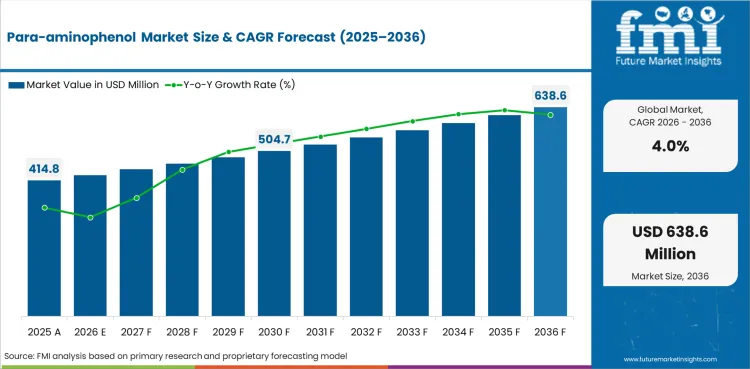
The para-aminophenol market was valued at an estimated USD 392.4 million in 2025, is projected to reach USD 414.8 million in 2026, and is forecast to expand to USD 612.7 million by 2036 at a CAGR of 4.0%.
| Parameter | Details |
|---|---|
| Market value (2026) | USD 414.8 million |
| Forecast value (2036) | USD 612.7 million |
| CAGR (2026 to 2036) | 4.0% |
| Estimated market value (2025) | USD 392.4 million |
| Incremental opportunity | USD 197.9 million |
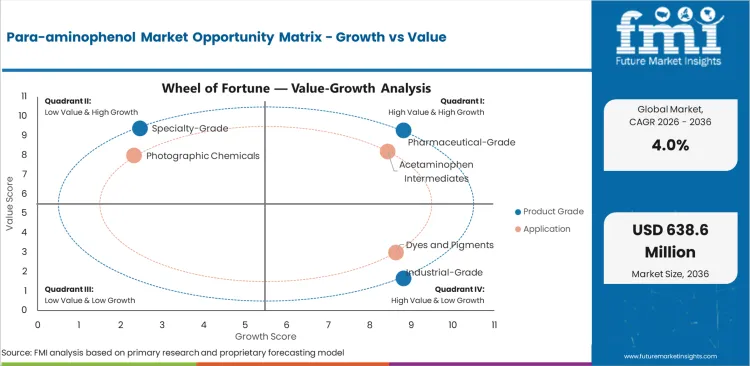
| Leading product | Pharmaceutical-grade para-aminophenol, 54.6% of product revenue |
| Leading application | Acetaminophen intermediates, 63.8% of application revenue |
| Leading end use | Pharmaceutical manufacturers, 58.2% of end-use revenue |
| Key players | Thermo Fisher Scientific, Merck, USP, specialty intermediate manufacturers in China and India |
Source: Analyst synthesis from authoritative sources, 2026.
Three factors keep market demand intact. Para-aminophenol remains directly tied to acetaminophen manufacturing economics, and paracetamol remains a globally essential medicine according to WHO. Pharmacopeial and USP controls maintain the relevance of 4-aminophenol in regulated quality systems because it is both an intermediate and a monitored impurity or degradation product. The market also retains a smaller but stable base in dye, photographic, and specialty chemical use, which supports baseline industrial demand even when pharmaceutical orders fluctuate.
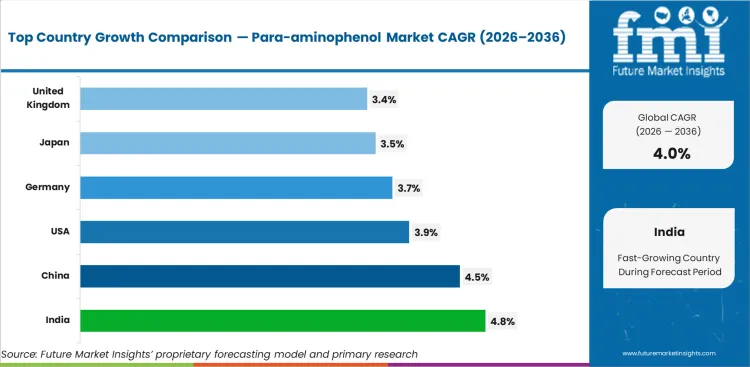
Across tracked geographies, India sets the pace at 4.8% CAGR. China follows at 4.5%. United States follows at 3.9%. Germany follows at 3.7%. Japan follows at 3.5%. United Kingdom follows at 3.4%.
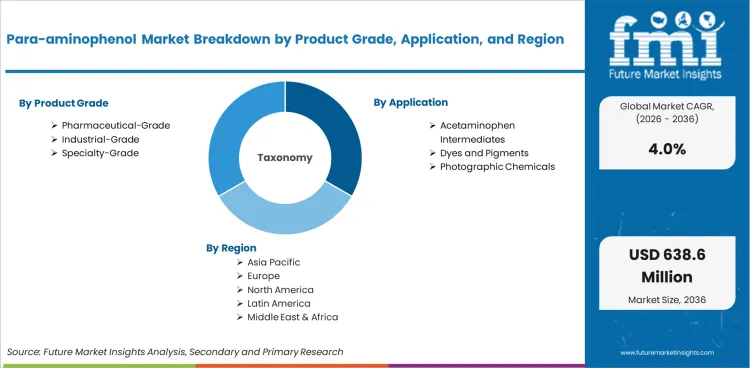
The para-aminophenol market covers commercial production and sale of para-aminophenol, also known as 4-aminophenol, used as a chemical intermediate in pharmaceuticals, dyes, photographic chemicals, and specialty synthesis. The market includes regulated and industrial grades sold for downstream manufacturing rather than finished dosage-form pharmaceutical revenue.
Market scope includes pharmaceutical-grade para-aminophenol, industrial-grade para-aminophenol, and specialty-grade material supplied for acetaminophen synthesis, dyestuff chemistry, photographic applications, and research or specialty synthesis. Revenue sizing spans direct chemical sales and intermediate use within regulated manufacturing chains where para-aminophenol is sold as a commercial input.
The scope excludes finished acetaminophen revenue, downstream formulated analgesics, unrelated aniline derivatives, and internal captive intermediate production not sold in the merchant market. It also excludes generic analytical testing revenue not tied to para-aminophenol sales.
The main driver is its role as a core intermediate in acetaminophen production. Acetaminophen remains one of the most widely used analgesic and antipyretic molecules globally, which keeps the para-aminophenol demand base structurally intact.
Quality controls also support market relevance. Para-aminophenol is not only an intermediate but also a monitored impurity in regulated pharmaceutical systems, which increases the need for reliable analytical standards and high-purity supply.
Industrial demand remains narrower than pharmaceutical demand, but it still contributes a useful floor through dye and specialty chemical applications.
The market is growing because para-aminophenol sits in a stable part of the pharmaceutical value chain. It does not depend on consumer branding or finished-dose promotion. Instead, it depends on the persistence of high-volume acetaminophen production and the continued need for controlled intermediates in regulated manufacturing.
Another source of resilience is that the material has multiple purity and application tiers. Pharmaceutical-grade supply benefits from impurity controls and process traceability. Industrial-grade material serves smaller non-pharma applications where pricing sensitivity is higher but demand remains recurring.
This combination creates moderate but steady growth rather than explosive expansion. The market benefits from durable pharmaceutical consumption, while regulatory requirements help preserve value in higher-purity grades.
The market is segmented by product grade, application, end use, distribution channel, and region. By product grade, the market includes pharmaceutical-grade para-aminophenol, industrial-grade para-aminophenol, and specialty-grade material. By application, the market includes acetaminophen intermediates, dyes and pigments, photographic chemicals, and specialty synthesis.
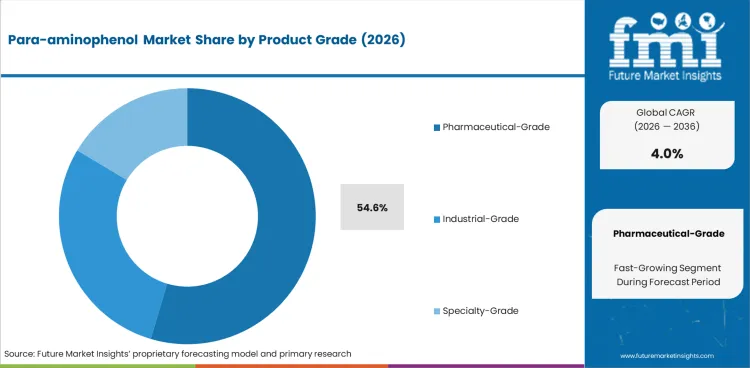
Pharmaceutical-grade para-aminophenol is projected to hold 54.6% of product revenue in 2026. This lead is supported by high-volume analgesic demand, stronger margin capture in regulated supply chains, and more stringent impurity and traceability requirements.
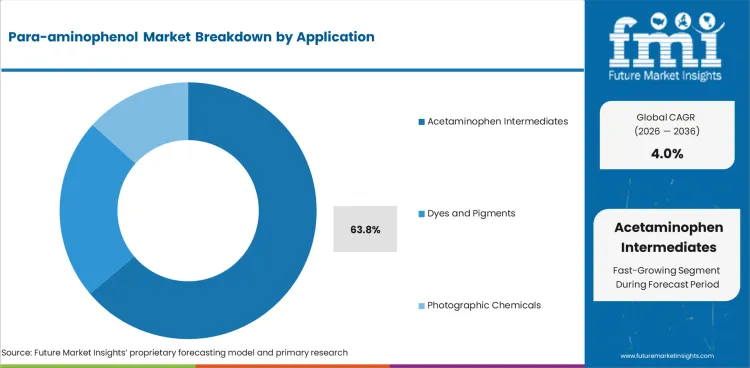
Acetaminophen intermediates are expected to capture 63.8% of application revenue in 2026. The segment leads because para-aminophenol remains directly linked to one of the most established and widely consumed analgesic API pathways in the world.
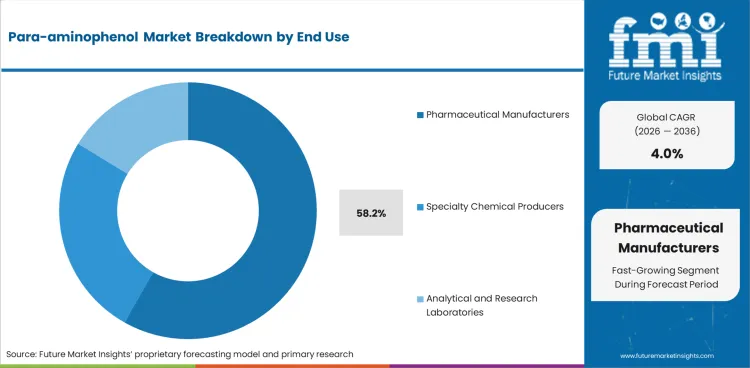
Pharmaceutical manufacturers are projected to account for 58.2% of end-use revenue in 2026. Merchant purchases are concentrated among regulated producers that require reliable intermediate quality, reproducibility, and documentation support.
Driver: stable global acetaminophen demand continues to support the core intermediate market.
Restraint: price sensitivity and feedstock volatility can compress margins in industrial and lower-purity supply tiers.
Trend: regulated buyers are placing greater weight on impurity control, documentation quality, and traceable supply.
The market is expanding steadily. Pharmaceutical supply chains remain the main growth driver because quality and continuity carry more weight than spot pricing. Industrial demand provides diversification and a stable secondary revenue base.
Growth is supported by the persistent scale of acetaminophen consumption. Because para-aminophenol is structurally tied to that value chain, its demand base remains more durable than many specialty chemical intermediates that depend on narrower end markets.
Cost pressure across chemical intermediates remains the main market constraint. Producers face margin compression when aromatic feedstock conditions tighten or when lower-cost industrial supply competes aggressively on price. This pressure is strongest in industrial-grade material.
Quality-led differentiation is strengthening. Buyers in regulated pharmaceutical chains place greater weight on impurity control, analytical support, and supply continuity, which supports suppliers with stronger documentation and quality systems.
.webp)
Asia remains the center of market growth because pharmaceutical intermediate manufacturing and chemical processing capacity are concentrated there. India and China are the most important demand and supply anchors.
The United States and Europe remain important through pharmaceutical consumption, analytical standards demand, and specialty chemical applications, but growth is slower than in Asia.
Japan supports a smaller but stable market through quality-intensive applications and specialty chemical use.
| Country | CAGR |
|---|---|
| India | 4.8% |
| China | 4.5% |
| United States | 3.9% |
| Germany | 3.7% |
| Japan | 3.5% |
| United Kingdom | 3.4% |
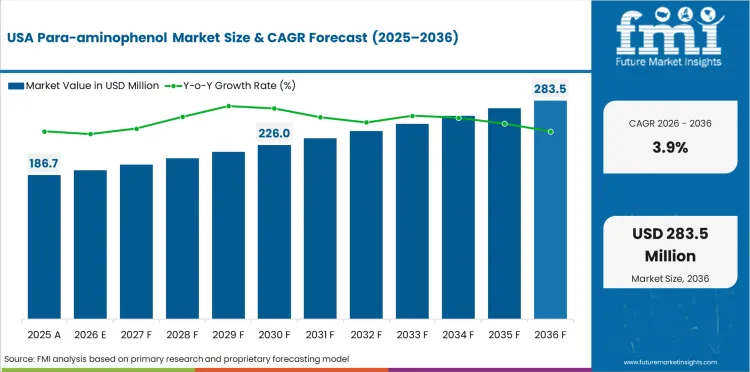
The United States is projected to grow at a CAGR of 3.9% through 2036. Growth is supported by persistent analgesic demand, strong analytical standards usage, and specialty chemical purchases.
The United Kingdom is projected to grow at a CAGR of 3.4% through 2036. Demand is supported by pharmaceutical manufacturing links and specialty chemical use, but scale remains smaller than major Asian markets.
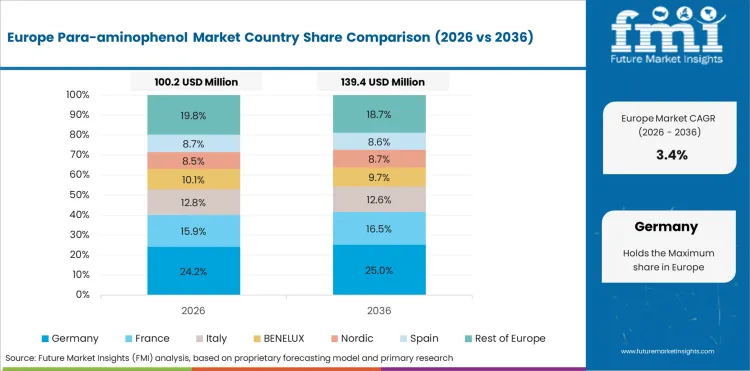
Germany is expected to grow at a CAGR of 3.7% through 2036. The market benefits from specialty chemical capability and pharmaceutical quality requirements.
Japan is projected to grow at a CAGR of 3.5% through 2036. The country supports steady demand in quality-intensive chemical and pharmaceutical applications.
China is expected to grow at a CAGR of 4.5% through 2036. The market benefits from chemical manufacturing scale, broad intermediate processing capability, and persistent pharmaceutical demand.
India is projected to grow at a CAGR of 4.8% through 2036. Growth is driven by strong API manufacturing capability and sustained demand from acetaminophen and related pharmaceutical value chains.
The competitive landscape combines global analytical and specialty chemical suppliers with regional intermediate manufacturers. Competitive strength depends on purity, documentation, batch consistency, and the ability to serve regulated pharmaceutical channels.
Merck, Thermo Fisher Scientific, and USP are important reference points in higher-purity and analytical supply. Merchant industrial and pharmaceutical intermediate supply is more fragmented and includes regional chemical producers, especially in Asia.
The market remains less brand-driven than many specialty chemicals. Buyers focus more on reliability, assay quality, impurity control, and supply continuity than on broad commercial marketing.
Competition is strongest in regulated supply chains, where differentiation depends on quality systems and analytical support. Lower-grade industrial supply remains more price-sensitive and fragmented.
Major Industry Players
Merck and Thermo Fisher Scientific are prominent in analytical and research supply because they offer reference-grade and catalog chemistry with broad global reach.
USP is relevant because it anchors pharmacopeial standards and quality control reference use around acetaminophen and 4-aminophenol-related systems.
Regional intermediate manufacturers in India and China remain important in merchant pharmaceutical and industrial supply because they are closer to API production chains and cost-competitive manufacturing bases.
| Company | Quality Positioning | Pharma Relevance | Industrial Supply Strength | Geographic Reach |
|---|---|---|---|---|
| Merck | High | High | Medium | Strong |
| Thermo Fisher Scientific | High | High | Medium | Strong |
| USP | High | High | Low | Strong |
| Regional Asian manufacturers | Medium | High | High | Moderate |
Source: Analyst synthesis from authoritative sources, 2026. Ratings reflect relative positioning based on disclosed capabilities and market presence.
Major Global Players:
Emerging Players/Startups
| Metric | Value |
|---|---|
| Quantitative Units | USD 414.8 million to USD 612.7 million, at a CAGR of 4.0% |
| Market Definition | Commercial production and sale of para-aminophenol used as a chemical intermediate in pharmaceuticals, dyes, photographic chemicals, and specialty synthesis. |
| Segmentation | Product grade: pharmaceutical-grade para-aminophenol, industrial-grade para-aminophenol, specialty-grade para-aminophenol. Application: acetaminophen intermediates, dyes and pigments, photographic chemicals, specialty synthesis. End use: pharmaceutical manufacturers, specialty chemical producers, analytical and research laboratories. Distribution channel: direct industrial sales, chemical distributors, catalog and laboratory supply channels. |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, Middle East and Africa |
| Countries Covered | United States, United Kingdom, Germany, Japan, China, India and others |
| Key Companies Profiled | Merck, Thermo Fisher Scientific, USP, regional Asian manufacturers |
| Forecast Period | 2026 to 2036 |
| Approach | Analyst-built hybrid top-down and bottom-up model using official chemical references, supplier pricing signals, and downstream pharmaceutical intermediate demand logic. |
| Historical Period | 2020 to 2025 |
How large is the demand for Para-aminophenol in the global market in 2026?
Demand for para-aminophenol in the global market is estimated at USD 414.8 million in 2026.
What will be the market size by 2036?
Market size is projected to reach USD 612.7 million by 2036.
What is the expected demand growth between 2026 and 2036?
Demand is expected to grow at a CAGR of 4.0% between 2026 and 2036.
Which product grade is poised to lead by 2026?
Pharmaceutical-grade para-aminophenol leads the market in 2026 with 54.6% of product revenue.
How are pharmaceutical manufacturers driving adoption?
Pharmaceutical manufacturers account for 58.2% of end-use revenue because para-aminophenol remains directly tied to acetaminophen intermediate demand and regulated supply chains.
What is driving demand in the United States?
The United States is projected to grow at a CAGR of 3.9% through 2036, supported by persistent analgesic demand, analytical standards use, and specialty chemical purchases.
What does the market definition mean?
The market covers commercial production and sale of para-aminophenol used as a chemical intermediate in pharmaceuticals, dyes, photographic chemicals, and specialty synthesis.
How does the analyst validate the forecast?
The forecast is validated through a hybrid model using official chemical references, supplier pricing signals, and downstream pharmaceutical intermediate demand logic.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.