Hydrocephalus Shunt Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
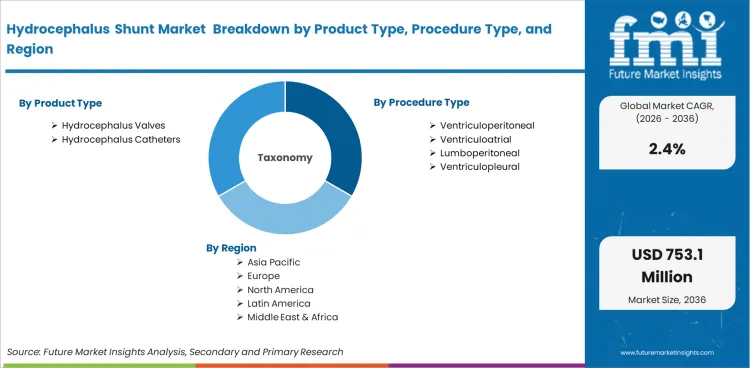
The Hydrocephalus Shunt Market is segmented by Product Type (Hydrocephalus Valves and Hydrocephalus Catheters), Procedure Type (Ventriculoperitoneal, Ventriculoatrial, Lumboperitoneal, and Ventriculopleural), Age Group (Pediatric and Adult), Valve Type (Programmable Valves and Fixed Pressure Valves), End User (Hospitals, Specialty Clinics, and Ambulatory Surgical Centers), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Hydrocephalus Shunt Market Size, Market Forecast and Outlook By FMI
Summary of the Hydrocephalus Shunt Market
- Demand and Growth Drivers
- Hydrocephalus shunt demand is supported by hospital use of reliable drainage systems for long-term cerebrospinal fluid management. Neurosurgical centers prefer systems with steady pressure regulation and established clinical familiarity.
- Programmable valve platforms are receiving clinical attention as care teams seek more control across follow-up visits. This supports demand for advanced hydrocephalus shunt products across pediatric and adult treatment use.
- Medical device standards influence supplier selection as hospitals review safety records and quality documentation. Cost pressure can slow premium adoption in countries with limited healthcare budgets.
- Product and Segment View
- Hydrocephalus valves dominate product type demand because pressure regulation requirements are important and programmable control is widely used. Hydrocephalus catheters support fluid transport across surgical drainage pathways.
- Established surgical use and widespread surgeon familiarity keep ventriculoperitoneal procedures at the front of procedure type demand. Alternative procedures serve patients with specific drainage constraints.
- Pediatric demand is important because of congenital hydrocephalus care and long follow-up cycles. Adult use supports steady demand across acquired hydrocephalus and normal pressure hydrocephalus treatment.
- Geography and Competitive Outlook
- India is expected to record 3.8% CAGR through 2036 the enhancing healthcare infrastructure and neurosurgical capacity.
- USA demand is forecast to advance at 2.5% CAGR through 2036 due to mature treatment access and medical technology adoption.
- Medtronic and Johnson & Johnson hold important positions across neurosurgical device supply.
- B. Braun and Sophysa support demand through focused shunt system capabilities.
- Integra LifeSciences competes through neurosurgical products with strong clinical use.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant at FMI says, “Hydrocephalus shunt demand is moving through a stable replacement cycle with stronger focus on pressure control and infection prevention. Hospitals value products with proven surgical use and reliable follow-up performance. Programmable valves are adding value though pricing can limit higher adoption.”
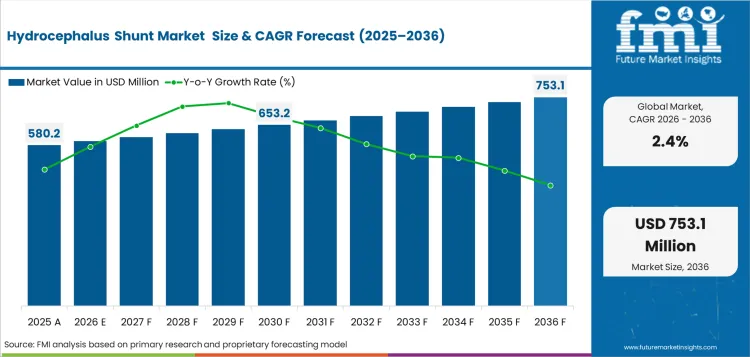
- Hydrocephalus Shunt Market Value Analysis
- The hydrocephalus shunt market is advancing from an established neurosurgical device category into a controlled long term treatment segment.
- Adoption is supported by the need for reliable cerebrospinal fluid drainage across pediatric and adult hydrocephalus care.
- The market benefits from clinical use of programmable valves for pressure adjustment during follow up treatment.
- Demand is supported by hospital preference for shunt systems with surgical reliability and infection control. Long term device performance adds more support.
Hydrocephalus Shunt Market Definition
The hydrocephalus shunt market includes implanted drainage systems used to divert cerebrospinal fluid from the brain into another body area. Product scope includes valves and catheters used across pediatric and adult hydrocephalus treatment. Device choice relies on drainage reliability, pressure control, and surgical pathway requirements.
Hydrocephalus Shunt Market Inclusions
Market scope includes hydrocephalus valves, hydrocephalus catheters, programmable valve systems, fixed pressure valves, and related shunt components. Procedure scope includes ventriculoperitoneal, ventriculoatrial, lumboperitoneal, and ventriculopleural procedures. End-user scope includes hospitals, specialty clinics, and ambulatory surgical centers with neurosurgical treatment capability.
Hydrocephalus Shunt Market Exclusions
Market scope excludes external ventricular drains used only for short-term emergency fluid management. Scope also excludes unrelated neurovascular implants and drainage products not designed for hydrocephalus care. General surgical tubes without hydrocephalus-specific clinical use are outside direct market sizing.
Hydrocephalus Shunt Market Research Methodology
- Primary research covers medical device manufacturers, hospital buyers, and neurosurgical specialists across major demand regions.
- Desk research reviews medical device regulations, treatment pathway references, and supplier product portfolios used in hydrocephalus care.
- Market sizing uses a hybrid bottom-up and top-down approach based on product type adoption and procedure type distribution.
- Data validation compares the 2026 value with the 2036 forecast and checks CAGR alignment across the ten-year window.
Why is the Hydrocephalus Shunt Market Growing?
- Neurosurgical centers need reliable drainage systems for chronic cerebrospinal fluid management across pediatric and adult patients.
- Programmable valve systems support pressure adjustment during follow-up care and reduce reliance on fixed settings.
- Antimicrobial catheter materials receive attention as hospitals focus on infection control and revision risk reduction.
- Asia Pacific demand improves as India and China add neurosurgical capacity and medical device access.
Hydrocephalus care needs durable products, as many patients require long-term monitoring after implantation. This creates steady demand for valves and catheters across hospitals with neurosurgical capability. Pressure regulation and clinical reliability give valve platforms higher product value. Catheters support every complete shunt system by enabling fluid movement from the ventricle to the target drainage area.
Clinical selection favors suppliers with proven device performance and surgical support capability. Hospitals consider documentation quality as medical device review rules tighten. Premium valve adoption can face limits in cost-sensitive markets. A measured growth outlook reflects steady treatment need and careful product selection across healthcare systems.
Market Segmentation Analysis
- Ventriculoperitoneal procedures lead procedure type demand with 93% share in 2026 due to routine surgical use.
- Strong pressure control needs position hydrocephalus valves as the leading product type with an 85% share in 2026.
- Congenital treatment demand places pediatric patients at 44.2% of age group share in 2026.
- Strong operating room infrastructure places hospitals at an estimated 72% share of end‑user demand in 2026.
- Programmable valves account for an estimated 58% valve type share in 2026, reflecting the importance of adjustable pressure settings.
The market for hydrocephalus shunts is divided into groups based on product type, procedure type, age group, valve type, end user, and region. There are hydrocephalus valves and hydrocephalus catheters under product type. By procedure type, the market includes ventriculoperitoneal, ventriculoatrial, lumboperitoneal, and ventriculopleural procedures. By age group, the market covers pediatric and adult patients. Based on valve type, it includes programmable valves and fixed pressure valves. By end user, the market is segmented into hospitals, specialty clinics, ambulatory surgical centers, rehabilitation centers, and others.
Insights into the Product Type Segment
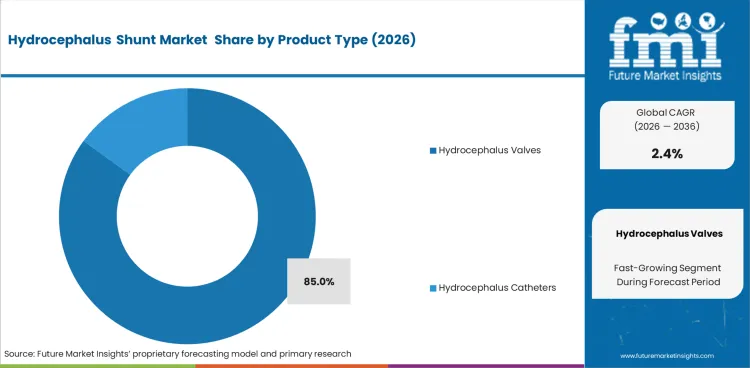
- In 2026, hydrocephalus valves account for 85.0% of product type demand. Programmable and pressure regulating valve systems form the main sub segment.
- Hydrocephalus catheters support steady demand as fluid transport and device positioning are core surgical requirements.
Insights into the Procedure Type Segment
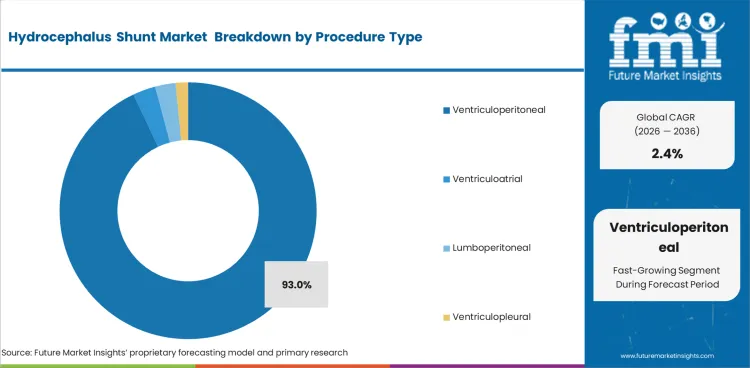
- Ventriculoperitoneal procedures account for 93.0% of procedure type demand in 2026. The biggest sub-segment is standard VP shunt placement. Surgeons prefer this pathway because it has broad clinical acceptance and strong familiarity across neurosurgical centers.
- Ventriculoatrial and lumboperitoneal procedures serve patient groups needing alternative drainage pathways under clinical limits.
Insights into the Age Group Segment
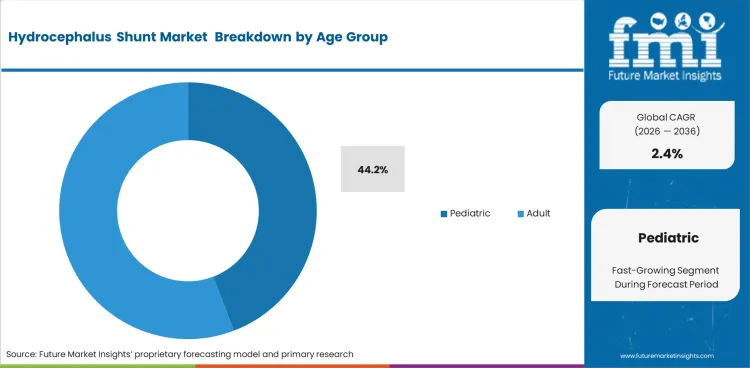
- Pediatric patients are forecast to comprise 44.2% of the total share in 2026, because congenital hydrocephalus creates early treatment needs. Long monitoring cycles add more support because many children require follow up checks through growth stages.
- Adult patients support demand through acquired hydrocephalus and age-related neurological care pathways.
Insights into the Valve Type Segment
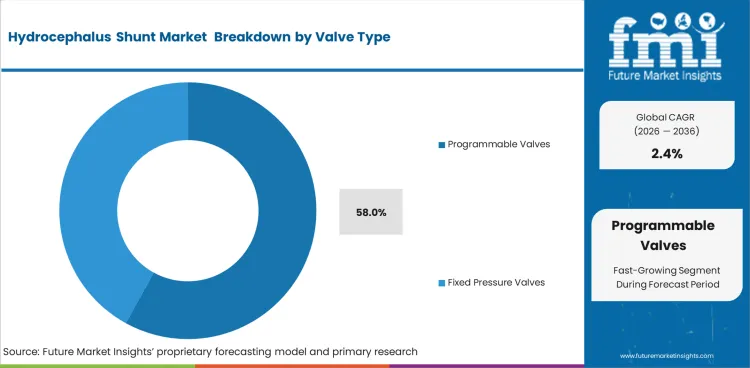
- Clinical preference for adjustable pressure control contributes to a 58% estimated share for programmable valves in 2026. Follow up pressure changes can be handled through valve adjustment, which supports use in complex long term cases.
- Fixed pressure valves maintain use in settings favoring simpler device choice and lower treatment cost.
Insights into the End User Segment
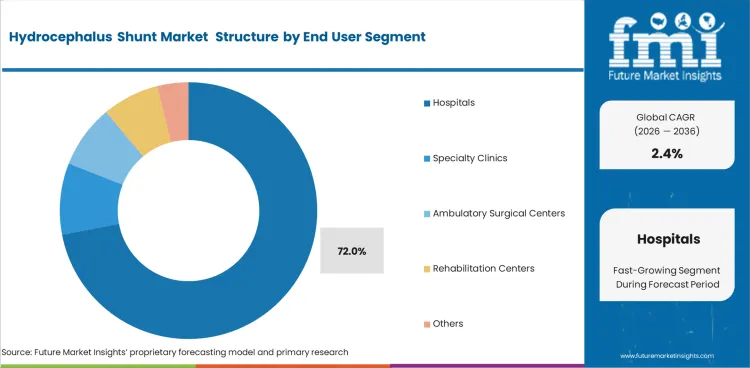
- Hospitals are forecast to command a 72% share in 2026, because shunt placement depends on neurosurgical support and operating room access. Tertiary hospitals and specialty surgical centers handle the main procedure concentration.
- Specialty clinics support follow-up visits and valve review after the primary surgical procedure is completed.
Hydrocephalus Shunt Market Drivers, Restraints, and Opportunities
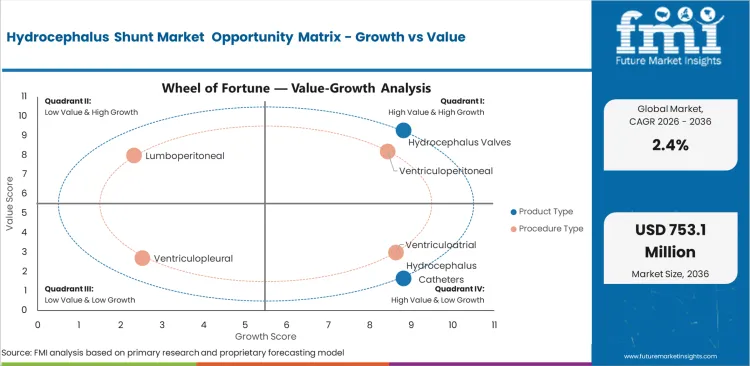
- Reliable cerebrospinal fluid drainage creates steady product need across neurosurgical centers and pediatric care use.
- Product cost can limit higher access to advanced valve systems in price-sensitive healthcare markets.
- Programmable valves create opportunity for suppliers with clinical support and device adjustment capability.
- Antimicrobial catheter adoption can increase as hospitals focus on infection prevention and revision reduction.
Hydrocephalus shunt systems are growing as hospitals need reliable cerebrospinal fluid drainage and precise pressure control across pediatric and adult care. Cost pressure and revision risk can slow adoption of advanced programmable valves. Growth is supported by programmable shunt systems and antimicrobial catheter applications.
Programmable Valve Adoption
Programmable valve systems support pressure adjustment after implantation and help clinicians manage long-term care. Advanced valves align with broader adoption of minimally invasive neurosurgery devices in modern neurosurgical departments. Supplier value rises as training support and product reliability help hospitals manage complex cases.
Infection Prevention Opportunity
Antimicrobial catheter support product differentiation across hospitals focused on complication reduction. Infection risk can raise revision burden and treatment cost across long-term care. Product selection favors material quality and clinical evidence. Enhanced use of ventricular catheters supports the role of catheter performance in neurocritical care.
Analysis of Hydrocephalus Shunt Market by Key Countries
.webp)
| Country | CAGR through 2036 |
|---|---|
| India | 3.8% |
| China | 3.5% |
| USA | 2.5% |
| Germany | 2.1% |
| Japan | 2.0% |
Source: Future Market Insights, 2026.
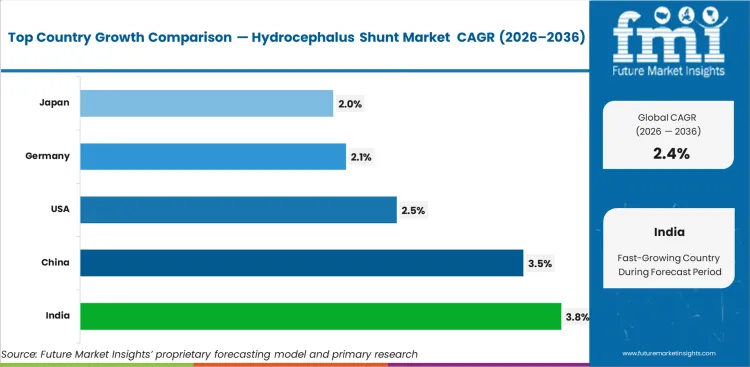
Hydrocephalus Shunt Market CAGR Analysis by Country
- India records the highest listed CAGR at 3.8% through 2036 as healthcare infrastructure expansion improves neurosurgical access.
- China follows at 3.5% CAGR between 2026 to 2036 as urban hospitals increase adoption of neurosurgical treatment.
- USA advances at 2.5% CAGR through 2036, supported by mature healthcare systems and device replacement demand.
- Germany records 2.1% CAGR by 2036, and Japan follows at 2.0% CAGR through 2036, reflecting mature clinical access.
Global hydrocephalus shunt demand is expected to expand at 2.4% CAGR from 2026 to 2036. Country-level demand varies with neurosurgical capacity and reimbursement access.
Hospital device budgets also influence adoption across different healthcare systems.
Emerging healthcare systems record a higher percentage expansion as treatment access improves. Mature countries follow a steadier path with existing adoption and strict product review.
Demand Outlook for Hydrocephalus Shunt Market in India
India is projected to expand at 3.8% CAGR through 2036 as hospitals add neurosurgical treatment capacity. Medical device access is improving across major cities and specialty care centers. Shunt suppliers benefit from use of advanced drainage systems across pediatric and adult care. Local adoption relies on service support and stable product availability.
- Neurosurgical capacity expansion supports increased use of shunt systems across specialty hospitals.
- Medical device adoption improves as hospital networks expand advanced treatment capability.
- Supplier access relies on training support and consistent distribution across high-volume centers.
Future Outlook for Hydrocephalus Shunt Market in China
China is estimated to register 3.5% CAGR during the forecast period due to healthcare modernization and neurosurgical access are improving. Urban medical centers are shifting toward standardized shunt systems for wider patient inclusion. Device suppliers must balance pricing needs with reliable product performance. Hospital purchasing favors established products with clear clinical documentation.
- Advances in healthcare modernization support expanded use of advanced shunt systems in urban hospitals.
- Medical device consumption increases as treatment capacity expands across neurosurgical departments.
- Standardized drainage systems gain preference across large treatment networks.
Demand Assessment for Hydrocephalus Shunt Market in the USA
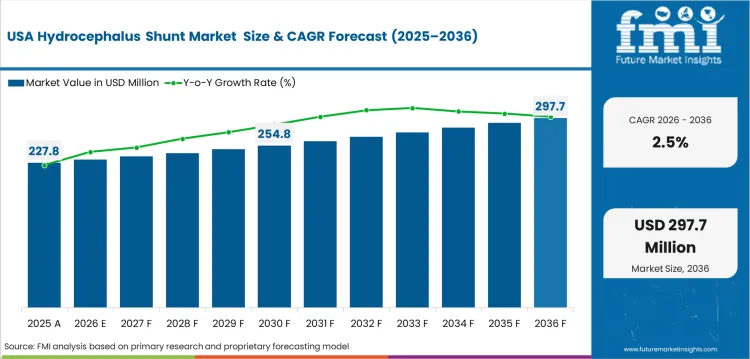
USA demand is expected to advance at 2.5% CAGR by 2036, supported by established neurosurgical care access. Hospitals use advanced valve and catheter systems across pediatric and adult procedures. Established market penetration and strict regulatory review keep demand measured. Supplier competition centers on product reliability and surgeon familiarity.
- Established healthcare infrastructure supports continued use of specialized shunt systems.
- Regulatory review keeps product evidence and safety documentation central to supplier selection.
- Medical device portfolios support hospital access across neurosurgical treatment centers.
Opportunity Analysis of Hydrocephalus Shunt Market in Germany
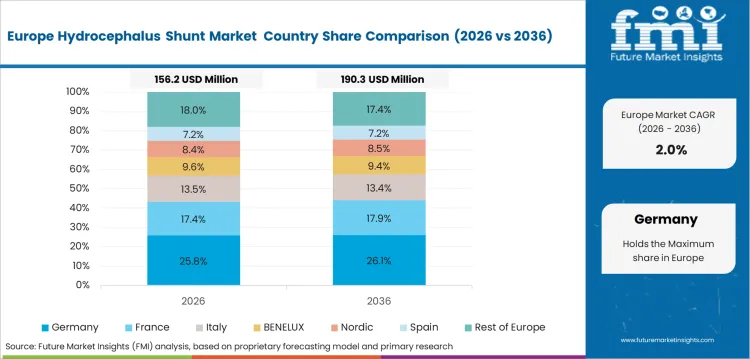
Germany is projected to expand at 2.1% CAGR from 2026 to 2036, pushed forward by advanced clinical infrastructure and high-quality expectations. Hospitals value validated products with reliable pressure control. Established treatment access keeps demand measured. Suppliers need clear documentation and technical support to meet hospital review requirements.
- Advanced medical infrastructure supports premium shunt systems across neurosurgical centers.
- Quality review requirements increase the value of validated device performance.
- Clinical networks favor suppliers with reliable support and consistent product quality.
In-depth Analysis of Hydrocephalus Shunt Market in Japan
Japan is expected to record 2.0% CAGR over the forecast years as adult neurological care supports stable demand. Aging demographics increase attention on normal pressure hydrocephalus treatment. Hospitals use advanced medical systems and careful device evaluation. Supplier performance relies on consistent quality and physician confidence.
- Aging patient needs support adult hydrocephalus care across established hospitals.
- Advanced healthcare systems favor reliable shunt technologies and documented device safety.
- Controlled medical device adoption supports steady use across long-term care pathways.
Competitive Landscape and Strategic Positioning
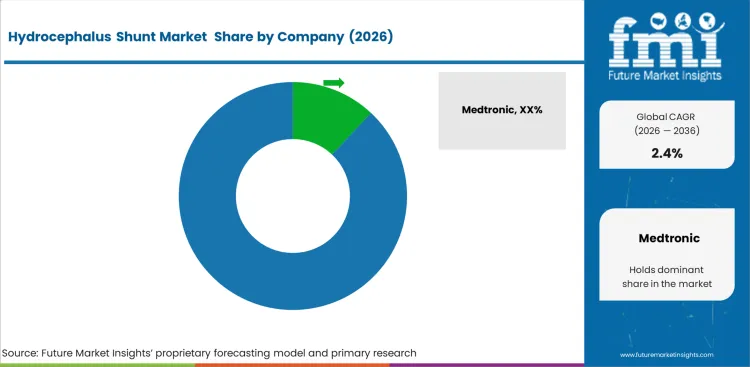
- Medtronic leads visibility through a broad neurosurgical device portfolio and global healthcare access.
- Johnson & Johnson and B. Braun compete through established medical device portfolios and hospital relationships.
- Sophysa and Integra LifeSciences focus on neurosurgical products with pressure control and clinical reliability.
Competition is moderately concentrated as established medical device companies hold supplier access across hospitals. Product differentiation emphasizes valve control and catheter reliability as core performance attributes. Clinical support is another factor that influences adoption and use over time. Large suppliers benefit from quality systems and global distribution depth. Regional specialists compete through focused valve platforms and technical service.
Hydrocephalus shunt suppliers face strict device review and high expectations for patient safety. Hospitals evaluate pressure regulation and material quality before making product selection decisions. Long-term clinical performance matters as well because hospitals need safe and sustained treatment outcomes. Broader medical device technology adoption supports supplier investment in validation and production control. Evidence-backed product performance is central to account retention.
Device makers compete through training support and reliable supply. Hospitals need products available across surgery and follow-up care settings. Shunt portfolios connected to neurotech devices stand to gain from broader neurology investment. Product quality and service responsiveness influence access across larger medical networks.
Key Companies in the Hydrocephalus Shunt Market
Key global companies leading the hydrocephalus shunt market include:
- Medtronic and Johnson & Johnson hold visible positions through broad medical device portfolios and neurosurgical access. Both companies support hospitals seeking reliable shunt products with established clinical pathways.
- B. Braun and Integra LifeSciences compete through healthcare device experience and neurosurgical product lines. Their strengths lie in quality systems, product availability, and hospital supply relationships.
- Sophysa, Natus Medical, and Spiegelberg serve focused neurosurgical demand through specialized device capabilities. These companies compete through product depth and technical understanding across selected treatment settings.
Competitive Benchmarking: Hydrocephalus Shunt Market
| Company | Valve Portfolio | Catheter Offering | Neurosurgery Access | Geographic Footprint |
|---|---|---|---|---|
| Medtronic | High | High | Strong | Global |
| Johnson & Johnson | Medium | Medium | Strong | Global |
| B. Braun | Medium | High | Strong | Global |
| Sophysa | High | Medium | Strong | Europe and Global |
| Integra LifeSciences | High | Medium | Strong | Global |
| Natus Medical | Medium | Medium | Moderate | North America and Global |
| Spiegelberg | Medium | Medium | Moderate | Europe |
Source: Future Market Insights, 2026.
Key Developments in Hydrocephalus Shunt Market
- In April 2025, CereVasc received FDA Breakthrough Device Designation for its investigational eShunt® System for communicating hydrocephalus in pediatric patients aged 12 and older.
- In June 2024, Anuncia Medical announced the first USA implant of its FDA-cleared ReFlow™ Mini Flusher with a sonolucent cranial plate to support noninvasive visualization and flushing of hydrocephalus shunts.
Key Players in the Hydrocephalus Shunt Market
Major Global Players:
- Medtronic
- Johnson & Johnson
- B. Braun Melsungen AG
- Sophysa
- Integra LifeSciences
Specialized and Adjacent Participants:
- Natus Medical
- Spiegelberg GmbH & Co. KG
- G. Surgiwear Ltd.
- Möller Medical GmbH
- Dispomedica GmbH
Report Scope and Coverage
| Parameter | Details |
|---|---|
| Quantitative Units | USD 580.2 million to USD 735.8 million at 2.4% CAGR |
| Market Definition | Implanted drainage systems used for cerebrospinal fluid diversion in hydrocephalus treatment |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | India, China, USA, Germany, Japan, and additional global countries |
| Key Companies Profiled | Medtronic, Johnson & Johnson, B. Braun, Sophysa, Integra LifeSciences, Natus Medical, Spiegelberg |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology based on product demand and procedure adoption |
Hydrocephalus Shunt Market by Segments
Hydrocephalus Shunt Market Segmented by Product Type:
- Hydrocephalus Valves
- Hydrocephalus Catheters
Hydrocephalus Shunt Market Segmented by Procedure Type:
- Ventriculoperitoneal
- Ventriculoatrial
- Lumboperitoneal
- Ventriculopleural
Hydrocephalus Shunt Market Segmented by Age Group:
- Pediatric
- Adult
Hydrocephalus Shunt Market Segmented by Valve Type:
- Programmable Valves
- Fixed Pressure Valves
Hydrocephalus Shunt Market Segmented by End User:
- Hospitals
- Specialty Clinics
- Ambulatory Surgical Centers
- Rehabilitation Centers
- Others
Hydrocephalus Shunt Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Europe
- Germany
- UK
- Italy
- Spain
- France
- Rest of Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East and Africa
- Saudi Arabia
- Other GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Sources and Bibliography
- Anuncia Medical. (2024, June 6). Anuncia Medical announces first successful US implant of the company’s ReFlow™ device with sonolucent cranial plate to noninvasively visualize flushing of hydrocephalus shunts.
- CereVasc. (2025, April 23). CereVasc, Inc. receives FDA breakthrough device designation for the eShunt® System for use in pediatric patients.
- Anwar, F., Zhang, K., Sun, C., Pang, M., Zhou, W., Li, H., He, R., Liu, X., & Ming, D. (2024, December). Hydrocephalus: An update on latest progress in pathophysiological and therapeutic research. Biomedicine & Pharmacotherapy, 181, 117702.
- Panagopoulos, D., Stranjalis, G., Gavra, M., Boviatsis, E., Korfias, S., & Karydakis, P. (2024). Current trends in the treatment of pediatric hydrocephalus: A narrative review centered on the indications, safety, efficacy, and long-term outcomes of available treatment modalities. Children, 11(11), 1334.
- Gencer, A. H., Schwarm, F. P., Nagl, J., Uhl, E., & Kolodziej, M. A. (2024, September 17). The benefits of ventriculoperitoneal shunting in normal pressure hydrocephalus patients: A follow-up of three years. Acta Neurochirurgica, 166(1), 372.
- Wach, J., Güresir, A., Güresir, E., & Vychopen, M. (2024, November 11). Survival after shunt therapy in normal-pressure hydrocephalus: A meta-analysis of 1614 patients. Neurology International, 16(6), 1438–1450.
This Report Answers
- What is the current and future size of the hydrocephalus shunt market through 2036?
- How fast is the hydrocephalus shunt market expected to expand between 2026 and 2036?
- Which product type is likely to lead the hydrocephalus shunt market by 2026?
- Which procedure type is expected to account for the highest demand by 2026?
- What factors are driving demand for hydrocephalus shunts across hospitals and specialty centers?
- How are programmable valves influencing device selection across long-term hydrocephalus care?
- Which countries are projected to record stronger CAGR through 2036?
- Who are the key companies active in the hydrocephalus shunt market?
- How does FMI estimate and validate the hydrocephalus shunt market forecast?
Frequently Asked Questions
What is the global market demand for Hydrocephalus Shunt in 2026?
In 2026, the global market for hydrocephalus shunt is expected to be worth USD 580.2 million.
How big will the market for Hydrocephalus Shunt be in 2036?
By 2036, the market for hydrocephalus shunt is expected to be worth USD 735.8 million.
How much is demand for Hydrocephalus Shunt expected to expand between 2026 and 2036?
Between 2026 and 2036, hydrocephalus shunt demand is expected to expand at 2.4% CAGR.
Which procedure type segment is likely to lead global demand by 2026?
Ventriculoperitoneal procedures will account for 93% share in 2026 because of broad clinical acceptance.
Which product type segment is expected to lead the hydrocephalus shunt market?
Hydrocephalus valves are expected to account for 85% share in 2026 due to pressure control needs.
What is supporting demand in India?
India is projected to expand at 3.8% CAGR through 2036 as neurosurgical access and healthcare infrastructure improve.
What does this report mean by Hydrocephalus Shunt Market definition?
The market includes implanted valve and catheter systems used for cerebrospinal fluid diversion in hydrocephalus treatment.
How does FMI make the Hydrocephalus Shunt forecast and check it?
Forecasting uses product adoption, procedure distribution, and regional demand checks to validate the 2026 to 2036 outlook.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Hydrocephalus Valves
- Hydrocephalus Catheters
- Hydrocephalus Valves
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Procedure Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Procedure Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Procedure Type, 2026 to 2036
- Ventriculoperitoneal
- Ventriculoatrial
- Lumboperitoneal
- Ventriculopleural
- Ventriculoperitoneal
- Y to o to Y Growth Trend Analysis By Procedure Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Procedure Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Age Group
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Age Group, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Age Group, 2026 to 2036
- Pediatric
- Adult
- Pediatric
- Y to o to Y Growth Trend Analysis By Age Group, 2021 to 2025
- Absolute $ Opportunity Analysis By Age Group, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Valve Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Valve Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Valve Type, 2026 to 2036
- Programmable Valves
- Fixed Pressure Valves
- Programmable Valves
- Y to o to Y Growth Trend Analysis By Valve Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Valve Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Specialty Clinics
- Ambulatory Surgical Centers
- Rehabilitation Centers
- Others
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Procedure Type
- By Age Group
- By Valve Type
- By End User
- Competition Analysis
- Competition Deep Dive
- Medtronic
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Johnson & Johnson
- B. Braun Melsungen AG
- Sophysa
- Integra LifeSciences
- Natus Medical
- Spiegelberg GmbH & Co. KG
- G. Surgiwear Ltd.
- Möller Medical GmbH
- Dispomedica GmbH
- Medtronic
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Procedure Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Age Group, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Valve Type, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Procedure Type, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Age Group, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Valve Type, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Procedure Type, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Age Group, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Valve Type, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Procedure Type, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Age Group, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Valve Type, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Procedure Type, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Age Group, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Valve Type, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Procedure Type, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Age Group, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Valve Type, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Procedure Type, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Age Group, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Valve Type, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Procedure Type, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Age Group, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Valve Type, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Procedure Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Procedure Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Procedure Type
- Figure 9: Global Market Value Share and BPS Analysis by Age Group, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Age Group, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Age Group
- Figure 12: Global Market Value Share and BPS Analysis by Valve Type, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Valve Type, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Valve Type
- Figure 15: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by End User
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Product Type
- Figure 32: North America Market Value Share and BPS Analysis by Procedure Type, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Procedure Type, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Procedure Type
- Figure 35: North America Market Value Share and BPS Analysis by Age Group, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Age Group, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Age Group
- Figure 38: North America Market Value Share and BPS Analysis by Valve Type, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Valve Type, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Valve Type
- Figure 41: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by End User
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Product Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Procedure Type, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Procedure Type, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Procedure Type
- Figure 51: Latin America Market Value Share and BPS Analysis by Age Group, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Age Group, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Age Group
- Figure 54: Latin America Market Value Share and BPS Analysis by Valve Type, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Valve Type, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Valve Type
- Figure 57: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by End User
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Procedure Type, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Procedure Type, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Procedure Type
- Figure 67: Western Europe Market Value Share and BPS Analysis by Age Group, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Age Group, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Age Group
- Figure 70: Western Europe Market Value Share and BPS Analysis by Valve Type, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Valve Type, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Valve Type
- Figure 73: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by End User
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Procedure Type, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Procedure Type, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Procedure Type
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Age Group, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Age Group, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Age Group
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Valve Type, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Valve Type, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Valve Type
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by End User
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Product Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Procedure Type, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Procedure Type, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Procedure Type
- Figure 99: East Asia Market Value Share and BPS Analysis by Age Group, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Age Group, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Age Group
- Figure 102: East Asia Market Value Share and BPS Analysis by Valve Type, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Valve Type, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Valve Type
- Figure 105: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by End User
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Procedure Type, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Procedure Type, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Procedure Type
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Age Group, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Age Group, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Age Group
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Valve Type, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Valve Type, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Valve Type
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Procedure Type, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Procedure Type, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Procedure Type
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Age Group, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Age Group, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Age Group
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Valve Type, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Valve Type, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Valve Type
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

