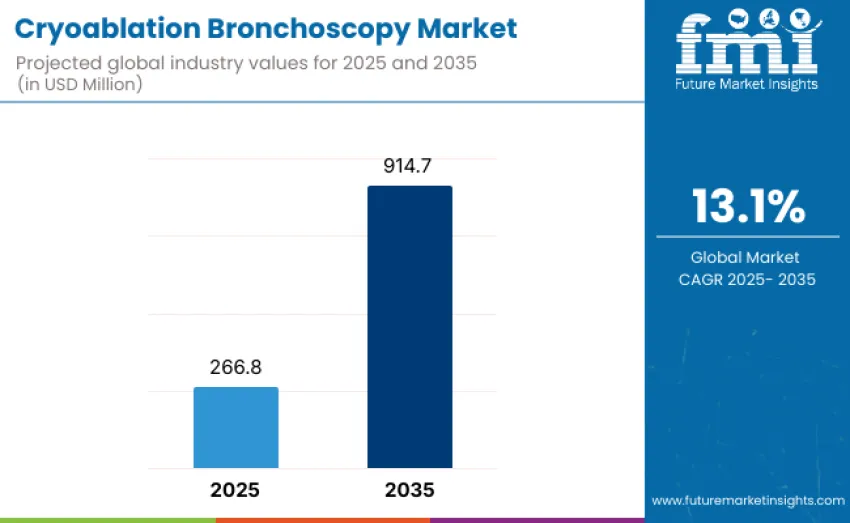
The global cryoablation bronchoscopy market is projected to reach USD 914.7 million by 2035, recording an absolute increase of USD 647.9 million over the forecast period. This market is valued at USD 266.8 million in 2025 and is set to rise at a CAGR of 13.1% during the assessment period. Expansion is driven by increasing adoption of minimally invasive therapies for lung cancer, bronchial tumors, and airway obstructions, coupled with rising investments in advanced interventional pulmonology infrastructure and healthcare digitalization projects globally.
Technological advancements in cryoprobe systems, imaging integration, and ergonomic bronchoscope design are reshaping the landscape. Contemporary systems feature high-performance cryoprobes capable of rapid tissue freezing, coupled with flexible bronchoscope platforms that allow precise navigation within complex airway anatomies. Integration with advanced imaging modalities, including fluoroscopy, endobronchial ultrasound, and real-time navigation systems, facilitates accurate lesion targeting and minimizes collateral tissue damage. Procedural enhancements such as improved suction, tip control, and ergonomic handles enhance clinician usability.
Government healthcare initiatives, reimbursement support, and clinical training programs accelerate growth. Public and private healthcare institutions globally are investing in interventional pulmonology infrastructure, including cryoablation-capable bronchoscopes, to improve access to minimally invasive lung therapies. Medical education programs and specialist training courses equip pulmonologists with the skills required to adopt cryoablation procedures safely and effectively. Rising prevalence of lung cancer and chronic airway diseases, coupled with patient preference for minimally invasive therapies with shorter recovery times, drives adoption.
Between 2025 and 2030, the cryoablation bronchoscopy market is projected to expand from USD 266.8 million to USD 494 million, resulting in a value increase of USD 227.2 million, which represents 35.1% of total forecast growth for the decade. This phase will be driven by increasing clinical adoption of minimally invasive bronchoscopic cryotherapy techniques, technological advancements in cryoprobe design and catheter flexibility, and expanding procedural utilization in both malignant and benign airway obstruction management.
From 2030 to 2035, growth is forecast from USD 494 million to USD 914.7 million, adding USD 420.7 million, which constitutes 64.9% of overall ten-year expansion. Growth during this phase will be characterized by wider adoption of next-generation cryoablation platforms featuring improved cooling efficiency, reduced procedural times, and integration with real-time imaging and navigation systems. The introduction of disposable cryoprobes, compact mobile cryo units, and hybrid bronchoscopic cryotherapy systems will enhance procedural accessibility across tertiary hospitals and ambulatory settings.
| Metric | Value |
|---|---|
| Market Value (2025) | USD 266.8 million |
| Market Forecast Value (2035) | USD 914.7 million |
| Forecast CAGR (2025 to 2035) | 13.1% |
The cryoablation bronchoscopy market is growing globally due to increasing adoption of minimally invasive procedures for management of pulmonary diseases, particularly lung cancer and airway obstructions. As global incidence of lung cancer continues to rise, healthcare providers are seeking advanced interventional solutions that can deliver precise tissue ablation with minimal damage to surrounding structures. Cryoablation offers distinct advantages such as reduced procedural risk, faster patient recovery, and improved visualization during bronchoscopy, making it an increasingly preferred alternative to traditional surgical interventions.
Technological advancements in cryoablation systems including enhanced probe flexibility, higher cooling efficiency, and compatibility with modern bronchoscopic platforms are driving adoption. The introduction of single-use cryoprobes and portable cryoablation consoles is expanding procedural accessibility across hospitals, outpatient centers, and interventional pulmonology units. Growing number of skilled interventional pulmonologists and increased availability of clinical training programs have strengthened procedural expertise, encouraging wider use of cryo-based therapies in respiratory care.
The cryoablation bronchoscopy market is segmented by product, technology, and region. By product, division includes cryo consoles/generators, single-use cryo catheters/probes, reusable cryo probes & handpieces, and gas supply sets & accessories. Based on technology, categorization covers nitrous oxide systems, carbon dioxide systems, and mixed-gas/dual-mode systems. Regionally, segmentation spans Asia Pacific, Europe, North America, Latin America, and Middle East & Africa.
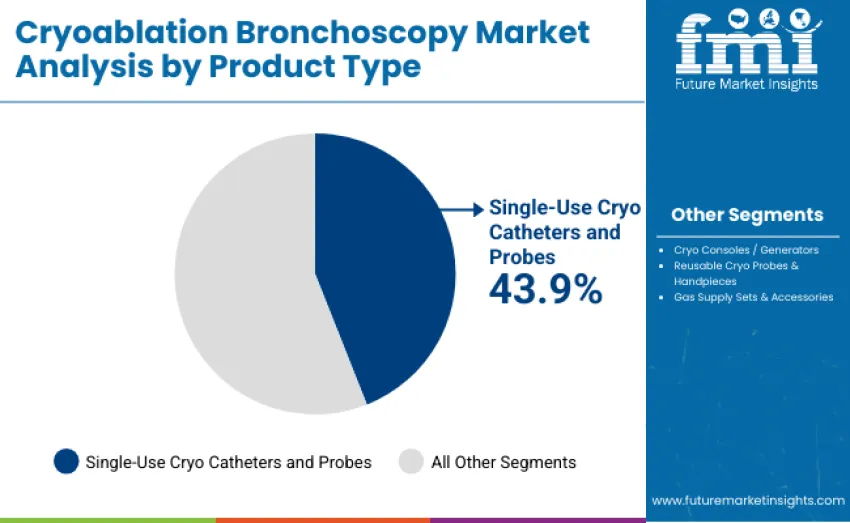
The single-use cryo catheters and probes segment represents the leading share, capturing 43.9% of total revenue in 2025. This category encompasses advanced disposable cryotherapy instruments designed for precision, safety, and sterility in interventional pulmonary procedures. Single-use systems have become the preferred option due to their ability to minimize cross-contamination, reduce sterilization turnaround times, and deliver consistent therapeutic cooling performance across multiple cases. Their adoption is reinforced by hospital infection control standards.
The cryo consoles and generators segment holds a 24.7% share, supported by integration of digital temperature control, gas flow regulation, and automated safety features within modern platforms. Reusable cryo probes and handpieces account for 18.6%, continuing to serve value-driven healthcare institutions seeking cost-efficient solutions. Gas supply sets and accessories represent 12.8% of overall revenue, comprising cryogenic gas regulators, connectors, tubing assemblies, and safety valves required to ensure consistent gas delivery and thermal stability.
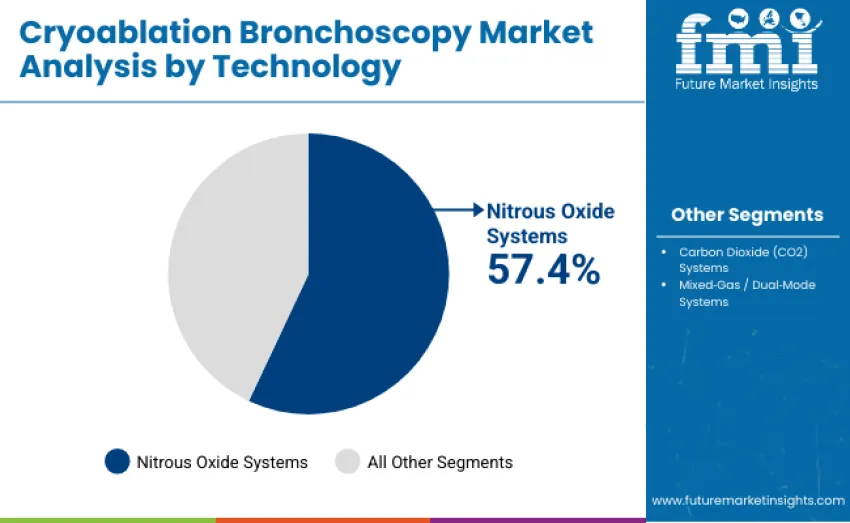
The nitrous oxide systems segment dominates, accounting for 57.4% of total revenue in 2025. Nitrous oxide systems deliver superior cooling efficiency, precise temperature control, and proven therapeutic outcomes, making them the gold standard for bronchial lesion ablation and cryobiopsy procedures. Their established clinical efficacy, operational stability, and compatibility with a broad range of cryo consoles reinforce their widespread preference among pulmonologists and interventional specialists.
The carbon dioxide systems segment follows with a 28.3% share, serving healthcare facilities seeking cost-effective cryoablation alternatives. Mixed-gas or dual-mode systems represent 14.3% of total revenue, characterized by flexibility and adaptability. These systems enable clinicians to alternate between nitrous oxide and carbon dioxide sources depending on procedural requirements, offering enhanced versatility for both superficial and deep tissue cryotherapy interventions. Key dynamics include superior cooling and procedural consistency positioning nitrous oxide as the reference standard.
The cryoablation bronchoscopy market is driven by three core demand factors linked to global expansion of minimally invasive pulmonary care and technological advancement in interventional devices. Increasing prevalence of lung cancer, central airway obstruction, and benign pulmonary lesions creates substantial demand for localized, minimally invasive ablation procedures that minimize patient trauma and hospital stay durations. With lung cancer remaining one of the leading causes of cancer-related mortality worldwide, hospitals are investing in cryoablation bronchoscopy systems to enhance procedural precision.
Key technological innovations driving market growth include advancements in cryo systems and probe design. The development of single-use cryoprobes enhances convenience and reduces cross-contamination risks, while digitally controlled consoles offer improved precision and ease of use. These innovations, combined with advanced temperature management mechanisms, ensure consistent cooling performance and enhanced safety for patients. The integration of these technologies simplifies the operation of cryotherapy systems, making them more efficient and accessible in clinical settings. As a result, these advancements are increasing the adoption of cryotherapy across various medical specialties.
Major restraints limiting adoption in the cryoablation bronchoscopy market include high equipment and procedure costs. Cryo consoles, gas systems, and disposable probes require significant capital investment and operational expenditures, which can be prohibitive for many healthcare facilities, especially smaller ones. Additionally, the need for specialized operator training presents another barrier, as cryoablation procedures require precise temperature control and advanced technical expertise. These factors can slow the widespread adoption of cryoablation in bronchoscopy, limiting its use to facilities with the resources for substantial investment and skilled personnel.
| Country/Region | CAGR (2025 to 2035)% |
|---|---|
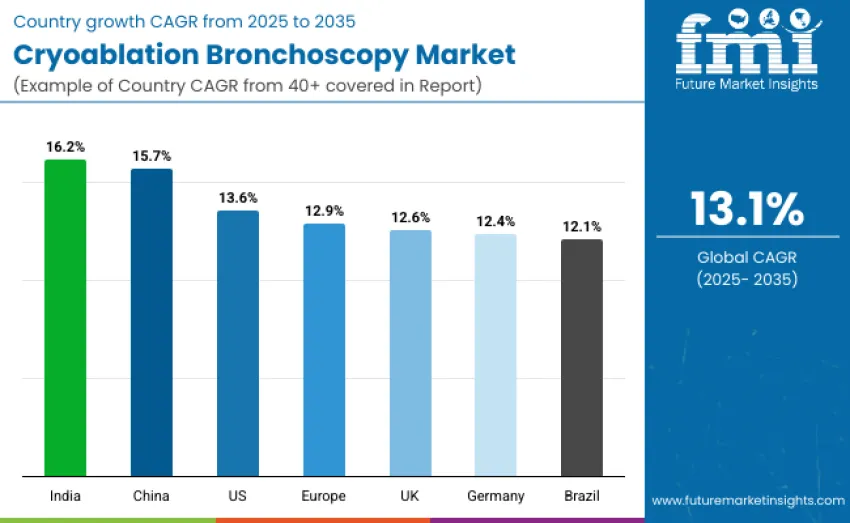
| USA | 13.6 |
| Brazil | 12.1 |
| China | 15.7 |
| India | 16.2 |
| Europe | 12.9 |
| Germany | 12.4 |
| UK | 12.6 |
The cryoablation bronchoscopy market is expanding globally, with India leading at a 16.2% CAGR due to rapid healthcare infrastructure advancements, growing access to interventional pulmonology, and government initiatives promoting early respiratory disease management. China follows with 15.7% growth, driven by hospital modernization and increased cryoablation adoption. The USA records 13.6% growth, supported by advanced healthcare systems and specialized pulmonary centers. Europe grows at 12.9%, with Germany at 12.4%, the UK at 12.6%, and France contributing to strong clinical adoption. Brazil demonstrates 12.1% growth, fueled by expanding healthcare networks and increasing awareness.
China demonstrates the strongest growth potential with a projected CAGR of 15.7% through 2035. Expansion is driven by rising prevalence of lung cancer, COPD, and other pulmonary diseases, alongside rapid modernization of tertiary hospital infrastructure in major cities such as Beijing, Shanghai, Guangzhou, and Chengdu. Government policies under the Healthy China 2030 framework provide financial incentives, subsidies, and structured training programs to encourage adoption of minimally invasive procedures.
Hospitals are increasingly integrating cryoablation-enabled bronchoscopic systems for airway tumor management and pulmonary obstruction relief, supported by standardized clinical protocols and outcome monitoring to optimize safety and efficacy. Domestic manufacturers are collaborating with international device companies to localize consoles and catheters, improving affordability, operational reliability, and after-sales clinical support. Interventional pulmonology centers are enhancing procedural capacity through structured physician and technician training programs, ensuring high operator competency.
India's cryoablation bronchoscopy market is projected to grow at a CAGR of 16.2% through 2035, supported by increasing prevalence of pulmonary diseases and expansion of interventional pulmonology services in urban healthcare centers. Tertiary hospitals in Bangalore, Hyderabad, Mumbai, and Delhi are establishing advanced bronchoscopy suites, enabling treatment of airway obstructions, early-stage tumors, and palliative care interventions. Government initiatives such as Ayushman Bharat and the National Health Mission are providing funding, procedural training grants, and financial support.
Private multispecialty hospitals are incorporating cryoablation-enabled systems for minimally invasive tumor ablation, while procedural standardization and clinical outcome monitoring ensure safety and efficacy. Collaboration between domestic manufacturers and international OEMs is promoting local production of cryoablation catheters and consoles, reducing costs and improving after-sales service. Structured training programs for pulmonologists and technicians are enhancing operator competency, and public-private partnerships facilitate equipment distribution and maintenance support.
Germany's cryoablation bronchoscopy market is expected to grow at a CAGR of 12.4% through 2035, reflecting a mature healthcare system with advanced interventional pulmonology capabilities, high clinical expertise, and robust reimbursement frameworks. University hospitals and tertiary care centers in Berlin, Munich, and Hamburg are equipping bronchoscopy suites with next-generation cryoablation consoles and catheters for minimally invasive management of malignant airway obstructions, pulmonary lesions, and biopsy procedures.
Procedural adoption is supported by structured training programs organized by national respiratory societies and device manufacturers, ensuring high operator competency and patient safety. European distributors maintain close partnerships with OEMs to guarantee device availability, preventive maintenance, and rapid clinical support, minimizing operational downtime. Hospitals leverage clinical registries and outcome monitoring to optimize procedural protocols and standardize care delivery. Investment in high-quality, durable equipment and adherence to EU medical device regulations reinforces long-term stability.
Brazil's cryoablation bronchoscopy market is anticipated to grow at a CAGR of 12.1% through 2035, driven by modernization of public and private hospital infrastructure, rising prevalence of respiratory diseases, and growing demand for minimally invasive procedures. Tertiary hospitals and regional reference centers in São Paulo, Rio de Janeiro, Brasília, and Porto Alegre are increasingly deploying cryoablation systems for tumordebulking, airway clearance, and diagnostic sampling.
Public-private partnerships between SUS hospitals, private healthcare networks, and pulmonology societies are facilitating procedural standardization, clinical training, and physician competency development. Regional distribution channels and Latin American subsidiaries of global medtech companies are ensuring reliable equipment availability, preventive maintenance, and technical support. Emerging reimbursement pathways and public health investments reduce barriers to adoption, enabling sustainable equipment deployment and improved patient access.
The United States cryoablation bronchoscopy market is projected to grow at a CAGR of 13.6% through 2035, reflecting widespread adoption across specialized pulmonary and thoracic oncology centers. Leading healthcare systems, including Mayo Clinic, Cleveland Clinic, and MD Anderson Cancer Center, are integrating cryoablation-enabled bronchoscopy for airway tumor ablation, palliative therapy, and diagnostic sampling. Procedural adoption is supported by broad reimbursement coverage under Medicare, Medicaid, and private insurance.
Hospitals are standardizing clinical workflows, monitoring outcomes, and optimizing equipment configurations to improve procedural safety, efficiency, and patient care. Ongoing research and development and FDA-cleared innovations in cryoablation probes, consoles, and delivery systems sustain technological competitiveness. Nationwide medical device supply chains and OEM partnerships ensure equipment availability, preventive maintenance, and rapid technical support. The combination of advanced healthcare infrastructure, regulatory support, and innovation drives sustained growth.
The United Kingdom's cryoablation bronchoscopy market is expected to grow at a CAGR of 12.6% through 2035, driven by NHS investments in minimally invasive oncology care, technology modernization, and growing interventional pulmonology expertise. Hospitals and university medical centers in London, Manchester, Bristol, and Birmingham are increasingly adopting cryoablation systems for airway tumor ablation, pulmonary lesion management, and diagnostic sampling.
Structured training programs led by academic institutions and professional societies ensure physician competency and procedural safety. NHS funding initiatives, procurement frameworks, and technology leasing arrangements enhance equipment accessibility and reliability across tertiary hospitals. Hospitals leverage clinical outcome registries and standardize procedural protocols to maintain consistent treatment efficacy, safety, and workflow efficiency. Collaboration with device manufacturers ensures preventive maintenance, post-market support, and integration of next generation cryoablation technology.
The cryoablation bronchoscopy market in Europe is projected to grow from USD 79.2 million in 2025 to USD 253.4 million by 2035, registering a CAGR of 12.9% over the forecast period. Germany is expected to maintain leadership with a 28.5% share in 2025, declining to 27.8% by 2035, supported by advanced healthcare infrastructure, strong interventional pulmonology ecosystem, and leading medical centers in Berlin, Munich, and Hamburg that are early adopters of cryotherapy systems.
France follows with a 22.3% share in 2025, projected to reach 22.6% by 2035, driven by a robust national oncology care framework, well-established thoracic surgery networks, and expanding adoption of minimally invasive cryoablation technologies across Paris and Lyon hospitals. The United Kingdom holds a 19.5% share in 2025, expected to reach 19.8% by 2035 through increased integration within NHS treatment pathways. Italy commands a 13.2% share in both 2025 and 2035, supported by growing demand for advanced bronchoscopy procedures.
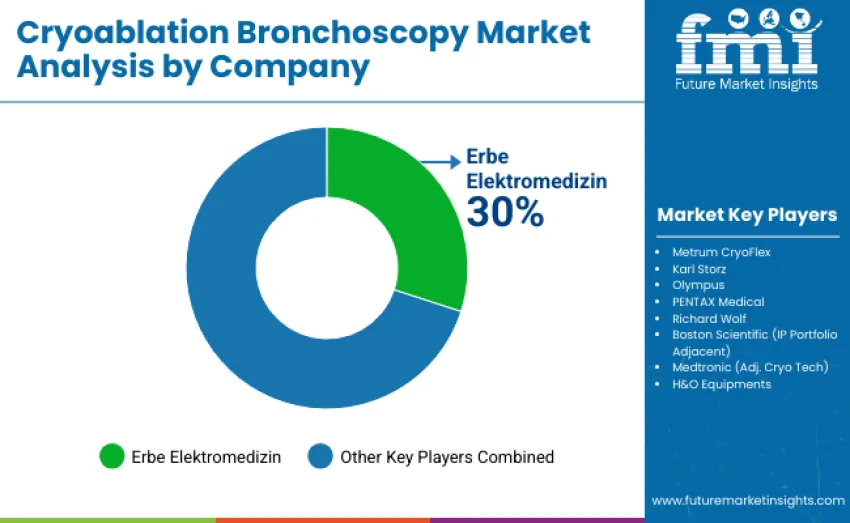
The global cryoablation bronchoscopy market is moderately concentrated, with 12–15 key players shaping competition. The top three companies, ErbeElektromedizin, Karl Storz, and Olympus, collectively control 55–60% of the global share, with ErbeElektromedizin leading at 30%. These leaders dominate through established clinical trust, brand recognition, and comprehensive device portfolios. Competition centers around cryoprobe performance, console integration, clinical workflow efficiency, and training support, with less emphasis on price competition alone.
ErbeElektromedizin, Karl Storz, and Olympus maintain competitive advantages through extensive patent portfolios covering cryotherapy catheters, consoles, and accessory systems, along with proven clinical reliability. These companies invest significantly in research and development to improve freezing efficiency, ergonomic console designs, and integration with bronchoscopic navigation platforms. They also support procedural training programs, hospital adoption initiatives, and evidence-based outcomes tracking to strengthen their leadership.
Challengers such as Richard Wolf, PENTAX Medical, and MetrumCryoFlex compete by offering differentiated products for specialized procedural applications, regional presence, and cost-effective solutions for mid-tier hospitals. Emerging players like Boston Scientific, Medtronic, and H&O Equipments increase competitive pressure through aggressive pricing, rapid product development, and local distribution strategies, especially in price-sensitive regions.
| Items | Values |
|---|---|
| Quantitative Units | USD 266.8 million |
| Product | Cryo Consoles/Generators, Single Use Cryo Catheters/Probes, Reusable Cryo Probes & Handpieces, Gas Supply Sets & Accessories |
| Technology | Nitrous Oxide Systems, Carbon Dioxide Systems, Mixed Gas/Dual Mode Systems |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Country Covered | USA, Brazil, China, India, Europe, Germany, France, UK |
| Key Companies Profiled | Erbe Elektromedizin , Metrum CryoFlex , Karl Storz, Olympus, PENTAX Medical, Richard Wolf, Boston Scientific, Medtronic, H&O Equipments |
| Additional Attributes | Dollar sales by product types and technology categories, regional trends across North America, Latin America, Europe, and Asia Pacific, competitive landscape of medical device manufacturers, integration with interventional pulmonology workflows, innovations in cryo console and catheter technology, and specialized system configurations for precision and safety |
How big is the cryoablation bronchoscopy market in 2025?
The global cryoablation bronchoscopy market is valued at USD 266.8 million in 2025.
What will be the size of the cryoablation bronchoscopy market in 2035?
The market is projected to reach USD 914.7 million by 2035.
How fast will the cryoablation bronchoscopy market grow between 2025 and 2035?
The market will grow at a CAGR of 13.1% from 2025 to 2035.
Which product type leads the cryoablation bronchoscopy market in 2025?
Single-use cryo catheters/probes lead the market with a 43.9% share in 2025.
Who are the major players in the cryoablation bronchoscopy market?
Key players include Erbe Elektromedizin, Metrum CryoFlex, Karl Storz, Olympus, PENTAX Medical, Richard Wolf, Boston Scientific, Medtronic, H&O Equipments, and others.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.