East & Africa Branded Generics Market Size (USD Billion), 2021-2036
- Middle East & Africa Branded Generics Market Share (%) By Country, 2025
- Middle East & Africa Branded Generics Market Share (%) By Country, 2036
- Middle East & Africa Branded Generics Market Share (%) By Drug Class, 2025
- Middle East & Africa Branded Generics Market Share (%) By Drug Class, 2036
- Middle East & Africa Branded Generics Market Share (%) By Therapy Area, 2025
- Middle East & Africa Branded Generics Market Share (%) By Therapy Area, 2036
- Middle East & Africa Branded Generics Market Share (%) By Formulation Type, 2025
- Middle East & Africa Branded Generics Market Share (%) By Formulation Type, 2036
- Middle East & Africa Branded Generics Market Attractiveness Analysis By Country
- Middle East & Africa Branded Generics Market Attractiveness Analysis By Drug Class
- Middle East & Africa Branded Generics Market Attractiveness Analysis By Therapy Area
- Middle East & Africa Branded Generics Market Attractiveness Analysis By Formulation Type
- USA Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- Canada Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- Mexico Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- Brazil Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- Chile Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- Germany Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- UK Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- Italy Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- Spain Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- France Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- India Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- ASEAN Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- Australia & New Zealand Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- China Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- Japan Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- South Korea Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- Russia Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- Poland Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- Hungary Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- Kingdom of Saudi Arabia Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- Turkiye Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- South Africa Branded Generics Pricing Trend Analysis (USD/Unit), 2021-2036
- Branded Generics Market Competitive Landscape
- Branded Generics Market Structure Analysis
- Branded Generics Market Competition Dashboard
- Branded Generics Market Competition Benchmarking Analysis
- Branded Generics Market Share Analysis of Top Players By Regional
- Branded Generics Market Share Analysis of Top Players By Drug Class
- Branded Generics Market Share Analysis of Top Players By Therapy Area
- Branded Generics Market Share Analysis of Top Players By Formulation Type
- Mylan Pharmaceuticals Inc. (Viatris Inc.) - Revenue Analysis (USD Billion), 2019-2025
- Novartis AG - Revenue Analysis (USD Billion), 2019-2025
- Teva Pharmaceuticals - Revenue Analysis (USD Billion), 2019-2025
- Pfizer Inc. - Revenue Analysis (USD Billion), 2019-2025
- Sun Pharmaceutical - Revenue Analysis (USD Billion), 2019-2025
- Mylan Pharmaceuticals Inc. (Viatris Inc.) - Market Position Analysis
- Novartis AG - Market Position Analysis
- Teva Pharmaceuticals - Market Position Analysis
- Pfizer Inc. - Market Position Analysis
- Sun Pharmaceutical - Market Position Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

