GLP-1 Receptor Agonist Market
GLP-1 Receptor Agonist Market The GLP-1 receptor agonist market is segmented by Drug (Semaglutide, Exenatide, Lixisenatide, Liraglutide, Dulaglutide, and Others), by Route of Administration (Oral and Parenteral), by Indication (Diabetes, Obesity, and Others), by Distribution Channel (Hospital Pharmacies, Retail Pharmacies, and Online Pharmacies), and by Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
GLP-1 Receptor Agonist Market Size, Market Forecast and Outlook By FMI
The GLP-1 receptor agonist market is expected to expand from USD 2.9 billion in 2026 to USD 7.8 billion by 2036. The market is anticipated to register a 11.7% CAGR during the forecast period. Exenatide is likely to lead with 24.4% share of the drug segment in 2025 as diabetes and obesity use cases widen across prescription channels.
Summary of the GLP-1 Receptor Agonist Market
- Demand and Growth Drivers
- Rising obesity and diabetes prevalence is expected to increase physician interest in therapies with metabolic and cardiovascular benefits.
- Expanding clinical evidence is likely to increase payer review of GLP-1 drugs across diabetes and weight management programs.
- Cardiovascular risk reduction claims are anticipated to raise treatment adoption across patients needing broader metabolic disease management.
- Product and Segment View
- Exenatide is expected to lead the drug segment through its established use across cost-sensitive treatment environments.
- Parenteral therapies are likely to keep strong adoption as injectable formats already have mature prescribing and device systems.
- Oral formulations are anticipated to gain faster acceptance among patients seeking non-injectable options for long-term therapy use.
- Geography and Competitive Outlook
- India is expected to record the fastest listed country expansion through rising diabetes prevalence and improving access to chronic care.
- China is likely to expand steadily as urban obesity awareness and diabetes treatment access increase across major provinces.
- Novo Nordisk and Eli Lilly are anticipated to strengthen competition through evidence generation and manufacturing capacity expansion.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant for Healthcare at FMI, suggests, “The GLP-1 receptor agonist market is expected to shift toward companies able to combine clinical evidence with reliable supply execution. Manufacturers with strong cardiometabolic data and scalable production are likely to gain advantage as payers review access across diabetes and obesity indications.”
- GLP-1 Receptor Agonist Market Value Analysis
- The GLP-1 receptor agonist market is moving from diabetes-centered prescribing toward broader cardiometabolic disease management.
- Demand is likely to rise as obesity treatment and cardiovascular risk reduction widen the eligible patient pool.
- Adoption is expected to improve as oral formulations reduce injection concerns among patients starting long-term therapy.
- Spending is anticipated to increase as manufacturers invest in outcome trials and larger production networks.
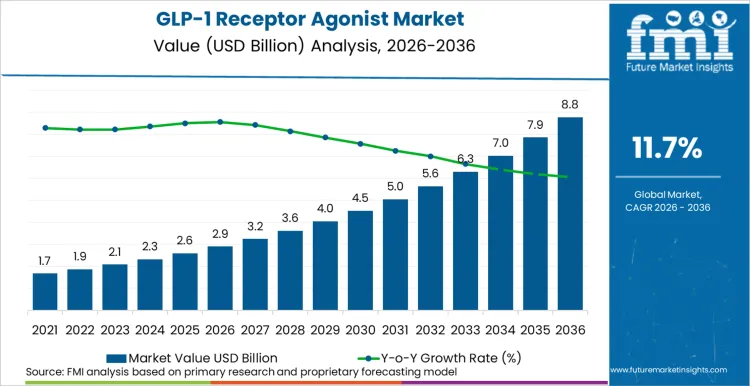
This performance reflects accelerating demand for high-efficacy, multi-indication GLP-1 molecules across North America, Europe, and Asia-Pacific, driven by blockbuster drugs such as semaglutide and tirzepatide. The therapeutic class is experiencing unprecedented demand as prescriber awareness of pleiotropic benefits, including kidney disease event reduction and major adverse cardiovascular event reduction, reshapes treatment algorithms.
North America leads global expansion, supported by high obesity prevalence and concentrated commercial infrastructure. Europe follows, driven by guideline-directed medical therapy adoption. Asia-Pacific emerges as a high-growth corridor, fueled by lifestyle disease burden and healthcare infrastructure modernization.
GLP-1 Receptor Agonist Market Definition
The GLP-1 receptor agonist market encompasses the global development, production, and supply of pharmaceutical agents that mimic the action of endogenous glucagon-like peptide-These injectable or oral therapies enhance glucose-dependent insulin secretion, suppress glucagon release, slow gastric emptying, and promote satiety. They are indicated for type 2 diabetes management, weight reduction in obesity, and cardiovascular risk reduction, with ongoing investigation into metabolic dysfunction-associated steatohepatitis, chronic kidney disease, and heart failure with preserved ejection fraction.
Market Inclusions
The report includes global and regional market size estimates in value terms, with a forecast period from 2026 to 2036. It provides segmentation by Drug, Route of Administration, Indication, Distribution Channel, and Region. The analysis incorporates manufacturing technology trends, clinical trial developments, regulatory approval pathways, and competitive positioning across therapeutic indications. Regional demand dynamics, country-level growth rates, and reimbursement landscape assessments are also evaluated as per FMI methodology.
Market Exclusions
The scope excludes over-the-counter weight loss supplements, non-prescription metabolic aids, and compounded drug products not subject to FDA new drug application approval. It also excludes revenues from delivery device manufacturing where separable from pharmaceutical product value. Biosimilar entrants not yet approved in major regulated markets are not considered in base year estimates due to regulatory uncertainty.
Research Methodology
- Primary Research: Interviews were conducted with endocrinologists, cardiologists, obesity medicine specialists, hospital pharmacy directors, managed care organization formulary managers, and regulatory affairs consultants.
- Desk Research: Public company filings, FDA and EMA approval documents, clinical trial registries, medical literature, and payer coverage policies were reviewed.
- Market-Sizing and Forecasting: A hybrid top-down and bottom-up model was constructed, reconstructing demand from prescription volume data, patient population estimates, and treatment penetration rates across indications.
- Data Validation and Update Cycle: Outputs were subjected to variance testing against epidemiological data, production capacity announcements, and structured internal review prior to publication.
GLP-1 Receptor Agonist Market Drivers, Restraints, and Opportunities
Future Market Insights analysis indicates the market has historically existed as a specialty diabetes category anchored in glycemic control. Its baseline scale is rooted in the rising global prevalence of type 2 diabetes. As per FMI, the market's valuation is increasingly uncoupled from diabetes prevalence alone, driven by regulatory endorsement of weight management indications and cardiovascular risk reduction claims that expand the addressable patient population by an order of magnitude [1].
FMI analysts observe a clear stratification between first-generation GLP-1s optimized for diabetes management and next-generation molecules engineered for weight loss superiority. Standard therapies face pressure where payers implement step therapy protocols favoring agents with cardiovascular outcomes data, while growth concentrates in molecules demonstrating superior weight reduction that justify premium pricing through obesity indication exclusivity. This shift nets out as accelerated value growth even as price competition intensifies in commoditized diabetes segments.
- Expanding Clinical Indications: Recent trial results have fundamentally reshaped the GLP-1 value proposition. Studies have demonstrated that GLP-1s reduce major adverse cardiovascular events and slow progression of kidney disease, transforming these agents from glucose-lowering drugs into cardiorenal-metabolic disease modifiers with expanded addressable populations [1]. The SELECT trial established cardiovascular risk reduction in overweight adults without diabetes, while the STEP HFpEF program showed symptomatic improvement in heart failure with preserved ejection fraction [2].
- Obesity Prevalence and Treatment Paradigm Shift: With over one billion people globally living with obesity and only a fraction receiving pharmacotherapy, the addressable market dwarfs diabetes alone [1]. Regulatory approvals for weight management indications, including expanded labels for cardiovascular risk reduction in overweight adults, have unlocked primary care prescribing and direct-to-consumer demand. Head-to-head studies demonstrating superior relative weight loss between competing molecules intensify competition for obesity market share [3].
- Regulatory Frameworks and Supply Dynamics: In December 2025, the FDA approved the first oral GLP-1 for weight loss-Novo Nordisk's once-daily semaglutide pill, which launched in January 2026, significantly expanding accessibility for patients averse to injections [3]. CMS has unveiled its BALANCE model, a voluntary Medicare and Medicaid coverage pathway for GLP-1s that includes price negotiations and supplemental lifestyle support, with coverage beginning in 2026 for Medicaid and 2027 for Medicare Part D [2]. Pricing pressures have surfaced across payer earnings, with some insurers expanding access with guardrails while others have dropped coverage entirely [3].
- Manufacturing Capacity and Supply Chain Investment: In a bid to avoid previous shortages of injectable GLP-1s, Eli Lilly has amassed a $1.5 billion pre-launch inventory of its oral weight loss drug orforglipron ahead of FDA decision expected in April 2026 [4]. This substantial inventory build signals management's confidence in regulatory approval and represents a decisive effort to avoid repeating the supply constraints that plagued earlier product rollouts [4]. Lilly is also cementing a long-term strategy by constructing new manufacturing facilities as part of a $27 billion investment drive, with at least three confirmed as production hubs for weight loss therapies [3].
- Pipeline Innovation and Differentiation: The development pipeline features next-generation mechanisms beyond single GLP-1 agonism. Eli Lilly's orforglipron is an investigational once-daily oral small molecule GLP-1 that does not carry restrictions on food and water intake, while retatrutide is a first-in-class once-weekly triple hormone receptor agonist activating GIP, GLP-1, and glucagon receptors [2]. Clinical results show retatrutide produced significant weight loss and improved pain and physical function among patients with obesity or overweight and knee osteoarthritis [1]. Analysts project orforglipron sales could reach significant figures in the coming decade, with retatrutide generating substantial revenue in the same timeframe [3].
Segment Analysis
GLP-1 Receptor Agonist Market Analysis by Drug
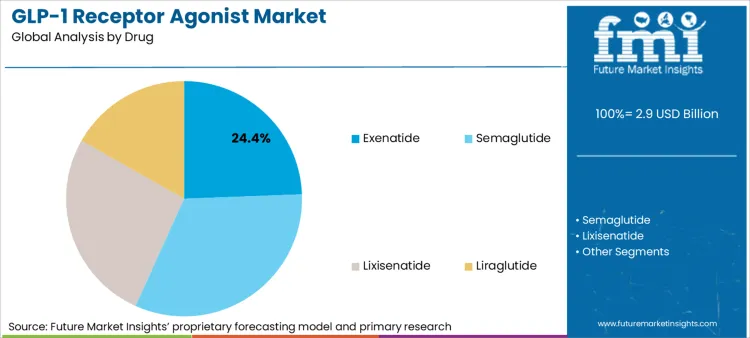
Based on FMI's GLP-1 receptor agonist market report, demand for exenatide is estimated to lead the drug segment, capturing 24.4% of the market share in 2025. Exenatide's established position reflects its first-to-market advantage and entrenched prescribing patterns in cost-sensitive healthcare systems. However, the competitive landscape is rapidly evolving with next-generation molecules demonstrating superior efficacy. Semaglutide, marketed as Ozempic for diabetes and Wegovy for obesity, has emerged as the commercial powerhouse, with the oral version launching in January 2026 following December 2025 FDA approval [3].
Tirzepatide, a dual GIP/GLP-1 agonist, has demonstrated superior weight loss efficacy, reshaping prescriber preferences in obesity management. The oral semaglutide formulation addresses injection-averse patient populations, capturing share in primary care settings where oral administration reduces treatment initiation barriers [2]. Looking ahead, orforglipron and retatrutide from Eli Lilly exemplify the next generation of metabolic therapies: oral formulations that promise improved adherence and triple-hormone mechanisms that deliver superior weight loss [4].
GLP-1 Receptor Agonist Market Analysis by Indication
Diabetes remains the foundation indication, accounting for the majority of current prescribing volume. However, obesity is the fastest-growing segment, with weight management indications driving incremental market expansion. The clinical rationale for obesity treatment extends beyond cosmetic benefit to meaningful cardiovascular risk reduction: Wegovy's label now includes cardiovascular risk reduction in overweight adults, fundamentally altering the value proposition for payers [1]. Emerging indications include metabolic dysfunction-associated steatohepatitis, where novel candidates are achieving statistically significant improvement with fibrosis improvement in studies. Chronic kidney disease represents another expansion opportunity, supported by data demonstrating renal protection independent of glycemic effects [2]. This indication diversification insulates GLP-1 manufacturers from reimbursement pressure in any single category while creating cross-indication pricing power.
GLP-1 Receptor Agonist Market Analysis by Route of Administration
Parenteral administration currently dominates, reflecting the biologic nature of peptide therapeutics and established delivery device infrastructure. However, oral formulations represent the most significant route-specific innovation, with the potential to expand the market by addressing injection anxiety and enabling primary care prescribing without specialized training. The FDA approval of the first oral GLP-1 for weight loss in December 2025-Novo Nordisk's once-daily semaglutide pill-represents a watershed moment for the category [3]. Analysts project significant prescription uptake for this oral formulation in early 2026, signifying strong adoption for the new treatment modality [4].
Next-generation oral candidates like orforglipron promise improved bioavailability and dosing flexibility without requiring special storage conditions or meal timing [2]. The convenience differential between weekly injectables and daily oral formulations creates distinct patient preference segments: some patients prioritize dosing frequency, others prefer avoiding injections entirely. Manufacturers are therefore maintaining dual formulation strategies to capture both segments while extending product life cycles through delivery innovation.
Regional Analysis
The GLP-1 receptor agonist market is segmented geographically across North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa. Growth dynamics differ based on regulatory approval timelines, reimbursement frameworks, obesity prevalence, and healthcare infrastructure development.
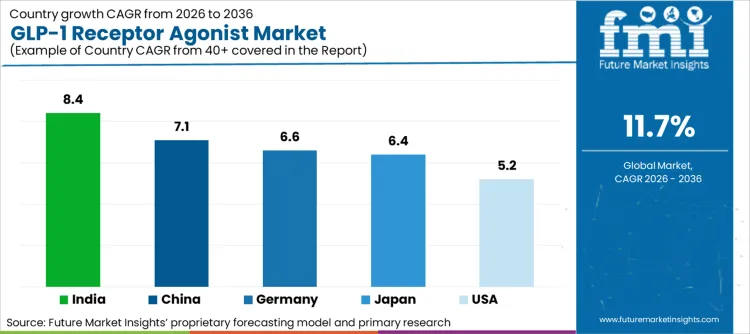
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 8.4% |
| China | 7.1% |
| Germany | 6.6% |
| Japan | 6.4% |
| United States | 5.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Market Analysis
North America functions as the commercial epicenter of the global GLP-1 market, characterized by high prescribing rates, direct-to-consumer advertising, and concentrated commercial infrastructure. Major players including Novo Nordisk and Eli Lilly maintain substantial commercial footprints, competing aggressively on efficacy claims, patient support programs, and contracting strategies.
United States
Demand for GLP-1 receptor agonists in the United States is forecasted to advance at 5.2% CAGR through 2036. The market is shaped by high obesity prevalence-over 100 million adults, coupled with reimbursement complexity across Medicare, Medicaid, and commercial plans [1]. According to recent data, GLP-1 prescriptions have penetrated beyond specialist prescribing into primary care. Unlike emerging markets where growth is volume-led, USA growth is driven by indication expansion into cardiovascular risk reduction and intensifying competition between semaglutide and tirzepatide.
Recent developments include CMS unveiling its BALANCE model, a voluntary Medicare and Medicaid coverage pathway that includes price negotiations and supplemental lifestyle support, with coverage beginning in 2026 for Medicaid and 2027 for Medicare Part D [2]. Pricing pressures have surfaced across payer earnings, with some insurers expanding access with guardrails while others have dropped coverage entirely [3].
FMI's analysis of the GLP-1 receptor agonist market in North America consists of country-wise assessment including the United States and Canada. Readers can find reimbursement policy tracking, competitive intelligence, and pipeline monitoring insights.
Europe Market Analysis
Europe acts as the regulatory-intensive adoption zone, characterized by health technology assessment requirements, national reimbursement negotiations, and guideline-driven prescribing patterns. Regional players align closely with EMA frameworks and country-specific access pathways.
Germany
Demand for GLP-1 receptor agonists in Germany is projected to rise at 6.6% CAGR through 2036. Growth is supported by Germany's early adoption of cardiovascular outcome data into treatment guidelines and the country's structured reimbursement pathways for innovative therapies [1]. Unlike markets where direct-to-consumer advertising drives demand, German growth is guideline-aligned and outcomes-dependent, with cardiometabolic evidence packages determining formulary positioning.
FMI's analysis of the GLP-1 receptor agonist market in Europe consists of country-wise assessment including Germany, United Kingdom, France, Italy, and Spain. Readers can find HTA outcome tracking, reimbursement landscape analysis, and prescribing pattern insights.
Asia-Pacific Market Analysis
Asia-Pacific represents the high-growth frontier, driven by rising diabetes prevalence, healthcare infrastructure expansion, and increasing obesity awareness. Multinational manufacturers are establishing commercial presence while domestic players advance biosimilar and next-generation programs.
China
Demand for GLP-1 receptor agonists in China is projected to rise at 7.1% CAGR through 2036. The country’s massive diabetes population supports growth, healthcare reform initiatives improving access to innovative therapies, and increasing obesity awareness among urban populations [2]. Recent developments include domestic players advancing next-generation molecules with potential regional market advantages.
FMI's analysis of the GLP-1 receptor agonist market in Asia-Pacific consists of country-wise assessment including China, India, Japan, and South Korea. Readers can find regulatory pathway tracking, domestic competitor intelligence, and access expansion modeling.
India
Demand for GLP-1 receptor agonists in India is projected to rise at 8.4% CAGR through 2036. Growth is supported by rising diabetes prevalence-over 100 million affected-increasing obesity awareness in urban populations, and expanding health insurance coverage for chronic disease management [1]. India's growth is driven by multinational product penetration and increasing affordability through tiered pricing strategies. Government initiatives strengthening the pharmaceutical manufacturing sector support local production capabilities for complex biologics.
Japan
Demand for GLP-1 receptor agonists in Japan is projected to rise at 6.4% CAGR through 2036. The country’s aging population, high diabetes prevalence among older adults, and strong regulatory pathways for innovative therapies [3], influences growth. Japan's national health insurance system provides broad access while premium pricing rewards demonstrated clinical value. The market's focus on oral formulations aligns with patient preference for non-injectable administration in older populations.
Competitive Landscape for Market Players
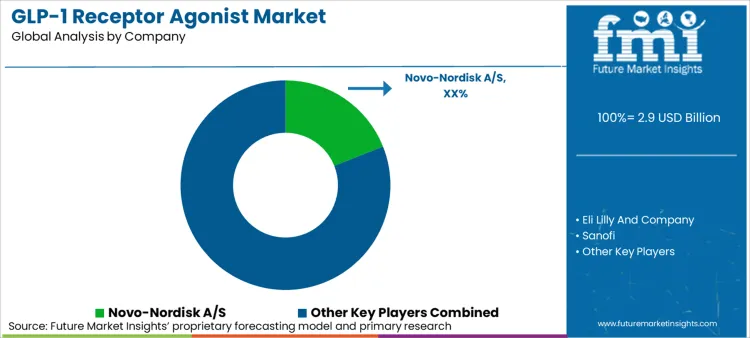
The GLP-1 receptor agonist market is characterized by a concentrated competitive landscape dominated by Novo Nordisk and Eli Lilly, with emerging challengers advancing next-generation molecules. As per FMI estimates, these two leaders collectively control a significant majority of market value, particularly in premium diabetes and obesity segments. The primary competitive variable is not pricing but clinical differentiation through head-to-head efficacy comparisons and outcomes trial results.
Novo Nordisk maintains first-to-market advantage with semaglutide across diabetes (Ozempic), obesity (Wegovy), and oral (Rybelsus) formulations, with the oral version launching in January 2026 following December 2025 FDA approval [3]. However, the company faces intensifying competition from Eli Lilly's tirzepatide (Mounjaro, Zepbound), which has demonstrated superior weight loss efficacy, reshaping prescriber preferences in obesity management [2].
Emerging competitors are advancing differentiated mechanisms beyond single GLP-1 agonism. Pfizer has struck a $2 billion deal with YaoPharma to develop and commercialize a phase 1 small-molecule GLP-1 receptor agonist for chronic weight management [1]. Amgen is advancing novel candidates, while Chinese innovators are developing next-generation molecules with potential regional market advantages.
Manufacturing capacity has emerged as a critical competitive differentiator. Eli Lilly has amassed a USD 1.5 billion pre-launch inventory of its oral weight loss drug orforglipron ahead of FDA decision, representing a decisive effort to avoid repeating supply constraints [4]. Lilly is also constructing new manufacturing facilities as part of a USD 27 billion investment drive, with at least three confirmed as production hubs for weight loss therapies [3].
Buyer behavior in this market reflects concentrated purchasing power through pharmacy benefit managers and national health systems. Formulary placement decisions increasingly depend on cardiovascular outcomes data, weight loss efficacy, and total cost of care calculations accounting for event reduction. Manufacturers therefore compete on evidence generation as much as commercial execution.
Recent Developments:
- Roche - Acquisition (December 2023): Roche acquired Carmot Therapeutics, adding dual GLP-1/GIP agonist assets to its pipeline, positioning to compete with Novo Nordisk and Eli Lilly in obesity [1].
- Novo Nordisk - Regulatory Approval (December 2025): FDA approved the first oral GLP-1 for weight loss-Novo Nordisk's once-daily semaglutide pill-which launched in January 2026 [3].
- Eli Lilly - Manufacturing Investment (February 2026): Eli Lilly reported $1.5 billion pre-launch inventory for orforglipron and announced new manufacturing facilities as part of $27 billion investment drive [4].
- Pfizer - Partnership (December 2025): Pfizer struck a $2 billion deal with YaoPharma to develop and commercialize a phase 1 small-molecule GLP-1 receptor agonist [1].
Key Players in the GLP-1 Receptor Agonist Market
- Novo-Nordisk A/S
- Eli Lilly And Company
- Sanofi
- AstraZeneca
- Pfizer Inc.
- Amgen, Inc.
- Innovent Biologics, Inc.
- PegBio Co., Ltd
- Sun Pharmaceutical Industries Ltd.
- Boehringer Ingelheim International GmbH
Scope of Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 2.9 billion (2026) to USD 7.8 billion (2036), at a CAGR of 11.7% |
| Market Definition | The GLP-1 receptor agonist market comprises the global development and supply of pharmaceutical agents mimicking glucagon-like peptide-1 for diabetes, obesity, and cardiovascular risk reduction. |
| Drug Segmentation | Semaglutide, Exenatide, Lixisenatide, Liraglutide, Dulaglutide, Others |
| Route of Administration Segmentation | Oral, Parenteral |
| Indication Segmentation | Diabetes, Obesity, Others |
| Distribution Channel Segmentation | Hospital Pharmacies, Retail Pharmacies, Online Pharmacies |
| Region Segmentation | North America, Latin America, Europe, Asia-Pacific, Middle East & Africa |
| Key Countries Covered | United States, Canada, Germany, United Kingdom, France, Italy, Spain, China, India, Japan, South Korea, and 40+ countries |
| Key Companies Profiled | Novo-Nordisk A/S, Eli Lilly And Company, Sanofi, AstraZeneca, Pfizer Inc., Amgen, Inc., Innovent Biologics, Inc., PegBio Co., Ltd, Sun Pharmaceutical Industries Ltd., Boehringer Ingelheim International GmbH |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling validated through primary interviews with endocrinologists, cardiologists, and payer formulary managers, supported by prescription audit data and clinical trial outcomes tracking |
GLP-1 Receptor Agonist Market Analysis by Segments
Drug:
- Semaglutide
- Exenatide
- Lixisenatide
- Liraglutide
- Dulaglutide
- Others
Route of Administration:
- Oral
- Parenteral
Indication:
- Diabetes
- Obesity
- Others
Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
Region:
- North America
- Latin America
- Europe
- Asia-Pacific
- Middle East & Africa
Bibliography
- Pharmaceutical Executive. (2026, January 8). Lilly's Orforglipron, Retatrutide Named as Defining GLP-1 Drugs of the Next Decade.
- Becker's ASC. (2026, January 27). What's new with GLP-1s?
- Drug Discovery World. (2026, February 2). Obesity treatment trends in 2026 and beyond. DDW Online.
- Pharmaceutical Technology. (2026, February 16). Eli Lilly builds orforglipron cache to avoid previous GLP-1RA shortages.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This Report Addresses
- Market intelligence to enable structured strategic decision-making across pharmaceutical manufacturers, contract research organizations, and healthcare investor portfolios.
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, including validated CAGR and absolute dollar opportunity sizing.
- Growth opportunity mapping across Drug, Indication, Route of Administration, and regional demand pockets, with emphasis on obesity indication expansion and oral formulation adoption.
- Segment and regional forecasting covering semaglutide, exenatide, and next-generation molecules, alongside indication-specific demand trajectories across diabetes, obesity, and cardiovascular risk reduction.
- Competition strategy assessment, including clinical trial investment, manufacturing capacity expansion, and payer negotiation dynamics shaping formulary placement.
- Product and regulatory tracking focused on FDA and EMA approvals, cardiovascular outcomes trial readouts, and combination product development for weight management.
- Reimbursement policy analysis covering Medicare price negotiation impacts, commercial payer coverage determinations, and international HTA framework evolution influencing market access.
- Report delivery in PDF, Excel, and presentation-ready formats to support executive planning, pipeline prioritization, and competitive benchmarking.
Frequently Asked Questions
How large is the demand for GLP-1 receptor agonists in the global market in 2026?
Demand for GLP-1 receptor agonists is estimated to be valued at USD 2.9 billion in 2026.
What will be the market size of GLP-1 receptor agonists in the global market by 2036?
Market size for GLP-1 receptor agonists is projected to reach USD 7.8 billion by 2036.
What is the expected demand growth for GLP-1 receptor agonists between 2026 and 2036?
Demand for GLP-1 receptor agonists is expected to grow at an 11.7% CAGR between 2026 and 2036.
Which drug segment is poised to lead global sales?
Exenatide is estimated to lead the drug segment, holding 24.4% share in 2025, reflecting its first-to-market advantage and established clinical profile in certain markets.
How significant is the role of obesity indication in driving market expansion?
Obesity is the fastest-growing indication segment, driven by high-efficacy weight loss data, expanded regulatory approvals, and direct-to-consumer demand [1].
What is driving demand in the United States?
High obesity prevalence, expanding cardiovascular indications, recent oral formulation approvals, and intensifying competition between semaglutide and tirzepatide are supporting U.S. demand growth [2].
What is the United States growth outlook in this report?
The United States is projected to expand at a 5.2% CAGR during 2026 to 2036.
Why is Germany described as a key market in Europe?
Germany's growth is supported by early adoption of cardiovascular outcomes data into treatment guidelines and structured reimbursement pathways for innovative therapies [1].
What is the Germany growth outlook in this report?
Germany is projected to expand at a 6.6% CAGR during 2026 to 2036.
Does the report cover China in its regional analysis?
Yes, China is included within Asia-Pacific and is a high-growth country market covered in the report.
What is the China growth outlook in this report?
China is projected to expand at a 7.1% CAGR during 2026 to 2036.
Does the report cover India in its regional analysis?
Yes, India is included within Asia-Pacific and is covered in the country-wise assessment.
What is the India growth outlook in this report?
India is projected to expand at an 8.4% CAGR during 2026 to 2036.
Does the report cover Japan in its regional analysis?
Yes, Japan is included within Asia-Pacific under the regional scope of analysis.
What is the Japan growth outlook in this report?
Japan is projected to expand at a 6.4% CAGR during 2026 to 2036.
What defines a GLP-1 receptor agonist as part of this market?
A product is included if it is an FDA or EMA-approved prescription GLP-1 receptor agonist therapy indicated for type 2 diabetes, obesity, or cardiovascular risk reduction.
What is included in the scope of this GLP-1 receptor agonist market report?
Scope includes market sizing and forecasting for 2026 to 2036, segmentation by drug, indication, route of administration, distribution channel, and region, along with competitive assessment and pipeline analysis.
What is excluded from the scope of this report?
The scope excludes over-the-counter supplements, compounded drug products, delivery device revenues separable from pharmaceutical value, and biosimilars not yet approved in major regulated markets.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Drug
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Drug , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Drug , 2026 to 2036
- Exenatide
- Semaglutide
- Lixisenatide
- Liraglutide
- Exenatide
- Y to o to Y Growth Trend Analysis By Drug , 2021 to 2025
- Absolute $ Opportunity Analysis By Drug , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Drug
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Drug
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Drug
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Drug
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Drug
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Drug
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Drug
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Drug
- Competition Analysis
- Competition Deep Dive
- Novo-Nordisk A/S
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Eli Lilly And Company
- Sanofi
- AstraZeneca
- Pfizer Inc.
- Amgen, Inc.
- Novo-Nordisk A/S
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Drug , 2021 to 2036
- Table 3: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Drug , 2021 to 2036
- Table 5: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: Latin America Market Value (USD Million) Forecast by Drug , 2021 to 2036
- Table 7: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Western Europe Market Value (USD Million) Forecast by Drug , 2021 to 2036
- Table 9: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Eastern Europe Market Value (USD Million) Forecast by Drug , 2021 to 2036
- Table 11: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: East Asia Market Value (USD Million) Forecast by Drug , 2021 to 2036
- Table 13: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: South Asia and Pacific Market Value (USD Million) Forecast by Drug , 2021 to 2036
- Table 15: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Middle East & Africa Market Value (USD Million) Forecast by Drug , 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Drug , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Drug , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Drug
- Figure 6: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Region
- Figure 9: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 10: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 11: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 12: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 17: North America Market Value Share and BPS Analysis by Drug , 2026 and 2036
- Figure 18: North America Market Y-o-Y Growth Comparison by Drug , 2026-2036
- Figure 19: North America Market Attractiveness Analysis by Drug
- Figure 20: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 21: Latin America Market Value Share and BPS Analysis by Drug , 2026 and 2036
- Figure 22: Latin America Market Y-o-Y Growth Comparison by Drug , 2026-2036
- Figure 23: Latin America Market Attractiveness Analysis by Drug
- Figure 24: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 25: Western Europe Market Value Share and BPS Analysis by Drug , 2026 and 2036
- Figure 26: Western Europe Market Y-o-Y Growth Comparison by Drug , 2026-2036
- Figure 27: Western Europe Market Attractiveness Analysis by Drug
- Figure 28: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: Eastern Europe Market Value Share and BPS Analysis by Drug , 2026 and 2036
- Figure 30: Eastern Europe Market Y-o-Y Growth Comparison by Drug , 2026-2036
- Figure 31: Eastern Europe Market Attractiveness Analysis by Drug
- Figure 32: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: East Asia Market Value Share and BPS Analysis by Drug , 2026 and 2036
- Figure 34: East Asia Market Y-o-Y Growth Comparison by Drug , 2026-2036
- Figure 35: East Asia Market Attractiveness Analysis by Drug
- Figure 36: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 37: South Asia and Pacific Market Value Share and BPS Analysis by Drug , 2026 and 2036
- Figure 38: South Asia and Pacific Market Y-o-Y Growth Comparison by Drug , 2026-2036
- Figure 39: South Asia and Pacific Market Attractiveness Analysis by Drug
- Figure 40: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Middle East & Africa Market Value Share and BPS Analysis by Drug , 2026 and 2036
- Figure 42: Middle East & Africa Market Y-o-Y Growth Comparison by Drug , 2026-2036
- Figure 43: Middle East & Africa Market Attractiveness Analysis by Drug
- Figure 44: Global Market - Tier Structure Analysis
- Figure 45: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

