CCR6 Antagonists Market
CCR6 Antagonists Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
CCR6 Antagonists Market Forecast and Outlook 2026 to 2036
The CCR6 antagonists market begins 2026 at USD 1 billion and is projected to grow to USD 4 billion by 2036 at a 12% CAGR. Adoption is guided by target patient populations, inflammatory disease prevalence, and clinical trial outcomes rather than brand recognition. Biopharma programs select molecules based on receptor specificity, pharmacokinetics, and safety profiles. Once a therapy enters a treatment protocol, switching requires regulatory review and new trial data. Physicians evaluate efficacy endpoints, dosing schedules, and combination potential. Supply planning considers batch stability, cold chain logistics, and expiration monitoring. Reimbursement frameworks and patient access influence adoption speed.
Commercial growth follows trial results, regulatory approvals, and guideline inclusion rather than promotional activity. Manufacturing focuses on API consistency, formulation reliability, and analytical verification. Release gates include potency, sterility, and stability testing. Distribution aligns with specialty centers and regional pharmacies to ensure uninterrupted therapy. Technical support guides administration, monitoring, and adverse event management. Profitability depends on predictable production, minimal batch failure, and patient uptake rather than frequent reformulation or price promotions. Program expansion follows indication approvals and adoption across specialty clinics.
Quick Stats for CCR6 Antagonists Market
- CCR6 Antagonists Market Value (2026): USD 1.0 billion
- CCR6 Antagonists Market Forecast Value (2036): USD 4.0 billion
- CCR6 Antagonists Market Forecast CAGR 2026 to 2036: 12.0%
- Leading Type in CCR6 Antagonists Market: Small-molecule CCR6 inhibitors
- Key Growth Regions in CCR6 Antagonists Market: North America, Asia Pacific, Europe
- Top Players in CCR6 Antagonists Market: Bristol Myers Squibb, Pfizer, AbbVie, Roche, Novartis

What Is the Growth Forecast for the CCR6 Antagonists Market through 2036?
Drug development pipelines and targeted therapy adoption drive the CCR6 antagonists market more than total autoimmune disease prevalence. In 2026, at around USD 1 billion, demand is concentrated in specialty hospitals, clinical trial centers, and early access programs where patient selection depends on biomarker expression and disease phenotype. Uptake is tied to regulatory approvals, physician adoption of novel immunomodulators, and integration into combination therapy regimens. Growth is propelled by increased clinical trial enrollment, expanded indications for autoimmune conditions, and incorporation into precision medicine frameworks. The trajectory reflects gradual market penetration among early adopters and progressive integration into standard therapeutic guidelines rather than immediate, broad scale deployment.
Commercial strategy and patient access planning define the later phase of the CCR6 antagonists market. As value approaches roughly USD 4 billion by 2036, stakeholders emphasize scalable manufacturing, supply chain robustness, and inclusion in reimbursement programs. Pharmaceutical companies that provide comprehensive support including diagnostic companion tests, patient adherence tools, and clinician education gain competitive advantage. The expansion from USD 1 billion to USD 4 billion reflects cumulative adoption across multi-site clinical networks, expanded global regulatory approvals, and therapy portfolio diversification rather than a single breakthrough event. Manufacturers that combine reliable production, consistent clinical support, and flexible distribution channels are positioned to secure long-term supply agreements.
CCR6 Antagonists Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1.0 billion |
| Forecast Value (2036) | USD 4.0 billion |
| Forecast CAGR 2026 to 2036 | 12.0% |
How Is the CCR6 Antagonists Market Supporting Targeted Immune Modulation in Inflammatory Diseases?
CCR6 antagonists are investigational or emerging therapies designed to block the chemokine receptor 6 pathway, which regulates migration of specific immune cells involved in autoimmune and inflammatory conditions. Traditional immunosuppressive treatments broadly suppress immune activity, leading to side effects and variable disease control. CCR6-targeted approaches aim to selectively modulate pathogenic T cell trafficking, offering potential efficacy with reduced systemic impact. Researchers and clinicians evaluate candidates based on receptor binding affinity, pharmacokinetics, tissue penetration, and safety in preclinical and clinical studies. Use is concentrated in conditions such as psoriasis, multiple sclerosis, and inflammatory bowel disease, where precise immune targeting can alter disease progression. Early adoption focuses on clinical trials and academic centers investigating mechanism-based therapy.
Manufacturers differentiate CCR6 antagonist candidates through chemical stability, receptor specificity, and scalability of production rather than pricing. Early programs emphasized laboratory synthesis and preclinical validation, with limited translational data. Current development emphasizes standardized formulations, biomarker-guided dosing, and integration with companion diagnostics to support clinical trial endpoints. Biopharmaceutical developers and contract manufacturing organizations compete on batch consistency, regulatory documentation, and supply chain reliability. Healthcare institutions evaluate total value through improved therapeutic indices, reduced adverse events, and potential for combination therapy rather than unit cost. Over time, market growth is linked to expansion of targeted immunotherapy research, increasing prevalence of autoimmune diseases, and demand for mechanism-based treatment options that enable precise modulation of pathogenic immune pathways.
What Factors Are Shaping the Demand for CCR6 Antagonists Market in 2026 by Products and Applications?
Adoption in the CCR6 Antagonists Market is influenced by disease severity, mechanism-specific efficacy, and regulatory compliance rather than short-term procurement trends. Healthcare providers prioritize therapies that offer targeted immune modulation, minimize adverse events, and integrate into treatment protocols for autoimmune and inflammatory conditions. Segment demand varies based on molecule type, route of administration, and patient monitoring requirements. Suppliers offering patient support programs, dosing guidance, and reliable supply chains are positioned to secure long-term adoption. Growth is concentrated in autoimmune diseases and inflammatory bowel disease, providing predictable demand for manufacturers capable of aligning therapeutic performance with clinical support and service reliability.
How Does Product Selection Influence Supplier Responsibilities and Clinical Integration in the CCR6 Antagonists Market?
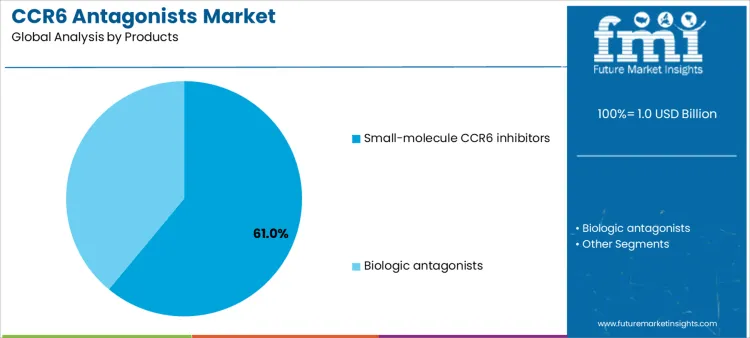
Small-molecule CCR6 inhibitors represent approximately 61% of demand due to their oral administration, targeted mechanism, and compatibility with chronic therapy regimens. Suppliers supporting these therapies must ensure consistent drug supply, adherence programs, and training for clinicians on dosing and monitoring. Biologic antagonists are used in more complex or refractory cases, requiring cold-chain logistics, clinical guidance for infusion protocols, and enhanced patient education. Product type selection defines supplier involvement in technical support, adherence monitoring, and inventory management. Manufacturers that provide comprehensive post-sale support and integration into clinical workflows can strengthen adoption and account retention across diverse healthcare settings.
Operational and therapeutic characteristics further shape supplier strategy. Small-molecule therapies require predictable supply, guidance on chronic dosing adherence, and integration into outpatient management programs. Biologic antagonists demand infusion infrastructure, monitoring of immune responses, and staff training for safe administration, increasing supplier responsibility for ongoing support. Hospitals standardizing on a particular product type rely on suppliers for consistent performance, patient education, and inventory logistics. Companies delivering training, technical assistance, and reliable supply channels can maximize adoption, improve treatment outcomes, and secure long-term contracts.
Why Does Application Mix Determine Volume Concentration and Supplier Strategy in the CCR6 Antagonists Market?

Autoimmune diseases account for approximately 66% of market demand because high prevalence and chronic treatment requirements create consistent, high-volume demand. Inflammatory bowel disease applications, while lower in volume, require specialized therapies, targeted dosing, and enhanced clinical support, presenting opportunities for high-value products and service offerings. Application-specific requirements shape inventory planning, staff training, and patient support programs. Aligning product capabilities and supplier services with both high-volume autoimmune conditions and specialized inflammatory bowel disease cases enables manufacturers to maximize adoption, revenue stability, and clinical impact.
Healthcare facility and patient program needs further influence supplier engagement. High-volume autoimmune treatment programs prioritize reliable supply, structured patient adherence support, and clinical training to ensure therapy effectiveness. Inflammatory bowel disease cases demand targeted guidance, infusion or administration support, and real-time monitoring. Niche or specialized applications require technical support, education, and precise supply logistics. Segment distribution is therefore driven by disease prevalence, therapy complexity, and integration into clinical workflows rather than short-term purchasing trends. Suppliers that provide training, technical support, and reliable product supply can secure broader adoption and long-term partnerships.
How is the CCR6 Antagonists Market Being Defined by Targeted Therapy and Immune Modulation?
The CCR6 Antagonists Market is shaped by hospitals, research institutions, and pharmaceutical developers focusing on immune-mediated diseases such as autoimmune disorders and inflammatory conditions. Treatments are evaluated based on specificity, efficacy, dosing regimen, and safety profile. Selection decisions involve clinicians, clinical pharmacologists, and hospital procurement teams. Suppliers that provide compounds with consistent pharmacological performance, regulatory-compliant documentation, and clinical support are preferred. This positions CCR6 antagonists as precision therapies rather than broad immunosuppressive agents, emphasizing clinical reliability, patient safety, and predictable outcomes.
How Are Standardized Clinical Protocols Influencing Supplier Expectations in the CCR6 Antagonists Market?
Healthcare providers and clinical trial sponsors increasingly adopt standardized treatment protocols to reduce variability and ensure reproducible outcomes. This drives demand in the CCR6 Antagonists Market toward therapies with well-characterized pharmacodynamics, predictable patient responses, and integration with monitoring protocols. Buyers prefer products with clinical data supporting mechanism of action and safety. Suppliers are evaluated on batch-to-batch consistency, regulatory compliance, and technical support rather than on experimental formulations. Therapies aligned with standardized protocols are more likely to be adopted across multiple clinical trials and care centers.
Where is Recurring Demand Being Built in the CCR6 Antagonists Market?
Recurring demand comes from hospitals, specialty clinics, and clinical trial sites managing patients with chronic immune-mediated conditions. Each treatment cycle generates predictable repeat orders, especially for multi-center studies. The CCR6 Antagonists Market also benefits from long-term therapy programs and ongoing trials where the same compound is used across multiple patient cohorts. Suppliers that provide dosing support, monitoring guidance, and reliable supply chains secure sustained demand. Embedding CCR6 antagonists into standard care pathways or trial protocols ensures consistent, repeat usage.
Why do Cost and Safety Considerations Limit Broader Adoption in the CCR6 Antagonists Market?
CCR6 antagonists carry high development and per-patient costs and require careful monitoring for immune-related adverse effects, which can limit adoption in smaller hospitals or low-volume clinics. The market also faces adoption constraints from regulatory approvals, clinical trial protocols, and formulary inclusion. Clinicians are cautious about switching to novel therapies without extensive data. Suppliers entering the market must provide clinical guidance, training, and consistent product availability. Adoption is concentrated in specialty centers and trial sites where precision therapy and patient safety are critical.
What is the Demand for CCR6 Antagonists by Country?
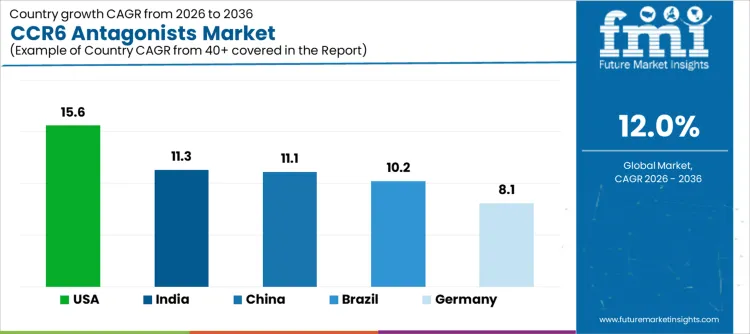
| Country | CAGR |
|---|---|
| USA | 15.6% |
| Brazil | 10.2% |
| China | 11.1% |
| India | 11.3% |
| Germany | 8.1% |
Demand for CCR6 antagonists is rising as pharmaceutical and biotech companies advance clinical programs targeting autoimmune, inflammatory, and immuno mediated diseases. India leads with an 11.3% CAGR, supported by growing clinical research activity, rising prevalence of autoimmune disorders, and adoption of novel targeted therapies. China follows at 11.1%, driven by expanding drug development pipelines, higher patient populations, and regulatory support for innovative biologics. Brazil records 10.2%, reflecting increased clinical trial activity and access to new therapies. The USA grows at 15.6%, shaped by high R&D investment, early adoption of next generation immunomodulatory agents, and focus on precision medicine. Germany, at 8.1%, reflects steady demand in a mature pharmaceutical environment with structured clinical adoption.
How Are Immunology and Specialty Therapy Programs Driving Demand in the CCR6 Antagonists Market in the United States?
United States demand for CCR6 antagonists is projected to expand along a 15.6% CAGR as hospitals and specialty clinics scale autoimmune and inflammatory disease programs. Procurement decisions are overseen by hospital pharmacy committees, clinical trial coordinators, and specialty physician boards. Adoption emphasizes precision therapy targeting, dosing flexibility, and integration with existing biologic regimens. Once validated, therapies are standardized across multiple hospital networks and specialty centers. Clinical trial participation, insurance coverage, and guideline adherence influence adoption. Import formulations dominate high specification programs, while domestic suppliers provide standard therapeutic options. Capital allocations follow program expansion and patient volume growth. Replacement cycles track therapy duration. Commercial success depends on inclusion in hospital formularies, network treatment protocols, and multi-year supply agreements.
Why Are Private Hospital Networks Driving Growth in the CCR6 Antagonists Market in Brazil?
Brazil is experiencing a 10.2% CAGR in CCR6 antagonist demand due to expansion of private hospital autoimmune programs and specialty care units. Procurement authority resides with network pharmacy committees, specialty department directors, and hospital purchasing offices. Adoption focuses on precision biologics for inflammatory and autoimmune conditions, including dosing monitoring and therapy management. Import products dominate premium regimens, while local suppliers provide standard therapies. Once approved, rollout extends across multiple affiliated hospitals. Physician training, clinical protocol adherence, and patient education influence utilization. Capital allocation follows hospital network expansion and specialty program launches. Replacement cycles align with therapy schedules. Commercial success depends on inclusion in network formularies, reference site adoption, and long-term supply contracts rather than isolated hospital purchases.
What Institutional Programs Are Driving Growth in the CCR6 Antagonists Market in China?
China’s CCR6 antagonists demand is increasing at an 11.1% CAGR due to expansion of tertiary hospital autoimmune programs, specialty immunology units, and clinical trial networks. Large hospital networks control procurement decisions. Adoption emphasizes targeted biologic therapy, patient monitoring, and integration with standard immunomodulatory regimens. Domestic suppliers cover routine therapies, while international brands retain positions in advanced protocols. Procurement follows tender systems and multi-year hospital planning. Once approved, therapies are deployed across multiple departments and hospitals. Training, protocol adherence, and clinical workflow influence uptake. Replacement cycles follow treatment duration. Capital allocation aligns with hospital expansion and program growth. Commercial success depends on inclusion in provincial and national hospital formularies and integration into standardized therapy frameworks rather than small clinic purchases.
Why Are Specialty Hospital Expansions Driving Growth in the CCR6 Antagonists Market in India?
India is experiencing an 11.3% CAGR in CCR6 antagonists due to expansion of specialty immunology centers, hospital autoimmune programs, and clinical trial networks. Procurement authority rests with hospital pharmacy boards, specialty committees, and capital planning teams. Device and therapy selection emphasizes efficacy, safety, dosing flexibility, and integration with existing biologic regimens. Import formulations dominate high specification cases while domestic suppliers cover standard therapies. Once approved, therapies are deployed across multiple affiliated hospitals and specialty centers. Training, protocol alignment, and workflow standardization influence adoption. Replacement cycles track therapy schedules and patient adherence. Budget allocation aligns with hospital expansion and program launches. Commercial success depends on inclusion in hospital formularies, reference site adoption, and multi-year supply contracts rather than isolated clinic or department purchases.
How Are Guidelines and Reimbursement Programs Shaping the CCR6 Antagonists Market in Germany?
Germany is experiencing an 8.1% CAGR in CCR6 antagonists due to structured clinical guidelines, reimbursement frameworks, and hospital protocol standardization. University hospitals and regional specialty centers control most procurement. Adoption focuses on targeted therapy, patient monitoring, and integration with existing autoimmune treatment regimens. Evaluation emphasizes efficacy, safety, and workflow integration. Procurement follows health technology assessment and hospital capital planning schedules. Import suppliers dominate premium applications, while domestic suppliers provide standard therapy options. Once approved, therapies are deployed across multiple departments and hospitals. Training, protocol adherence, and monitoring influence utilization. Replacement cycles follow therapy schedules. Capital allocation aligns with hospital specialty program expansion. Commercial success depends on guideline inclusion, framework agreements, and reference site adoption rather than isolated hospital or clinic purchases.
How Do Biopharma Companies Compete for Clinical Development in the CCR6 Antagonists Market?

CCR6 antagonist adoption follows clinical trial protocol design, target patient population selection, and mechanistic rationale rather than routine formulary review. Investigators define indication, dosing schedule, biomarker endpoints, and combination therapy potential before selecting compounds. Bristol Myers Squibb, Pfizer, AbbVie, Roche, Novartis compete during protocol planning, investigator engagement, and regulatory strategy stages. Once a candidate enters a study arm, trial design, safety monitoring, and data collection anchor its use. Bristol Myers Squibb leverages pipeline experience in inflammatory and autoimmune conditions. Pfizer focuses on small molecule programs integrated with immunology portfolios. AbbVie emphasizes clinical efficacy data and multi-center trial networks. Roche aligns candidates with biomarker guided patient selection and translational research. Novartis targets accelerated development through combination studies and global trial access.
Competitive positioning depends on clinical proof of mechanism, patient recruitment feasibility, and regulatory alignment rather than pricing. Study sponsors assess target engagement, pharmacokinetics, adverse event profile, and trial logistics during protocol evaluation. CROs and sites value compounds with predictable handling, dosing, and monitoring procedures. Bristol Myers Squibb secures adoption through established investigator networks and trial infrastructure. Pfizer gains traction by linking CCR6 antagonists with other immunomodulatory programs. AbbVie competes where efficacy and endpoint robustness drive investigator preference. Roche holds share in biomarker rich trials with translational research integration. Novartis participates where multi arm and global trials require compound availability. Market positions vary by therapeutic indication, trial design, patient population, and regulatory environment.
Key Players in the CCR6 Antagonists Market
- Bristol Myers Squibb
- Pfizer
- AbbVie
- Roche
- Novartis
- Others
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Product Type | Small-molecule CCR6 inhibitors; Biologic antagonists |
| Application | Autoimmune diseases; Inflammatory bowel disease; Others |
| End User / Sales Channel | Hospital pharmacies; Specialty pharmacies |
| Region | Asia Pacific; Europe; North America; Latin America; Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Brazil, Chile, Saudi Arabia, Turkey, South Africa, Rest of regions |
| Key Companies Profiled | Bristol Myers Squibb; Pfizer; AbbVie; Roche; Novartis |
| Additional Attributes | Dollar by sales by product and application, small molecule versus biologic mix, autoimmune versus IBD case split, clinical trial driven demand, cold chain and specialty distribution needs, batch stability and shelf life impact, reimbursement and access pathway influence, adherence and monitoring workload, guideline and regulatory milestone effects |
CCR6 Antagonists Market Segmentation
Products:
- Small-molecule CCR6 inhibitors
- Biologic antagonists
Applications:
- Autoimmune diseases
- Inflammatory bowel disease
- Others
End User / Sales Channel:
- Hospital pharmacies
- Specialty pharmacies
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- Wasilko, D. J., et al. (2024). Structural basis for CCR6 modulation by allosteric antagonists. Nature Communications, 15, Article 52045.
- Meghwani, H., et al. (2025). CCL20/CCR6 signaling modulates disease severity during inflammatory responses. mBio.
- Gómez-Melero, S., & Caballero-Villarraso, J. (2023). CCR6 as a potential target for therapeutic antibodies. Antibodies, 12(2), Article 30.
- Allodi, M., et al. (2025). Effects of CCL20/CCR6 modulators in T-cell adoptive transfer models. Pharmaceutics, 18(9), 1327.
Frequently Asked Questions
How big is the ccr6 antagonists market in 2026?
The global ccr6 antagonists market is estimated to be valued at USD 1.0 billion in 2026.
What will be the size of ccr6 antagonists market in 2036?
The market size for the ccr6 antagonists market is projected to reach USD 3.1 billion by 2036.
How much will be the ccr6 antagonists market growth between 2026 and 2036?
The ccr6 antagonists market is expected to grow at a 12.0% CAGR between 2026 and 2036.
What are the key product types in the ccr6 antagonists market?
The key product types in ccr6 antagonists market are small-molecule ccr6 inhibitors and biologic antagonists.
Which applications segment to contribute significant share in the ccr6 antagonists market in 2026?
In terms of applications, autoimmune diseases segment to command 66.0% share in the ccr6 antagonists market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Products
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Products , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Products , 2026 to 2036
- Small-molecule CCR6 inhibitors
- Biologic antagonists
- Small-molecule CCR6 inhibitors
- Y to o to Y Growth Trend Analysis By Products , 2021 to 2025
- Absolute $ Opportunity Analysis By Products , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Applications
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Applications, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Applications, 2026 to 2036
- Autoimmune diseases
- Inflammatory bowel disease
- Autoimmune diseases
- Y to o to Y Growth Trend Analysis By Applications, 2021 to 2025
- Absolute $ Opportunity Analysis By Applications, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospital pharmacies
- Specialty pharmacies
- Others
- Hospital pharmacies
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Products
- By Applications
- By End User
- Competition Analysis
- Competition Deep Dive
- Bristol Myers Squibb
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Pfizer
- AbbVie
- Roche
- Novartis
- Others
- Bristol Myers Squibb
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Products
- Figure 6: Global Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Applications
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Products
- Figure 26: North America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Applications
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Products
- Figure 36: Latin America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Applications
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Products
- Figure 46: Western Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Applications
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Products
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Applications
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Products
- Figure 66: East Asia Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Applications
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Products
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Applications
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Products
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Applications
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

