Human Milk Oligosaccharides Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
The Human Milk Oligosaccharides Market is segmented by Product Type (Fucosylated HMOs, Sialylated HMOs, Non-Fucosylated Neutral HMOs), Form (Powder, Liquid), End Use (Infant Formula, Nutraceutical Supplements, Functional Food and Beverage, Others), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Human Milk Oligosaccharides Market Size, Market Forecast and Outlook By FMI
The human milk oligosaccharides market size crossed a valuation of USD 339.7 million in 2025. The sector is set to surpass USD 394.5 million in 2026 at a CAGR of 17.20% during the forecast period. Continued investment is expected to bring the market to USD 1,925.6 million by 2036 as regulatory pathways support the commercial scaling of precision fermentation-derived strains. This progress allows pediatric brands to advance human milk oligosaccharides market trends that narrow functional gaps without creating prohibitive unit economics.
Infant nutrition formulation teams face a critical choice regarding their synthetic oligosaccharide strategies. They must decide whether to treat these ingredients as a premium-tier exclusive or a baseline functional requirement across all product lines. Delays in HMO adoption in infant nutrition risk total displacement by competitors positioning bio-identical immunity profiles as a non-negotiable standard. Evaluating human milk oligosaccharides requires mapping the downstream processing bottleneck. Formulators balance inclusion rates against margin erosion to maintain profitability.
Summary of Human Milk Oligosaccharides Market
- Human Milk Oligosaccharides Market Definition:
- This category covers precision-fermented, bio-identical carbohydrate polymers that replicate human breast milk structures. It separates advanced microbiome-modulating ingredients from generic plant-based prebiotics, establishing the functional boundary for premium pediatric and clinical nutrition globally.
- Demand Drivers in the Market:
- Accelerated novel food regulatory clearances force pediatric brand managers to upgrade baseline nutritional profiles globally.
- Bio-synthetic yield optimizations compel procurement teams to replace legacy dietary fibers with structurally identical alternatives.
- Evolving pediatric gastroenterology protocols drive clinical dietitians to specify formulations containing multi-strain bio-identical complexes.
- Key Segments Analyzed in the FMI Report:
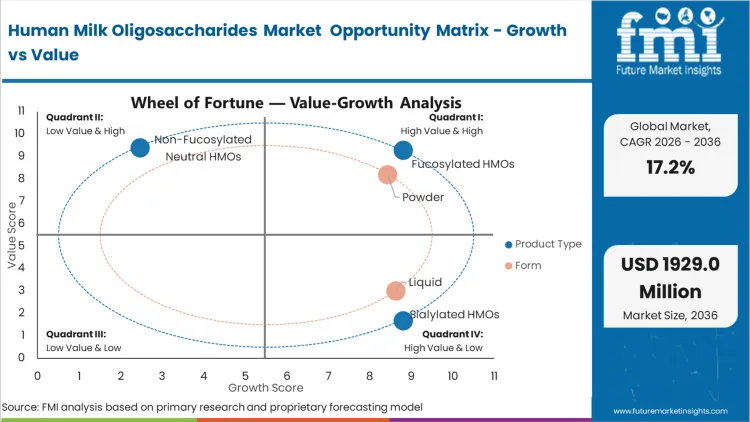
- Product Type: Fucosylated HMOs is expected to grab 69.1% share in 2026. This is driven by the structural reality that 2'-FL is the most abundant natural oligosaccharide and the first to achieve cost parity for mass application.
- Form: Powder is expected to grab 84.5% share in 2026. Thermal stability constraints mandate dry blending during the final stages of formula manufacturing.
- End Use: Infant Formula is expected to grab 68.9% share in 2026. This dominance is dictated by the immediate regulatory focus on closing the functional gap with natural human breast milk.
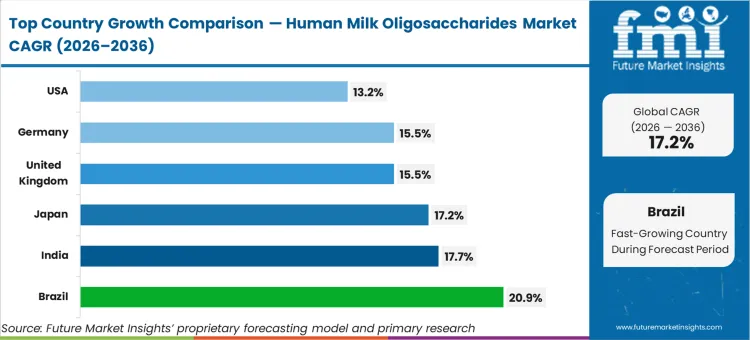
- Brazil: 20.9% compound growth. Rapid urbanization pushes dual-income households directly into premium nutritional tiers.
- Analyst Opinion at FMI:
- Nandini Roy Choudhury, Principal Analyst, Food and Beverage, at FMI, opines, "Standard industry metrics track regulatory approvals to map expansion. Authorization dossiers miss the physical extraction bottleneck entirely. The actual constraint defining volume availability centers on downstream purification capacity. Removing fermentation byproducts at industrial scale without degrading the oligosaccharide structure matters far more than theoretical regulatory timelines in forecasting actual commercial adoption."
- Strategic Implications / Executive Takeaways:
- Formulation directors must map continuous fermentation yield improvements to time their transition from single-strain to multi-strain blends accurately.
- Fermentation capacity planners should secure long-term downstream purification partnerships to avoid stranded bio-synthetic production volumes.
- Procurement heads face structural reliance on a concentrated biotech supplier base. This dynamic requires multi-year off-take agreements to justify capital expansion.
- Methodology:
- Primary Research: Chief formulation officers and senior procurement directors at tier-1 pediatric nutrition companies.
- Desk Research: Novel food authorization dossiers from regulatory bodies and industrial biotech strain patent filings.
- Market-Sizing and Forecasting: Baselines anchored to installed commercial precision fermentation capacity and verifiable off-take agreements.
- Data Validation and Update Cycle: Forecasts triangulated against capital expenditure timelines for downstream purification facility expansions.
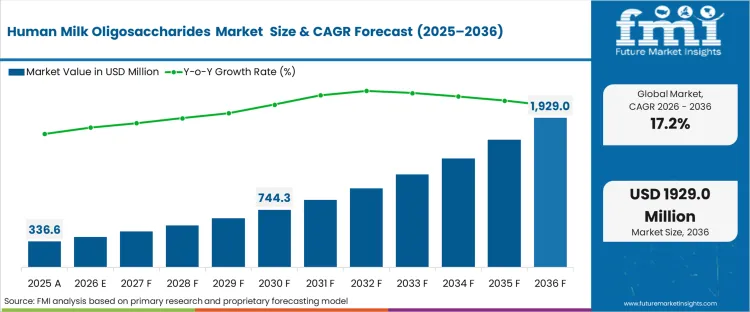
Growth is likely to accelerate once the transition from single-strain supplementation to complex multi-strain blends becomes cost-effective for standard formula tiers. Biotech suppliers support this shift when advanced continuous fermentation yields reach parity with legacy dietary fibers. As this threshold is reached, share analysis models point to a faster phase-out of conventional prebiotics across middle-tier pediatric portfolios. Companies monitoring HMO market share by company are watching this transition closely.
Brazil stands at 20.9%, India reaches 17.7%, and Japan follows at 17.2%. The United Kingdom and Germany each register 15.5%, while the United States records 13.2%. Growth momentum is stronger in developing regions, where rapid urbanization and the rise of dual-income households are supporting demand for premium formula tiers. Many buyers in these markets are adopting newer offerings without moving through earlier product generations. This regional gap reflects the difference between markets supported by local dairy joint ventures and those facing longer domestic novel food qualification timelines.
Human Milk Oligosaccharides Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 394.5 million |
| Industry Value (2036) | USD 1,925.6 million |
| CAGR (2026 to 2036) | 17.20% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Human Milk Oligosaccharides Market Definition
The HMO market encompasses the industrial-scale synthesis, purification, and commercialization of structurally identical carbohydrate polymers naturally found in breast milk. This sector functions as the critical bio-synthetic bridge between foundational pediatric nutrition and advanced microbiome modulation. It defines the performance ceiling for next-generation dietary formulations.
Human Milk Oligosaccharides Market Inclusions
Scope incorporates commercial volumes of fucosylated and sialylated oligosaccharides. It also includes non-fucosylated neutral variants produced via precision fermentation or enzymatic synthesis. The analysis covers distinct forms optimized for solubility and thermal stability during downstream food processing. This establishes functional baselines for Asean human milk oligosaccharides implementation. Dry powder concentrates and liquid stabilized variants explicitly fall within this boundary.
Human Milk Oligosaccharides Market Exclusions
Conventional prebiotics remain strictly excluded. This covers standard galacto-oligosaccharides and fructo-oligosaccharides extracted from bovine milk or plant sources. Legacy fibers cannot replicate the specific receptor-binding and anti-adhesive pathogen mechanisms characteristic of true human-identical oligosaccharide structures.
Human Milk Oligosaccharides Market Research Methodology
- Primary Research: Chief formulation officers and senior procurement directors at tier-1 pediatric nutrition companies.
- Desk Research: Novel food authorization dossiers from regulatory bodies and industrial biotech strain patent filings.
- Market-Sizing and Forecasting: Baselines anchored to installed commercial precision fermentation capacity and verifiable off-take agreements.
- Data Validation and Update Cycle: Forecasts triangulated against capital expenditure timelines for downstream purification facility expansions.
Segmental Analysis
Human Milk Oligosaccharides Market Analysis by Product Type
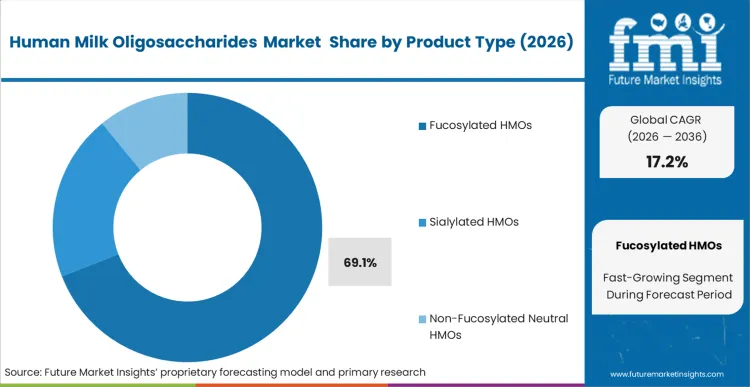
Fucosylated HMOs account for a 69.1% share of the market. 2'-FL is the most abundant oligosaccharide in human breast milk and has been among the earliest to clear complex novel food pathways across global markets. According to FMI analysis, biotech suppliers are focusing on yield improvements for this molecule to support cost-in-use parity in wider formula applications. For pediatric nutrition brands, this structure has become an important benchmark in HMO ingredient selection for infant formula. Suppliers of 2'-FL are supporting formulators as they incorporate these components into premium-positioned products. Delayed adoption may weaken brand positioning as verifiable fucosylated ingredients gain importance across higher-tier formula offerings.
- Receptor Saturation: 2'-FL molecules structurally block pathogen adhesion in the infant gut. Clinical formulators capture immediate product differentiation by claiming bio-identical defense mechanisms.
- Yield Stabilization: Advanced fermentation pipelines stabilize production costs for this specific variant. Procurement directors offset the initial premium by phasing out redundant legacy prebiotics.
- Regulatory Precedent: Extensive toxicological dossiers establish clear safety profiles for fucosylated strains. Product development teams utilize this established pathway to accelerate time-to-market for new pediatric lines.
Human Milk Oligosaccharides Market Analysis by Form
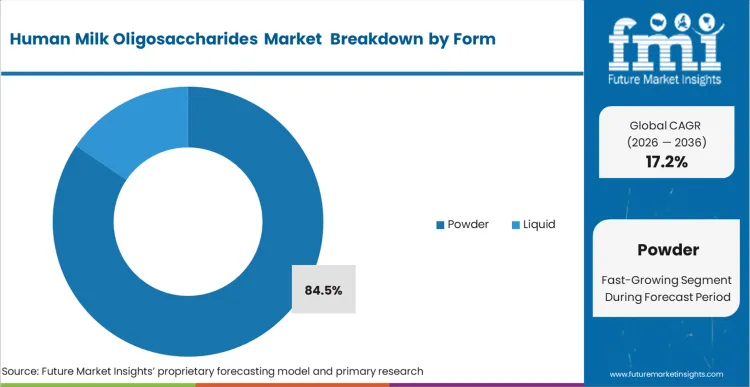
Thermal degradation during high-heat wet processing forces ingredient handlers to rely overwhelmingly on powder formats. Spray-drying realities dictate that introducing bio-active oligosaccharides too early in the standard liquid phase destroys their complex structural integrity. Powder secures 84.5% share in 2026 precisely because it aligns with dry-blending protocols at the final stages of manufacturing. Maintaining prebiotic ingredient viability requires bypassing the aggressive pasteurization steps inherent to dairy base preparation. Fermentation facilities outputting crystalline powder allow formulation plants to integrate these sensitive molecules without overhauling their existing thermal infrastructure. Facilities attempting to cut costs via liquid concentrate integration face unacceptable bioactive degradation rates and subsequent batch failures.
- Thermal Bypass: Dry formats allow operators to circumvent aggressive pasteurization heat profiles. Plant managers maintain oligosaccharide integrity by limiting thermal exposure during final blending operations.
- Hygroscopic Management: Advanced spray-drying techniques control the inherent moisture-attracting properties of pure oligosaccharides. Packaging engineers prevent caking and ensure flowability in automated dosing equipment.
- Supply Stability: Dehydrated variants dramatically extend functional shelf life compared to aqueous solutions. Supply chain directors decouple fermentation production schedules from immediate formula blending demands.
Human Milk Oligosaccharides Market Analysis by End Use
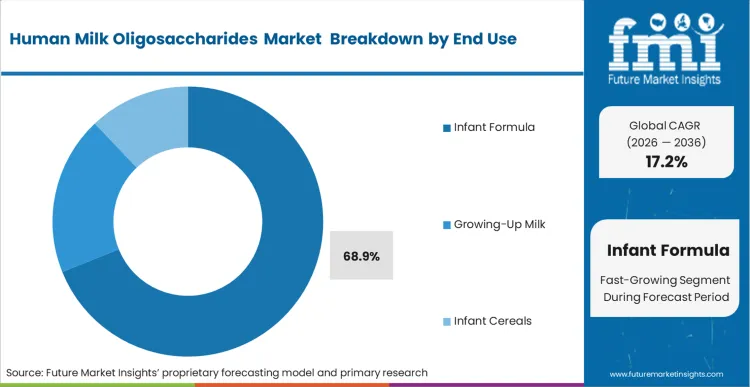
Product development teams at tier-1 dairy conglomerates are currently deciding whether to restrict complex oligosaccharides to specialized therapeutic lines or absorb the margin hit to standardize them across everyday pediatric lines. The functional performance gradient determines this path. While galacto oligosaccharide provides generic gut modulation, true bio-identical structures deliver targeted immune signaling. Infant Formula captures 68.9% share in 2026 as brands scramble to close the ultimate performance gap with natural breast milk. Evaluating the broader infant formula landscape reveals that regulatory bodies are increasingly receptive to health claims linked specifically to these complex sugars. Procurement leaders who fail to secure long-term oligosaccharide supply agreements risk watching their core HMOs in infant formula product lines become technically obsolete within a single product life cycle.
- Clinical Validation: Bio-identical structures cross the threshold from generic nutrition to targeted immune modulation. Brand managers secure premium retail shelf space by validating these specific clinical endpoints.
- Substrate Specificity: Only true human-identical oligosaccharides feed specialized bacterial strains selectively. Microbiome researchers isolate these precise interactions to substantiate next-generation health claims.
- Standardization Pressure: Global regulatory frameworks shift to recognize these molecules as standard requirements rather than novel additions. Compliance officers mandate inclusion to maintain product parity in highly regulated pediatric categories.
Human Milk Oligosaccharides Market Drivers, Restraints, and Opportunities
Accelerated novel food regulatory clearances force pediatric brand managers to upgrade baseline nutritional profiles globally. This structural pressure compels formulation teams to integrate bio-identical structures to avoid competitive disqualification in premium retail tiers. The commercial stakes involve capturing dominant share in the highly lucrative probiotic infant formula space before requisite biotech supply chains reach full capacity. Companies operating within the broader infant formula ingredients market recognize this shift as a fundamental category realignment.
Yield optimization within downstream processing creates severe batch cost volatility. This friction forces procurement teams to constantly balance inclusion rates against formula margin erosion. Continuous fermentation technologies stabilize output to some extent across the microbial fermentation HMO production market. Massive capital requirements still restrict adoption to a handful of elite biotech conglomerates, keeping bulk material costs structurally elevated.
Opportunities in the Human Milk Oligosaccharides Market
- Multi-strain Complexing: The commercialization of sialylated and non-fucosylated variants enables complete bio-identical profile matching. Advanced clinical formulation teams capture multi-HMO blend commercialization opportunities by launching elite product tiers that mimic natural milk composition entirely.
- Adult Therapeutic Application: Clinical data mapping oligosaccharide efficacy in mature microbiomes opens adjacent therapeutic categories. Medical nutrition directors bypass pediatric volume constraints by targeting adult health applications of HMOs within adult gastroenterology protocols.
- Synbiotic Pairing: Combining specific HMO strains with complementary probiotic ingredients creates self-sustaining microbiome ecosystems. Product architects capture unique early life microbiome solutions by patenting these precise strain-to-substrate combinations.
Regional Analysis
Based on the regional analysis, the human milk oligosaccharides market is segmented into The Americas, Europe, and Asia Pacific across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| Brazil | 20.9% |
| India | 17.7% |
| Japan | 17.2% |
| United Kingdom | 15.5% |
| Germany | 15.5% |
| United States | 13.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The Americas Human Milk Oligosaccharides Market Analysis
Regulatory authorization pathways shape the specific adoption timelines across the Americas. The geographic polarity between rapid GRAS notifications and complex localized novel food authorizations dictates where formulation teams deploy their capital. According to FMI's estimates, biotech suppliers anchor their primary precision fermentation capacity near established agricultural hubs to secure continuous carbon feedstock supplies. This infrastructure centralization forces pediatric nutrition brands to align their procurement strategies with highly consolidated regional biotech outputs.
- Brazil: Rapid urbanization in Brazil pushes dual-income households toward premium formula tiers. This demographic shift accelerates the phase-out of legacy products. Localized dairy joint ventures integrate these advanced prebiotics to capture ascending middle-class demographics. Brazil is poised for a Compound Annual Growth Rate of 20.9%. Early-moving formulation teams secure lucrative retail distribution contracts that remain closed to brands relying on outdated dietary fiber profiles.
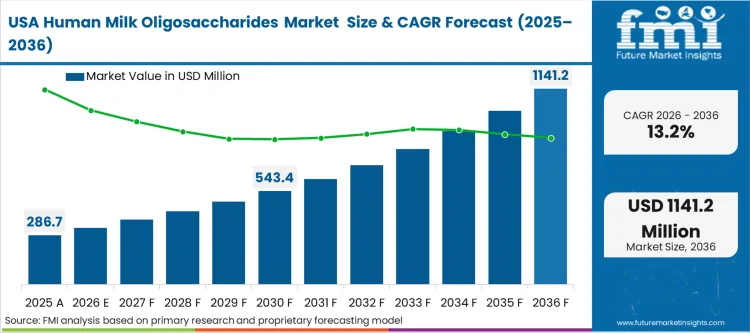
- United States: The GRAS notification process in the United States enables rapid commercial deployment of newly synthesized oligosaccharide strains. Regulatory affairs teams utilize this streamlined pathway to validate complex multi-strain blends faster than their European counterparts. FMI estimates the United States human milk oligosaccharides market to expand at an annual growth rate of 13.2%. Formulators who delay their GRAS-aligned product launches face severe market share penalties as competitors establish new baseline functional standards.
FMI's report includes Canada, Mexico, and emerging Latin American markets. The expanding footprint of regional biotech subsidiaries allows local nutrition brands to bypass costly intercontinental shipping constraints for specialized bio-ingredients.
Europe Human Milk Oligosaccharides Market Analysis
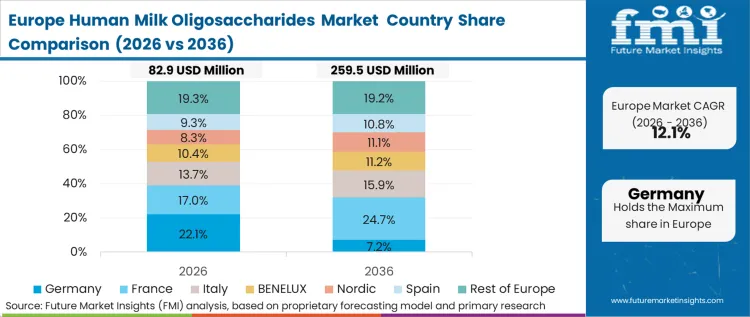
Procurement practices and stringent novel food qualification standards heavily influence European adoption patterns. The structural imperative centers on EFSA's comprehensive toxicological dossier requirements. These regulations effectively lock out low-tier or emerging biotech suppliers lacking massive regulatory capital. As per FMI's projection, this creates a highly concentrated vendor landscape where European formulation teams must secure long-term off-take agreements with established bioscience incumbents. Analyzing the Europe human milk oligosaccharides market ecosystem reveals that brand differentiation relies less on novel molecule introduction and more on securing reliable purification capacity.
- United Kingdom: Post-Brexit regulatory adaptations in the United Kingdom force pediatric brands to navigate parallel authorization frameworks. Compliance officers manage duplicated dossier submissions to maintain product parity across regional borders. Over the forecast period, demand for united kingdom human milk oligosaccharides is set for a CAGR of 15.5%. Supply chain directors spend significant resources mapping HMO regulatory approvals by countries to manage distinct label requirements efficiently.
- Germany: Germany's concentrated clinical nutrition sector demands unparalleled purity metrics and extensive clinical substantiation for pediatric claims. Procurement directors at tier-1 domestic brands enforce some of the strictest downstream quality audits in the global industry. The German landscape is set to achieve a CAGR of 15.5%. This stringent environment shields compliant domestic incumbents from low-cost international ingredient imports.
FMI's report includes France, Italy, Spain, and the broader EU bloc. Harmonized EFSA approvals eventually trigger synchronized formula upgrades across these member states, creating massive, localized demand spikes that test regional fermentation capacity.
Asia Pacific Human Milk Oligosaccharides Market Analysis
Biotech infrastructure maturity and localized clinical validation drive the transition timeline throughout the Asia Pacific. The rapid scaling of domestic precision fermentation facilities fundamentally alters the cost structure. This localized production allows regional formula brands to challenge legacy Western imports directly. In FMI's view, the integration of alpha glucan oligosaccharide alongside advanced HMOs signals a shift toward highly complex microbiome modulators. Facilities here leapfrog basic single-strain production to focus immediately on multi-strain industrial viability.
- India: Aggressive modernization of India's dairy processing infrastructure enables the integration of highly sensitive bio-ingredients into domestic product lines. Plant managers upgrade their thermal and dry-blending capabilities specifically to handle these advanced oligosaccharides without degradation. The India HMO market is forecast to register a CAGR of 17.7%. This physical infrastructure upgrade permanently transitions the domestic landscape away from basic commodity nutrition toward globally competitive pediatric formulations.
- Japan: Japan's deeply established functional food sector applies rigorous FOSHU standards to any oligosaccharide integration. Research and development teams leverage decades of advanced fermentation expertise to optimize downstream purification yields. The Japan sector is poised to expand at 17.2%. Passing these intense qualification protocols immediately simplifies secondary manufacturing processes for local brands seeking highly substantiated functional additive profiles.
FMI's report includes China, South Korea, Australia, and the ASEAN bloc. The rapid expansion of the China human milk oligosaccharides market highlights a broader regional push to decouple from Western biotech supply chains entirely by building localized Australia human milk oligosaccharides pathways.
Competitive Aligners for Market Players
Competition in the Human Milk Oligosaccharides market is increasingly centered on a relatively small group of science-led suppliers with strengths in fermentation capability, regulatory execution, and customer access in infant nutrition. dsm-firmenich, Kyowa Hakko Bio, and FrieslandCampina Ingredients stand out as key competitive anchors, as each combines HMO development with broader ingredient, formulation, or commercial infrastructure that supports scale-up and multinational supply. Novonesis also merits attention through its biosolutions strength following the combination of Novozymes and Chr. Hansen, though its role should be framed carefully depending on the product scope being discussed. Smaller or more specialized participants remain relevant in selected molecules, research-grade supply, or regional opportunities, but market influence still rests largely with companies that can move from molecule development to regulatory clearance and long-term customer integration. As a result, competition is defined less by the number of participants and more by who can translate technical capability into dependable, approval-backed commercial supply.
Key Players in Human Milk Oligosaccharides Market
- dsm-firmenich
- Kyowa Hakko Bio
- FrieslandCampina Ingredients
- Novonesis
- Novozymes
- Chr. Hansen
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 394.5 million to USD 1,925.6 million, at a CAGR of 17.20% |
| Market Definition | This category covers precision-fermented, bio-identical carbohydrate polymers that replicate human breast milk structures. It establishes the functional boundary for advanced pediatric nutrition. |
| Product Type Segmentation | Fucosylated HMOs, 2'-Fucosyllactose (2'-FL), 3-Fucosyllactose (3-FL), 2 FL DFL, Sialylated HMOs, 3'-Sialyllactose (3'-SL), 6'-Sialyllactose (6'-SL), Non-Fucosylated Neutral HMOs, Lacto-N-tetraose (LNT), Lacto-N-neotetraose (LNnT) |
| Form Segmentation | Powder, Liquid |
| End Use Segmentation | Infant Formula, Standard Infant Formula, Follow-On Formula, Specialty Infant Formula, Growing-Up Milk, Infant Cereals, Baby Foods, Nutraceutical Supplements, Gut Health Supplements, Immune Support Supplements, Cognitive Health Supplements, General Wellbeing Supplements, Sports Nutrition Products, Sports Recovery Supplements, Hydration and Electrolyte Supplements, Performer Enhancers, Protein Support Supplements, Functional Food and Beverage, Probiotic Yogurt and Dairy Products, Functional Beverages, Snacks and Baked Goods, Others (Personal Care, Research & Therapeutics, etc.) |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | US, UK, Germany, India, Brazil, Japan, and 40+ countries |
| Key Companies Profiled | dsm-firmenich, Kyowa Hakko Bio, FrieslandCampina Ingredients, Novonesis, Novozymes, Chr. Hansen |
| Forecast Period | 2026 to 2036 |
| Approach | Chief formulation officers and senior procurement directors at tier-1 pediatric nutrition companies were interviewed to map adoption timelines. Baselines anchor to installed commercial precision fermentation capacity and verifiable off-take agreements. Forecasts were triangulated against capital expenditure timelines for downstream purification facility expansions. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Human Milk Oligosaccharides Market Analysis by Segments
Product Type:
- Fucosylated HMOs
- 2'-Fucosyllactose (2'-FL)
- 3-Fucosyllactose (3-FL)
- 2 FL DFL
- Sialylated HMOs
- 3'-Sialyllactose (3'-SL)
- 6'-Sialyllactose (6'-SL)
- Non-Fucosylated Neutral HMOs
- Lacto-N-tetraose (LNT)
- Lacto-N-neotetraose (LNnT)
Form:
-
- Powder
- Liquid
End Use:
- Infant Formula
- Standard Infant Formula
- Follow-On Formula
- Specialty Infant Formula
- Growing-Up Milk
- Infant Cereals
- Baby Foods
- Nutraceutical Supplements
- Gut Health Supplements
- Immune Support Supplements
- Cognitive Health Supplements
- General Wellbeing Supplements
- Sports Nutrition Products
- Sports Recovery Supplements
- Hydration and Electrolyte Supplements
- Performer Enhancers
- Protein Support Supplements
- Functional Food and Beverage
- Probiotic Yogurt and Dairy Products
- Functional Beverages
- Snacks and Baked Goods
- Others (Personal Care, Research & Therapeutics, etc.)
Region:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- European Commission. (2024, July 29). Commission Implementing Regulation (EU) 2024/2090 of 29 July 2024 authorising the placing on the market of Lacto-N-fucopentaose I and 2'-Fucosyllactose mixture produced using a derivative strain of Escherichia coli K-12 DH1 as a novel food and amending Implementing Regulation (EU) 2017/2470. Official Journal of the European Union.
- USA Food and Drug Administration. (2026). GRAS Notice (GRN) No. 001261: Agency response letter.
- FrieslandCampina Ingredients. (2025, August 27). Ask the Expert: How can milk oligosaccharide diversity unlock elevated support for infant gut health?
- van der Woude, H., Pelgrom, S. M. J. G., Buskens, C., Hoffmans, R., Krajcs, N., & Delsing, D. J. (2024). Pre-clinical safety assessment of biotechnologically produced lacto-N-tetraose (LNT). Regulatory Toxicology and Pharmacology, 148, 105580.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Market intelligence to support strategic decision making across bio-synthetic pediatric nutrition and precision fermentation categories
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, supported by installed capacity mapping and off-take agreement validation
- Growth opportunity mapping across Fucosylated and Sialylated structures with emphasis on the transition from single-strain supplementation to multi-strain blends
- Segment and regional revenue forecasts covering dry-blended powder formats across rapid GRAS environments and complex novel food qualification zones
- Competition strategy assessment including downstream purification economics, yield stability, and localized regulatory capital
- Capability development tracking including continuous fermentation pipelines, thermal bypass blending, and multi-strain bio-identical complexes
- Market access analysis covering EFSA toxicological dossiers, FDA GRAS notifications, and FOSHU compliance standards
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive strategy, fermentation capacity planning, and operational benchmarking use
Frequently Asked Questions
How large is the demand for Human Milk Oligosaccharides in the global market in 2026?
Demand for Human Milk Oligosaccharides in the global market is estimated to be valued at USD 394.5 million in 2026.
What will be the market size of Human Milk Oligosaccharides in the global market by 2036?
Market size for Human Milk Oligosaccharides is projected to reach USD 1,925.6 million by 2036.
What is the expected demand growth for Human Milk Oligosaccharides in the global market between 2026 and 2036?
Demand for Human Milk Oligosaccharides is expected to grow at a CAGR of 17.20% between 2026 and 2036.
Which product type is poised to lead global sales by 2026?
Fucosylated HMOs account for 69.1% in 2026 as 2'-FL remains the most commercially advanced and cost-optimized structure for formula applications.
Which form is expected to dominate the global market by 2026?
Powder holds 84.5% share in 2026 as dry blending helps manufacturers protect oligosaccharide integrity during final-stage processing.
Which end use is expected to lead Human Milk Oligosaccharides demand in 2026?
Infant Formula is expected to account for 68.9% share in 2026 as pediatric brands work to narrow the functional gap with natural breast milk.
What is driving demand in Brazil?
Rapid urbanization and rising dual-income households are accelerating demand for premium infant nutrition products in Brazil.
What is the Brazil growth outlook in this report?
Brazil is projected to grow at a CAGR of 20.9% during 2026 to 2036.
What is driving demand in India?
Modernization of dairy processing infrastructure and improved dry-blending capabilities are supporting the use of advanced bio-identical oligosaccharides in India.
What is India's growth outlook in this report?
India is projected to expand at a CAGR of 17.7% during 2026 to 2036.
What is driving demand in Japan?
Japan’s strong functional food ecosystem and advanced fermentation expertise are supporting higher-value HMO integration across substantiated nutrition products.
What is the Japan growth outlook in this report?
Japan is projected to grow at a CAGR of 17.2% during 2026 to 2036.
What compliance standards or regulations are referenced for Germany?
Germany is discussed within the European framework shaped by stringent novel food qualification standards and extensive toxicological dossier requirements.
What is the Germany growth outlook in this report?
Germany is projected to expand at a CAGR of 15.5% during 2026 to 2036.
Does the report cover the United States in its regional analysis?
Yes, the United States is included within the Americas under the regional scope of analysis.
What are the sources referred to for analyzing the United States?
The analysis refers to GRAS notification pathways, regulatory clearances, installed fermentation capacity, and commercial product deployment trends.
What is the main demand theme linked to the United States in its region coverage?
Demand centers on rapid commercialization of newly synthesized oligosaccharide strains through streamlined GRAS-aligned product development.
What is the United States growth outlook in this report?
The United States is projected to grow at a CAGR of 13.2% during 2026 to 2036.
Does the report cover the United Kingdom in its regional analysis?
Yes, the United Kingdom is included within Europe under the regional coverage framework.
Yes, the United Kingdom is included within Europe under the regional coverage framework.
Post-Brexit regulatory adaptations and parallel authorization requirements are shaping HMO adoption timelines in the United Kingdom.
Post-Brexit regulatory adaptations and parallel authorization requirements are shaping HMO adoption timelines in the United Kingdom.
The United Kingdom is projected to expand at a CAGR of 15.5% during 2026 to 2036.
Why is Asia Pacific described as an important region in this report?
Asia Pacific is gaining importance as local fermentation capacity expands and regional formula brands build alternatives to Western ingredient supply chains.
Which product formats or configurations are strategically important for Asia Pacific supply chains?
Powder-based HMO formats are strategically important as they align with upgraded dry-blending and thermal-control infrastructure across regional dairy plants.
What is Human Milk Oligosaccharides and what is it mainly used for?
Human Milk Oligosaccharides are bio-identical carbohydrate structures modeled on breast milk. They are mainly used in infant formula, pediatric nutrition, and microbiome-focused nutritional products.
What does Human Milk Oligosaccharides mean in this report?
The report defines Human Milk Oligosaccharides as industrial-scale synthesized and purified oligosaccharide structures that replicate those found naturally in human milk.
What is included in the scope of this Human Milk Oligosaccharides report?
The report covers fucosylated, sialylated, and non-fucosylated neutral HMOs produced through precision fermentation or enzymatic synthesis in powder and liquid forms across infant formula, nutraceutical supplements, and functional food and beverage uses.
What is excluded from the scope of this report?
Conventional prebiotics such as galacto-oligosaccharides and fructo-oligosaccharides derived from bovine milk or plant sources are excluded from the scope.
What does market forecast mean on this page?
The market forecast represents a model-based projection built on installed commercial capacity, off-take validation, regulatory approvals, and downstream purification expansion assumptions.
How does FMI build and validate the Human Milk Oligosaccharides forecast?
FMI uses primary interviews with formulation and procurement executives, maps installed fermentation capacity, reviews regulatory dossiers and patent filings, and triangulates forecasts against purification facility expansion timelines.
What does zero reliance on speculative third-party market research mean here?
It means the analysis is grounded in primary research, regulatory documentation, patent activity, installed capacity mapping, and verifiable commercial agreements rather than unverified syndicated estimates.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Fucosylated HMOs
- Sialylated HMOs
- Non-Fucosylated Neutral HMOs
- Fucosylated HMOs
- Y-o-Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Form, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Form, 2026 to 2036
- Powder
- Liquid
- Powder
- Y-o-Y Growth Trend Analysis By Form, 2021 to 2025
- Absolute $ Opportunity Analysis By Form, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Infant Formula
- Growing-Up Milk
- Infant Cereals
- Infant Formula
- Y-o-Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Form
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Form
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Form
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Form
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Form
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Form
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Form
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Form
- By End Use
- Competition Analysis
- Competition Deep Dive
- dsm-firmenich
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Kyowa Hakko Bio
- FrieslandCampina Ingredients
- Novonesis
- Novozymes
- Chr. Hansen
- dsm-firmenich
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Form
- Figure 9: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End Use
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Product Type
- Figure 26: North America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Form
- Figure 29: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End Use
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Product Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Form
- Figure 39: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End Use
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Form
- Figure 49: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by End Use
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Form
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Product Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Form
- Figure 69: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by End Use
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Form
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Form
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

