IV Bag Market
IV Bag Market Forecast and Outlook 2026 to 2036
IV Bag Market Size, Market Forecast and Outlook By FMI
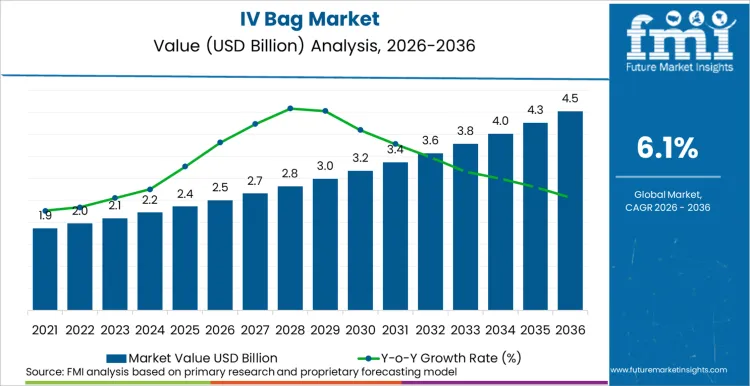
The IV Bag market is projected to reach a valuation of USD 2.5 billion in 2026, driven by the escalating global burden of chronic diseases necessitating parenteral nutrition and hydration. The industry is estimated to expand at a steady CAGR of 6.1% through 2036, pushing total sector worth beyond USD 3.8 billion by the conclusion of this assessment period. As per FMI's projection, manufacturers are transitioning towards non-DEHP PVC and polyolefin-based films, with material costs for these advanced polymers averaging 15-20% higher than traditional PVC, yet necessary to meet strict EU MDR and USA FDA safety guidelines regarding plasticizers.
Summary of IV Bag Market
- Market Snapshot
- The IV Bag Market is estimated at USD 2.5 billion in 2026.
- The IV Bag Market is projected to reach USD 3.8 billion by 2036 at a CAGR of 6.1%.
- The market growth is driven by rising demand for parenteral nutrition and hydration due to chronic diseases.
- Demand and Growth Drivers
- Transition to non-DEHP PVC and polyolefin materials increases costs by 15-20% but ensures regulatory compliance.
- Ready-to-administer (RTA) IV bags reduce medication errors by approximately 30%, driving hospital adoption.
- Expansion of home healthcare and infusion therapies is increasing demand for portable and durable IV bags.
- Product and Segment View
- Single chamber IV bags hold a 58% share due to high-volume use in fluid resuscitation and basic therapies.
- Intravenous therapies account for 60% of market share driven by widespread hospital use.
- PVC-based materials lead with a 46% share due to cost efficiency and established manufacturing systems.
- Geography and Competitive Outlook
- India leads with a CAGR of 7.2%, followed by China at 7.0% and the United States at 5.0%.
- Germany and Japan are expected to grow at CAGRs of 4.6% and 3.3%, respectively.
- Key players include Baxter, B. Braun, ICU Medical, Fresenius Kabi, and Terumo focusing on vertical integration and premixed drug solutions.
Companies are shifting focus from standard saline bags to premixed drug formulations, redefining procurement habits in major hospital networks where ready-to-administer (RTA) formats reduce medication errors by approximately 30%. Such behavioral changes encourage converters to introduce comprehensive multi-chamber portfolios utilizing radio-frequency (RF) welding technologies for precise compartmentalization.
Key Industry Insights
How does the shift to non-PVC materials impact manufacturing equipment capability?
Transitioning to polyolefins (PP/PE) often requires upgrading from radio-frequency (RF) welding to thermal contact or laser welding systems, as non-polar materials do not respond well to traditional RF sealing. This capital expenditure is a critical consideration for legacy PVC manufacturers aiming to enter the eco-friendly or oncology segments.
What are the sterilization bottlenecks for premixed multi-chamber bags?
Premixed bags containing heat-sensitive amino acids or lipids cannot withstand standard steam autoclaving cycles (121°C). Manufacturers must invest in aseptic filling lines or lower-temperature sterilization technologies, which significantly increases the "Production Cycle Time" and operational complexity compared to standard saline bags.
How can supply chain resilience be maintained for critical resin inputs?
Dependence on specific medical-grade resin grades creates vulnerability. Key players are establishing dual-source agreements for medical-grade PVC and PP resins to prevent production stoppages, ensuring continuous supply to hospitals even during petrochemical market fluctuations.
Healthcare infrastructure expansion and home infusion trends are significantly influencing industry expansion. Packaging engineers emphasize barrier properties, utilizing multi-layer laminates to prevent oxidation of sensitive parenteral nutrition formulations during shelf life. As per FMI's estimates, clinical trials investigating the efficacy of home-based chemotherapy are creating needs for portable, high-durability elastomeric bags, instilling confidence among oncology providers regarding future product pipelines.
Does Gross Margin % Absorb Raw Material Volatility?
Gross margins in the IV bag sector typically range between 35% and 45%, but are sensitive to petrochemical price shifts. Manufacturers utilizing vertical integration for film extrusion can better protect these margins. The shift towards higher-value iv bag and tubing recycling initiatives and non-PVC materials also allows for premium pricing, buffering against commoditized saline bag margins.
How Critical is Production Cycle Time for Sterility?
Production cycle time is heavily influenced by the sterilization phase, which can take several hours. optimizing autoclave load configurations and cool-down periods is essential for throughput. For iv infusion gravity bags, reducing cycle time directly correlates to higher asset utilization of expensive cleanroom facilities.
Why is Inventory Turnover Ratio Vital for Premixed Drugs?
Inventory turnover is critical for premixed therapeutic bags due to shorter shelf lives compared to empty containers. Efficient management ensures that drug-filled bags reach hospitals well before expiry. High turnover is particularly important for pressure infusion bags distributors handling time-sensitive emergency supplies.
Companies are launching infusion fluid holder compatible designs that integrate seamlessly with smart pumps. There is a notable trend towards acquiring regional film extruders to control the quality of the primary packaging material. Manufacturers are also refining intravenous packaging labeling to include barcodes for medication safety tracking. This strategic pivot towards integrated, safety-engineered fluid delivery systems ensures long-term relevance in a patient-centric healthcare landscape.
IV Bag Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 2.5 Billion |
| Industry Value (2036) | USD 3.8 Billion |
| CAGR (2026-2036) | 6.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
What Drivers Are Accelerating Non-PVC Adoption?
The shift toward eco-friendly and safe medical plastics is serving as a significant catalyst for broader acceptance of Polypropylene (PP) and Polyethylene (PE) bags. Recent breakthroughs in iv disposables manufacturing have allowed for the production of multi-layer non-PVC films that match the flexibility of PVC without the risk of DEHP leaching. This holistic approach appeals to pediatric and neonatal units seeking to minimize toxin exposure in vulnerable patients. Clinical success of non-PVC containers in preventing drug adsorption (common with oncology drugs and PVC) has expanded application possibilities for complex compounding. Education programs highlighting the environmental benefits of incineration-safe non-PVC materials are driving procurement proactivity. FMI analysts opine that converters are capitalizing on these trends by introducing iv poles compatible hanging systems integrated directly into the bag design. Integrating self-sealing ports is becoming considered standard, ensuring infection control mandates are met without additional components.
How Is the IV Bag Market Segmented Across Key Categories?
The global landscape is intricately segmented to address diverse requirements of critical care, oncology, and general medicine, categorizing the sector by material, chamber type, and application. Structural division allows stakeholders to identify specific growth pockets, such as rising demand for medical specialty bag solutions in dialysis or preference for multiple chamber bags in parenteral nutrition. By analyzing these segments, manufacturers can tailor production lines to meet distinct needs of high-volume saline production versus high-value drug delivery systems.
Why Do Single Chamber IV Bags Maintain Dominance?
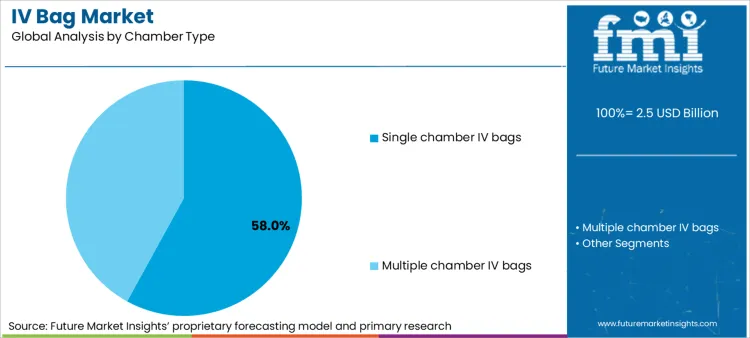
Single chamber IV bags account for 58% of share in 2026, driven by their foundational role in fluid resuscitation and basic drug delivery. Leadership position is underpinned by widespread adoption in emergency rooms and general wards for saline, dextrose, and Ringer's lactate solutions. Segment benefits from continuous manufacturing efficiencies, allowing for low unit costs essential for high-volume disposables. Research focused on blood bags similarities suggests that the single-chamber format remains the standard for simplicity and ease of use. Critical role in acting as a diluent carrier for concentrated medications further solidifies standing. As supply chains prioritize cost-efficiency, demand for robust single-chamber formats is expected to remain robust.
Why Is Intravenous Therapies the Primary Revenue Stream?
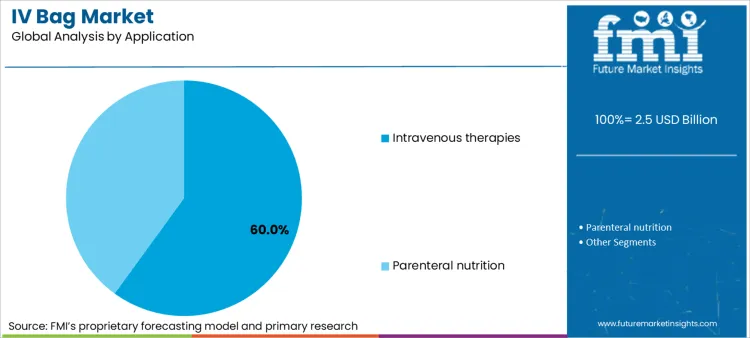
Intravenous therapies command 60% share of sector in 2026, reflecting the universal reliance on IV delivery for rapid medication onset. Dominance is inextricably linked to the treatment of dehydration, infections (antibiotics), and pain management in hospital settings. Manufacturers rely on specialized blood bag tube sealer technology to ensure leak-proof closure of these critical fluid containers. These settings are essential for handling acute care scenarios, driving priority status in procurement budgets. Relentless pursuit of patient safety ensures packaging designed with clear graduation marks and tamper-evidence remains top priority. High consumption volume necessitates dynamic material supply, bolstering sustained revenue streams for medical packaging converters.
How Will Home Healthcare Trends Redefine Design?
Ambulatory care adoption is set to reshape sales landscapes by demanding portable and durable fluid containers. As patients move therapies home, industries access infusion pumps compatible bags that can withstand movement and varied storage conditions. Shift toward patient-administered care creates demand for bags with intuitive connecting systems to prevent errors. Manufacturers failing to upgrade ergonomic designs risk losing visibility among home-care providers. Availability of pre-filled, long-stability bags drives complimentary consumption in the chronic care sector. Rise of tele-health monitoring stimulates innovation, with companies differentiating offerings by providing smart bags that track fluid usage.
What Role Does Sustainability Play in Medical Plastics?
Circular economy adoption is set to reshape manufacturing priorities by demanding recyclable or reduced-plastic solutions. As hospital waste management costs mount, brands demand blood collection devices and IV bags that fit into recycling streams. Shift toward PVC-free materials creates demand for polyolefins that can be incinerated safely without releasing dioxins. Manufacturers failing to optimize material selection for environmental impact risk losing visibility among green-hospital initiatives. Availability of take-back programs drives complimentary consumption of sustainable medical plastics. Rise of "Green OT" certifications stimulates innovation, with companies differentiating offerings by providing lifecycle assessment data.
How Are Premixed Formulations Evolving?
Ready-to-administer adoption is set to reshape manufacturing priorities by demanding barrier films that protect drug stability. As pharmacy compounding regulations tighten, hospitals demand rapid infuser ready bags pre-filled with antibiotics or analgesics. Shift toward multi-chamber bags creates demand for peelable seals that separate unstable components until administration. Manufacturers failing to optimize barrier properties against oxygen and moisture risk losing visibility among pharmaceutical partners. Availability of custom-compounded bags drives complimentary consumption in the specialty pharmacy sector. Rise of labor-saving devices stimulates innovation, with companies differentiating offerings by providing plug-and-play solutions for busy nursing staff.
How Will the IV Bag Industry Evolve in Major Nations?
The global landscape for parenteral packaging is characterized by diverse growth patterns, influenced by healthcare spending and disease prevalence. Established markets prioritize safety features and premixed drugs, whereas emerging economies rely on volume expansion of basic fluids. Asia-Pacific is emerging as a rapid growth hub due to improving hospital infrastructure and rising chronic disease rates. Conversely, North American sectors pivot towards high-value specialty nutrition and drug delivery systems. Government initiatives in developing nations supporting domestic medical manufacturing ensure sustained demand for reliable fluid therapy solutions across all geographies.
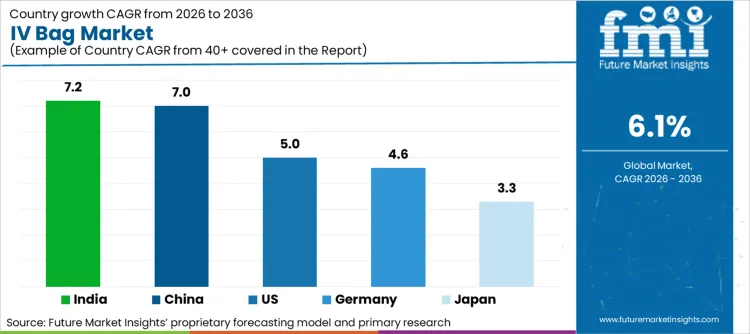
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 7.2% |
| China | 7.0% |
| United States | 5.0% |
| Germany | 4.6% |
| Japan | 3.3% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Why is the IV Bag Sector in India Exploding?
Demand for fluid therapy in India is projected to rise at 7.2% CAGR during assessment period. Initiatives emphasizing "Ayushman Bharat" universal health coverage foster favorable environment for domestic medical device vendors. Increasing investment in hospital chains in tier-2 cities creates massive supply channel for saline and dextrose bags. Rising prevalence of lifestyle diseases prompts local firms to seek reliable blood banking equipment and IV solutions. As per FMI's projection, shift is supported by government policies promoting local manufacturing of medical disposables, accelerating uptake of cost-effective IV bags across country and fueling sector expansion.
Why is the IV Bag Industry in China Expanding Rapidly?
The IV Bag industry in China is poised to expand at 7.0% CAGR. Surge is largely attributed to the aging population and expansion of critical care facilities. Government focus on modernizing medical consumables encourages facilities to opt for non-PVC and multi-chamber bags. Increasing partnerships between local pharma giants and packaging firms necessitate availability of high-quality barrier films. High volume of inpatient admissions attract significant investment in automated IV production lines. Strict adherence to NMPA safety regulations further drives adoption of advanced medical polymers.
Why is the IV Bag Industry in the United States Showing Strong Growth?
Sales of IV containers in United States are set to grow at 5.0% CAGR through 2036. Expansion is underpinned by the high prevalence of chronic conditions requiring parenteral nutrition and chemotherapy. Growth is fueled by widespread shift towards premixed solutions to mitigate compounding errors. Rapid adoption of united states blood collection devices and advanced infusion therapies drives revenue generation per unit. Presence of major healthcare conglomerates within country streamlines supply chains. According to FMI's estimates, emphasis on infection control and nurse safety continues to be primary driver, maintaining United States' position as critical innovation generator.
Why is the IV Bag Industry in Germany Showing Stability?
The IV Bag industry in Germany is poised to expand at 4.6% CAGR. Surge is largely attributed to robust healthcare system and early adoption of non-PVC standards. Government focus on sustainability in healthcare encourages facilities to opt for eco-friendly packaging materials. Increasing partnerships between medical technology firms and hospitals necessitate availability of specialized blood bank reagent compatible bags. High standards for patient safety combined with aging demographics attract significant investment in nutritional therapy bags. Strict adherence to EU Medical Device Regulations (MDR) further drives adoption of compliant solutions.
Why is the IV Bag Sector in Japan Expanding?
Demand for geriatric care fluids in Japan is projected to rise at 3.3% CAGR during assessment period. Initiatives emphasizing home-based care for the elderly foster favorable environment for portable IV solutions. Increasing investment in nutritional support products creates steady supply channel for multi-chamber parenteral bags. Rising awareness regarding medical waste prompts local administrators to seek low-volume, high-efficiency packaging. Shift is supported by government policies improving long-term care infrastructure, accelerating uptake of user-friendly bag designs across country and fueling sector expansion.
What is the Competitive Outlook for the IV Bag Market?
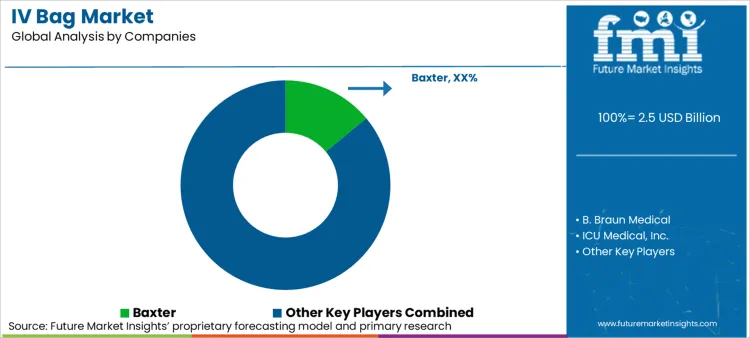
Market participants are increasingly focusing on vertical integration to capture value from resin extrusion to final filling. FMI analysts opine that leading players like Baxter and B. Braun are directing investment toward expanding global manufacturing footprints to reduce supply chain risks. Significant trend involves developing proprietary film formulations that offer superior drug compatibility. Companies actively engage in strategic collaborations with pharmaceutical companies to develop premixed drug bags. Sustainability emerges as competitive frontier, with providers exploring bio-based polymers for medical use. Consolidation activities remain high as large med-tech conglomerates acquire regional manufacturers to broaden their capacity against regional competitors. Baxter holds a significant 14% share, leveraging its diversified portfolio of renal and hospital products.
Key Players in IV Bag Market
- Baxter
- B. Braun Medical
- ICU Medical, Inc.
- Fresenius Kabi Group
- Hospira
- Terumo Corporation
- Wipak Group Management
- Poly Medicure
- Renolit
- Mountainside Medical Equipment
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Material Type | Polyvinyl chloride (PVC), Polyethylene (PE), Ethylene vinyl acetate (EVA), Polypropylene (PP), Copolyester ether, Other materials |
| Chamber Type | Single chamber IV bags, Multiple chamber IV bags |
| Application | Intravenous therapies, Parenteral nutrition |
| IV Bag Size | Less than 250 ml, 250 ml to 500 ml, 500 ml to 1000 ml, More than 1000 ml |
| Regions | North America, Europe, East Asia, South Asia, Latin America, MEA |
IV Bag Market by Segments
Material Type
- Polyvinyl chloride (PVC)
- Polyethylene (PE)
- Ethylene vinyl acetate (EVA)
- Polypropylene (PP)
- Copolyester ether
- Other materials
Chamber Type
- Single chamber IV bags
- Multiple chamber IV bags
Application
- Intravenous therapies
- Parenteral nutrition
IV Bag Size
- Less than 250 ml
- 250 ml to 500 ml
- 500 ml to 1000 ml
- More than 1000 ml
Bibliography
- USA Food and Drug Administration. (2025, August 7). Statement from FDA Commissioner Marty Makary, M.D., M.P.H., announcing resolution of the IV saline solutions shortage. USA Food and Drug Administration, United States.
- World Health Organization. (2024). Injection safety. World Health Organization, Switzerland.
- USA Environmental Protection Agency. (2024). Sustainable materials management and plastics recycling in healthcare facilities. USA Environmental Protection Agency, United States.
- Association for the Advancement of Medical Instrumentation. (2024). ANSI/AAMI ST79: Comprehensive guide to steam sterilization and sterility assurance in health care facilities. Association for the Advancement of Medical Instrumentation, United States.
Frequently Asked Questions
How large is the demand for IV Bags in the global market in 2026?
Demand for IV Bags in global market is estimated to be valued at USD 2.5 billion in 2026.
What will be the market size of IV Bags in the global market by 2036?
Market size for IV Bags is projected to reach USD 3.8 billion by 2036.
What is the expected demand growth for IV Bags in the global market between 2026 and 2036?
Demand for IV Bags in global market is expected to grow at CAGR of 6.1% between 2026 and 2036.
Which Chamber Type is poised to lead global sales by 2026?
Single chamber IV bags are expected to be dominant chamber type, capturing approximately 58% of global market share in 2026 due to general ward usage.
How significant is the role of Intravenous Therapies End Use in driving IV Bag adoption in 2026?
Intravenous Therapies End Use represents critical application, projected to hold substantial 60% share of total market in 2026 as hospital admissions rise.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Roots Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended A9oach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Material Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Material Type , 2026 to 2036
- Polyvinyl chloride (PVC)
- Polyethylene (PE)
- Ethylene vinyl acetate (EVA)
- Polypropylene (PP)
- Copolyester ether
- Other materials
- Polyvinyl chloride (PVC)
- Y to o to Y Growth Trend Analysis By Material Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Material Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Chamber Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Chamber Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Chamber Type, 2026 to 2036
- Single chamber IV bags
- Multiple chamber IV bags
- Single chamber IV bags
- Y to o to Y Growth Trend Analysis By Chamber Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Chamber Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Intravenous therapies
- Parenteral nutrition
- Intravenous therapies
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By IV Bag Size
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By IV Bag Size, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By IV Bag Size, 2026 to 2036
- Less than 250 ml
- 250 ml to 500 ml
- 500 ml to 1000 ml
- More than 1000 ml
- Less than 250 ml
- Y to o to Y Growth Trend Analysis By IV Bag Size, 2021 to 2025
- Absolute $ Opportunity Analysis By IV Bag Size, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Material Type
- By Chamber Type
- By Application
- By IV Bag Size
- Competition Analysis
- Competition Deep Dive
- Baxter
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- B. Braun Medical
- ICU Medical, Inc.
- Fresenius Kabi Group
- Hospira
- Terumo Corporation
- Wipak Group Management
- Poly Medicure
- Renolit
- Mountainside Medical Equipment
- Baxter
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Chamber Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by IV Bag Size, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Chamber Type, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by IV Bag Size, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Chamber Type, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by IV Bag Size, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Chamber Type, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by IV Bag Size, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Chamber Type, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by IV Bag Size, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Chamber Type, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by IV Bag Size, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Chamber Type, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by IV Bag Size, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Chamber Type, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by IV Bag Size, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Material Type
- Figure 6: Global Market Value Share and BPS Analysis by Chamber Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Chamber Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Chamber Type
- Figure 9: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Application
- Figure 12: Global Market Value Share and BPS Analysis by IV Bag Size, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by IV Bag Size, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by IV Bag Size
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Material Type
- Figure 29: North America Market Value Share and BPS Analysis by Chamber Type, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Chamber Type, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Chamber Type
- Figure 32: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Application
- Figure 35: North America Market Value Share and BPS Analysis by IV Bag Size, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by IV Bag Size, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by IV Bag Size
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Material Type
- Figure 42: Latin America Market Value Share and BPS Analysis by Chamber Type, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Chamber Type, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Chamber Type
- Figure 45: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Application
- Figure 48: Latin America Market Value Share and BPS Analysis by IV Bag Size, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by IV Bag Size, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by IV Bag Size
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by Material Type
- Figure 55: Western Europe Market Value Share and BPS Analysis by Chamber Type, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Chamber Type, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Chamber Type
- Figure 58: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Application
- Figure 61: Western Europe Market Value Share and BPS Analysis by IV Bag Size, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by IV Bag Size, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by IV Bag Size
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Material Type
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Chamber Type, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Chamber Type, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Chamber Type
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Application
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by IV Bag Size, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by IV Bag Size, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by IV Bag Size
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by Material Type
- Figure 81: East Asia Market Value Share and BPS Analysis by Chamber Type, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Chamber Type, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Chamber Type
- Figure 84: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Application
- Figure 87: East Asia Market Value Share and BPS Analysis by IV Bag Size, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by IV Bag Size, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by IV Bag Size
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Material Type
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Chamber Type, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Chamber Type, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Chamber Type
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by IV Bag Size, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by IV Bag Size, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by IV Bag Size
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Material Type
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Chamber Type, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Chamber Type, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Chamber Type
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by IV Bag Size, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by IV Bag Size, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by IV Bag Size
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

