Femtech Market
Femtech Market Size and Share Forecast Outlook 2026 to 2036
Femtech Market Forecast and Outlook 2026 to 2036
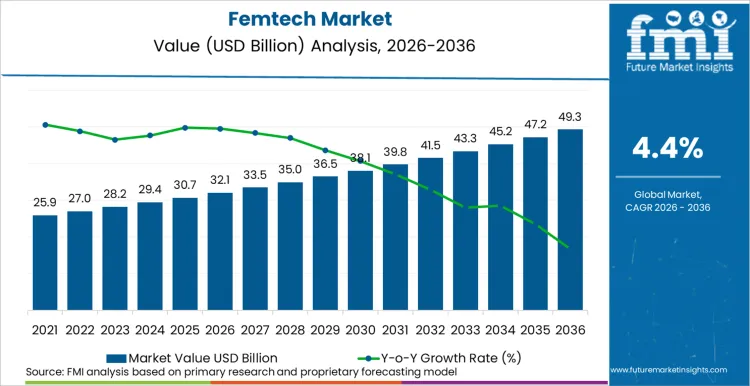
The global femtech market is likely to total USD 32.1 billion in 2026, and expected to advance to USD 49.3 billion by 2036, at a 4.4% CAGR. Expansion is increasingly tied to women’s health moving into formal agendas, such as the USA Executive Order 14120 elevated women’s health research and translation into real-world care, while NHS England required women’s health hubs across ICBs to improve access and care experience, pulling femtech demand toward standardized pathways and measurable outcomes.
From 2026 to 2036, scale economics for femtech will be shaped less by feature velocity and more by governance and pathway-fit. HIPAA reproductive-health privacy changes, including a February 16, 2026 compliance date for remaining Notice of Privacy Practices modifications after a June 18, 2025 court order vacated most provisions, signals sustained scrutiny on how sensitive women’s health data is handled, which is raising value of auditable consent, disclosure controls, and vendor operating discipline.
Quick Stats in the Global Femtech Market
- Femtech Market Value (2026): USD 32.1 billion
- Femtech Market Forecast Value (2036): USD 49.3 billion
- Femtech Market Forecast CAGR (2026-2036): 4.4%
- Leading Product Type Segment in Femtech Market: Diagnostics (54.0%)
- Leading Application Segment in Femtech Market: General Health and Wellness (63.0%)
- Leading End User Segment in Femtech Market: Direct to Consumer (56.0%)
- Key Companies Profiled: Flo Health Inc., Clue by BioWink GmbH, Natural Cycles USA Corp., Elvie (Chiaro Technology Ltd.), Willow Innovations, Inc., Others
How are Policy and Compliance Frameworks Shaping Femtech Adoption in Key Regions?
| Region | 2026 Metrics |
|---|---|
| North America | D2C platforms extend into employer and provider ecosystems. OCR issued a 2024 HIPAA reproductive-health privacy Final Rule; a June 18, 2025 court order vacated most provisions, while notices-of-privacy-practices updates carry a February 16, 2026 compliance date in industry guidance. Differentiation shifts to auditable consent and disclosure controls that can be configured per partner workflow. |
| Latin America | Wellness tools are moving into regulated diagnostic and monitoring modules. Brazil’s RDC 848/2024, effective September 4, 2024, updates essential safety and performance requirements across medical device lifecycle, raising expectations for technical documentation, risk controls, and post-market routines in regulated women’s health products. |
| Europe | Reimbursement and evidence gating shape scale. Germany’s DiGA fast-track guidance sets assessment-facing expectations for evidence and data protection in reimbursable digital health applications. England’s 2024 women’s health priorities expand women’s health hubs and reinforce demand around menstrual health, menopause care, and maternity support. Vendors separate consumer UX from reimbursable pathways, with evidence artifacts and governance controls becoming launch prerequisites. |
| Asia Pacific | National digital rails reduce onboarding friction and improve continuity. India’s ABDM updates show expanding ABHA identity and broad facility and professional registration, accelerating interoperability at ecosystem level. Integration readiness becomes a growth lever, lowering acquisition cost via ecosystem distribution while tightening expectations around consent-led data exchange. |
| Middle East & Africa | Government-led modernization drives procurement-led adoption. UAE Federal Decree-Law No. (38) of 2024 introduces governance mechanisms such as national tracking, coding, and record requirements. Players with documentation maturity and compliant commercialization playbooks gain procurement credibility faster. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Femtech Market Key Metrics
| Metric | Value |
|---|---|
| Industry Size (2026E) | USD 32.1 billion |
| Industry Value (2036F) | USD 49.3 billion |
| CAGR (2026 to 2036) | 4.4% |
Why is the Femtech Market Expanding?
Women’s health is now being treated as a delivery-system problem, not only an awareness gap. UK 2024 women’s health priorities explicitly spotlight menstrual problems, menopause, maternity care, and birth trauma support-areas that translate into demand for triage, diagnostics, and monitored pathways rather than standalone self-tracking.
Similar momentum is visible in the USA, with policy signals such as those from Executive Order 14120, which pushes research translation and disparity closure. This is raising appetite for evidence-ready femtech solutions. Execution is aligned with decision structures associated with digital health, telehealth and telemedicine, and digital health tracking apps, especially where clinical partners require traceability, consent clarity, and auditable workflows.
How is the Femtech Market Segmented?
Why are Women’s Health Hub Pathway Design and One-Visit Service Integration reinforcing Diagnostics leadership?
Diagnostics accounts for a 54.0% share because women’s health hubs are being operationalised as intermediate-care models that explicitly target fragmentation and prioritise earlier intervention, fewer avoidable referrals, and more care delivered in a single appointment. Hub core specification is explicit on integrated, multi-need visits.
Examples include combining cervical screening with LARC fitting/removal, and managing heavy menstrual bleeding in a consolidated workflow. This redesign changes demand shape, vendors that support rule-out logic, structured documentation, and referral-ready outputs fit commissioning intent better than tools optimised only for engagement.
Policy signals also pull demand toward diagnostics-supported journeys, because 2024 priorities for England’s Women’s Health Strategy highlight menstrual problems, menopause, and maternity care and birth trauma support. These are areas where pathway throughput depends on consistent assessment, escalation criteria, and tracked follow-up rather than isolated content experiences. The net effect is diagnostics-led propositions sit closer to system objectives than standalone wellness propositions, which preserves share as hub models move from pilots into baseline delivery expectations.
Why does General Health and Wellness remain dominant as a Life-course Entry Layer?
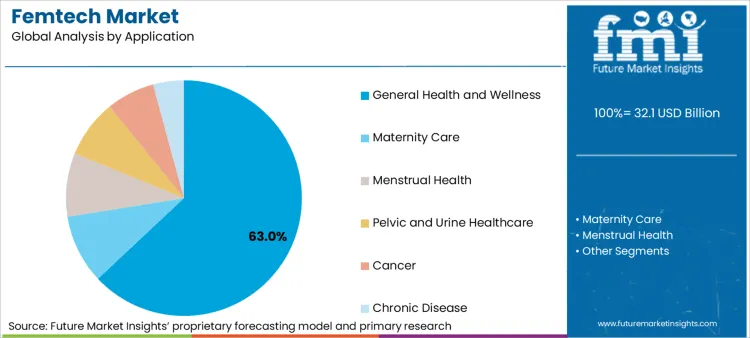
General health and wellness holds a 63.0% share because women’s health hubs are defined around a life-course approach and wrapped care, which is explicitly not limited to single-condition interventions. These are such broad entry-layer journeys remain structurally advantaged for onboarding, continuity, and routing into condition-specific services when risk increases. NHS operational planning guidance reinforces this logic by requiring at least one women’s health hub in every ICB by December 2024, with minimum requirements and a virtual option. These conditions favor scalable, generalist access layers that can triage and direct demand efficiently.
Women’s health is being positioned as a long-horizon outcomes agenda, not a short-cycle engagement theme. For instance, Executive Order 14120 frames federal policy around advancing women’s health research, closing disparities, and translating research into real-world clinical benefit across lifespans, which supports platform strategies that start with broad health-and-wellbeing entry points and then deepen into validated modules. The UK government priorities for 2024 similarly span multiple life-stage domains which keeps general health and wellness as the connective tissue across journeys rather than a single application silo.
Why does Direct to Consumer lead End User Share?
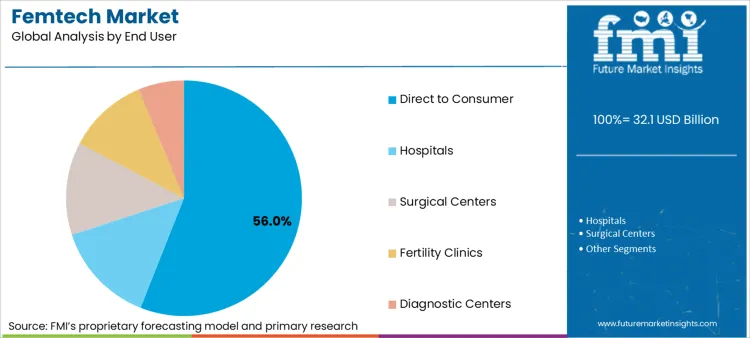
Direct to consumer holds a 56.0% share as subscription-led engagement still funds acquisition and iteration. Even so, D2C leaders increasingly design provider-compatible workflows, including data export, consent controls, and clear claims discipline, to keep doors open for employer and clinical channel expansion.
Femtech market is segmented by product type as diagnostics, monitoring, therapeutic, and consumer products. Femtech market is segmented by application as general health and wellness, maternity care, menstrual health, pelvic and urine healthcare, cancer, and chronic disease. End user as direct to consumer, hospitals, surgical centers, diagnostic centers, fertility clinics, and others segments Femtech market.
Wearables are reinforcing monitoring-led femtech propositions, especially as device ecosystems mature. Workflow fit improves when signals arrive through regulated or semi-regulated channels aligned with wearable medical devices, wearable healthcare devices, and telemedicine equipment, which supports higher-frequency follow-up and clearer escalation logic.
What are the key Femtech Market dynamics?
Privacy and governance are moving from legal review into product design constraints. USA reproductive-health privacy actions under HIPAA signal that sensitive women’s health data handling will face continued scrutiny, pushing vendors toward explicit consent states, role-based access, and defensible disclosure pathways—especially when femtech touches regulated services or clinical partnerships.
Public-system service redesign is also reframing demand. Women’s health hubs in England create structured access points that reward solutions capable of triage support, referral routing, and measurable care experience uplift-shifting vendor value from engagement toward pathway performance, with interoperability increasingly shaped by broader telehealth and telemedicine operating models.
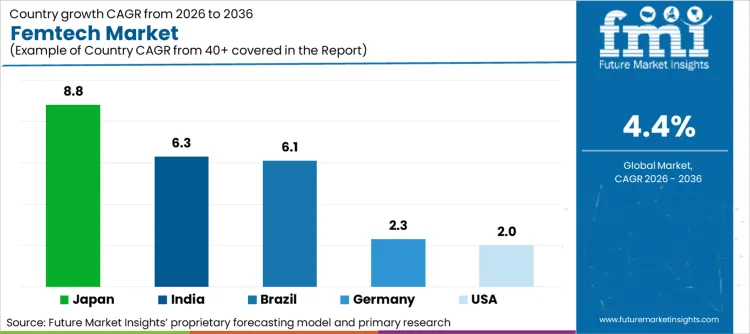
Femtech Market Country-wise Analysis
| Country | CAGR (2026-2036) |
|---|---|
| USA | 2.0% |
| Germany | 2.3% |
| India | 6.3% |
| Japan | 8.8% |
| Brazil | 6.1% |
How will the Femtech Landscape in USA evolve as it Leverages Key Advancements in Women’s Health?
The femtech landscape in USA is expected to progress at a 2.0% CAGR from 2026 to 2036. This growth reflects a tightening linkage between women’s health products and data-governance expectations.
HHS notes a June 18, 2025 court order vacated most of the HIPAA reproductive health privacy rule, while remaining NPP modifications still require compliance by February 16, 2026. This helps keep privacy-by-design and disclosure defensibility central for femtech partners operating with providers and employers.
Momentum also tracks women’s health elevation in federal priorities. The Executive Order 14120 strengthens research translation intent, which increases demand for solutions that can support evidence generation, cohort analytics, and clinically meaningful endpoints rather than engagement-only KPIs.
How is Germany maintaining Measured, Evidence-led scaling for Femtech?
Projected to record a 2.3% CAGR, the femtech market’s expansion in Germany is shaped by reimbursement-adjacent discipline and documentation rigor. BfArM’s DiGA Fast-Track guidance codifies process expectations and evidence orientation, influencing how vendors design studies, claims language, and submission readiness for women’s health applications that seek formal channel access.
Commercial success increasingly depends on packaging product capabilities into payer-readable artifacts. These include outcomes logic, risk management, and data processing clarity. This shifts competitive advantage from feature breadth to execution quality in regulated documentation and post-market discipline, including positioning that remains compatible with digital health solutions used by health systems.
How is India accelerating Femtech adoption through National Digital Rails?
The femtech market in India is projected to advance at a 6.3% CAGR from 2026 to 2036. Growth is reinforced by ABDM scale-up creating structural interoperability incentives. Government updates report large-scale ABHA creation and substantial health record linkage, making consent-led identity and data portability more actionable across hospital networks and app ecosystems.
Femtech vendors that align onboarding, consent capture, and record-sharing workflows with ABDM rails are positioned to move faster from standalone D2C adoption to provider-connected deployments, especially for monitoring and maternal pathways that benefit from continuity. This alignment increasingly mirrors operating patterns seen across digital health workflows in other scaled public-infrastructure settings.
What is enabling Japan’s High-growth Shift toward Regulated Modules in the Femtech Industry?
Japan’s growth is supported by regulatory modernization aimed at software-driven care. A CAGR of 8.8% is forecasted for the femtech market in Japan. The USA International Trade Administration notes a two-stage approval system for medical device software (SaMD) which commenced in 2024, creating a clearer route for monitoring and therapeutic modules that rely on real-world evidence progression.
This supports vendor roadmaps that start with bounded claims and controlled rollouts then expand indications with accumulated evidence. These favour organizations that can operate disciplined post-market analytics, safety monitoring, and iterative validation without diluting clinical credibility.
How is Brazil pushing Femtech Expansion from Wellness into Regulated Performance?
With an expected CAGR of 6.1%, Brazil’s femtech industry growth is shaped by stronger conformity expectations for medical devices and related software. ANVISA notes that regulations have a major influence in pushing femtech prospects ahead. An example of this is the RDC 848/2024, which entered into force on September 4, 2024. The regulation updated essential safety and performance requirements, raising documentation and design-control requirements that affect regulated women’s health diagnostics and monitoring modules.
Vendor scale increasingly depends on technical file maturity, risk management, performance justification, and lifecycle controls rather than rapid claim-driven expansion. Companies with regulator-ready documentation and structured validation processes can expand faster across institutional buyers.
Femtech Market Competitive Landscape
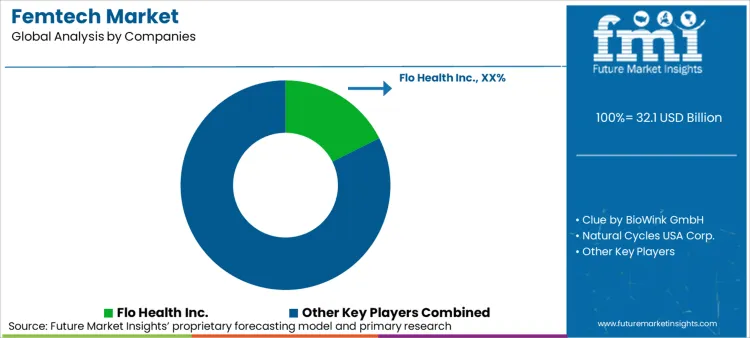
Competitive advantage is consolidating around trust, evidence readiness, and channel strategy. Governance expectations are rising as women’s health data becomes more legally and reputationally sensitive; recent legal developments around reproductive health privacy reinforce a need for auditable consent and defensible disclosure logic in partner-led deployments.
Consolidation and accountability signals are also visible in operator moves and privacy outcomes. Willow’s acquisition of Elvie assets following Elvie’s administration illustrates portfolio consolidation pressure in device-led maternal health, while reported settlements tied to period-tracking data-sharing allegations underline persistent scrutiny on data practices in women’s health apps.
Workflow design is often aligned with decision structures associated with healthcare digital experience platforms and digital transformation in healthcare, especially where delivery systems demand standardized intake, referral routing, and measurable patient experience outcomes.
Femtech Market Key Players
- Flo Health Inc.
- Clue by BioWink GmbH
- Natural Cycles USA Corp.
- Elvie (Chiaro Technology Ltd.)
- Willow Innovations, Inc.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Product Type | Diagnostics; Monitoring; Therapeutic; Consumer Products |
| Application | General Health and Wellness; Maternity Care; Menstrual Health; Pelvic and Urine Healthcare; Cancer; Chronic Disease |
| End User | Direct to Consumer; Hospitals; Surgical Centers; Diagnostic Centers; Fertility Clinics; Others |
| Countries covered | USA; Germany; India; Japan; Brazil |
Key Segments
By Product Type
- Diagnostics
- Monitoring
- Therapeutic
- Consumer Products
By Application
- General Health and Wellness
- Maternity Care
- Menstrual Health
- Pelvic and Urine Healthcare
- Cancer
- Chronic Disease
By End User
- Direct to Consumer
- Hospitals
- Surgical Centers
- Diagnostic Centers
- Fertility Clinics
- Others
By Region
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia & Pacific
- Middle East & Africa
Bibliography
- USA Department of Health & Human Services. (2025). HIPAA Privacy Rule Final Rule to Support Reproductive Health Care Privacy: Fact Sheet.
- UK Department of Health and Social Care. (2024). Health Secretary announces new women’s health priorities for 2024.
- Federal Register. (2024). Advancing Women’s Health Research and Innovation.
- Agência Nacional de Vigilância Sanitária (ANVISA). (2024). Norma sobre requisitos de segurança para dispositivos médicos entra em vigor (RDC 848/2024).
- Ministry of Health and Family Welfare, Government of India. (2024). Update on Ayushman Bharat Digital Mission.
- Federal Institute for Drugs and Medical Devices (BfArM). (2025). Digital Health Applications (DiGA) and DiGA guide resources.
Frequently Asked Questions
How Opportunistic are Growth Prospects for the Global Femtech Market in 2026?
In 2026, the global femtech market is projected to attain a valuation of USD 32.1 billion.
At what rate is the Global Femtech Market expected to rise from 2026 to 2036?
The femtech industry is forecasted to advance at a CAGR of 4.4% from 2026 to 2036.
How much value will the Global Femtech Market generate by 2036?
The femtech landscape is anticipated to total USD 49.3 billion by 2036.
Which countries are the Frontrunners of the Femtech Industry?
USA, Germany, India, Japan, and Brazil emerge as the main engines of the femtech industry’s growth, as per FMI’s findings.
Which Product Category is expected to capture the largest share of the Global Femtech Industry?
Femtech diagnostics dominates by product type, expected to capture 54% of total revenues in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Roots Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended A9oach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Alcohol-Free
- Low Alcohol
- Alcohol-Free
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Category
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Category, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Category, 2026 to 2036
- Flavored
- Plain
- Flavored
- Y to o to Y Growth Trend Analysis By Category, 2021 to 2025
- Absolute $ Opportunity Analysis By Category, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Material, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Material, 2026 to 2036
- Malted Grains
- Hops
- Yeasts
- Enzymes
- Others
- Malted Grains
- Y to o to Y Growth Trend Analysis By Material, 2021 to 2025
- Absolute $ Opportunity Analysis By Material, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sales Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sales Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sales Channel, 2026 to 2036
- Supermarkets
- Convenience Stores
- Liquor Stores
- Restaurants & Bars
- Online Stores
- Supermarkets
- Y to o to Y Growth Trend Analysis By Sales Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Sales Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Category
- By Material
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Category
- By Material
- By Sales Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Category
- By Material
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Category
- By Material
- By Sales Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Category
- By Material
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Category
- By Material
- By Sales Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Category
- By Material
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Category
- By Material
- By Sales Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Category
- By Material
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Category
- By Material
- By Sales Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Category
- By Material
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Category
- By Material
- By Sales Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Category
- By Material
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Category
- By Material
- By Sales Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Category
- By Material
- By Sales Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Category
- By Material
- By Sales Channel
- Competition Analysis
- Competition Deep Dive
- Anheuser-Busch InBev
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Heineken N.V.
- Carlsberg A/S
- Suntory Beer
- Big Drop Brewing Co.
- Athletic Brewing
- Erdinger Weibbrau
- Anheuser-Busch InBev
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Category, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Category, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Category, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Category, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Category, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Category, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Category, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Category, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Category, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Category, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Category
- Figure 9: Global Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Material
- Figure 12: Global Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Sales Channel
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Product Type
- Figure 29: North America Market Value Share and BPS Analysis by Category, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Category, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Category
- Figure 32: North America Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Material
- Figure 35: North America Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Sales Channel
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Product Type
- Figure 42: Latin America Market Value Share and BPS Analysis by Category, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Category, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Category
- Figure 45: Latin America Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Material
- Figure 48: Latin America Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Sales Channel
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by Product Type
- Figure 55: Western Europe Market Value Share and BPS Analysis by Category, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Category, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Category
- Figure 58: Western Europe Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Material
- Figure 61: Western Europe Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Sales Channel
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Category, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Category, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Category
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Material
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Sales Channel
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by Product Type
- Figure 81: East Asia Market Value Share and BPS Analysis by Category, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Category, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Category
- Figure 84: East Asia Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Material
- Figure 87: East Asia Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by Sales Channel
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Category, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Category, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Category
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Material
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Sales Channel
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Category, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Category, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Category
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Material
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Sales Channel
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

