Competent Cells Market
Competent Cells Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Competent Cells Market Size and Share Forecast Outlook 2025 to 2035
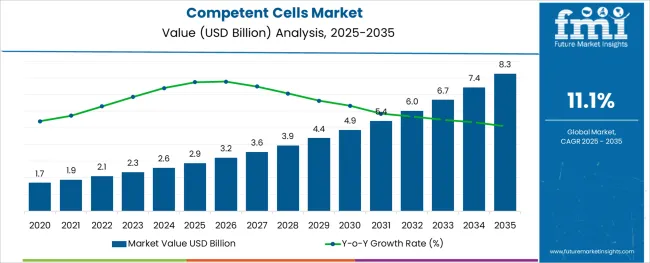
The Competent Cells Market is estimated to be valued at USD 2.9 billion in 2025 and is projected to reach USD 8.3 billion by 2035, registering a compound annual growth rate (CAGR) of 11.1% over the forecast period.
Quick Stats for Competent Cells Market
- Industry Value (2025): USD 2.9 billion
- Forecast Value (2035): USD 8.3 billion
- Forecast CAGR: 11.1%
- Leading Segment: Chemically Competent Cells (61.0%)
- Key Growth Region: North America, Asia-Pacific, Europe
- Top Key Players: Agilent Technologies Inc., Genescript Biotech Corporation, Bio-Rad Laboratories Inc, Merck & Co Inc, OriGene Technologies, Inc, New England Biolabs Inc, Illumina, Inc., Thermo Fisher Scientific, Inc., QIAGEN N.V., Zymo Research Corporation, Promega Corporation, HiMedia Laboratories, Avantor, Inc., Scarab Genomics, LLC, Lucigen, Tonbo Biosciences, Transgen Biotech, Codex DNA, Inc., bluebird bio, Inc.
Rationale for Segmental Growth in the Competent Cells Market
Market Overview
The competent cells market is expanding steadily, supported by the increasing demand for advanced molecular biology tools in biotechnology, genetic engineering, and academic research. Rising investments in synthetic biology, gene therapy, and biologics manufacturing have created favorable conditions for the adoption of high-efficiency cell transformation systems.
The surge in research activities involving plasmid propagation, gene expression, and recombinant protein production has driven the need for reliable and efficient competent cells. Further, advancements in transformation protocols and optimized media formulations are reducing process variability and improving yield consistency.
Regulatory emphasis on reproducibility and standardization in research, particularly in academic and government-funded laboratories, is also enhancing market uptake. As the focus intensifies on personalized medicine and CRISPR-based technologies, the demand for specialized competent cell lines tailored for high-throughput and precise cloning applications is projected to ris.
Segmental Analysis
The market is segmented by Product Type, Application Type, and End User and region. By Product Type, the market is divided into Chemically Competent Cells and Electrocompetent Cells. In terms of Application Type, the market is classified into Cloning, Protein Expression, and Other Applications. Based on End User, the market is segmented into Academic Research Institutes, Pharmaceutical & Biotechnology Companies, and Contract Research Organizations. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Chemically Competent Cells Product Type Segment
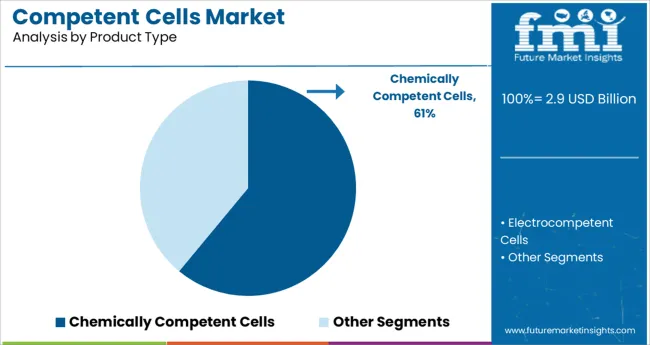
Chemically competent cells are projected to account for 61.0% of total market revenue in 2025, establishing them as the leading product type segment. Their dominance is attributed to ease of preparation, cost-effectiveness, and compatibility with standard transformation techniques such as heat shock.
These cells are widely adopted in both basic and applied research due to their ability to deliver consistent transformation efficiencies for plasmid uptake in bacterial systems. The broad availability of chemically competent cell strains optimized for various applications-such as high-copy plasmids or blue-white screening—has strengthened their position in molecular biology workflows.
Additionally, their extended shelf life, simple storage requirements, and rapid scalability make them the preferred choice in academic labs and small-scale production environments.
Insights into the Cloning Application Type Segment
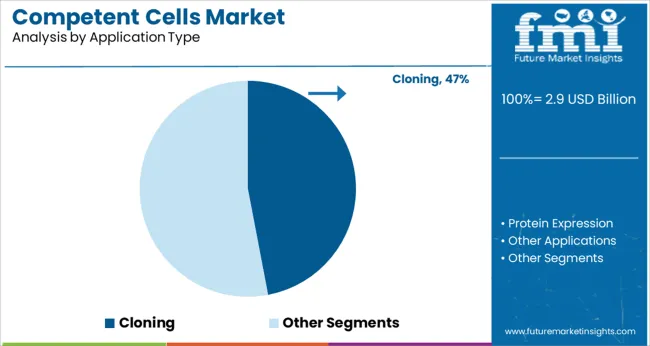
Cloning is expected to account for 47.0% of the market share by 2025, making it the dominant application segment within the competent cells market. This segment’s growth is being driven by the increasing use of recombinant DNA technologies, gene synthesis, and vector construction in drug discovery, diagnostics, and functional genomics.
Competent cells optimized for cloning provide high transformation efficiency, minimizing the need for repeated experiments and reducing time-to-results. Their widespread use in protocols involving PCR product insertion, site-directed mutagenesis, and subcloning has solidified their essential role in molecular workflows.
Furthermore, academic and commercial research institutions continue to prioritize high-quality, validated cell lines to ensure reproducibility and accuracy in gene function studies, further reinforcing the segment’s lead.
Insights into the Academic Research Institutes End User Segment
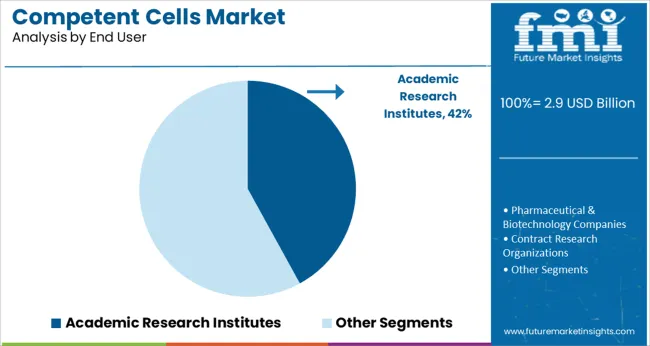
Academic research institutes are projected to contribute 42.0% of the overall market revenue in 2025, ranking as the top end-user segment. This position is supported by sustained funding in life sciences education, basic research, and genomic studies across universities and public research organizations.
The demand for competent cells in these institutions is closely linked to curriculum-driven laboratory work, student-led research projects, and faculty-led innovation programs. Institutions often favor chemically competent cells due to their affordability, reliability, and adaptability across a broad spectrum of cloning and expression applications.
Additionally, the rise of interdisciplinary research initiatives and bio-entrepreneurship programs within universities has accelerated the procurement of competent cell kits for proof-of-concept and early-phase molecular biology work.
Cloning Applications to open up New Avenues during the Forecast Period
Cloning is expected to expand significantly in the application area due to the growing demand for molecularly cloned items. The cloning sector has benefited from COVID-19 as a result of corporations investing more money in research and development. Furthermore, it is anticipated that the segment would develop faster because of increased cloning research and financial assistance from a number of organizations.
Techniques for cell engineering, such as somatic cell nuclear transfer (SCNT), have demonstrated a variety of uses, including the creation of transgenic animals, the preservation of endangered species, and therapeutic cloning. Since competent cells may be employed in therapeutic cloning research, these fields are anticipated to experience renewed attention, which will lead to an increase in demand for these cells.
Due to rising Research and Development expenditures and the commercialization of proteomics and genomics-based goods, the biotechnology sector is also anticipated to see a significant growth rate. The need for cutting-edge product advancements in the treatment of different ailments is also anticipated to support the segment's expansion. Additionally, it is projected that increased investment in research into DNA cloning methods at academic research facilities and the usage of these products in therapies will accelerate market growth in the near future.
APAC to bring in more demand during 2022 to 2032
During the projected period, it is predicted that the Asia-Pacific market would account for a large portion of the worldwide competent cells market. Extensive research on DNA cloning methods in the area and the existence of significant market participants are credited with the expansion.
For instance, Takara Bio Inc. stated in January 2024 that its new facility, the Center for Gene and Cell Processing II, for manufacturing and research purposes, had been completed in Shiga, Japan.
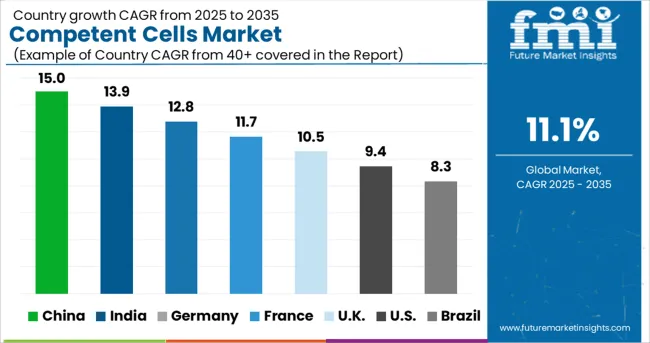
China is expected to develop at a profitable CAGR of 10.1% throughout the projected period and will hold a market share of around 54.2% in East Asia in 2024. The competent cells market will grow as research funding and activity increase.
For instance, the State Key Laboratory of Microbial Application, the Natural Science Foundation of China, and SDU's Qilu Youth Scholar Start-up Funding all provided funding for a research study on Escherichia coli BW25113 competent cells made using a straightforward chemical method that has unparalleled transformation and cloning efficiencies in 2025.
Another regional company, HiMedia Solutions, is situated in India and produces a wide range of goods for the market for competent cells. The HiPurA BL21(DE3) Competent Cells, Bacterial Transformation Kit, HiPurA XL1-Blue Competent Cells, and HiPurA Competent Cells are its four main items for this market. The business is India's top producer of trustworthy cells. Its goods made cloning experiments simpler, such as the HiPurA Competent Cells, which were the first competent DH5a cells marketed on the Indian market. The existence of such businesses is probably going to benefit market growth.
Competent cells are bacterial cells, which can utilize foreign DNA from their surroundings through a process known as transformation. Griffith described it initially in Streptococcus pneumoniae. If the cell walls of E.coli are changed, the cells have a greater tendency to absorb the DNA.
Calcium chloride and temperature shock therapy can make the cells competent. Rapidly growing cells can be rendered more competent than cells at other phases of development. Following transformation, the cells may express the acquired genetic information.
Sales Analysis of Competent Cells Market from 2017 to 2021 Vs Demand Outlook for 2022 to 2032
As per Future Market Insights (FMI), the competent cells market was approximately 12.8% of the overall USD 2.6 Billion global cell expansion market in 2024. Overall sales of competent cells expanded at a CAGR of 9.9% from 2020 to 2024, on the back of the rising expansion of the healthcare industry.
Increasing adoption of molecular cloned products, recombinant proteins, advancement in molecular cloning research and development with the emergence of new technologies, and numerous government initiatives and government grants available for life science research are the key factors driving industry growth during the forecast period.
Gene therapy can potentially treat a wide range of ailments. The increasing prevalence of cancer, cardiovascular diseases, cystic fibrosis, blood disorders, and immunocompromised cases is rising, which will boost the market in the forecast period. Gene therapy attempts to cure an ailment or enhance your body's capacity to fight disease by replacing a malfunctioning gene or introducing a new gene.
According to World Health Organization (WHO) 2025, cancer is the largest cause of mortality around the globe, accounting for over 2.6 million deaths in 2024, or roughly one in every six. Colon, lung, breast, rectum, and prostate cancers are the most prevalent. Tobacco use, obesity, alcohol consumption, a lack of consumption of fruits and vegetables, and an insufficient level of physical activity account for almost one-third of cancer fatalities.
In addition, cystic fibrosis is a widespread hereditary condition throughout the white population in the USA. The condition affects one in every 2,500 to 3,500 white babies. Cystic fibrosis affects around one in every 17,000 African Americans and 31,000 Asian Americans, as stated by MedlinePlus in 2024. These conditions require gene therapy, which will positively impact the competent cells market. Backed by these aforementioned factors, the global competent cells market is expected to grow at a CAGR of 11.1% during the forecast period.
What are the Key Opportunities for the Competent Cells Market Manufacturers?
Favorable Government Initiatives for Gene Therapy Products to Push the Sales
Growth in the market is predicted to surge on the back of favourable initiatives by governments in emerging countries, increasing FDA approval for gene therapy products, and advancements in research and developmental activities. These factors will boost the demand for better technologies, creating a conducive environment in the competent cells market.
Recombinant DNA technology is one of the most commonly used techniques in introducing a specific gene or a healthy gene into a vector. These vectors can be plasmodial, viral, or nanostructured; the latter is the most commonly used because of its efficacy in colonizing cells and incorporating their genetic material.
Recent advances in recombinant DNA synthesis have enabled the effective production of recombinant proteins at industrial sizes with higher yields and lower production costs. This has enabled the development of new treatments, vaccines, and diagnostic reagents.
Certain nutrients are allowed to clone human genes, which reduces the likelihood of unfavourable immune system reactions in patients, allows for the creation of highly specialized, functional, and active proteins, increases productivity and cost-effectiveness, and reduces the possibility of unknown pathogen transmission from animals or humans.
What are the Factors Restraining the Demand for Competent Cells?
The rising costs of research and developmental activities and stringent regulations in the field might hamper the growth in the forecast period. Competent cells are utilized in molecular cloning procedures, protein expression, and various plasmid DNA-based applications.
However, depending on the transformation method, for instance, developing the E. coli competent cells can be time-consuming, needing exceptionally pure water, high-grade reagents, specified autoclaved glassware, and or even specialized equipment (electroporators).
Numerous protocols available describe the technique and buffers needed to produce competent cells. There are even kits available that include pre-made buffers to expedite this process. Furthermore, it is more expensive to use electroporation.
A specialized apparatus is required to implement the charge, in addition to cuvettes to transport the charge to the cell solution. Excess salt in the cuvette can cause electroporation to fail and result in the loss of valuable samples. The most widely mentioned disadvantage of chemical transformation is its reduced efficiency.
Country-wise Insights
What Makes the USA a Lucrative Competent Cells Market?
Growing Investment in Gene Cell Therapy Products to Boost the Demand
The USA dominated the North American competent cells market and accounted for nearly 84.8% in 2024. Demand in the market is anticipated to surge on the back of rising investment in gene therapy products. Further, increasing research and development by government bodies, grants, and clinical trials for disease treatment and others, will improve the growth of the competent cells market.
Will China Retain its Dominance in the East Asia Competent Cells Market?
China Competent Cells Market to Benefit from Rising Research & Development for Cloning Efficiency
China held approximately 54.2% share in the East Asia competent cells market in 2024. Demand for competent cells in China will rise at a CAGR of 10.1% during the forecast period. Increasing research activities and grants will propel the competent cells market.
For instance, the Natural Science Foundation of China, the funding by the State Key Laboratory of Microbial Application, and the Qilu Youth Scholar Start-up Funding of SDU, funded a research study on Escherichia coli BW25113 competent cells prepared using a simple chemical method having unmatched transformation and cloning efficiencies, in 2025.
What is the Demand Outlook in Germany's Competent Cells Market?
Introduction of Different Strains for Gene Transformation to Propel Sales
As per FMI, the overall sales in the German competent cells market are projected to expand at 9.3% CAGR in the Europe competent cells market during the forecast period. Increasing production activities by manufacturers to develop different types of strains, which allows easy gene transformation, will propel the market. For instance, New England Biolabs GmbH has products specific to cloning & subcloning and strains for protein expression, with each type having quality specifications.
Category-wise Insights
Which Competent Cells Product is Driving Market Growth?
Nearly 7 out of 10 Sales to be Contributed by Chemically Competent Cells
Chemically competent cells are expected to present high growth at a CAGR of 15.3% between 2025 and 2035, with a share of about 67.8% in 2024. Cloning, DNA sequencing, and the creation of DNA libraries are just a few of the many molecular biology techniques that frequently include transformation. Competent cells must be used to perform the transformation. Chemically competent cells hold a bigger market share because chemical transformation is quicker and less error-prone than electroporation.
Which is the Chief Application in Competent Cells Market?
Cloning Segment to Account for Around 3/5th of Competent Cells Sales
In terms of application, the cloning segment held a revenue share of 57.8% in 2024 and is expected to display steady growth over the forecast period. Competent cells are frequently employed in molecular cloning to increase and preserve cloned DNA in plasmids because they are engineered to absorb foreign DNA from the environment more efficiently. With growing investments in cell and gene therapy, the cloning segment holds a higher market share in terms of application within the overall competent cells market, globally.
Based on end users, the academic research institutes held the highest market share value of 51.2% in 2024. Increasing research and development activities in life sciences are propelling sales of the overall competent cells market.
Competitive Landscape
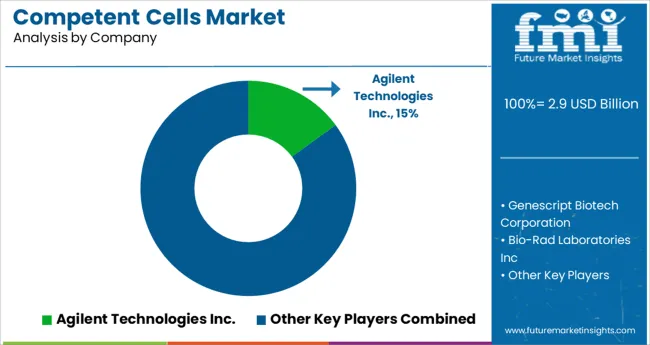
With several competitors in the competent cells market, the overall market is fragmented. To expand their customer base, leading companies are implementing methods such as mergers and acquisitions, partnerships and collaborations, and new product launches.
For instance:
- The VmaxTM X2 Competent Cells were introduced in January 2024 by SGI-DNA, the company behind the BioXpTM 3200 System and Gibson Assembly® Reagents. They are intended to produce two to four times as much soluble protein in half the time as current methods that rely on E. coli.
- Bluebird Bio announced the introduction of ZYNTEGLOTM Gene Therapy for Patients with Transfusion-Dependent -Thalassemia Who Do Not Have β?/β0 Genotype in Germany in January 2024.
Report Scope as per Competent Cells Industry Analysis
| Attribute | Details |
|---|---|
| Estimated Market Size 2025 | USD 2.9 billion |
| Projected Market Value 2035 | USD 8.3 billion |
| CAGR 2025 to 2035 | 11.1% |
| Forecast Period | 2020 to 2024 |
| Historical Data Available for | 2025 to 2035 |
| Market Analysis | million for Value |
| Key Regions Covered | North America; Latin America; Europe; South Asia; East Asia; Oceania; and Middle East & Africa |
| Key Countries Covered | USA, Canada, Brazil, Mexico, Argentina, UK, Germany, Italy, Russia, Spain, France, BENELUX, India, Thailand, Indonesia, Malaysia, Japan, China, South Korea, Australia, New Zealand, Turkey, GCC Countries, South Africa, and North Africa |
| Key Market Segments Covered | Product, Application, End User, and Region |
| Key Companies Profiled | Agilent Technologies Inc.; Genescript Biotech Corporation; Bio-Rad Laboratories Inc; Merck & Co Inc; OriGene Technologies, Inc; New England Biolabs Inc; Illumina, Inc.; Thermo Fisher Scientific, Inc.,; QIAGEN N.V.,; Zymo Research Corporation; Promega Corporation; HiMedia Laboratories; Avantor, Inc.; Scarab Genomics, LLC; Lucigen; bluebird bio, Inc.; Tonbo Biosciences; TransGen Biotech; Codex DNA, Inc. |
Key Market Segments Covered in Competent Cells Industry Research
By Product:
- Chemically Competent Cells
- Electrocompetent Cells
By Application:
- Cloning
- Protein Expression
- Other Applications
By End User:
- Academic Research Institutes
- Pharmaceutical & Biotechnology Companies
- Contract Research Organizations
By Region:
- North America
- Latin America
- Europe
- South Asia
- East Asia
- Oceania
- Middle East and Africa (MEA)
Frequently Asked Questions
How big is the competent cells market in 2025?
The global competent cells market is estimated to be valued at USD 2.9 billion in 2025.
What will be the market size for the competent cells market in 2035?
It is projected to reach USD 8.3 billion by 2035.
How much will be the CAGR of the competent cells market between 2025 and 2035?
The market is expected to grow at a 11.1% CAGR between 2025 and 2035.
What are the key product types in the competent cells market?
The key product types are chemically competent cells and electrocompetent cells.
Which cloning segment is expected to dominate the competent cells market in 2025?
cloning segment is expected to dominate with a 47.0% industry share in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020-2024 and Forecast, 2025-2035
- Historical Market Size Value (USD Million) Analysis, 2020-2024
- Current and Future Market Size Value (USD Million) Projections, 2025-2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020-2024 and Forecast 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2020-2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2025-2035
- Chemically Competent Cells
- Electrocompetent Cells
- Chemically Competent Cells
- Y-o-Y Growth Trend Analysis By Product Type , 2020-2024
- Absolute $ Opportunity Analysis By Product Type , 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Application Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application Type, 2020-2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application Type, 2025-2035
- Cloning
- Protein Expression
- Other Applications
- Cloning
- Y-o-Y Growth Trend Analysis By Application Type, 2020-2024
- Absolute $ Opportunity Analysis By Application Type, 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2020-2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2025-2035
- Academic Research Institutes
- Pharmaceutical & Biotechnology Companies
- Contract Research Organizations
- Academic Research Institutes
- Y-o-Y Growth Trend Analysis By End User, 2020-2024
- Absolute $ Opportunity Analysis By End User, 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020-2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025-2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application Type
- By End User
- Key Takeaways
- Latin America Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Application Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application Type
- By End User
- Key Takeaways
- Western Europe Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Application Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application Type
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Application Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application Type
- By End User
- Key Takeaways
- East Asia Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application Type
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Application Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application Type
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Application Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application Type
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By Application Type
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application Type
- By End User
- Competition Analysis
- Competition Deep Dive
- Agilent Technologies Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Genescript Biotech Corporation
- Bio-Rad Laboratories Inc
- Merck & Co Inc
- OriGene Technologies, Inc
- New England Biolabs Inc
- Illumina, Inc.
- Thermo Fisher Scientific, Inc.
- QIAGEN N.V.
- Zymo Research Corporation
- Promega Corporation
- HiMedia Laboratories
- Avantor, Inc.
- Scarab Genomics, LLC
- Lucigen
- Tonbo Biosciences
- Transgen Biotech
- Codex DNA, Inc.
- bluebird bio, Inc.
- Agilent Technologies Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020-2035
- Table 2: North America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 3: Latin America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 4: Western Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 5: Eastern Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 6: East Asia Market Value (USD Million) Forecast by Country, 2020-2035
- Table 7: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020-2035
- Table 8: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental $ Opportunity, 2025-2035
- Figure 7: Latin America Market Incremental $ Opportunity, 2025-2035
- Figure 8: Western Europe Market Incremental $ Opportunity, 2025-2035
- Figure 9: Eastern Europe Market Incremental $ Opportunity, 2025-2035
- Figure 10: East Asia Market Incremental $ Opportunity, 2025-2035
- Figure 11: South Asia and Pacific Market Incremental $ Opportunity, 2025-2035
- Figure 12: Middle East & Africa Market Incremental $ Opportunity, 2025-2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis
- Figure 21: Global Market - Company Share Analysis

