Resveratrol Market
The resveratrol market is segmented by Form (Powder, Liquid), Product Claim (Natural, Synthetic), End Use Application (Nutraceutical/Dietary Supplements, Personal Care Cosmetics, Functional Food & Beverages, Animal Feed, Pharmaceutical, Pet Food), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Resveratrol Market Size, Market Forecast and Outlook By FMI
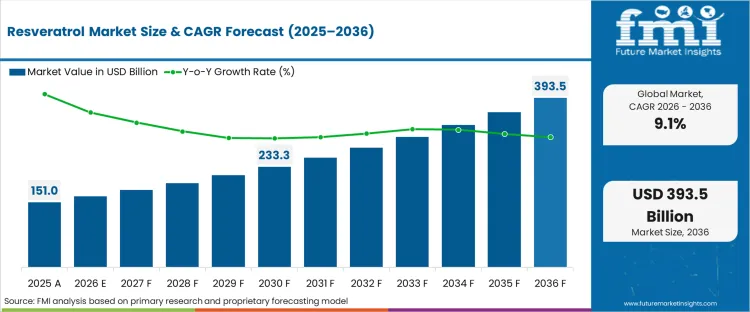
The resveratrol market surpassed a value of USD 151.8 billion in 2025. The industry is expected to reach USD 164.7 billion in 2026 at a CAGR of 9.1% during the forecast period. Demand outlook carries the market valuation to USD 394.0 billion by 2036 as advancements in encapsulation technologies improve bioavailability and stability of stilbenoid compounds across nutraceutical and pharmaceutical applications.
Shelf-stability realities force procurement directors to rewrite their entire qualification criteria for polyphenols. Purchasing managers previously evaluated pure compound cost based on extraction origin. They now demand comprehensive pharmacokinetic profiles proving gastrointestinal survival. Delaying the transition to lipid-based delivery systems within herbal supplements guarantees immediate delisting from major pharmacy chains. Retailers require verified absorption data before granting shelf space. Analyzing the resveratrol demand outlook shows that efficacy claims mean nothing without structural protection against oxidation.
Standardizing lipid-based microencapsulation across contract manufacturing lines acts as the critical gate for this sector. Once major production facilities integrate these specialized matrices at scale, raw material purchasing shifts away from simple price-per-kilo metrics. Buyers prioritize actual absorption-yield ratios instead. Passing this threshold establishes a new baseline for the resveratrol B2B supply chain. Competition shifts entirely toward cellular delivery guarantees.
Summary of Resveratrol Market
- Resveratrol Market Definition
- Polyphenolic phytoalexins synthesized by specific plant species function as core commercial bioactives. Trans-resveratrol requires precise stabilization processing to survive human metabolic pathways.
- Demand Drivers in the Market
- Clinical validation requirements compel formulation scientists to adopt advanced liposomal delivery systems.
- Quality degradation risks force procurement directors to demand guaranteed stability data prior to raw material purchase.
- Regulatory scrutiny pushes compliance officers to mandate verified synthetic fermentation pathways over vulnerable agricultural sourcing.
- Key Segments Analyzed in the FMI Report
- Form: Powder is expected to account for 92.1% share in 2026, driven by superior shelf-life stability during extended warehousing cycles.
- Product Claim: Natural is projected to capture 86.2% share in 2026, reflecting consumer resistance toward synthetic lab-grown analogues.
- End Use Application: Nutraceutical/Dietary Supplements are anticipated to hold 71.9% share in 2026, sustained by high-dosage clinical protocols demanding concentrated inputs.
- India: 9.8% compound growth, sustained by rapid domestic scale-up of knotweed extraction infrastructure.
- Analyst Opinion at FMI
- Nandini Roy Choudhury, Principal Analyst for Food and Beverages, at FMI, points out, "Standard metrics capture raw extraction volumes accurately. They miss the massive volume of active compounds destroyed during standard encapsulation entirely. The true constraint determining the resveratrol market growth trajectory centers on cellular delivery survival. Buyers prioritizing raw material price over stabilization architecture lose their entire investment to gastric degradation. Supply chain value rests almost entirely on lipid matrices rather than botanical purity."
- Strategic Implications / Executive Takeaways
- Formulation scientists face immediate pressure to pivot from standard capsules to liposomal formats.
- Procurement directors must secure locked pricing on knotweed extracts before regional agricultural disruptions spike raw material costs.
- Contract manufacturers scaling synthetic fermentation pathways capture significant margin advantage over agricultural extraction competitors.
- Methodology
- Primary Research: Direct input from formulation scientists establishes ground-level usage realities.
- Desk Research: Clinical trial registries confirm moving baseline efficacy standards.
- Market-Sizing and Forecasting: Extraction volumes mapped against yield ratios dictate core sizing parameters.
- Data Validation and Update Cycle: Import logs provide cross-validation against domestic consumption patterns.
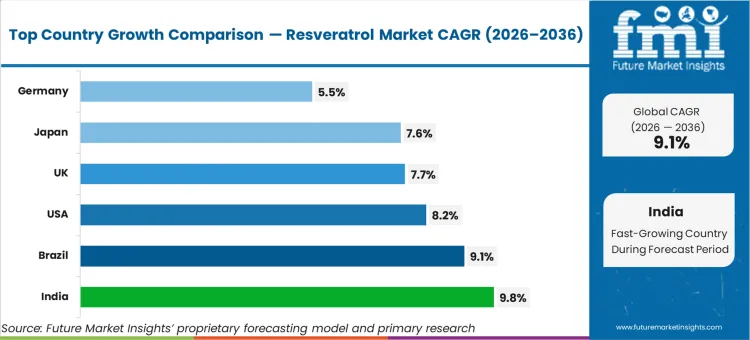
Reviewing the resveratrol regional outlook, India leads global expansion at 9.8% compound growth as domestic extraction facilities scale capacity rapidly. Brazil tracks closely at 9.1% driven by expanding agricultural processing infrastructure targeting export channels. Demand in America expands at 8.2% on stringent clinical validation protocols dictating formulation choices. Japan advances at 7.6% alongside United Kingdom at 7.7%. Germany maintains steady 5.5% progression as strict European safety authorities dictate permissible dosage limits.
Resveratrol Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 164.7 billion |
| Industry Value (2036) | USD 394.0 billion |
| CAGR (2026 to 2036) | 9.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Supply Chain Analysis in the Resveratrol Market
The resveratrol supply chain is built around a relatively concentrated processing core that sits between fragmented agricultural sourcing and diversified downstream demand. At the front end, fruits and knotweed serve as the main feedstocks, supplied by raw material producers and farmers to processing companies for extraction and moisture removal. Value creation is concentrated in the manufacturing stage, where resveratrol plants convert botanical inputs into usable ingredients and then channel output to a broad set of end-use manufacturers, including dietary supplement, cosmetic, and personal care companies. From there, the market splits into two parallel routes: one serving domestic retail networks and another feeding export markets before products reach retail units and final consumers. This structure shows that competitive strength in resveratrol depends less on raw material ownership alone and more on processing efficiency, ingredient standardization, and the ability to maintain reliable links with downstream formulation industries and retail channels.
Resveratrol Market Definition
Polyphenolic phytoalexins synthesized naturally by plants under pathogenic stress form the foundation of this commercial category. Trans-resveratrol functions as the primary bioactive isomer targeted for commercial extraction. This compound interacts specifically with human sirtuin pathways. Isolates derived from grape seed extracts require intense stabilization processing before commercial deployment. Tracking the broader resveratrol market requires understanding these structural vulnerabilities.
Resveratrol Market Inclusions
Standardized resveratrol derivatives from Japanese knotweed root and grape skins fall within precise analytical boundaries. Synthetic analogues engineered via yeast fermentation protocols qualify for comprehensive evaluation. Commercial formulations featuring verified trans-isomer concentrations above baseline threshold requirements represent core tracked volume. Breaking down the resveratrol market by application confirms these specific purity standards across end uses.
Resveratrol Market Exclusions
Raw unprocessed agricultural commodities lack necessary concentration metrics for inclusion. Finished consumer beverages containing trace incidental polyphenols do not qualify for tracking. Crude botanical extracts without verified trans-resveratrol standardization fail to meet minimum qualification parameters.
Resveratrol Market Research Methodology
- Primary Research: Formulation scientists and clinical researchers navigating supply chain constraints provide direct qualification metrics.
- Desk Research: Clinical trial registries and FDA GRAS notification databases supply baseline safety validation data.
- Market-Sizing and Forecasting: Verified bulk extraction volumes tracked against commercial yield ratios establish initial valuation baselines.
- Data Validation and Update Cycle: Import-export customs logs cross-referenced with synthetic fermentation batch records validate ongoing resveratrol market forecast modeling.
Segmental Analysis
Resveratrol Market Analysis by Form
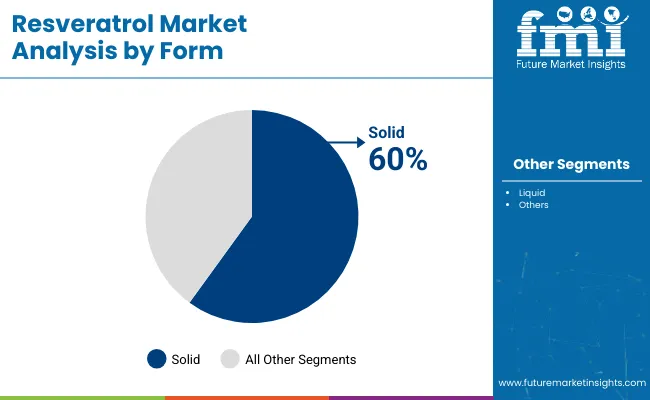
Ambient humidity inside manufacturing facilities dictates bulk material handling protocols globally. Powder formats hold 92.1% share, and FMI's analysis indicates production managers depend entirely on granular stability to survive unpredictable international shipping delays. Liquid formats present severe oxidation risks. They require complex nitrogen-flushed packaging systems that erode commercial margins. Granular inputs flow seamlessly through high-speed encapsulation equipment. Operations managers avoid mechanical blockages common with viscous liquids. What procurement directors rarely consider is how varying particle sizes within powder batches drastically alter dissolution rates post-consumption. Facilities relying on generic flavonoid blends or crude grape skin extract face catastrophic batch failures when hygroscopic clumping forces mid-run equipment shutdowns. This dynamic shapes the broader resveratrol powder market landscape.
- Handling Stability: Granular physical structures resist ambient humidity during open-air processing cycles. Production managers avoid costly environmental control upgrades by prioritizing dry inputs. They guarantee smooth operation across varied climate zones.
- Flow Dynamics: Uniform particle distributions prevent hopper bridging during high-speed capsule filling operations. Equipment operators maintain optimal throughput velocity without manual intervention. Falling below required mesh specifications causes immediate production delays.
- Dissolution Variability: Particle size inconsistency alters gastric release timing unpredictably. Formulation scientists must enforce strict specifications to pass disintegration testing protocols. Failing these tests results in immediate product recalls.
Resveratrol Market Analysis by Product Claim
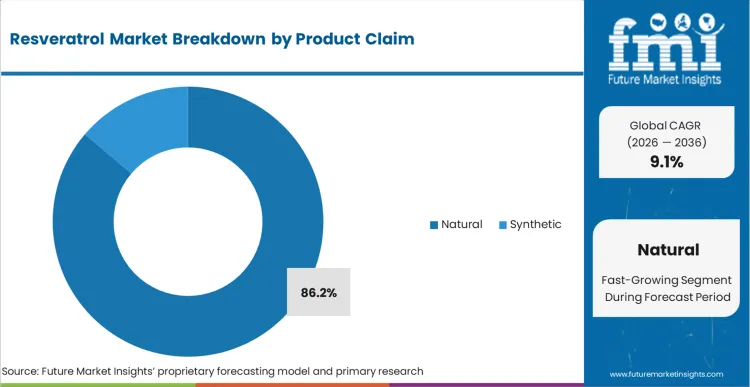
Natural and synthetic sourcing continue to create a clear tension between consumer preference and manufacturing efficiency. This demand pattern places natural variants at an 86.2% share of the market. FMI assessment suggests that marketing teams often continue to favor natural positioning even when synthetic alternatives offer the same molecular structure and stronger purity consistency. Fermentation-derived versions can perform well on technical metrics, though label transparency rules may reduce consumer acceptance when synthetic wording appears on pack. Japanese knotweed extraction remains a practical route for maintaining natural positioning with workable unit economics. At the same time, sourcing teams must manage seasonal harvest variability in natural antioxidants, while synthetic production avoids that constraint. This tension continues to shape strategy across the natural resveratrol market.
- Label Positioning: Agricultural extraction allows clean-label declarations that consumers demand. Marketing directors leverage botanical origins to justify premium retail pricing tiers. Moving away from these claims destroys brand equity instantly.
- Yield Constraints: Natural knotweed processing generates massive botanical waste volumes requiring costly disposal. Operations managers face escalating facility overhead as environmental regulations tighten. Waste management costs erode agricultural margin advantages significantly.
- Purity Limits: Plant-derived batches contain variable secondary compounds complicating standardization efforts. Quality control technicians reject entire shipments when trace contaminants exceed allowable maximums. These rejections disrupt entire quarterly production schedules.
Resveratrol Market Analysis by End Use Application
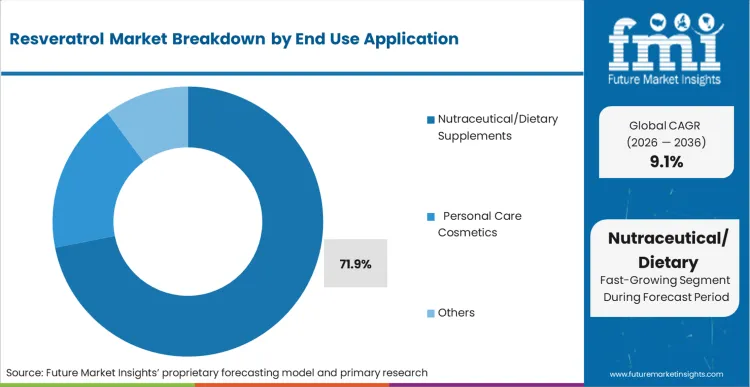
Therapeutic efficacy usually requires more concentrated delivery than food-based applications can provide. This demand pattern gives Nutraceutical and Dietary Supplements a 71.9% share of the market. In this segment, clinical practitioners place greater importance on verified milligrams per serving than on trace-level functional food inclusion. Efficacy thresholds demand dedicated healthy aging supplement formats designed specifically for intestinal absorption. Integrating bioactives into functional beverages destroys molecular integrity through prolonged liquid suspension. Surface-level analysis misses how nutraceutical ingredients deployed in skin health supplement ranges require completely different stabilizing co-factors than cardiovascular formulations. Brand owners pursuing botanical supplements risk severe regulatory penalties if label claims exceed clinically verified absorption data. These dynamics heavily influence the resveratrol supplement market trajectory.
- Dosage Density: Hard-shell capsules accommodate massive active payloads impossible to mask in functional beverages. Formulation scientists achieve therapeutic thresholds without compromising consumer sensory experience. Trying to disguise these dosages in food formats causes severe taste rejection.
- Suspension Failure: Aqueous environments trigger rapid polyphenol degradation before expiration dates approach. Beverage formulators abandon projects when active compound half-lives fall below acceptable distribution timelines. Liquid stability remains a critical limiting factor.
- Absorption Targeting: Specialized enteric coatings protect actives from harsh gastric acids. Product developers ensure survival until compounds reach optimal intestinal absorption zones. Unprotected formulations lose active potency entirely during early digestion stages.
Resveratrol Market Drivers, Restraints, and Opportunities

Strict clinical validation protocols force formulation scientists to abandon cheap unstandardized extracts. Premium pharmacy chains now demand third-party assay results proving trans-resveratrol content before approving shelf space. Securing these vital retail channels requires massive upgrades in raw material sourcing quality. Failing to adopt verified botanical ingredients leaves major brands vulnerable to consumer watchdog litigation over false efficacy claims. This pressure reshapes resveratrol market trends significantly. Ingredient buyers face immense pressure to secure stable pricing before regulatory tightening restricts available supply networks.
Gastric degradation presents a massive operational friction slowing therapeutic adoption rates. Unprotected polyphenols face near-total destruction during digestion before reaching cellular targets. Liposomal encapsulation provides partial protection. It introduces significant manufacturing complexity and cost burdens. Operations managers struggle to balance these enhanced delivery costs against rigid consumer price ceilings. Evaluating the resveratrol market analysis highlights this specific technical hurdle holding back broader mass-market penetration.
Opportunities in the Resveratrol Market
- Liposomal Integration: Advanced delivery matrices shield active compounds from digestive destruction. Formulation scientists capture premium pricing by guaranteeing verifiable cellular absorption rates.
- Fermentation Scaling: Synthetic yeast pathways eliminate agricultural supply chain vulnerabilities entirely. Production managers secure predictable pricing untethered from unpredictable harvest yields.
- Topical Stabilization: Integrating botanical bioactives into antioxidant skincare demands novel microencapsulation. Cosmetic chemists unlock lucrative anti-aging channels by solving dermal penetration challenges.
Regional Analysis
Based on regional analysis, resveratrol market is segmented into Americas, Asia Pacific, and Europe across 40+ countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| USA | 8.2% |
| India | 9.8% |
| Brazil | 9.1% |
| Japan | 7.6% |
| UK | 7.7% |
| Germany | 5.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Americas Resveratrol Market Analysis
Stringent clinical validation protocols dictate North American commercial environments. Major retail pharmacies now reject supplements lacking independent third-party assay verification. Buyers across this region prioritize verified trans-isomer concentrations over basic botanical origin claims. South American processing facilities increasingly target premium export standards rather than domestic consumption. FMI analysts note that rigorous testing mandates separate sophisticated contract manufacturers from basic agricultural processors. Tracking the United States resveratrol market reveals a definitive shift toward premium verified sourcing.
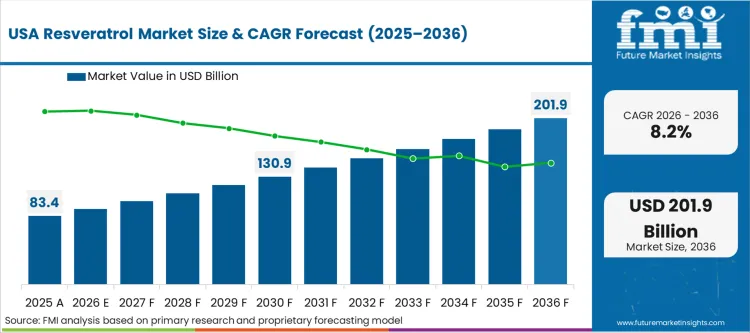
- USA: In the United States, strict FDA scrutiny around novel structure-function claims is pushing brand owners toward more rigorous clinical testing pathways. Formulators rely on verified materials to reduce the risk of costly warning letters. Demand in the country is expected to grow at a CAGR of 8.2%. GRAS approval for newer synthetic variants can also support entry into food-additive applications that are still largely served by natural extracts.
- Brazil: In Brazil, expanding agricultural infrastructure is supporting larger-scale domestic extraction capacity. Processing teams are using local biomass availability to improve pricing competitiveness against Asian suppliers. The market is forecast to grow at a CAGR of 9.1%. Continued facility modernization is also helping local producers strengthen their position in premium export contracts that have traditionally been served by established European companies.
FMI's report includes Canada and Mexico. North American buyers consistently demand comprehensive pharmacokinetic data accompanying every bulk material purchase.
Asia Pacific Resveratrol Market Analysis
Domestic extraction scale-up initiatives transform Asian supply chain dynamics fundamentally. Regional producers leverage massive knotweed harvests to dominate global bulk material export volumes. Sophisticated buyers navigate complex supplier networks to verify actual trans-resveratrol concentrations against declared label values. Mature aging populations drive intense demand for clinically validated formulations. In FMI's view, aggressive Japan innovation pushes delivery system technology forward rapidly. This momentum heavily influences China resveratrol market export strategies.
- India: Investment in extraction capacity is changing the role of local suppliers. Many are moving beyond raw biomass exports and building capabilities in refined isolates, which is improving access to high-purity material for downstream buyers. Revenue is projected to grow at a 9.8% CAGR. European export opportunities are adding another layer of pressure, especially around solvent recovery standards, where faster upgrades can make the difference in qualifying for higher-value contracts.
- Japan: Demand in Japan is being shaped by an older consumer base that is more focused on scientifically supported healthy aging products. That is pushing developers toward water-soluble formats that fit easily into everyday beverages and routine consumption. Sales are anticipated to rise at a 7.7% CAGR. The market is also staying important from a formulation standpoint, particularly in lipid-based bioavailability enhancement, where Japanese players continue to influence product development.
FMI's report includes China and Australia. Asian facilities increasingly adopt European quality standards to secure lucrative international export agreements.
Europe Resveratrol Market Analysis
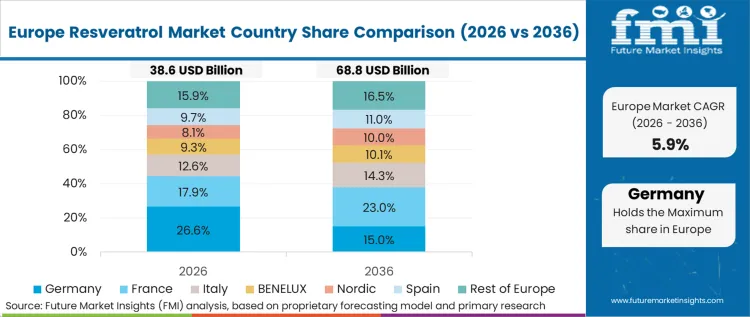
Strict European Food Safety Authority dosage limits strictly govern regional product formulations. Compliance officers reject high-dose imported supplements exceeding legally permissible daily intake thresholds. Regional formulators focus entirely on maximizing bioavailability from lower absolute dosages to maintain clinical efficacy within legal bounds. Western Europe dictates global safety standards. According to FMI's estimates, demand in EU strongly favors sustainable synthetic fermentation over ecologically taxing agricultural extraction. Tracking the Germany resveratrol market confirms this regulatory influence.
- UK: Post-Brexit regulatory independence allows slight deviations from strict continental dosage maximums. Brand owners exploit this flexibility to launch higher-potency variants targeting serious biohackers. Demand is set to grow at 7.7% CAGR. Securing domestic contract manufacturing capacity shields brands from unpredictable cross-channel import delays.
- Germany: Relentless EFSA oversight restricts permissible claims regarding cardiovascular or anti-aging benefits. Marketing directors must utilize approved generic health claims rather than specific therapeutic promises. This sector progresses at 5.5% CAGR. Success requires flawless documentation tracking raw material origins straight back to initial harvest locations.
FMI's report includes France and Italy. European buyers prioritize verified sustainable sourcing methodologies alongside stringent molecular purity requirements.
Competitive Aligners for Market Players
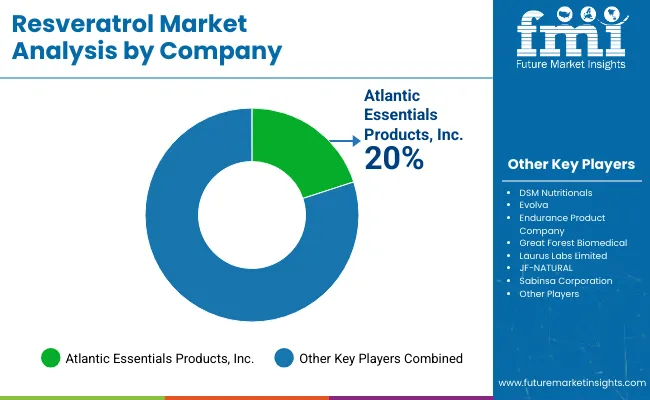
In the Resveratrol Market, competition is shaped primarily by the strength of ingredient standardization, bioavailability positioning, formulation support, and channel reach. dsm-firmenich holds a strong position through resVida®, a high-potency trans-resveratrol platform backed by global nutrition infrastructure, while Sabinsa Corporation competes through its science-led botanical portfolio, including Resvenox®, with a stronger emphasis on branded extract credibility. Maypro is differentiated less by upstream scale and more by specialty distribution and delivery-format value, particularly through MicroActive® Resveratrol, which is positioned around sustained release and absorption support. Layn Natural Ingredients adds pressure through botanical extraction depth and commercial offerings based on Polygonum cuspidatum, including enhanced-bioavailability positioning for resveratrol ingredients. Taken together, the market is competitive but not flat: leadership depends less on commodity supply and more on branded science, formulation performance, and the ability to serve supplement manufacturers with reliable quality, technical documentation, and application-specific support.
Key Players in Resveratrol Market
- dsm-firmenich
- Sabinsa Corporation
- Maypro
- Layn Natural Ingredients
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 164.7 billion to USD 394.0 billion, at a CAGR of 9.1% |
| Market Definition | Polyphenolic phytoalexins synthesized naturally form core commercial bioactives requiring precise stabilization processing. |
| Segmentation | Form, Product Claim, End Use Application |
| Regions Covered | North America, Latin America, Europe, Asia Pacific, Middle East and Africa |
| Countries Covered | U.S., India, Brazil, Japan, UK, Germany |
| Key Companies Profiled | dsm-firmenich, Sabinsa Corporation, Maypro, Layn Natural Ingredients |
| Forecast Period | 2026 to 2036 |
| Approach | Verified bulk extraction volumes tracked against commercial yield ratios establish initial valuation baselines. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Resveratrol Market Analysis by Segments
Form
- Powder
- Liquid
Product Claim
- Natural
- Synthetic
End Use Application
- Nutraceutical/Dietary Supplements
- Personal Care Cosmetics
- Functional Food & Beverages
- Animal Feed
- Pharmaceutical
- Pet Food
Region:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- Brown, K., Wong, J., Rorato, R. C., & Liu, Y. (2024). Resveratrol for the Management of Human Health: How Far Have We Come? A Systematic Review of Resveratrol Clinical Trials to Highlight Gaps and Opportunities.
- Molani-Gol, R., Bagheri, A., Askari, M., Barati-Boldaji, R., Farsi, F., Mirmiran, P., & Hekmatdoost, A. (2024). Effects of resveratrol on the anthropometric indices and inflammatory markers in obesity: An umbrella meta-analysis.
- Chan, L. K. W., Lee, K. W. A., Lee, C. H., Lam, K. W. P., Lee, K. F. V., Wu, R., Wan, J., Shivananjappa, S., Sky, W. T. H., Choi, H., & Yi, K.-H. (2024). Cosmeceuticals in photoaging: A review. Skin Research and Technology, 30(9), e13730.
- Salla, M., Karaki, N., El Kaderi, B., Ayoub, A. J., Younes, S., Abou Chahla, M. N., Baksh, S., & El Khatib, S. (2024). Enhancing the bioavailability of resveratrol: Combine it, derivatize it, or encapsulate it? Pharmaceutics, 16(4), 569.
- Dikmetas, D. N., Yenipazar, H., & Karaca, A. C. (2024). Recent advances in encapsulation of resveratrol for enhanced delivery. Food Chemistry, 460(Pt. 1), 140475.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Formulation scientists transitioning encapsulation protocols to prevent rapid polyphenol oxidation.
- Procurement directors requiring guaranteed stability data before authorizing bulk ingredient contracts.
- Quality control managers tracking specific trans-isomer degradation rates across extended warehousing cycles.
- Supply chain officers diversifying sourcing away from unpredictable agricultural knotweed yields.
- Compliance directors navigating strict European Food Safety Authority dosage limitation protocols.
- Operations managers solving hygroscopic clumping issues within high-speed powder handling machinery.
- Brand managers seeking clinical data supporting premium retail shelf-space positioning.
- Contract manufacturers scaling synthetic fermentation systems to bypass botanical extraction bottlenecks.
Frequently Asked Questions
What factors influence resveratrol ingredient pricing?
Clinical validation protocols force formulators to abandon cheap unstandardized extracts. Procurement directors demand guaranteed stability data prior to raw material purchase. This premium documentation requirement drives higher aggregate pricing.
Why does the resveratrol powder market dominate commercial sourcing?
Moisture sensitivity dictates bulk material handling protocols globally. Production managers depend entirely on granular stability to survive unpredictable international shipping delays. Powder avoids expensive nitrogen-flushing requirements.
What shapes the resveratrol cosmetic ingredient market?
Integrating bioactives into skincare demands novel microencapsulation technologies. Cosmetic chemists unlock lucrative anti-aging channels by solving dermal penetration challenges. Superior topical stability commands significant pricing premiums.
How is the Japanese knotweed resveratrol market performing?
Agricultural extraction allows clean-label declarations that consumers demand. Marketing directors leverage botanical origins to justify premium retail pricing tiers. Operations managers must navigate massive botanical waste volumes generated during processing.
What limits agricultural extraction efficiency?
Natural knotweed processing generates massive botanical waste volumes requiring costly disposal. Operations managers face escalating facility overhead as environmental regulations tighten. These costs erode natural extraction profit margins.
Which entities represent the top resveratrol companies globally?
Dominant producers command internal yeast fermentation capabilities. Identifying reliable commercial partners remains a top priority for global brands. Major buyers prioritize suppliers offering proprietary clinical data packages.
What defines the trans-resveratrol market premium?
Clinical practitioners demand verified milligrams per serving rather than functional food trace elements. Efficacy thresholds demand dedicated formats designed specifically for intestinal absorption. Buyers prioritize trans-isomer verification above basic botanical origins.
Why do functional beverages fail stability testing?
Integrating bioactives into functional beverages destroys molecular integrity through prolonged liquid suspension. Compound degradation accelerates exponentially in unprotected aqueous environments. Liquid instability limits broad beverage category expansion.
What drives the resveratrol for healthy aging market?
Mature demographics drive intense demand for scientifically validated formulations. Product developers prioritize water-soluble variants for seamless integration into daily wellness routines. Consumer awareness regarding cellular longevity sustains premium demand.
How large is the resveratrol nutraceutical market?
Hard-shell capsules accommodate massive active payloads impossible to mask in functional beverages. Formulation scientists achieve therapeutic thresholds without compromising consumer sensory experience. High-dose dietary supplements drive the vast majority of commercial volume.
What characterizes Japanese consumer demand patterns?
Mature demographics drive intense demand for scientifically validated healthy aging formulations. Product developers prioritize water-soluble variants for seamless integration into daily functional beverages. Formulators here push delivery system innovations globally.
How do resveratrol applications in supplements differ from skincare?
Specialized enteric coatings protect actives from harsh gastric acids internally. Topical applications require complex dermal penetration enhancers. The stabilization co-factors differ entirely between ingestion and topical routes.
Why does Germany exhibit slower compound growth?
Strict European Food Safety Authority dosage limits govern regional product formulations. Compliance officers reject high-dose supplements exceeding legally permissible daily intake thresholds. This forces formulators to prioritize enhanced bioavailability over total dosage.
What protects incumbent suppliers from challengers?
Major producers maintain massive libraries of independent clinical trials backing specific branded extracts. Challengers face significant hurdles replicating years of longitudinal human efficacy data. This data advantage shields incumbents from rapid substitution.
How do large buyers avoid vendor lock-in?
Consumer brands qualify minimum three distinct suppliers per required bioactive. Sourcing directors negotiate aggressive volume discounts by threatening to shift production toward synthetic analogues. Patent protections on delivery systems complicate this strategy.
What mechanism solves gastric degradation challenges?
Liposomal matrices shield active compounds from digestive destruction. Formulation scientists capture premium pricing by guaranteeing verifiable cellular absorption rates. This technology remains the primary competitive differentiator.
How does India achieve rapid compound growth?
Massive investments in modern extraction infrastructure transition local suppliers into refined isolate producers. Procurement directors leverage these new facilities to secure reliable supply chains. Scale-up initiatives capture significant export volume.
What limits rapid synthetic fermentation scaling?
Securing GRAS status for novel synthetic variants requires expensive longitudinal safety data. Regulatory approval delays slow commercial rollout significantly. Consumer resistance toward synthetic label declarations also slows adoption.
How do particle sizes alter clinical outcomes?
Particle size inconsistency alters gastric release timing unpredictably. Formulation scientists must enforce strict mesh-size requirements or risk failing disintegration testing protocols. Failing regulatory dissolution tests forces immediate product recalls.
What triggers raw material shipment rejections?
Plant-derived batches contain variable secondary compounds complicating standardization efforts. Quality control technicians reject entire shipments when trace contaminants exceed allowable maximums. These rejections disrupt manufacturing schedules completely.
Why do premium pharmacies reject generic supplements?
Major retail chains demand third-party assay results proving trans-resveratrol content before approving shelf space. Retailers protect themselves from consumer watchdog litigation. Unverified extracts lose retail access entirely.
What shifts purchasing from price to absorption?
Once contract manufacturers integrate microencapsulation commercially, stability ceases functioning as a competitive differentiator. Raw material purchasing shifts immediately toward actual absorption-yield ratios. Buyers prioritize cellular delivery guarantees.
Table of Content
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Desk Research Programme (Secondary Evidence)
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Form , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Form , 2026 to 2036
- Powder
- Liquid
- Y to o to Y Growth Trend Analysis By Form , 2021 to 2025
- Absolute $ Opportunity Analysis By Form , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Claim
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Claim, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Claim, 2026 to 2036
- Natural
- Synthetic
- Y to o to Y Growth Trend Analysis By Product Claim, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Claim, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use Application, 2026 to 2036
- Nutraceutical/Dietary Supplements
- Personal Care Cosmetics
- Others
- Y to o to Y Growth Trend Analysis By End Use Application, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Form
- By Product Claim
- By End Use Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By Product Claim
- By End Use Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Form
- By Product Claim
- By End Use Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By Product Claim
- By End Use Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Form
- By Product Claim
- By End Use Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By Product Claim
- By End Use Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Form
- By Product Claim
- By End Use Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By Product Claim
- By End Use Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Form
- By Product Claim
- By End Use Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By Product Claim
- By End Use Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Form
- By Product Claim
- By End Use Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By Product Claim
- By End Use Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Form
- By Product Claim
- By End Use Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By Product Claim
- By End Use Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Product Claim
- By End Use Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Form
- By Product Claim
- By End Use Application
- Competition Analysis
- Competition Deep Dive
- dsm-firmenich
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Sabinsa Corporation
- Maypro
- Layn Natural Ingredients
- dsm-firmenich
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Product Claim, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Use Application, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Product Claim, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End Use Application, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Product Claim, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End Use Application, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Product Claim, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End Use Application, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Product Claim, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End Use Application, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Product Claim, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End Use Application, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Product Claim, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End Use Application, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Product Claim, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End Use Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Form
- Figure 6: Global Market Value Share and BPS Analysis by Product Claim, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Product Claim, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Product Claim
- Figure 9: Global Market Value Share and BPS Analysis by End Use Application, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Use Application, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End Use Application
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Form
- Figure 26: North America Market Value Share and BPS Analysis by Product Claim, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Product Claim, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Product Claim
- Figure 29: North America Market Value Share and BPS Analysis by End Use Application, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End Use Application, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End Use Application
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Form
- Figure 36: Latin America Market Value Share and BPS Analysis by Product Claim, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Product Claim, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Product Claim
- Figure 39: Latin America Market Value Share and BPS Analysis by End Use Application, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End Use Application, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End Use Application
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Form
- Figure 46: Western Europe Market Value Share and BPS Analysis by Product Claim, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Product Claim, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Product Claim
- Figure 49: Western Europe Market Value Share and BPS Analysis by End Use Application, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End Use Application, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by End Use Application
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Form
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Product Claim, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Product Claim, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Product Claim
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End Use Application, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End Use Application, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End Use Application
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Form
- Figure 66: East Asia Market Value Share and BPS Analysis by Product Claim, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Product Claim, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Product Claim
- Figure 69: East Asia Market Value Share and BPS Analysis by End Use Application, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End Use Application, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by End Use Application
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Form
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Product Claim, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Claim, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Product Claim
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End Use Application, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use Application, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End Use Application
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Form
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Product Claim, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Product Claim, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Product Claim
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End Use Application, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End Use Application, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End Use Application
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

