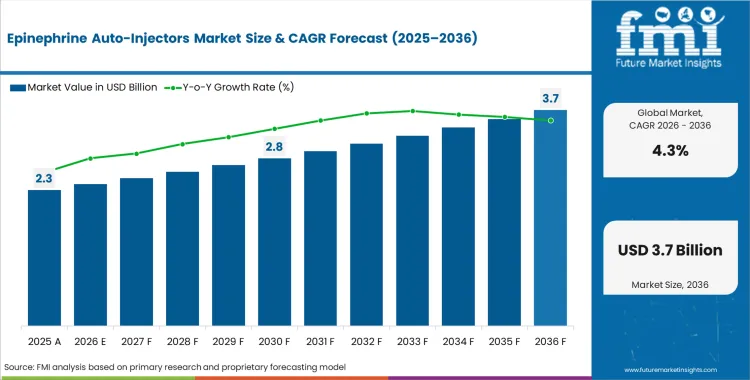
The epinephrine auto-injectors market was valued at USD 2.30 billion in 2025, projected to reach USD 2.40 billion in 2026, and is forecast to expand to USD 3.65 billion by 2036 at a 4.3% CAGR. As per FMI, the incremental opportunity over the forecast period amounts to USD 1.25 billion. The market trajectory is shaped by evolving application requirements, tightening regulatory frameworks, and specification-driven procurement patterns that favour verified, performance-certified products.
FMI analysts observe that the epinephrine auto-injector market operates under a durable demand floor set by anaphylaxis prevalence rates and regulatory stock mandates. Schools, airlines, restaurants, and public venues in multiple jurisdictions are now required to maintain emergency epinephrine supplies, creating a recurring institutional procurement cycle independent of individual prescription volumes. Generic and authorized generic entrants have introduced price competition in North America and Europe, compressing unit margins for originator brands while expanding volume access. The net effect is a market where unit volumes grow steadily but revenue growth lags volume growth in mature regions, while emerging markets contribute incremental revenue from first-time access expansion.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 2.40 billion |
| Industry Value (2036) | USD 3.65 billion |
| CAGR (2026 to 2036) | 4.3% |
Source: Future Market Insights, 2026
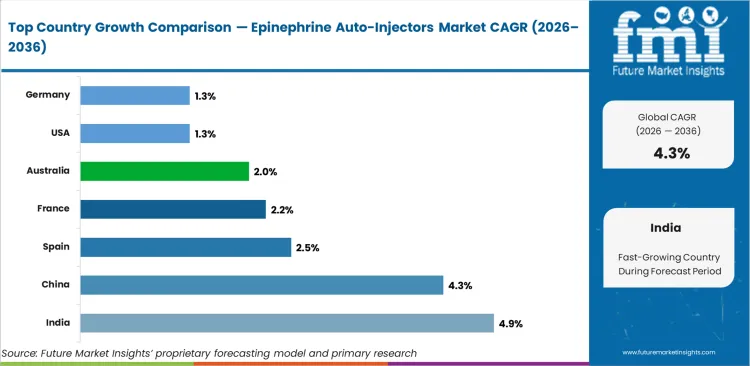
Country-level growth rates reflect distinct development trajectories. India at 4.9% CAGR, China at 4.3% CAGR, South Korea at 3.4% CAGR, Spain at 2.5% CAGR, France at 2.2% CAGR, Australia at 2.0% CAGR, USA at 1.3% CAGR, Germany at 1.3% CAGR. India leads the expansion driven by accelerating infrastructure investment, rising institutional demand, and supportive policy environments. Mature markets sustain steady procurement volumes through replacement cycles and compliance-driven specification upgrades. FMI is of the opinion that regional supply chain realignment and localization mandates will increasingly shape demand distribution through 2036.
Epinephrine auto-injectors are pre-filled, single-use injectable devices designed to deliver a measured dose of epinephrine for emergency treatment of severe allergic reactions (anaphylaxis). The market covers devices in 0.15 mg, 0.3 mg, and other dosage configurations intended for use across hospitals, clinics, home settings, and institutional environments. Based on Future Market Insights analysis, demand is anchored by anaphylaxis prevalence rates, institutional stocking mandates, and expanding access programs in emerging markets.
Market scope includes all commercially traded epinephrine auto-injectors products segmented by dosage, age group, end user, and region. Coverage extends to global and regional market sizing, forecast projections from 2026 to 2036, segment-level breakdowns, pricing analysis, and procurement pattern assessment across major consuming markets.
The scope excludes bulk pharmaceutical-grade epinephrine active ingredient, manual pre-filled syringes without auto-injection mechanisms, epinephrine formulations for cardiac arrest or asthma indications not packaged in auto-injector format, and veterinary epinephrine products. Training devices without active drug content are also excluded.
Rising Prevalence of Anaphylaxis Is Propelling the Growth for Epinephrine Auto-Injectors
Anaphylaxis represents the most severe and life-threatening allergic reaction, which is mediated by most known allergens-food, medications, insect bites, and even atmospheric conditions. It is an acute and sometimes lethal condition that demands immediate treatment.
It is the only treatment ever shown to arrest symptoms and prevent further complications. The global incidence of anaphylaxis has continued to rise, whereby, according to predictions, the demand for epinephrine autoinjectors-a stab-in-time and lifesaving treatment-is on the increase.
According to the Allergy & Asthma Network 2023 statistics, it is estimated that 1.6% to 5.1% of the US population has experienced anaphylaxis in 2023. The most common causes of anaphylaxis include medications at 34%, food at 31%, and insect sting envenomation at 20%.
The medications, particularly NSAIDs and antibiotics, are considered very common causes, while food allergens, with special mention of peanuts and tree nuts, make up a rather fair share of reactions, especially among children. These are the statistics underlining the increasing demand for epinephrine auto-injectors to serve as a vital lifesaver for the treatment of this growing health problem.
Accordingly, with growing diagnoses for high-level severity allergies and greater awareness by the public of the dangers of anaphylactic shock, demand for the auto-injector has grown.
Rising Number of Patients Suffering from Asthma is Affecting the Demand for Epinephrine Auto-Injectors Market
Asthma is a chronic respiratory condition resulting from inflammation and narrowing of the human respiratory airways, affecting millions across the globe. Its prevalence is increasingly spreading due to both environmental and genetic factors.
Commonly, asthma symptoms are controlled by bronchodilators and inhaled corticosteroids; however, some patients experience severe asthma attacks or exacerbation that can develop into life-threatening anaphylaxis, especially for those suffering from allergies.
According to 2026 statistics of the World Health Organization, an estimated 262 million people with asthma worldwide are expected in 2026, and this prevalence is likely to continue. It may also be that poor air quality and a higher pollen count suggest that a shift in environmental factors could make those suffering from asthma more vulnerable to picking up respiratory complications, which also include severe allergic reactions.
This is the increasing incidence of asthma, mostly among children and young adults, which requires better access to epinephrine autoinjectors in light of emergency management.
With the increased awareness of asthma and its association with anaphylaxis, demand for the epinephrine injector has similarly been in force. Thus, physicians have now become increasingly observant in diagnosing asthma and associated allergies in patients, issuing prescription one after another for auto-injectors.
In summary, the increasing prevalence of asthma, further coupled with its strong association with anaphylaxis, constitutes one of the major driving factors for epinephrine auto-injectors. A rise in asthma patients, particularly with co-existing allergies, has turned these devices into a lifeline for the management of any emergency situation.
Epinephrine Auto-Injectors Adoption Is Poised to Increase Due to its Mandate Use in School and Workplace.
These are driving factors in the epinephrine auto-injector market, as awareness of the risks from severe allergic reactions drives requirements for stocking these life-saving treatments in schools and the workplace.
Laws enacted in many parts of the world, especially in the USA, Europe, and Australia, require all schools and workstations to keep epinephrine injectors at hand for the protection of students, employees, or visitors from a sudden fatal anaphylactic reaction.
The legislators stress that education and training programs are necessary to make the non-medical staff aware of symptoms of an allergic reaction and how to effectively administer epinephrine. With this increasing need, the manufacturers along with health care providers offer the training programs as part of their services hence increasing demand for auto injectors.
In conclusion, epinephrine auto-injectors have increasingly been mandated for schools and workplaces, creating a steadily greater demand for the devices. Not only have these mandates given rise to manufacturers' selling opportunities for supplying institutions with medical devices, but they also create innovation incentives for design and functionalities in epinephrine auto-injectors.
Alternative Drug Delivery Systems Emerging as a Challenge for the Epinephrine Auto-Injectors Market
The growing field of alternative drug delivery systems presents a significant restraint toward the epinephrine auto-injectors market Although devices like the EpiPen have been the go-to solution for treating anaphylactic reactions, alternative methods of epinephrine administration are gaining momentum.
New delivery systems aim at overcoming certain drawbacks attributed to conventional auto-injectors, including high costs, ease of use, and accessibility.
The most notable developments in drug delivery technology is the development of needle-free systems for epinephrine administration. This design will help to overcome the fear and other issues in regard to needles associated with injectors.
Commonly, needle-free devices rely on a high pressure mechanism for the delivery of epinephrine under the skin using no traditional needles. These systems also eliminate the risks of needle use, such as needle phobia, accidental injury, or improper injection technique.
This, therefore, makes them very appealing, especially for kids and their caregivers who might feel apprehensive in an emergency situation with the use of needle-based devices.
One such example is the in-development Nasacort, or nasal spray epinephrine. This could give epinephrine nasally, allowing it to act quickly without being as invasive as an auto-injector. Epinephrine auto-injectors are still the most applied and the best-known treatment against anaphylaxis. Nevertheless, other types of drug delivery systems really compete with epinephrine autoinjectors for leading positions.
The global epinephrine auto-injectors industry recorded a CAGR of 3.3% during the historical period between 2021 and 2025. The growth of epinephrine auto-injectors industry was positive as it reached a value of USD 2,136.2 million in 2025 from USD 1,806.2 million in 2021.
Since the incorporation of the epinephrine auto-injector market, it has seen considerable development. Initially designed for military use, the technology was adapted for medical purposes to provide a quick, effective solution for treating anaphylaxis as the prevalence of food allergies and other triggers of anaphylaxis grew, demand for auto-injectors increased.
Some of the developments that ensued with increased awareness of severe allergic reactions included increased market penetration of epinephrine auto-injectors, especially in schools, workplaces, and public places. Various governments and health organizations helped in this call by passing legislation that ensured access to auto-injectors in public spaces.
In the years to come, technological innovations are expected to mark the face of the market. Newer, smarter auto-injectors with various digital capabilities in the form of mobile app connectivity, dosage tracking, and other user-friendly improvements are expected to enhance patient confidence and adherence.
This will allay the fears of improper usage or accidental injury, thus making the devices more user-friendly for a wide range of users.
Tier 1 companies comprise market leaders with a market revenue of above USD 100 million capturing significant market share of 62.5% in global market. Tier 1 companies focus on expanding their market presence in emerging economies by establishing local manufacturing facilities, distribution networks, and partnerships.
This strategy helps them tap into growing healthcare markets and address local demands more effectively. These companies implement rigorous quality control processes and ensure their products comply with international standards to build trust and credibility in the market. Prominent companies in tier 1 include Adamis Pharmaceuticals Corporation and Alk-Abello A/S.
Tier 2 companies include mid-size players with revenue of USD 50 to 100 million having presence in specific regions and highly influencing the local market and holds around 26.5% market share. Tier 2 companies often specialize in specific segments of the market, such as developing epinephrine auto-injectors for particular types of surgeries or materials.
This targeted approach allows them to become experts in niche areas and cater to specific needs that larger players might overlook. Prominent companies in tier 2 include Impax Laboratories, Inc. and Mylan N.V.
Finally, Tier 3 companies, such as Antares Pharma and others These market players specialize in specific products and cater to niche markets, adding diversity to the industry.
Overall, while Tier 1 companies are the primary drivers of the market, Tier 2 and 3 companies also make significant contributions, ensuring the Epinephrine Auto-Injectors sales remains dynamic and competitive.
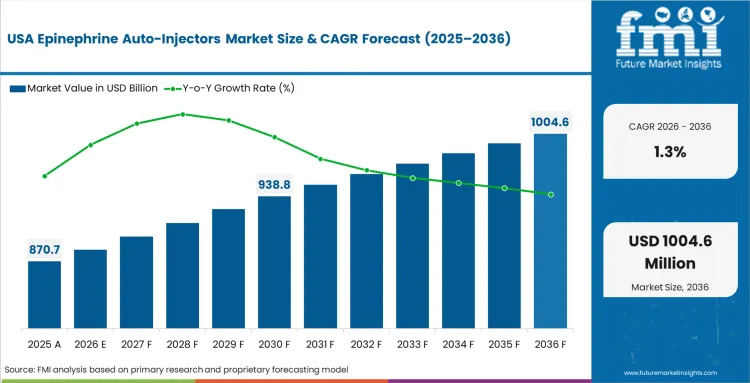
The section below covers the industry analysis for the epinephrine auto-injectors market for different countries. Market demand analysis on key countries in several regions of the globe, including North America, Asia Pacific, Europe, and others, is provided. The United States is anticipated to remain at the forefront in North America, with higher market share through 2036. In Asia Pacific, India is projected to witness a CAGR of 4.9% by 2036.
.webp)
| Countries | Value CAGR (2026 to 2036) |
|---|---|
| USA | 1.3% |
| Germany | 1.3% |
| China | 4.3% |
| France | 2.2% |
| India | 4.9% |
| Spain | 2.5% |
| Australia | 2.0% |
| South Korea | 3.4% |
Source: FMI analysis based on primary research and proprietary forecasting model
China epinephrine auto-injectors market is poised to exhibit a CAGR of 4.3% between 2026 and 2036. Currently, it holds the highest share in the East Asia market, and the trend is expected to continue during the forecast period.
The low costs of production methods in China, combined with lower labor costs, contribute to the affordable cost of epinephrine auto-injectors. This cost advantage will enable Chinese manufacturers to quote competitive prices in both the domestic and international markets, thereby attracting healthcare providers and distributors.
China has been able to corner a strong share in the global epinephrine auto-injectors export market by leveraging the advantage brought in by its manufactural potential to supply healthcare providers across the globe. This reach has given impetus to China's position in the international epinephrine auto-injectors market.
Supported by infrastructure development in the Chinese healthcare industry, such as building new hospitals and surgical centers, epinephrine auto-injectors have kept up their rapid growth. This growing market opens ample opportunities for local manufacturers to supply their products.
With enhanced healthcare standards globally, there will be a continuous demand for the highest quality drug delivery systems, including epinephrine auto-injectors. The increasing demand is based upon the requirement of precision, safety, and efficiency in drug delivery.
Epinephrine Auto-Injectors manufacturers view this as a challenge and substantially invest in research and development activities to come out with new products meeting the emerging requirements of health institutions. Improved infrastructure in the healthcare sector, primarily in emerging economies, has translated to better access to drug delivery systems and, consequently, increased demand for epinephrine auto-injectors.
Although there are opportunities galore in the market, some challenges could be the dampener for growth. Rigorous regulatory approvals in the case of medical devices are expected to serve as a barrier in the adoption of new technologies associated with epinephrine auto-injectors, hence delaying their launch.
India’s epinephrine auto-injectors market is poised to exhibit a CAGR of 4.9% between 2026 and 2036. Currently, it holds the highest share in the Asia Pacific market, and the trend is expected to continue during the forecast period.
According to, 2023 research article published by Medires, India contributes to 12.9% of Global Asthma cases and 42.4% of global Asthma Deaths This highlights the significant impact of asthma on the country’s health burden. The condition sometimes causes extreme respiratory distress and allergic reactions including life-threatening anaphylaxis.
Asthma patients are continuously increasing in rural and urban areas of India because of the continuous deterioration of air quality due to pollution, industrialization, and urbanization. The rise increased demand for emergency medications like epinephrine that help avoid fatal outcomes because of a severe allergic reaction or exacerbation of asthma.
In addition, the main factors contributing to the high demand in the auto-injector market include increasing awareness among patients and healthcare providers about anaphylaxis and the requirement for emergency preparedness. Doctors also recommend better availability of epinephrine auto-injectors, especially for patients showing more tendency toward severe asthma attacks or other allergic reactions.
The section contains information about the leading segments in the industry. By dosage, the 0.3 mg segment holds the highest market share of 46.6% in 2026.
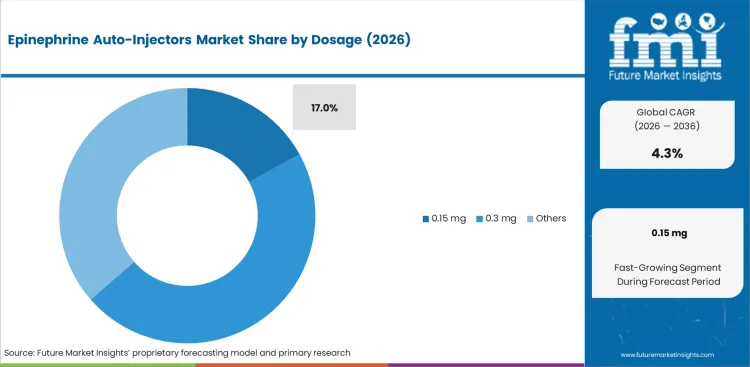
| By Dosage | 0.3 mg |
|---|---|
| Value Share (2026) | 46.6% |
The 0.3 mg dosage segment dominates the dosage segment of the epinephrine auto-injectors market, as the dosages are appropriate for treating anaphylactic reactions in adults and people weighing over 30 kg (66 lbs).
The 0.3 mg dosage was considered a standard dosage for the effective delivery of epinephrine to counteract severe allergic reactions, such as symptoms related to anaphylaxis like difficulty in breathing, swelling, and shock, since the medication is lifesaving. Epinephrine constricts the blood vessels and relaxes the smooth muscles of the airways, returning breathing and blood pressure to normal.
The broad spread of the 0.3 mg dosage results from the fact that clinically it acts on anaphylaxis-a life-threatening condition that calls for immediate attention. It is commonly recommended for adult patients and, for that reason, falls in the category of emergency preparedness among people who are vulnerable to allergic attacks.
Efficiency in handling, portability, and ease of handling allow 0.3 mg epinephrine autoinjectors to be the most approached option by healthcare providers, emergency response people, and individuals managing allergies.
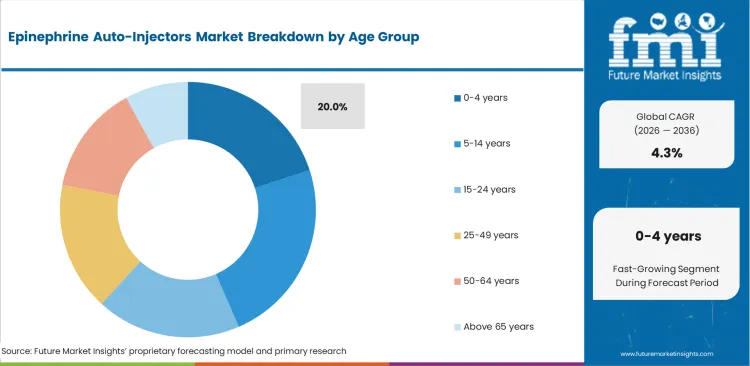
| By Age Group | 5-14 Years |
|---|---|
| Value Share (2026) | 23.5% |
Children are particularly vulnerable to developing allergies, and the early school years are often when many allergic reactions are first identified and diagnosed. Common allergens in this age group include foods like peanuts, tree nuts, dairy, and shellfish, as well as insect stings and medications.
Another key factors driving the dominance of the 5-14 years age group in this market is the increasing awareness of anaphylaxis in schools, daycare centers, and among parents. Public health campaigns with education about the condition lead to increased identification of severe allergic reactions, which in turn has driven more adoptions of epinephrine auto-injector adoption for at-risk children.
Schools have become key players, especially in ensuring availability for such emergencies through the implementation of policies in many countries that make it necessary for institutions to stock these lifesaving devices.
Additionally, the rise in the number of children with multiple allergies has further fueled demand for epinephrine auto-injectors.
The market players are using strategies to stay competitive, such as product differentiation through innovative formulations, strategic partnerships with healthcare providers for distribution. Another key strategic focus of these companies is to actively look for strategic partners to bolster their product portfolios and expand their global market presence.

| Metric | Value |
|---|---|
| Quantitative Units | USD 2.40 billion to USD 3.65 billion, at a CAGR of 4.3% |
| Market Definition | Epinephrine auto-injectors encompass pre-filled emergency injection devices in 0.15 mg, 0.3 mg, and other dosages for anaphylaxis treatment across hospital, clinic, home, and institutional settings. |
| Dosage Segmentation | 0.15 mg, 0.3 mg, Others |
| Age Group Segmentation | 0 to 4 Years, 5 to 14 Years, 15 to 24 Years, 25 to 49 Years, 50 to 64 Years, Above 65 Years |
| End User Segmentation | Hospitals, Clinics, Home Based, Others |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | India, China, South Korea, Spain, France, Australia, USA, Germany, and 40 plus countries |
| Key Companies Profiled | Adamis Pharmaceuticals Corporation, Alk-Abello A/S, Impax Laboratories, Inc., Viatris, Antares Pharma, Amgen Inc., Biocon, Bio-Rad Laboratories Inc., F. Hoffmann-La Roche Ltd |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with verified procurement and shipment data, cross-validated against publicly reported capacity metrics and financial guidance. |
In terms of dosage, the industry is segregated into 0.15 mg, 0.3 mg and others.
In terms of age group, the industry is divided into 0-4 years, 5-14 years, 15-24 years, 25-49 years, 50-64 years and above 65 years.
In terms of end user, the industry is divided into hospitals, clinics, home based and others.
Key countries of North America, Latin America, Western Europe, Eastern Europe, South Asia & Pacific, East Asia and Middle East and Africa (MEA) have been covered in the report.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
How large is the demand for Epinephrine Auto-Injectors in the global market in 2026?
Demand for Epinephrine Auto-Injectors in the global market is estimated to be valued at USD 2.40 billion in 2026.
What will be the market size of Epinephrine Auto-Injectors in the global market by 2036?
Market size for Epinephrine Auto-Injectors is projected to reach USD 3.65 billion by 2036.
What is the expected demand growth for Epinephrine Auto-Injectors in the global market between 2026 and 2036?
Demand for Epinephrine Auto-Injectors is expected to grow at a CAGR of 4.3% between 2026 and 2036.
Which Dosage is poised to lead global sales by 2026?
0.3 mg accounts for 46.6% in 2026 as dosage-specific procurement preferences favour established performance-certified configurations.
How is the role of 5 to 14 Years in driving Epinephrine Auto-Injectors adoption in 2026?
5 to 14 Years represents 23.5% of age group share as application requirements standardize around verified performance benchmarks.
What is driving demand in India?
India leads with a 4.9% CAGR driven by infrastructure expansion, rising institutional requirements, and supportive regulatory frameworks that accelerate adoption of specification-grade products.
What is Epinephrine Auto-Injectors and what is it mainly used for?
Epinephrine auto-injectors are pre-filled devices that deliver a measured dose of epinephrine for emergency anaphylaxis treatment. They are used by patients, caregivers, and institutions to manage severe allergic reactions in hospital, clinic, home, and public settings.
What does market forecast mean on this page?
The market forecast represents a model-based projection built on verified procurement data, capacity metrics, and regulatory compliance assumptions for strategic planning purposes.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.