Zoonotic Antivirals Market
Zoonotic Antivirals Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Zoonotic Antivirals Market Forecast and Outlook 2026 to 2036
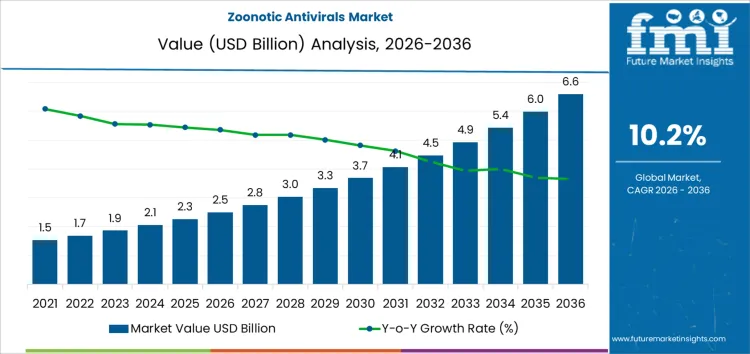
The zoonotic antivirals market is expected to be valued at USD 2.5 billion in 2026 and reach USD 6.6 billion by 2036 at a CAGR of 10.2%. Value formation in this space reflects preparedness-oriented purchasing rather than steady therapeutic consumption. Procurement decisions are concentrated within public health agencies that prioritize antivirals with cross-pathogen activity, long shelf stability, and rapid deployment profiles. Spending is episodic, tied to surveillance alerts, risk assessments, and policy mandates rather than seasonal treatment cycles. Development pipelines emphasize mechanisms conserved across viral families to preserve utility against uncertain spillover threats. Manufacturing strategies balance surge readiness with obsolescence risk, shaping batch sizing and inventory rotation practices.
Stockpile composition, regulatory acceleration pathways, and intergovernmental coordination largely determine realized revenue timing. Hospitals represent secondary consumption nodes, activating antiviral use only when case confirmation and treatment protocols align.
Pricing dynamics differ from conventional infectious disease therapeutics, since volume commitments are negotiated in advance under preparedness frameworks. Clinical trial design remains constrained by sporadic incidence, influencing approval routes and post-authorization evidence requirements. Capital allocation favors platforms adaptable to multiple outbreak scenarios, reinforcing concentration among suppliers with scalable manufacturing and policy-facing capabilities. Funding cycles further affect contract timing and replenishment frequency globally aligned.
Quick Stats for Zoonotic Antivirals Market
- Zoonotic Antivirals Market Value (2026): USD 2.5 billion
- Zoonotic Antivirals Market Forecast Value (2036): USD 6.6 billion
- Zoonotic Antivirals Market Forecast CAGR 2026 to 2036: 10.2%
- Leading Product Category by Utilization: Broad-spectrum antivirals
- Fastest-Growing Countries: India, China, Brazil, USA, UK
- Top Players in Global Demand: Gilead Sciences, Roche, Pfizer, Merck, Johnson & Johnson
Zoonotic Antivirals Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 2.5 billion |
| Market Forecast Value (2036) | USD 6.6 billion |
| Forecast CAGR 2026 to 2036 | 10.2% |
Why is the Demand for Zoonotic Antivirals Growing?
Demand for zoonotic antivirals is increasing as public health authorities and pharmaceutical developers respond to rising incidence and recognition of viral diseases transmitted between animals and humans. Pathogens such as influenza A viruses, certain coronaviruses, and zoonotic flaviviruses pose ongoing risks for spillover events that can lead to outbreaks with significant morbidity and mortality. Healthcare systems and research institutions are prioritizing antiviral agents that target conserved viral replication mechanisms to reduce viral load, limit disease progression, and shorten infectious periods in affected individuals.
Procurement teams within hospitals and national stockpile programs evaluate antiviral efficacy against multiple zoonotic strains, resistance profiles, and pharmacokinetic properties because broad utility across potential outbreak scenarios supports preparedness planning and treatment continuity. Clinical adoption is influenced by regulatory frameworks that encourage accelerated review of therapeutic candidates with demonstrated activity against high-priority zoonotic viruses.
Growth in global surveillance efforts and increased reporting of zoonotic disease clusters drives investment in antiviral discovery and clinical pipelines that can be deployed rapidly when novel or re-emerging agents are identified. Collaboration between veterinary health and human public health agencies reinforces development of antivirals that may serve dual use in outbreak control, particularly where animal reservoirs contribute to sustained transmission cycles. Real-world data on treatment outcomes inform clinical guidelines and support reimbursement decisions that influence hospital formulary inclusion. These application-specific clinical and preparedness requirements are contributing to sustained demand growth in the zoonotic antivirals market.
How Is the Zoonotic Antivirals Industry Segmented?
The zoonotic antivirals industry is influenced by outbreak preparedness planning, cross-species transmission risk, and public health response frameworks. Treatment strategies prioritize rapid deployment, broad viral coverage, and compatibility with emergency use protocols. Adoption patterns reflect surveillance intensity, regulatory readiness, and stockpiling policies rather than routine prescribing volumes. Therapy selection considers spectrum of activity, resistance risk, and scalability during outbreaks. Segmentation highlights how antiviral design, infection context, and institutional procurement channels shape utilization, inventory management, and deployment timing across zoonotic disease control efforts.
Which Product Category Shows the Highest Utilization in Zoonotic Antivirals?
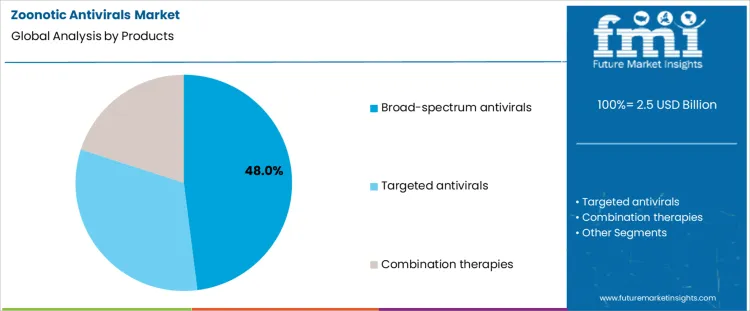
Utilization concentrates around broad-spectrum antivirals at 48.0%, reflecting their role in early-stage outbreak response before pathogen-specific confirmation. These agents support empirical treatment where viral identity and mutation profile remain uncertain. Flexibility across multiple virus families enhances suitability for emergency deployment and stockpiling. Targeted antivirals at 32.0% are used once diagnostic clarity improves and pathogen-specific efficacy is established. Combination therapies at 20.0% address resistance management and severe cases under controlled protocols. Product segmentation reflects prioritization of versatility and readiness over precision during initial zoonotic threat containment.
Key Points
- Broad-spectrum agents support early, uncertainty-driven response.
- Targeted options follow diagnostic confirmation.
- Combination use focuses on resistance or severe infection scenarios.
How Do Emerging and Endemic Zoonoses Influence Application-Level Demand?
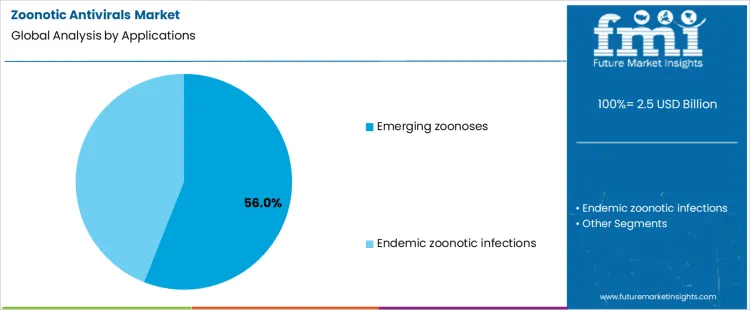
Application demand is weighted toward emerging zoonoses at 56.0%, driven by unpredictable spillover events and limited preexisting immunity. Rapid transmission potential necessitates immediate antiviral availability under emergency frameworks. Surveillance-driven alerts accelerate deployment before full epidemiological characterization. Endemic zoonotic infections at 44.0% involve recurring exposure patterns with established treatment protocols. These infections allow more structured antiviral use and procurement planning. Application segmentation reflects higher urgency and volume concentration where outbreak uncertainty, rapid spread risk, and containment priorities dominate public health decision-making.
Key Points
- Emerging threats require immediate antiviral readiness.
- Surveillance triggers early deployment decisions.
- Endemic infections follow structured treatment pathways.
Which End User Dominates Zoonotic Antiviral Procurement and Deployment?
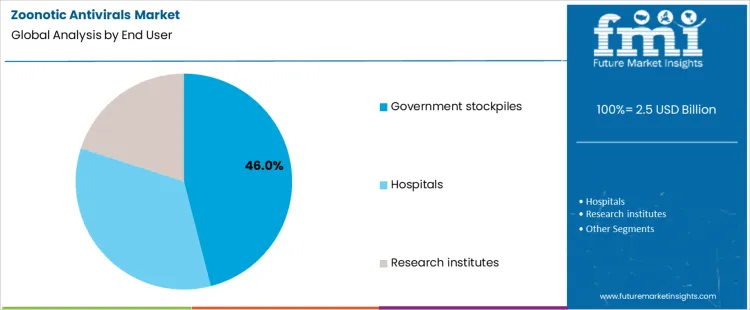
Government stockpiles account for 46.0% of antiviral utilization, driven by national preparedness and emergency response mandates. Centralized procurement supports rapid distribution during outbreaks and cross-border health events. Inventory management emphasizes shelf-life, rotation policies, and surge capacity. Hospitals at 34.0% administer antivirals during active case management and containment phases. Research institutes at 20.0% support clinical evaluation, resistance monitoring, and protocol development. End-user segmentation reflects concentration where policy authority, funding control, and large-scale deployment capability determine antiviral utilization patterns.
Key Points
- Governments prioritize preparedness and rapid deployment capacity.
- Hospitals focus on active case treatment.
- Research centers support evaluation and protocol refinement.
What are the Key Dynamics in the Zoonotic Antivirals Market?
Demand for zoonotic antivirals reflects public health and clinical need to treat viral infections transmitted from animals to humans with outbreak potential. Adoption concentrates in hospital infectious disease units, public health stockpiles, and emergency response systems. Global scope aligns with surveillance-driven preparedness and outbreak containment strategies. Usage centers on broad-spectrum and pathogen-specific antiviral agents deployed for treatment, post-exposure prophylaxis, and limited preventive use during zoonotic spillover events.
How do outbreak preparedness requirements and clinical risk profiles shape adoption?
Zoonotic viral infections often emerge unpredictably, with rapid human-to-human transmission risk following animal spillover. Demand increases where early antiviral intervention reduces disease severity, transmission window, or mortality. Health systems prioritize agents with activity against high-consequence viruses and acceptable safety profiles under emergency use. Treatment protocols emphasize rapid deployment, simplified dosing, and compatibility with critical care settings. Stockpiling decisions depend on spectrum of activity, shelf life, and ease of administration. Use cases extend to healthcare workers and exposed populations during containment phases. Coordination with surveillance data informs targeted deployment. Adoption reflects operational need for readiness and immediate therapeutic response rather than routine chronic use.
How do development uncertainty, regulatory complexity, and economic constraints influence market scalability?
Zoonotic antivirals face uncertain demand due to sporadic outbreak occurrence. Demand sensitivity rises where development investment lacks predictable utilization pathways. Clinical trials remain challenging due to limited case numbers and ethical constraints during outbreaks. Regulatory approvals often rely on accelerated or emergency frameworks, increasing post-authorization obligations. Manufacturing scale-up must balance surge capacity with inventory obsolescence risk. Pricing and reimbursement depend heavily on government procurement rather than commercial channels. Resistance development remains a concern for broad-spectrum agents. Scalability remains constrained by episodic demand patterns, high development risk, and dependence on public sector funding and preparedness policies.
How Is Demand for Zoonotic Antivirals Evolving Globally?
Sales of zoonotic antivirals are expanding globally as surveillance intensifies at the human-animal interface and preparedness planning advances. Public health agencies prioritize stockpiling and rapid deployment for spillover-prone pathogens. Hospital formularies integrate antivirals aligned with outbreak protocols, while research pipelines emphasize broad-spectrum activity. Manufacturing scale-up and regional procurement frameworks influence access. Growth rates in India at 11.9%, China at 11.7%, Brazil at 11.4%, USA at 10.8%, and the UK at 9.7% indicate sustained expansion driven by preparedness mandates, surveillance-triggered utilization, and protocolized response rather than routine seasonal prescribing.
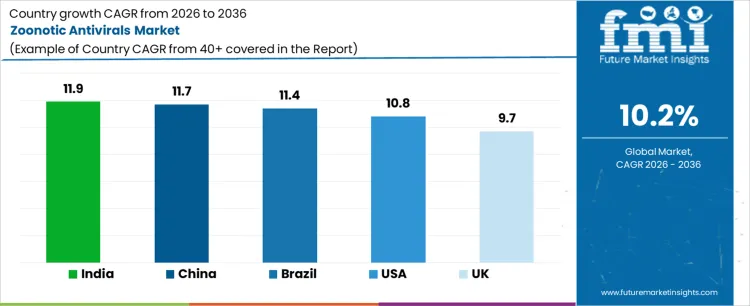
| Country | CAGR (%) |
|---|---|
| India | 11.9% |
| China | 11.7% |
| Brazil | 11.4% |
| USA | 10.8% |
| UK | 9.7% |
What Is Driving Expansion of Zoonotic Antiviral Demand in India?
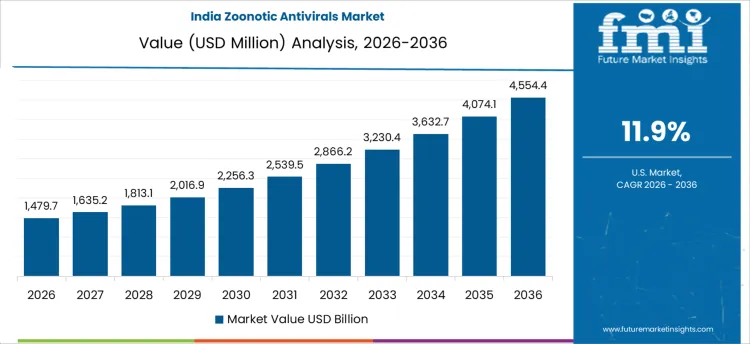
Demand for zoonotic antivirals in India is growing at a CAGR of 11.9%, shaped by surveillance expansion and public health procurement. National programs strengthen detection across high-risk interfaces involving livestock, wildlife, and dense urban populations. Government stockpiling supports readiness for spillover events. Public laboratories and tertiary hospitals align formularies with response guidelines. Domestic pharmaceutical capacity improves availability and price stability. Growth reflects preparedness-oriented purchasing and protocol alignment rather than continuous outpatient utilization.
- Expanded zoonotic surveillance coverage
- Government-led strategic stockpiling programs
- Alignment of hospital formularies with protocols
- Strong domestic manufacturing capacity
Why Is China Experiencing Rapid Uptake of Zoonotic Antivirals?
Zoonotic antiviral demand in China is expanding at a CAGR of 11.7%, supported by integrated surveillance and centralized procurement. National monitoring systems prioritize early containment of cross-species transmission. Public hospitals maintain antivirals for rapid deployment during alerts. State-supported manufacturing ensures supply continuity. Research institutions collaborate on broad-spectrum candidates. Growth reflects system-level preparedness and rapid-response logistics rather than routine therapeutic substitution.
- Integrated national surveillance systems
- Centralized procurement for rapid deployment
- State-supported manufacturing reliability
- Research collaboration on broad-spectrum agents
How Is Brazil Sustaining Growth in Zoonotic Antiviral Utilization?
Demand for zoonotic antivirals in Brazil is growing at a CAGR of 11.4%, influenced by biodiversity exposure and public health response capacity. Federal programs prioritize antivirals for emerging infections linked to wildlife reservoirs. Public healthcare networks incorporate antivirals within contingency plans. Technology transfer agreements support local production. Regional laboratory expansion improves detection and response timing. Growth reflects preparedness investments and access scaling rather than steady-state clinical prescribing.
- Biodiversity-linked spillover risk management
- Federal contingency planning and procurement
- Local production via technology transfer
- Expanded regional laboratory capacity
What Factors Are Driving Zoonotic Antiviral Demand in United States?
Zoonotic antiviral demand in the United States is expanding at a CAGR of 10.8%, driven by preparedness frameworks and biodefense priorities. Strategic national stockpiles maintain antiviral inventories. Hospital systems integrate response protocols aligned with federal guidance. Advanced surveillance and genomic monitoring inform targeted deployment. Public-private partnerships accelerate manufacturing readiness. Growth reflects readiness spending and protocolized activation rather than continuous high-volume clinical use.
- Strategic national stockpile maintenance
- Hospital protocol integration for rapid response
- Advanced surveillance and genomic monitoring
- Public-private manufacturing partnerships
Why Is the UK Showing Sustained Growth in Zoonotic Antivirals?
Demand for zoonotic antivirals in United Kingdom is growing at a CAGR of 9.7%, supported by centralized health security planning. National agencies coordinate procurement and distribution for outbreak readiness. NHS trusts align formularies with contingency guidance. Research funding supports evaluation of antiviral effectiveness across scenarios. Managed access frameworks balance cost and readiness. Growth remains sustained, reflecting structured preparedness and coordinated response capacity rather than routine therapeutic expansion.
- Centralized health security procurement
- NHS formulary alignment with contingencies
- Research funding for effectiveness evaluation
- Managed access balancing readiness and cost
What is the competitive landscape of zoonotic antivirals market globally?
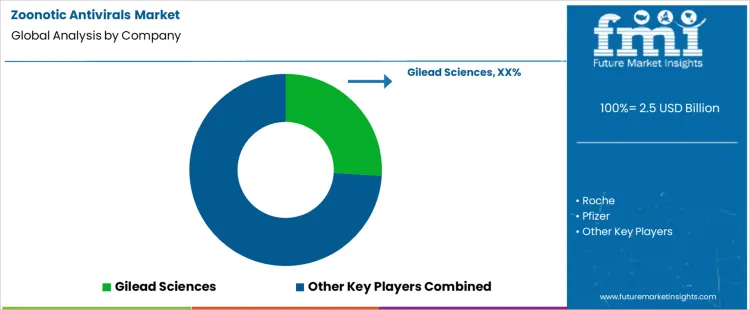
Demand for zoonotic antivirals is shaped by outbreak preparedness, cross species transmission risk, and rapid response requirements. Public health agencies and hospitals evaluate spectrum coverage, resistance barriers, stockpile stability, and scalability under surge conditions. Buyer assessment includes regulatory readiness, emergency use pathways, manufacturing resilience, cold chain needs, and global distribution reach. Procurement behavior prioritizes advance purchase agreements, shelf life predictability, quality assurance documentation, and rapid deployment capability.
Trend in the zoonotic antivirals market reflects preparedness funding cycles and integration with surveillance and diagnostics. Usage concentrates around respiratory and hemorrhagic threats with emphasis on early treatment windows.
Gilead Sciences leads competitive positioning through antiviral platforms with proven efficacy and scalable manufacturing for emergency demand. Roche competes via antiviral and diagnostic integration supporting rapid case identification and treatment alignment. Pfizer supports demand with oral antivirals, global commercialization strength, and experience navigating emergency authorizations.
Merck maintains relevance through broad antiviral research depth and established relationships with public sector buyers. Johnson and Johnson participates through diversified infectious disease capabilities and supply reliability across multiple geographies. Competitive differentiation centers on speed to scale, resistance management, access frameworks, and coordination with outbreak response systems.
Key Players in the Zoonotic Antivirals Market
- Gilead Sciences
- Roche
- Pfizer
- Merck
- Johnson & Johnson
- Others
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Products | Broad-spectrum antivirals; Targeted antivirals; Combination therapies |
| Applications | Emerging zoonoses; Endemic zoonotic infections |
| End User | Government stockpiles; Hospitals; Research institutes |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, Brazil, USA, UK, and 40+ countries |
| Key Companies Profiled | Gilead Sciences; Roche; Pfizer; Merck; Johnson & Johnson; Others |
| Additional Attributes | Dollar sales by product type, application, end user, and sales channel; antiviral spectrum breadth and resistance management across emerging and endemic zoonotic threats; role of government preparedness programs and strategic stockpiling; clinical deployment in hospital settings during outbreak response; research-driven demand linked to surveillance, pathogen characterization, and therapeutic development; procurement dynamics shaped by public health funding cycles and emergency authorization frameworks. |
Zoonotic Antivirals Market by Segment
Products:
- Broad-spectrum antivirals
- Targeted antivirals
- Combination therapies
Applications:
- Emerging zoonoses
- Endemic zoonotic infections
End User:
- Government stockpiles
- Hospitals
- Research institutes
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- World Health Organization. (2024). WHO research and development blueprint for epidemics: Priority zoonotic viral diseases. World Health Organization.
- World Health Organization. (2024). Clinical management of emerging and re-emerging viral zoonoses. World Health Organization.
- Centers for Disease Control and Prevention. (2024). Zoonotic diseases and emerging viral threats at the human–animal interface. U.S. Department of Health and Human Services.
- Centers for Disease Control and Prevention. (2024). Antiviral drugs: Mechanisms of action and role in outbreak response. U.S. Department of Health and Human Services.
- Food and Agriculture Organization of the United Nations. (2024). Zoonotic viruses and spillover prevention at the animal–human interface. FAO Animal Health Division.
- European Centre for Disease Prevention and Control. (2024). Preparedness planning for emerging zoonotic viral diseases. European Centre for Disease Prevention and Control.
Frequently Asked Questions
How big is the zoonotic antivirals market in 2026?
The global zoonotic antivirals market is estimated to be valued at USD 2.5 billion in 2026.
What will be the size of zoonotic antivirals market in 2036?
The market size for the zoonotic antivirals market is projected to reach USD 6.6 billion by 2036.
How much will be the zoonotic antivirals market growth between 2026 and 2036?
The zoonotic antivirals market is expected to grow at a 10.2% CAGR between 2026 and 2036.
What are the key product types in the zoonotic antivirals market?
The key product types in zoonotic antivirals market are broad-spectrum antivirals, targeted antivirals and combination therapies.
Which applications segment to contribute significant share in the zoonotic antivirals market in 2026?
In terms of applications, emerging zoonoses segment to command 56.0% share in the zoonotic antivirals market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Products
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Products , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Products , 2026 to 2036
- Broad-spectrum antivirals
- Targeted antivirals
- Combination therapies
- Broad-spectrum antivirals
- Y to o to Y Growth Trend Analysis By Products , 2021 to 2025
- Absolute $ Opportunity Analysis By Products , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Applications
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Applications, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Applications, 2026 to 2036
- Emerging zoonoses
- Endemic zoonotic infections
- Emerging zoonoses
- Y to o to Y Growth Trend Analysis By Applications, 2021 to 2025
- Absolute $ Opportunity Analysis By Applications, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Government stockpiles
- Hospitals
- Research institutes
- Government stockpiles
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Products
- By Applications
- By End User
- Competition Analysis
- Competition Deep Dive
- Gilead Sciences
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Roche
- Pfizer
- Merck
- Johnson & Johnson
- Others
- Gilead Sciences
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Products
- Figure 6: Global Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Applications
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Products
- Figure 26: North America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Applications
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Products
- Figure 36: Latin America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Applications
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Products
- Figure 46: Western Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Applications
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Products
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Applications
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Products
- Figure 66: East Asia Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Applications
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Products
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Applications
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Products
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Applications
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

