Liposomes Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Liposomes Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Liposomes Market Forecast and Outlook 2025 to 2035
The global liposomes demand is valued at USD 28.4 million in 2025 and is projected to reach USD 68.9 million by 2035, registering a CAGR of 9.3%. Demand increases as formulators seek efficient delivery systems capable of enhancing penetration, stability, and controlled release of active ingredients in topical products. Liposomal encapsulation helps preserve ingredient potency and reduce irritation risk in formulations containing sensitive or high-efficacy actives.
Skin care remains the leading application. Liposomes are used in serums, creams, and targeted treatments to support moisturisation, barrier reinforcement, and antioxidant delivery. Their use is widespread in anti-ageing, brightening, and dermatology-adjacent products where measurable improvements in dermal absorption are prioritized. Brands highlight liposomal systems as a scientific differentiator in premium product segments.
Quick Stats for Global Liposomes Demand
- Global Liposomes Sales Value (2025): USD 28.4 million
- Global Liposomes Forecast Value (2035): USD 68.9 million
- Global Liposomes Forecast CAGR (2025 to 2035): 9.3%
- Leading Application in Global Liposomes Demand: Skin Care
- Key Growth Regions in Global Liposomes Demand: Asia Pacific, Europe, North America
- Top Players in Global Liposomes Demand: BASF SE, Croda International, Evonik Industries, Symrise, Dow
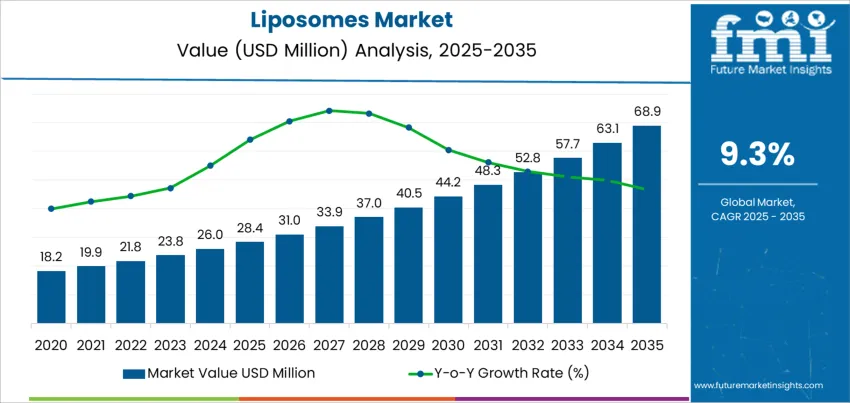
What is the Growth Forecast for the Liposomes Market through 2035?
Asia Pacific, Europe, and North America represent key growth regions driven by advanced cosmetic manufacturing capabilities, rising investment in dermatological innovation, and continued adoption of encapsulation technologies. Uptake is strongest in regions with demand for clinically validated ingredients and structured formulation performance claims. Key suppliers include BASF SE, Croda International, Evonik Industries, Symrise, and Dow. Their current focus includes scalable lipid-encapsulation platforms, improved stability under cosmetic formulation stressors, and compatibility with natural-origin ingredients to align with evolving regulatory and clean-label expectations.
A ten-year growth comparison for the global liposomes industry shows a shift from foundational expansion to more diversified and innovation-driven progress. Over the early part of the period, growth is primarily supported by established pharmaceutical applications such as oncology drug delivery and vaccines, where liposomal formulations improve therapeutic stability and targeted delivery. This stage reflects consistent volume expansion tied to regulatory approvals and broader clinical adoption.
Later-period growth is influenced by accelerated penetration into nutraceuticals, dermatology, and cosmetic actives. These categories introduce new revenue channels with shorter development cycles and greater product variation, resulting in stronger incremental gains. Consumer preference for enhanced bioavailability in vitamins, anti-aging serums, and botanical actives allows growth to remain resilient following the early-stage base establishment.
The comparison indicates that the growth slope steepens toward the second half of the forecast timeline as the industry transitions from specialized, high-cost pharmaceutical uses to wider accessibility across consumer health markets. The ten-year view highlights compound gains driven by manufacturing scale-up and advances in encapsulation technologies.
Liposomes Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2025) | USD 28.4 million |
| Market Forecast Value (2035) | USD 68.9 million |
| Forecast CAGR (2025-2035) | 9.3% |
Why is the Demand for Liposomes Growing Globally?
Demand for liposomes is increasing worldwide because pharmaceutical and cosmetic manufacturers require delivery systems that improve stability and targeted absorption of active ingredients. Liposomes encapsulate drugs or nutrients within a lipid bilayer structure, which enhances bioavailability and reduces irritation for sensitive compounds. This technology supports controlled release in cancer therapy, vaccines and anti-inflammatory treatments, expanding adoption across clinical applications.
Cosmetic brands integrate liposomes into serums, moisturizers and eye care products to deliver vitamins, peptides and botanical extracts deeper into the skin. The trend toward premium skincare with scientifically supported mechanisms strengthens demand in both advanced and consumer level product lines. Nutraceutical developers also use liposomal formats to improve uptake of vitamins and antioxidants in liquid supplements intended for daily use.
Growth in biologics manufacturing encourages investment in liposome based drug carriers that protect fragile molecules during transport within the body. Research collaborations between pharmaceutical companies and material scientists continue to broaden therapeutic uses. Constraints include high production cost, complex quality control requirements and the need for specialized equipment. Regulatory evaluation can extend development timelines in drug applications, particularly when seeking approval for new formulations.
Which End-Use Applications and Product Forms Lead Global Demand for Liposomes?
Global demand for liposomes is driven by rising formulation needs for improved skin penetration, enhanced ingredient stability, and controlled release of active molecules. Liposomal delivery supports anti-aging, hydration, barrier repair, and sensitive-skin performance. Premium skincare and hybrid wellness segments continue to adopt encapsulated actives for measurable clinical benefits. Expansion in men’s grooming, baby care, and professional dermatology treatments supports broader product diversification, while innovation in bio-based and non-irritating lipid carriers strengthens clean-label positioning across mass and luxury categories.
By End-Use Application, Skin Care Leads Global Demand
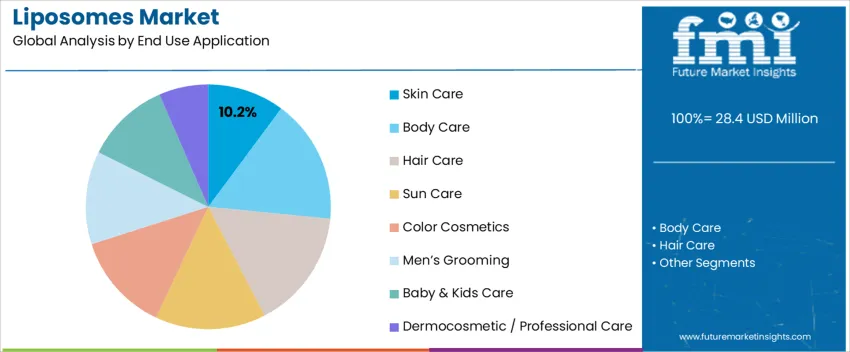
Skin care holds 10.2%, reflecting liposomes’ strong role in dermal absorption enhancement for retinoids, vitamins, peptides, and botanical extracts. These encapsulated systems improve stability and reduce irritation, supporting fast growth in anti-aging and sensitive-skin products. Body-care products leverage liposomes for firming and moisture-locking functions, while hair-care formulations use them in scalp therapies and follicle-nourishing serums. Sun-care utilizes liposomes for UV-filter uniformity and soothing actives. Color cosmetics benefit from enhanced pigment dispersion and longer wear. Men’s grooming emphasizes barrier strengthening for post-shave sensitivity, and baby/kids care integrates gentler encapsulation for mild actives. Dermocosmetic intervention remains key for targeted treatment regimens.
Key Points:
- Anti-aging and sensitivity care drive adoption in skin-focused SKUs
- Lipid-encapsulation improves delivery of unstable active ingredients
- Growing clinical use supports advanced dermocosmetic positioning
By Product Form, Powder Format Represents the Largest Share
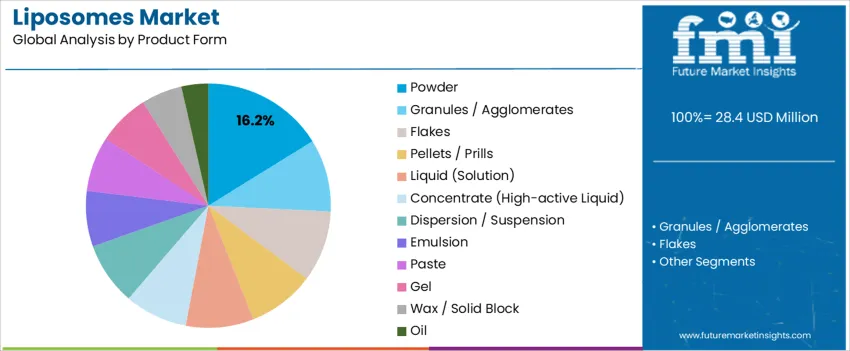
Powder format accounts for 16.2%, supported by long-term stability, simplified storage, and efficient transport for global manufacturing networks. Powders provide flexible incorporation in serums, emulsions, and masks without rapid degradation risks. Liquid solutions enable direct infusion in high-precision dermocosmetic systems requiring immediate bioavailability. Concentrates deliver potent dosing for professionals and high-efficacy products. Emulsions, dispersions, and gels enhance sensorial aesthetics and maintain encapsulation during application. Oil-based forms serve barrier repair and hair-conditioning systems. Form selection is guided by active sensitivity, delivery pathway, and compatibility with preservative-light formulations.
Key Points:
- Powders lead due to stability and easier formulation handling
- Liquids and concentrates support premium, fast-acting skincare
- Form choice depends on release control and ingredient protection
What are the Drivers, Restraints, and Key Trends of the Global Liposomes Market?
Growth of advanced drug delivery demand, rising investment in cosmetic and personal care formulations and expansion of nutraceutical encapsulation are driving demand.
Globally, liposomes are increasingly used in pharmaceuticals to deliver drugs with improved bioavailability, targeted release and reduced toxicity. Encapsulation helps protect sensitive molecules such as biologics, RNA-based therapies and hydrophobic drugs, during systemic delivery. Cosmetic and skincare industries worldwide adopt liposomal creams and serums because liposomes enhance skin absorption of actives like vitamins, antioxidants and moisturizing agents. Nutraceutical manufacturers use liposome encapsulation for omega-3s, herbal extracts and probiotics to improve stability and absorption in functional foods and dietary supplements. These widespread applications support sustained procurement across multiple sectors from clinical pharmaceuticals to consumer wellness.
Complex manufacturing requirements, high production costs and regulatory challenges around stability and reproducibility restrain growth.
Manufacturing liposomes requires strict process control, sterile production conditions and specialized equipment such as homogenizers and ultracentrifuges. These technical demands increase cost compared with standard formulations. Ensuring batch-to-batch reproducibility and stability during storage or transport presents challenges that complicate scale-up. Regulatory bodies in different regions often require extensive data on pharmacokinetics, safety and stability before approving liposomal drug or supplement claims. These complexity and compliance burdens can discourage smaller manufacturers from entering or expanding liposome-based product lines.
Shift toward targeted and controlled-release liposomes, increased demand in mRNA and gene therapies, and rising use in cosmeceuticals and functional foods define key trends.
Research and development investments in liposome formulations for mRNA vaccines, gene therapy delivery and cancer treatments accelerate adoption in pharmaceutical sectors. Controlled-release liposomes tailored for tissue-specific delivery are gaining clinical value, especially in oncology and chronic disease management. Cosmetic and cosmeceutical brands expand liposome-based topical products claiming improved transdermal delivery of active compounds.
Functional-food producers integrate liposome-encapsulated nutrients to boost absorption while preserving taste and shelf life. Hybrid liposome-nanoparticle systems combining benefits of both platforms emerge for advanced therapy development. These developments indicate strong, innovation-driven demand for liposomes in pharmaceutical, personal care and wellness industries globally.
Analysis of the Liposomes Market by Key Country
The global liposomes market is expanding due to increasing deployment of nanocarrier drug-delivery systems, skin-care encapsulation, and improved bioavailability for active pharmaceutical ingredients. Growth is reinforced by oncology therapy adoption, rising prevalence of chronic diseases, and continued investment in lipid nanoparticle (LNP) technologies. India leads at 12.5% CAGR, followed by China at 11.6% due to strong bio manufacturing infrastructure. Japan grows at 10.7% with advanced clinical research programs, while the United Kingdom records 9.7% driven by pharmaceutical innovation. Germany expands at 8.8% with high GMP standards and hospital procurement, and the United States grows at 7.9% through personalized-medicine advancements and regenerative dermatology products.
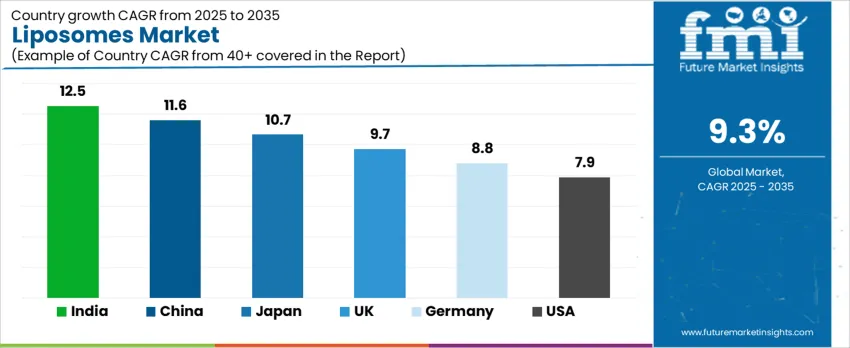
| Country | CAGR (%) |
|---|---|
| India | 12.5% |
| China | 11.6% |
| Japan | 10.7% |
| UK | 9.7% |
| Germany | 8.8% |
| USA | 7.9% |
How is India leading the growth of the Liposomes Market?
India grows at 12.5% CAGR, driven by rapid expansion of pharmaceutical manufacturing and rising integration of liposomal formulations in cancer therapy and infectious-disease treatment. Domestic drug developers apply liposome delivery systems to enhance bioavailability of chemotherapeutics, antifungals, and vaccines, reducing toxicity and improving targeted release. Dermatology companies incorporate liposome-encapsulated actives in anti-aging, hydration, and depigmentation product lines to strengthen cosmeceutical penetration in retail channels.
Contract development and manufacturing organizations (CDMOs) offer competitive export pricing with scale-up capabilities for lipid-nanoparticle systems aligned to global quality standards. Government healthcare initiatives increase adoption of advanced therapeutics across metropolitan hospitals. Clinical studies progress toward heat-sensitive and pH-responsive liposomes enabling site-specific delivery.
- Oncology formulations and antifungals driving volume
- Cosmeceutical demand raising liposome-encapsulation needs
- CDMO capacity supporting global outsourcing
- Hospital adoption aided by expanding reimbursement coverage
How is China strengthening liposomal drug-delivery capabilities?
China achieves 11.6% CAGR, supported by a strong biologics ecosystem and national investment into nanomedicine and vaccine delivery platforms. Pharmaceutical companies integrate liposomal carriers for targeted chemotherapy and sustained-release formulations addressing high cancer incidence rates. Domestic skincare brands adopt liposomal actives to improve skin-penetration efficacy in beauty serums and dermal patches, expanding mass-market acceptance.
Government programs enhance GMP capability for lipid-nanoparticle production used in mRNA and prophylactic vaccines, creating strategic manufacturing depth post-pandemic. Export-oriented suppliers scale production of encapsulated nutraceuticals and bioactives with validated diffusion profiles. Procurement groups emphasize batch-consistency controls, removing adoption barriers in hospital networks.
- Nanomedicine investment growing clinical applications
- Dermatology formulations improving consumer access
- LNP capability rising with vaccine-linked innovation
- Manufacturing scale supporting export competitiveness
How is Japan advancing precision therapeutics with liposomal systems?
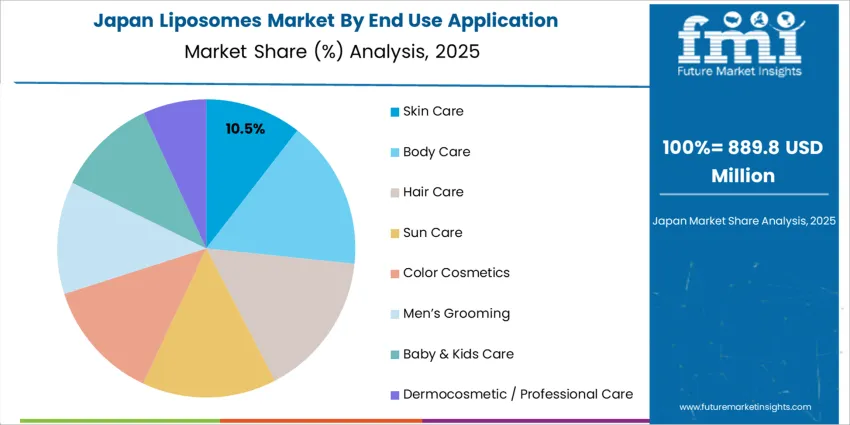
Japan expands at 10.7% CAGR, driven by pharmaceutical innovation aligned with precision oncology, CNS disorders, and metabolic-disease therapies. Hospitals use liposome-enabled targeted delivery to reduce systemic toxicity and improve response rates in complex treatment regimens. Research institutions validate long-circulation liposomes and PEG-stabilized systems designed for controlled pharmacokinetics. Cosmeceutical manufacturers deploy liposomal vitamin complexes and biomimetic lipids in aging-care products suited to Japan’s senior population focus. Partnerships between drug makers and universities accelerate clinical-translation timelines. Procurement teams evaluate sterility, particle-size uniformity, and durability of encapsulation when selecting platforms for therapeutic pipelines.
- Precision-medicine research strengthening clinical deployment
- Highly regulated production maintaining formulation integrity
- Senior demographic fueling anti-aging dermal innovations
- Academic-industry collaboration advancing targeted therapies
How is the United Kingdom shaping nanocarrier adoption?
The United Kingdom records 9.7% CAGR, propelled by strong pharmaceutical research clusters and government support for next-generation delivery technologies. Liposomes are used across oncology, rare-disease management, and gene-therapy enabling platforms. Contract research organizations (CROs) evaluate performance in long-acting injectables and responsive lipid-bilayer systems optimized for localized release. Consumer brands expand encapsulated cosmetic formulations in brightening, hydration, and antioxidant segments. Healthcare providers adopt liposomal drugs backed by NICE-approved clinical-outcome data. Sustainability-focused packaging and micro-encapsulation innovation create differentiation among cosmeceutical suppliers.
- High-science validation sustaining medical-grade adoption
- CRO ecosystem supporting preclinical-to-commercial transition
- Retail cosmetics using encapsulation to enhance product efficacy
- National reimbursement shaping hospital uptake
How is Germany reinforcing regulated clinical deployment?
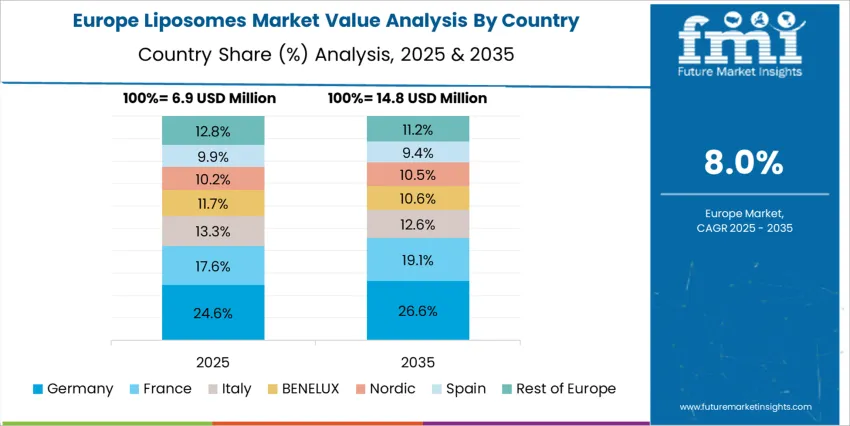
Germany posts 8.8% CAGR, driven by strong hospital-based prescription of liposomal chemotherapies and antimicrobial drugs. GMP manufacturing emphasizes sterility assurance, vesicle stability, and endotoxin control, guiding supplier qualification. Biotech firms evaluate liposomes as adjuvants and delivery systems for DNA/RNA therapeutics. Dermatological brands incorporate encapsulated botanical actives designed for controlled transdermal absorption. Physicians prioritize liposomal options for patients with heightened sensitivity to systemic toxicity. Germany’s export-compliant manufacturing standards support European distribution of high-purity liposomal pharmaceuticals.
- Hospital oncology fueling stable demand
- GMP-led controls ensuring clinical-grade quality
- Growth in transdermal cosmetics and medical skincare
- Biotech partnerships advancing RNA-delivery potential
How is the United States scaling innovation in lipid-based delivery?
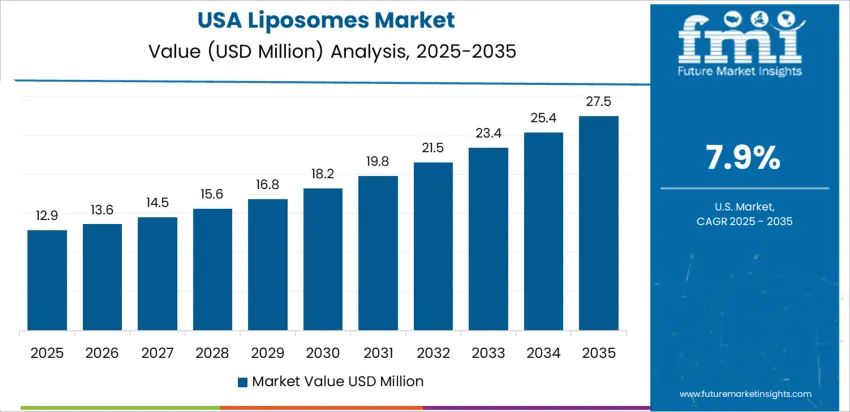
The United States grows at 7.9% CAGR, supported by leading expertise in lipid nanoparticle (LNP) platforms, mRNA therapeutics, and personalized oncology. Bio-pharma companies integrate electrostatic-optimized liposomes to enhance tumor targeting and drug-resistance management. Dermatology markets adopt encapsulation for sensitive-skin formulations and sustained moisturization technologies. Industrial IP portfolios in advanced encapsulation, such as stealth liposomes, expand commercialization opportunities. Retail channels support OTC cosmeceuticals with clinical-grade claims. Regulatory workflows focus on stability data, encapsulation efficiency, and immune-response profiles.
- mRNA and cancer-therapeutics R&D leading innovation
- High adoption in regenerative and sensitive-skin dermatology
- Intellectual property strengthening competitive advantage
- Clinical validation shaping reimbursement pathways
What does the global competitive structure for liposome demand show?
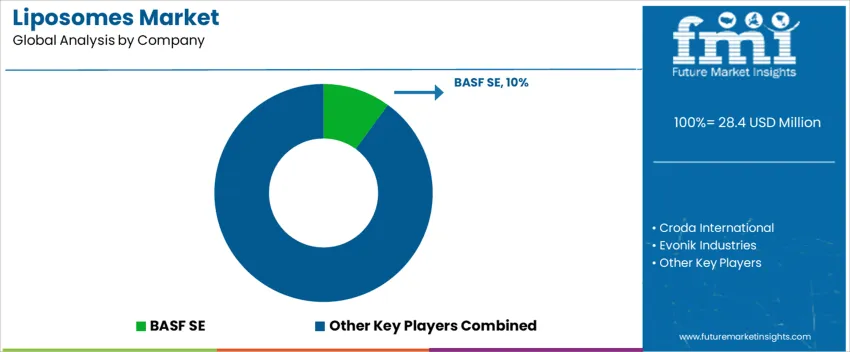
Global demand for liposomes supports cosmetic actives, topical dermal delivery, and regulated pharmaceutical formulations requiring enhanced penetration, stability, and ingredient protection. Procurement decisions depend on phospholipid purity, reproducible particle-size control, and compatibility with sensitive biomolecules. Growth concentrates in dermatology and oncology administration forms, together with rapid interest in encapsulated botanicals and vitamin systems in skin care.
BASF SE holds about 10.0% share worldwide. It focuses on phosphatidylcholine-based systems with controlled oxidation profiles for personal-care and OTC applications. Croda International maintains strong positions through high-grade lecithin sources and formulation assistance that supports actives delivery across facial serums and scalp-care concepts. Evonik Industries contributes with pharmaceutical-grade capabilities across parenteral and controlled-release formats where regulatory documentation and stability data determine supplier approvals.
Symrise focuses on cosmetic encapsulation solutions integrating plant-derived claims and sensory-driven performance. Dow addresses larger-volume consumer ranges requiring emulsification compatibility and extended shelf-life support. Ashland and Clariant supply specialized liposomal forms aligned with sustainable sourcing, solvent transparency, and analytical verification. Broader participation comes from Seppic, Lubrizol, Lonza, Inolex and peers offering encapsulation options for antioxidants, peptides, and nutricosmetic ingredients. Competitive factors include phospholipid traceability, nanostructure stability, and proven release behaviour into targeted skin and mucosal environments.
Key Players in the Liposomes Market
- BASF SE
- Croda International
- Evonik Industries
- Symrise (incl. IFF/Givaudan actives)
- Dow / Dow Inc.
- Ashland
- Clariant
- Others (Seppic, Lubrizol, Lonza, Inolex etc.)
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| End Use Application | Skin Care, Body Care, Hair Care, Sun Care, Color Cosmetics, Men’s Grooming, Baby & Kids Care, Dermocosmetic / Professional Care |
| Product Form | Powder, Granules / Agglomerates, Flakes, Pellets / Prills, Liquid (Solution), Concentrate (High-active Liquid), Dispersion / Suspension, Emulsion, Paste, Gel, Wax / Solid Block, Oil |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, USA, Germany, South Korea, Japan, Italy, and 40+ countries |
| Key Companies Profiled | BASF SE, Croda International, Evonik Industries, Symrise (incl. IFF/Givaudan actives), Dow / Dow Inc., Ashland, Clariant, Others (Seppic, Lubrizol, Lonza, Inolex etc.) |
| Additional Attributes | Dollar sales by application and formulation type; penetration in encapsulated actives delivery for cosmetic and personal care products; rapid adoption of nanoliposome and multilamellar vesicle technologies; strong demand in anti-aging, hydration, and targeted skin barrier repair formulations; regional growth driven by premium skincare in Asia Pacific and North America; regulatory compliance and safety validation for improved bioavailability of active ingredients. |
Liposomes Market by Segment
End Use Application:
- Skin Care
- Body Care
- Hair Care
- Sun Care
- Color Cosmetics
- Men’s Grooming
- Baby & Kids Care
- Dermocosmetic / Professional Care
Product Form:
- Powder
- Granules / Agglomerates
- Flakes
- Pellets / Prills
- Liquid (Solution)
- Concentrate (High-active Liquid)
- Dispersion / Suspension
- Emulsion
- Paste
- Gel
- Wax / Solid Block
- Oil
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the liposomes market in 2025?
The global liposomes market is estimated to be valued at USD 28.4 million in 2025.
What will be the size of liposomes market in 2035?
The market size for the liposomes market is projected to reach USD 68.9 million by 2035.
How much will be the liposomes market growth between 2025 and 2035?
The liposomes market is expected to grow at a 9.3% CAGR between 2025 and 2035.
What are the key product types in the liposomes market?
The key product types in liposomes market are skin care, body care, hair care, sun care, color cosmetics, men’s grooming, baby & kids care and dermocosmetic / professional care.
Which product form segment to contribute significant share in the liposomes market in 2025?
In terms of product form, powder segment to command 16.2% share in the liposomes market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End Use Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use Application , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use Application , 2025 to 2035
- Skin Care
- Body Care
- Hair Care
- Sun Care
- Color Cosmetics
- Men’s Grooming
- Baby & Kids Care
- Dermocosmetic / Professional Care
- Y-o-Y Growth Trend Analysis By End Use Application , 2020 to 2024
- Absolute $ Opportunity Analysis By End Use Application , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Product Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Form, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Form, 2025 to 2035
- Powder
- Granules / Agglomerates
- Flakes
- Pellets / Prills
- Liquid (Solution)
- Concentrate (High-active Liquid)
- Dispersion / Suspension
- Emulsion
- Paste
- Gel
- Wax / Solid Block
- Y-o-Y Growth Trend Analysis By Product Form, 2020 to 2024
- Absolute $ Opportunity Analysis By Product Form, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By End Use Application
- By Product Form
- By Country
- Market Attractiveness Analysis
- By Country
- By End Use Application
- By Product Form
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By End Use Application
- By Product Form
- By Country
- Market Attractiveness Analysis
- By Country
- By End Use Application
- By Product Form
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By End Use Application
- By Product Form
- By Country
- Market Attractiveness Analysis
- By Country
- By End Use Application
- By Product Form
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By End Use Application
- By Product Form
- By Country
- Market Attractiveness Analysis
- By Country
- By End Use Application
- By Product Form
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By End Use Application
- By Product Form
- By Country
- Market Attractiveness Analysis
- By Country
- By End Use Application
- By Product Form
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By End Use Application
- By Product Form
- By Country
- Market Attractiveness Analysis
- By Country
- By End Use Application
- By Product Form
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By End Use Application
- By Product Form
- By Country
- Market Attractiveness Analysis
- By Country
- By End Use Application
- By Product Form
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By End Use Application
- By Product Form
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By End Use Application
- By Product Form
- Competition Analysis
- Competition Deep Dive
- BASF SE
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Croda International
- Evonik Industries
- Symrise (incl. IFF/Givaudan actives)
- Dow / Dow Inc.
- Ashland
- Clariant
- Others (Seppic, Lubrizol, Lonza, Inolex etc.)
- BASF SE
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020 to 2035
- Table 2: Global Market Value (USD Million) Forecast by End Use Application , 2020 to 2035
- Table 3: Global Market Value (USD Million) Forecast by Product Form, 2020 to 2035
- Table 4: North America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 5: North America Market Value (USD Million) Forecast by End Use Application , 2020 to 2035
- Table 6: North America Market Value (USD Million) Forecast by Product Form, 2020 to 2035
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 8: Latin America Market Value (USD Million) Forecast by End Use Application , 2020 to 2035
- Table 9: Latin America Market Value (USD Million) Forecast by Product Form, 2020 to 2035
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 11: Western Europe Market Value (USD Million) Forecast by End Use Application , 2020 to 2035
- Table 12: Western Europe Market Value (USD Million) Forecast by Product Form, 2020 to 2035
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 14: Eastern Europe Market Value (USD Million) Forecast by End Use Application , 2020 to 2035
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Product Form, 2020 to 2035
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 17: East Asia Market Value (USD Million) Forecast by End Use Application , 2020 to 2035
- Table 18: East Asia Market Value (USD Million) Forecast by Product Form, 2020 to 2035
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by End Use Application , 2020 to 2035
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Product Form, 2020 to 2035
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by End Use Application , 2020 to 2035
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Product Form, 2020 to 2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value Share and BPS Analysis by End Use Application , 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by End Use Application , 2025-2035
- Figure 5: Global Market Attractiveness Analysis by End Use Application
- Figure 6: Global Market Value Share and BPS Analysis by Product Form, 2025 and 2035
- Figure 7: Global Market Y-o-Y Growth Comparison by Product Form, 2025-2035
- Figure 8: Global Market Attractiveness Analysis by Product Form
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2025-2035
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2025-2035
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2025-2035
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2025-2035
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2025-2035
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: North America Market Value Share and BPS Analysis by End Use Application , 2025 and 2035
- Figure 21: North America Market Y-o-Y Growth Comparison by End Use Application , 2025-2035
- Figure 22: North America Market Attractiveness Analysis by End Use Application
- Figure 23: North America Market Value Share and BPS Analysis by Product Form, 2025 and 2035
- Figure 24: North America Market Y-o-Y Growth Comparison by Product Form, 2025-2035
- Figure 25: North America Market Attractiveness Analysis by Product Form
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 27: Latin America Market Value Share and BPS Analysis by End Use Application , 2025 and 2035
- Figure 28: Latin America Market Y-o-Y Growth Comparison by End Use Application , 2025-2035
- Figure 29: Latin America Market Attractiveness Analysis by End Use Application
- Figure 30: Latin America Market Value Share and BPS Analysis by Product Form, 2025 and 2035
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Product Form, 2025-2035
- Figure 32: Latin America Market Attractiveness Analysis by Product Form
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 34: Western Europe Market Value Share and BPS Analysis by End Use Application , 2025 and 2035
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by End Use Application , 2025-2035
- Figure 36: Western Europe Market Attractiveness Analysis by End Use Application
- Figure 37: Western Europe Market Value Share and BPS Analysis by Product Form, 2025 and 2035
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Product Form, 2025-2035
- Figure 39: Western Europe Market Attractiveness Analysis by Product Form
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by End Use Application , 2025 and 2035
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by End Use Application , 2025-2035
- Figure 43: Eastern Europe Market Attractiveness Analysis by End Use Application
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Product Form, 2025 and 2035
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Product Form, 2025-2035
- Figure 46: Eastern Europe Market Attractiveness Analysis by Product Form
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 48: East Asia Market Value Share and BPS Analysis by End Use Application , 2025 and 2035
- Figure 49: East Asia Market Y-o-Y Growth Comparison by End Use Application , 2025-2035
- Figure 50: East Asia Market Attractiveness Analysis by End Use Application
- Figure 51: East Asia Market Value Share and BPS Analysis by Product Form, 2025 and 2035
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Product Form, 2025-2035
- Figure 53: East Asia Market Attractiveness Analysis by Product Form
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by End Use Application , 2025 and 2035
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use Application , 2025-2035
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by End Use Application
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Product Form, 2025 and 2035
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Form, 2025-2035
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Product Form
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by End Use Application , 2025 and 2035
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by End Use Application , 2025-2035
- Figure 64: Middle East & Africa Market Attractiveness Analysis by End Use Application
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Product Form, 2025 and 2035
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Product Form, 2025-2035
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Product Form
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

