Antibody Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Antibody Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Antibody Market Outlook for 2025 to 2035
The antibody market is expanding steadily due to growing demand for targeted therapies, rising prevalence of chronic and infectious diseases, and rapid advancements in biopharmaceutical research. The current scenario is defined by strong investment in biologics development and a growing pipeline of antibody-based treatments aimed at improving efficacy and safety profiles.
Market growth is being supported by technological innovations in monoclonal antibody production, recombinant DNA technology, and antibody–drug conjugate platforms that enhance therapeutic outcomes. Increasing healthcare expenditure, favorable regulatory frameworks, and strategic collaborations between pharmaceutical and biotechnology companies are also contributing to market expansion.
The future outlook is marked by continued integration of precision medicine, expansion of biosimilar portfolios, and greater penetration of antibody therapeutics across emerging economies Growth rationale is anchored in the increasing clinical success rates of antibody therapies, their expanding indications across multiple disease types, and the scalability of biomanufacturing technologies, collectively supporting strong and sustained market growth over the coming years.
Quick Stats for Antibody Market
- Antibody Market Industry Value (2025): USD 288.4 billion
- Antibody Market Forecast Value (2035): USD 628.5 billion
- Antibody Market Forecast CAGR: 8.1%
- Leading Segment in Antibody Market in 2025: Cancer (36.8%)
- Key Growth Region in Antibody Market: North America, Asia-Pacific, Europe
- Top Key Players in Antibody Market: F. Hoffmann-La Roche AG, Novartis AG, Johnson & Johnson, Takeda Pharmaceutical Company Limited, Amgen Inc.
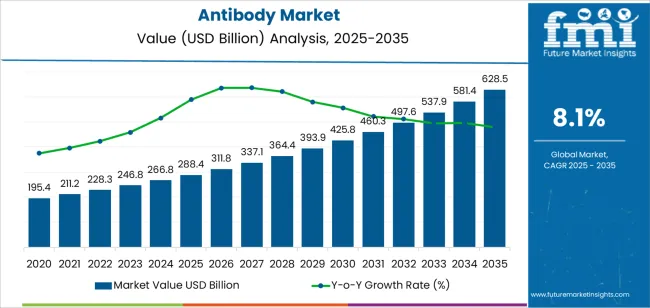
| Metric | Value |
|---|---|
| Antibody Market Estimated Value in (2025 E) | USD 288.4 billion |
| Antibody Market Forecast Value in (2035 F) | USD 628.5 billion |
| Forecast CAGR (2025 to 2035) | 8.1% |
Segmental Analysis
The market is segmented by Disease Indication, Product Type, and End User and region. By Disease Indication, the market is divided into Cancer, Cardiovascular Diseases, CNS Disorders, Autoimmune Disorders, and Other Disease Indications. In terms of Product Type, the market is classified into Monoclonal Antibodies, Polyclonal Antibodies, and Antibody-Drug Complexes. Based on End User, the market is segmented into Hospitals, Long-Term Care Facilities, Research Institutes, and Other End Users. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Disease Indication Segment
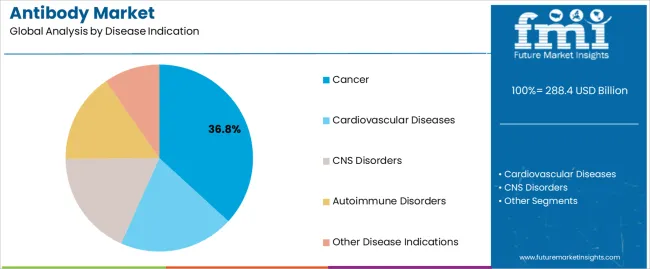
The cancer segment, accounting for 36.80% of the disease indication category, has emerged as the leading contributor to market revenue. Growth has been driven by the high prevalence of cancer globally and the growing acceptance of antibody-based targeted therapies as a standard treatment option.
The ability of antibodies to selectively bind and neutralize cancer cells while minimizing off-target effects has significantly increased their clinical adoption. Strong research funding, rapid approval of novel oncology antibodies, and improvements in biomarker-guided therapy selection have reinforced market leadership.
Rising demand for personalized treatment regimens and favorable reimbursement policies for advanced cancer therapies are expected to sustain the dominance of this segment Continuous innovation in immunotherapy and the development of bispecific and conjugated antibodies are anticipated to further strengthen its position in the global antibody market.
Insights into the Product Type Segment
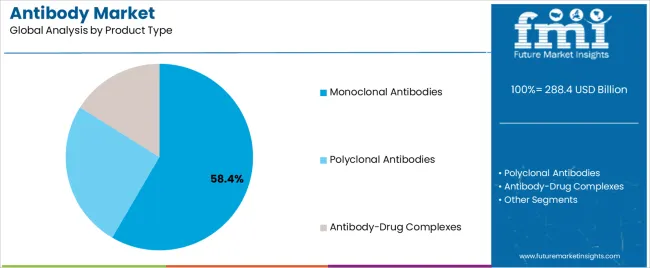
The monoclonal antibodies segment, representing 58.40% of the product type category, has maintained its leadership position due to broad therapeutic applications and high clinical efficacy. Its growth has been supported by increasing R&D investment, technological advancements in antibody engineering, and expanding indications across oncology, autoimmune, and infectious diseases.
Monoclonal antibodies are being increasingly integrated into first-line treatment regimens owing to their targeted action and reduced adverse effects. The introduction of humanized and fully human antibodies has enhanced safety profiles, boosting market confidence.
Manufacturing scalability and cost optimization achieved through advanced bioprocessing techniques have further strengthened commercial viability As biosimilar monoclonal antibodies gain regulatory approvals, market accessibility is expected to expand, supporting consistent revenue growth and reinforcing this segment’s dominant share in the antibody market.
Insights into the End User Segment
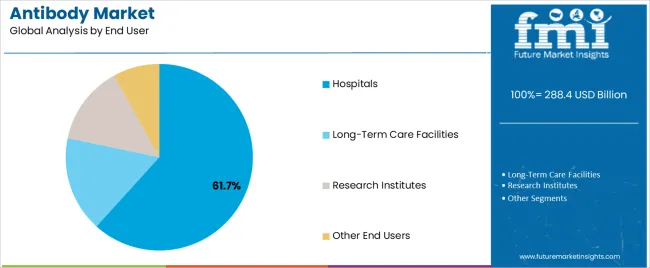
The hospitals segment, holding 61.70% of the end user category, continues to dominate the antibody market due to its critical role in patient care, treatment administration, and clinical diagnostics. Hospitals serve as the primary distribution and usage centers for antibody-based therapeutics, particularly in oncology and immunology.
The presence of advanced healthcare infrastructure, skilled medical professionals, and comprehensive diagnostic facilities has facilitated high adoption rates within hospital settings. Strategic partnerships between hospitals and pharmaceutical manufacturers have enabled efficient supply chain management and rapid access to new antibody therapies.
Increasing hospitalization rates for chronic diseases and the rise in inpatient biologic treatments have further strengthened this segment’s position Continuous improvement in hospital-based infusion centers and integration of digital healthcare systems are expected to sustain dominance and drive future growth within the antibody market.
Antibody Market Trends & Analysis
- As the prevalence of autoimmune diseases is on the rise, the demand for antibody drugs has strengthened over the last couple of years. Moreover, the rising application of antibodies in chronic disease treatment is anticipated to create more opportunities for antibody market expansion in the near future.
- Novel biosimilar antibody manufacturers are slashing their prices to compete with branded antibody manufacturers, especially in the European market. With the expiration of patents of renowned products, a boost in demand for biosimilar antibodies can be expected in upcoming years.
Global Antibody Market Historical Analysis (2020 to 2024) Vs Forecast Outlook (2025 to 2035)
The global antibody market was valued at USD 195.4 billion in 2020. The market overview of antibody drugs progressed at an annual rate of 12.5% between 2020 and 2025. The overall market value was about USD 288.4 billion by 2025.
| Attributes | Details |
|---|---|
| Antibody Market Value (2020) | USD 195.4 billion |
| Market Revenue (2025) | USD 288.4 billion |
| Market Historical Growth Rate (CAGR 2020 to 2025) | 12.5% CAGR |
Growth Opportunities Witnessed by the Global Antibody Market Participants
Research and Development Activities Impact Antibodies Sales Outlook
A surge in research and development activities among pharmaceutical market leaders to develop economical and effective antibodies is driving growth opportunities for diagnostic specialty antibody suppliers.
In February 2024, Abingdon Health collaborated with Abcam to explore opportunities for their service and portfolio expansion which includes Abcam's reagent portfolio and Abingdon Health's rapid test contract development and manufacturing services.
Rising cases of multiple sclerosis and Parkinson's disease among the older population is a key factor fueling the demand for antibodies. There is an urgency to develop personalized and cheap medicines, advanced therapeutics, advanced genetic engineering technology, and novel and effective treatments in middle-income countries.
In 2024, Berkeley Lights, a digital cell biology company, joined hands with Genovac, an antibody discovery company, to readily access the right antibodies in the shortest amount of time with the help of modern technology.
Increasing Production of Bio-similar Antibodies is Driving Demand
Biosimilar antibodies are experiencing heavy demand as they are inexpensive in comparison with human monoclonal antibodies, and they have very few side effects. In 2024, Johnson and Johnson reported USD 266.8 Billion achieved through the sales of Remicade. According to Pfizer, Inflectra, the biosimilar competitor of Remicade, generated USD 659 million in 2024.
USA Food and Drug Administration (FDA) approved a biosimilar of Remicade called Avsola in December 2020 and is now eligible for producing biologics products all over the world. Increasing demand for biosimilar antibodies in the treatment of rheumatoid arthritis, along with a high patient acceptance rate, is anticipated to boost the market growth for antibodies.
One of the leaders in prescription medicines, Pfizer, received EU approval for biosimilar antibody therapeutics Amsparity for Crohn’s disease, psoriasis, and psoriatic arthritis in February 2024. Elevated new drug approval rates and products in clinical trials in pharmaceutical companies are anticipated to further drive the market in the upcoming years.
Country-wise Insights
The table below mentions the countries that are expected to witness higher growth opportunities in this market.
| Regional Market Comparison | CAGR (2025 to 2035) |
|---|---|
| United States | 9.6% |
| Germany | 9.4% |
| United Kingdom | 10.1% |
| India | 9.5% |
| China | 6.5% |
Advancement in Therapeutics is driving the Market in the United States
The United States therapeutic antibodies market is poised to advance at a rate of 10.1% per year till 2035.
- Modern healthcare infrastructural facilities and the presence of leading market players in the United States.
- Autoimmune diseases are the third most common reason for chronic ailments creating a higher demand for antibody drugs in the country.
The Medical Tourism Sector has boosted Antibody Consumption in the United Kingdom
Consumption of antibody products in the United Kingdom is projected to rise at a rate of 10.1% during the forecast years.
- Higher disease prevalence as well as the United Kingdom citizens’ capacity for spending on cutting-edge medicines.
- The rapidly improving medical tourism sector of the country is projected to drive the demand for antibodies in the coming days.
Rising Elderly Population Percentage is fueling Antibodies Sales Outlook in China
The antibodies industry in China is projected to thrive at a CAGR of 5.8% through the projected years.
- Growing awareness about the benefits of antibodies for various medical ailments like rheumatoid arthritis, cancer, and infections.
- The rising geriatric population and associated ailments are anticipated to drive market growth for antibodies in the upcoming years.
Germany Antibodies Therapeutics Market Size Expanding with Rising Cancer Cases
Sales of antibodies in Germany are anticipated to increase at a rate of 9.4% from 2025 to 2035.
- A surge in cancer patients is anticipated to drive the demand for antibodies for cancer treatment.
- Germany's antibody drugs market has strengthened significantly with the presence of top companies, whose products are in great demand worldwide.
Government Funding has Bolstered Antibodies Development and Production in India
Demand for antibodies in India is expected to rise at a rate of 9.5% per year between 2025 and 2035.
- Rising cases of cancer and cardiovascular diseases along with the development of the healthcare system in the country.
- Increasing government funding to make the treatment affordable, and increasing consumer spending on healthcare are anticipated to create more opportunities.
Category-wise Insights
Monoclonal Antibodies are More Effective than Polyclonal Antibodies
Based on product type, the monoclonal antibodies market segment is estimated to account for 95% of the market share in 2025.
| Attributes | Details |
|---|---|
| Top Product Type or Segment | Monoclonal Antibodies |
| Total Market Share in 2025 | 95% |
- This segment is bolstering in response to the increasing cases of rheumatoid arthritis, psoriasis, and asthma in the geriatric population across the globe.
- Hospitals are anticipated to remain the dominant end user of monoclonal antibodies on account of rising cases of various types of chronic ailments.
Recombinant Antibodies are Primarily Used for Cancer Treatment
Based on disease indication, the use of antibodies for treating cancer is expected to account for 57% of the total sales in 2025.
| Attributes | Details |
|---|---|
| Top Disease Indication Type or Segment | Cancer |
| Total Market Share in 2025 | 57% |
- The rising prevalence of non-Hodgkin lymphoma and other types of cancer has fuelled the demand for cancer antibodies for developing non-invasive and safe treatment options.
- With the focus on cancer research increasing, the antibodies sales outlook for the upcoming decade is anticipated to remain positive.
Competitive Landscape for Antibody Market
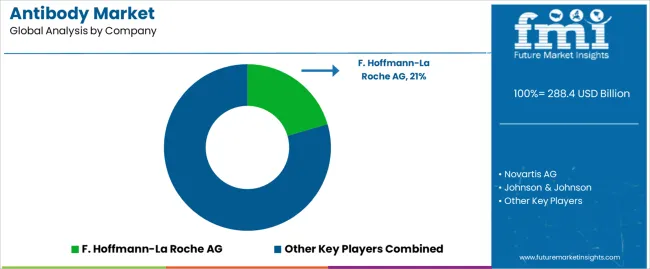
Leading market players are working towards developing new antibodies to remain competitive in the present market. They are also researching to create long-acting antibody drugs to reduce healthcare costs and attract chronic patients.
In order to gain a competitive edge in the antibody testing market, key players are developing oral and noninvasive treatments using antibodies to treat chronic ailments. Strategic mergers and acquisitions for portfolio expansion are in the cards as well.
Startups Should Concentrate on Increasing the Scope of Their Research to Promote Innovation
Future market players should concentrate on acquiring additional money through financing rounds to broaden their research and company scope. New businesses are looking into how to leverage cutting-edge technologies to streamline the creation of therapeutic antibodies, including nanobodies and monoclonal antibodies.
- Great Bay Bio (GBB), a start-up with headquarters in Hong Kong, declared in September 2025 that it had successfully raised USD 15 million in its Pre-series B investment round. The financing is to be put to use by the business to quicken the creation of bioprocessing platforms that are powered by artificial intelligence (AI) in order to enhance the CMC process for a variety of biological pharmaceuticals, such as monoclonal antibodies and fusion proteins.
- Capstan Therapeutics, a startup with the goal of creating treatments for conditions with unmet medical needs, declared its launch in September 2025 and provided USD 165 million for in-vivo cell engineering. The business is creating in-vivo CAR treatments, which allow for cell engineering while the patient is still receiving treatment.
- Versant Ventures, a well-known investor in biotechnology startups, announced a new relationship with AbCellera, a business specializing in antibody development, in July 2025. Through this collaboration, Versant has significantly sped up the research and development process by supplying start-ups with antibodies from AbCellera that are tailored to their target disease.
Recent Developments in the Global Antibody Market
- Research: Investigational Oral Treatment
On June 11, 2024, Novartis published its new Phase II data of an investigational oral treatment iptacopan, developed for treating paroxysmal nocturnal hemoglobinuria (PNH). The study indicated that during 12 weeks of iptacopan monotherapy treatment, no unexpected safety issues were reported which led to rapid and durable transfusion-free improvement of hemoglobin levels in the majority of patients.
- Acquisition: Johnson & Johnson and Momenta Pharmaceuticals Inc.
In August 2024, Johnson & Johnson entered into a final agreement to acquire Momenta Pharmaceuticals Inc. for around USD 6.5 Billion. Momenta Pharmaceuticals Inc. is a market leader in developing novel therapies for immune-mediated diseases.
The acquisition may allow the Janssen Pharmaceutical Companies of Johnson & Johnson to expand its product portfolio in immune-mediated diseases while driving growth opportunities through expansion into autoantibody-driven disease.
- Partnership: Sanofi and IGM Biosciences
Leading pharmaceutical company Sanofi announced in March 2025 that it will collaborate with IGM Biosciences to research, produce, and market six novel immunoglobulin M antibody agonists. Sanofi has agreed to approximately USD 6 billion in possible milestone payments through this agreement.
Key Players Profiled in the Antibody Market Report
- Novartis AG
- F. Hoffmann-La Roche AG
- Johnson & Johnson
- Takeda Pharmaceutical Company Limited
- Amgen Inc.
Key Coverage in the Antibody Market Report
- Adjacent study on research antibodies & reagents market.
- Comparative analysis of antibody vs. antigen market.
- Antibody market potential in African nations.
Antibody Market Report Scope
| Attribute | Details |
|---|---|
| Estimated Market Size (2025) | USD 266.83 billion |
| Projected Market Size (2035) | USD 581.42 billion |
| Anticipated Growth Rate (2025 to 2035) | 8.1% |
| Forecast Period | 2025 to 2035 |
| Historical Data Available for | 2020 to 2025 |
| Market Analysis | USD million or billion for Value and Units for Volume |
| Key Regions Covered | North America; Latin America; Europe; Middle East & Africa (MEA); East Asia; South Asia and Oceania |
| Key Countries Covered | United States, Canada, Brazil, Mexico, Germany, Spain, Italy, France, United Kingdom, Russia, China, India, Australia & New Zealand, GCC Countries, and South Africa |
| Key Segments Covered | By Product Type, By Disease Indication, By End Use Verticals, and By Region |
| Key Companies Profiled | Novartis AG; F. Hoffmann-La Roche AG; Johnson & Johnson; Takeda Pharmaceutical Company Limited; Amgen Inc. |
| Report Coverage | Market Forecast, Company Share Analysis, Competition Intelligence, DROT Analysis, Market Dynamics and Challenges, and Strategic Growth Initiatives |
| Customization & Pricing | Available upon Request |
Key Segments Covered by Antibodies Industry Survey Report
By Disease Indication:
- Cardiovascular Diseases
- CNS Disorders
- Cancer
- Autoimmune Disorders
- Other Disease Indications
By Product Type:
- Monoclonal Antibodies
- Polyclonal Antibodies
- Antibody-drug Complexes
By End User:
- Hospitals
- Long-term Care Facilities
- Research Institutes
- Other End Users
By Region:
- North America
- Latin America
- Europe
- East Asia
- South Asia and Pacific
- Middle East and Africa (MEA)
Frequently Asked Questions
How big is the antibody market in 2025?
The global antibody market is estimated to be valued at USD 288.4 billion in 2025.
What will be the size of antibody market in 2035?
The market size for the antibody market is projected to reach USD 628.5 billion by 2035.
How much will be the antibody market growth between 2025 and 2035?
The antibody market is expected to grow at a 8.1% CAGR between 2025 and 2035.
What are the key product types in the antibody market?
The key product types in antibody market are cancer, cardiovascular diseases, cns disorders, autoimmune disorders and other disease indications.
Which product type segment to contribute significant share in the antibody market in 2025?
In terms of product type, monoclonal antibodies segment to command 58.4% share in the antibody market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Disease Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Disease Indication , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Disease Indication , 2025 to 2035
- Cancer
- Cardiovascular Diseases
- CNS Disorders
- Autoimmune Disorders
- Other Disease Indications
- Y to o to Y Growth Trend Analysis By Disease Indication , 2020 to 2024
- Absolute $ Opportunity Analysis By Disease Indication , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type, 2025 to 2035
- Monoclonal Antibodies
- Polyclonal Antibodies
- Antibody-Drug Complexes
- Y to o to Y Growth Trend Analysis By Product Type, 2020 to 2024
- Absolute $ Opportunity Analysis By Product Type, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2025 to 2035
- Hospitals
- Long-Term Care Facilities
- Research Institutes
- Other End Users
- Y to o to Y Growth Trend Analysis By End User, 2020 to 2024
- Absolute $ Opportunity Analysis By End User, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Disease Indication
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Indication
- By Product Type
- By End User
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Disease Indication
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Indication
- By Product Type
- By End User
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Disease Indication
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Indication
- By Product Type
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Disease Indication
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Indication
- By Product Type
- By End User
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Disease Indication
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Indication
- By Product Type
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Disease Indication
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Indication
- By Product Type
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Disease Indication
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Indication
- By Product Type
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Disease Indication
- By Product Type
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Disease Indication
- By Product Type
- By End User
- Competition Analysis
- Competition Deep Dive
- F. Hoffmann-La Roche AG
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Novartis AG
- Johnson & Johnson
- Takeda Pharmaceutical Company Limited
- Amgen Inc.
- F. Hoffmann-La Roche AG
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020 to 2035
- Table 2: Global Market Value (USD Million) Forecast by Disease Indication , 2020 to 2035
- Table 3: Global Market Value (USD Million) Forecast by Product Type, 2020 to 2035
- Table 4: Global Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 5: North America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 6: North America Market Value (USD Million) Forecast by Disease Indication , 2020 to 2035
- Table 7: North America Market Value (USD Million) Forecast by Product Type, 2020 to 2035
- Table 8: North America Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 10: Latin America Market Value (USD Million) Forecast by Disease Indication , 2020 to 2035
- Table 11: Latin America Market Value (USD Million) Forecast by Product Type, 2020 to 2035
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 14: Western Europe Market Value (USD Million) Forecast by Disease Indication , 2020 to 2035
- Table 15: Western Europe Market Value (USD Million) Forecast by Product Type, 2020 to 2035
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Disease Indication , 2020 to 2035
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Product Type, 2020 to 2035
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 22: East Asia Market Value (USD Million) Forecast by Disease Indication , 2020 to 2035
- Table 23: East Asia Market Value (USD Million) Forecast by Product Type, 2020 to 2035
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Disease Indication , 2020 to 2035
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Product Type, 2020 to 2035
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Disease Indication , 2020 to 2035
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Product Type, 2020 to 2035
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2020 to 2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value Share and BPS Analysis by Disease Indication , 2025 and 2035
- Figure 4: Global Market Y to o to Y Growth Comparison by Disease Indication , 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Disease Indication
- Figure 6: Global Market Value Share and BPS Analysis by Product Type, 2025 and 2035
- Figure 7: Global Market Y to o to Y Growth Comparison by Product Type, 2025-2035
- Figure 8: Global Market Attractiveness Analysis by Product Type
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 10: Global Market Y to o to Y Growth Comparison by End User, 2025-2035
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 13: Global Market Y to o to Y Growth Comparison by Region, 2025-2035
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2025-2035
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2025-2035
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2025-2035
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2025-2035
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2025-2035
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 23: North America Market Value Share and BPS Analysis by Disease Indication , 2025 and 2035
- Figure 24: North America Market Y to o to Y Growth Comparison by Disease Indication , 2025-2035
- Figure 25: North America Market Attractiveness Analysis by Disease Indication
- Figure 26: North America Market Value Share and BPS Analysis by Product Type, 2025 and 2035
- Figure 27: North America Market Y to o to Y Growth Comparison by Product Type, 2025-2035
- Figure 28: North America Market Attractiveness Analysis by Product Type
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 30: North America Market Y to o to Y Growth Comparison by End User, 2025-2035
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 33: Latin America Market Value Share and BPS Analysis by Disease Indication , 2025 and 2035
- Figure 34: Latin America Market Y to o to Y Growth Comparison by Disease Indication , 2025-2035
- Figure 35: Latin America Market Attractiveness Analysis by Disease Indication
- Figure 36: Latin America Market Value Share and BPS Analysis by Product Type, 2025 and 2035
- Figure 37: Latin America Market Y to o to Y Growth Comparison by Product Type, 2025-2035
- Figure 38: Latin America Market Attractiveness Analysis by Product Type
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 40: Latin America Market Y to o to Y Growth Comparison by End User, 2025-2035
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 43: Western Europe Market Value Share and BPS Analysis by Disease Indication , 2025 and 2035
- Figure 44: Western Europe Market Y to o to Y Growth Comparison by Disease Indication , 2025-2035
- Figure 45: Western Europe Market Attractiveness Analysis by Disease Indication
- Figure 46: Western Europe Market Value Share and BPS Analysis by Product Type, 2025 and 2035
- Figure 47: Western Europe Market Y to o to Y Growth Comparison by Product Type, 2025-2035
- Figure 48: Western Europe Market Attractiveness Analysis by Product Type
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 50: Western Europe Market Y to o to Y Growth Comparison by End User, 2025-2035
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Disease Indication , 2025 and 2035
- Figure 54: Eastern Europe Market Y to o to Y Growth Comparison by Disease Indication , 2025-2035
- Figure 55: Eastern Europe Market Attractiveness Analysis by Disease Indication
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Product Type, 2025 and 2035
- Figure 57: Eastern Europe Market Y to o to Y Growth Comparison by Product Type, 2025-2035
- Figure 58: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 60: Eastern Europe Market Y to o to Y Growth Comparison by End User, 2025-2035
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 63: East Asia Market Value Share and BPS Analysis by Disease Indication , 2025 and 2035
- Figure 64: East Asia Market Y to o to Y Growth Comparison by Disease Indication , 2025-2035
- Figure 65: East Asia Market Attractiveness Analysis by Disease Indication
- Figure 66: East Asia Market Value Share and BPS Analysis by Product Type, 2025 and 2035
- Figure 67: East Asia Market Y to o to Y Growth Comparison by Product Type, 2025-2035
- Figure 68: East Asia Market Attractiveness Analysis by Product Type
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 70: East Asia Market Y to o to Y Growth Comparison by End User, 2025-2035
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Disease Indication , 2025 and 2035
- Figure 74: South Asia and Pacific Market Y to o to Y Growth Comparison by Disease Indication , 2025-2035
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Disease Indication
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Product Type, 2025 and 2035
- Figure 77: South Asia and Pacific Market Y to o to Y Growth Comparison by Product Type, 2025-2035
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 80: South Asia and Pacific Market Y to o to Y Growth Comparison by End User, 2025-2035
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Disease Indication , 2025 and 2035
- Figure 84: Middle East & Africa Market Y to o to Y Growth Comparison by Disease Indication , 2025-2035
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Disease Indication
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Product Type, 2025 and 2035
- Figure 87: Middle East & Africa Market Y to o to Y Growth Comparison by Product Type, 2025-2035
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 90: Middle East & Africa Market Y to o to Y Growth Comparison by End User, 2025-2035
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

