The global UTI therapeutics landscape is characterized by fragmentation across generic manufacturers and limited differentiation among branded products. Major pharmaceutical companies including GSK, Pfizer, Bayer, Viatris, Cipla, Sun Pharma and Teva maintain UTI therapy portfolios spanning prescription antibiotics and over-the-counter urinary analgesics like phenazopyridine. However, specific revenue segmentation for UTI products remains largely undisclosed in public financial statements, as these agents typically constitute small portions of broader anti-infective or generics divisions.
Treatment reliance on generic formulations stems from the inclusion of nitrofurantoin, trimethoprim-sulfamethoxazole and amoxicillin-clavulanate on the WHO Model List of Essential Medicines, which guides national formularies and procurement policies across over 100 countries. This essential medicine designation creates downward pricing pressure, as public health systems negotiate volume-based tenders with multiple qualified suppliers competing primarily on cost. Generic penetration exceeds 90 percent in most developed economies for first-line agents, with branded products maintaining presence only in specific niches like extended-release formulations or combination products with novel claims.
The competitive landscape for premium innovation remains nascent. Companies developing novel anti-infectives for complicated UTI face lengthy clinical development timelines, regulatory pathways requiring non-inferiority or superiority trials against established agents, and reimbursement challenges in jurisdictions accustomed to low-cost generic alternatives. This dynamic has limited new product launches, with most innovation focused on reformulation, dosing convenience or diagnostic-therapeutic bundles rather than genuinely novel mechanisms.
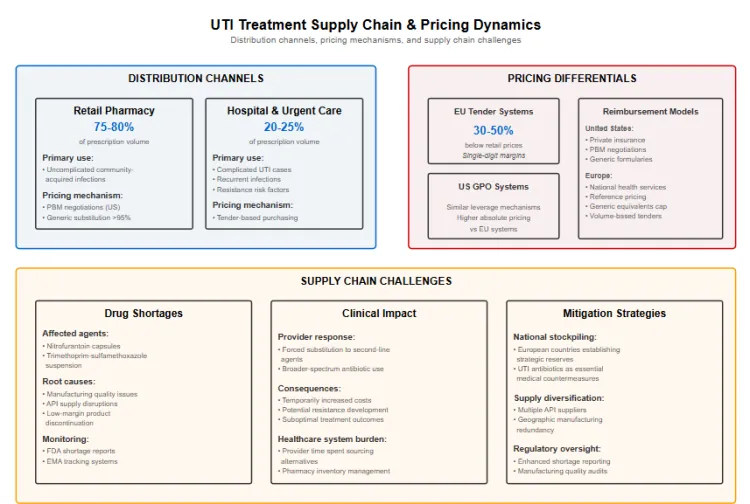
UTI treatment distribution operates through distinct channels with divergent pricing dynamics. Retail pharmacy represents the primary route for uncomplicated community-acquired infections, accounting for approximately 75 to 80 percent of global UTI prescription volume. These prescriptions typically flow through private insurance reimbursement in the United States, where pharmacy benefit managers negotiate generic substitution rates exceeding 95 percent for common agents, or through national health service formularies in Europe where reference pricing systems cap reimbursement at generic equivalents.
Hospital and urgent care channels serve complicated UTI cases, recurrent infections and patients with resistance risk factors. Hospital procurement operates predominantly through tender-based purchasing, where public health systems and hospital groups aggregate demand to negotiate volume discounts. Tender processes in European countries frequently achieve prices 30 to 50 percent below retail for identical formulations, compressing manufacturer margins to single-digit percentages for commodity generics. Private hospital systems in the United States employ group purchasing organizations using similar leverage, though pricing remains higher in absolute terms.
Drug shortages have emerged as a recurring supply chain challenge. The USA Food and Drug Administration and European Medicines Agency periodically report shortages of key UTI antibiotics including nitrofurantoin capsules and trimethoprim-sulfamethoxazole suspension, driven by manufacturing quality issues, API supply disruptions or commercial decisions to discontinue low-margin products. These shortages force providers to substitute second-line agents, temporarily increasing costs and potentially contributing to resistance development through broader-spectrum antibiotic use. National stockpiling strategies in some European countries now include UTI antibiotics as essential medical countermeasures.
Nitrofurantoin, fosfomycin, ciprofloxacin and cephalosporins constitute the core API inputs for UTI therapeutics, with each facing distinct supply dynamics. Nitrofurantoin API production is concentrated among approximately five major suppliers globally, with significant manufacturing capacity in India and China. The relatively simple synthesis and stable demand have maintained API pricing below USD 100 per kilogram for standard grades, though quality variability has contributed to finished-dose product recalls in recent years.
Fosfomycin trometamol, used for single-dose UTI treatment, commands substantially higher API pricing due to more complex synthesis and lower production volumes. While specific pricing data is not disclosed in public documents reviewed, industry sources indicate fosfomycin API costs multiple times higher per treatment course than nitrofurantoin, contributing to retail prices for single-dose sachets reaching USD 20 to 40 in developed economies compared to USD 5 to 10 for seven-day generic alternatives.
Fluoroquinolones including ciprofloxacin face increasing supply constraints unrelated to production capacity. Antimicrobial stewardship policies across Europe, North America and parts of Asia have restricted fluoroquinolone prescribing for uncomplicated UTI due to resistance concerns and adverse event profiles. The European Medicines Agency issued recommendations in 2019 limiting fluoroquinolone use to situations where alternative antibiotics are inappropriate, directly reducing demand volumes and creating excess API capacity. This policy-driven volume contraction has led some manufacturers to exit production, paradoxically creating potential shortage risk for complicated cases where these agents remain appropriate.
Cephalosporin APIs, particularly those used in oral formulations like cefixime and cefpodoxime, originate predominantly from Chinese and Indian manufacturers. Trade data from UN Comtrade indicates these countries supply over 80 percent of global cephalosporin API exports by volume. Cost pressures from environmental compliance requirements, particularly wastewater treatment standards for beta-lactam production facilities, have contributed to API price increases of 15 to 30 percent since 2020 according to industry reporting, though these increases remain modest in absolute terms for mature molecules.
Single-dose fosfomycin trometamol represents the most commercially successful premium UTI innovation, achieving pricing premiums of 200 to 400 percent compared to multi-day generic regimens despite similar or marginally superior efficacy. The convenience value proposition of single-dose therapy resonates with patients and providers, supporting sustained volume share in the 10 to 15 percent range for uncomplicated UTI despite lack of formulary preference in most public health systems. Fosfomycin maintains activity against many extended-spectrum beta-lactamase producing organisms, providing clinical differentiation beyond convenience.
Pipeline anti-infectives targeting resistant uropathogens remain limited despite significant unmet need. Novel beta-lactamase inhibitor combinations and agents with gram-negative activity have entered late-stage development, but most target serious infections requiring intravenous administration rather than oral outpatient UTI treatment. The economics of developing oral agents for a landscape dominated by generic pricing create challenging return-on-investment scenarios, discouraging pharmaceutical investment unless differential reimbursement mechanisms emerge.
Rapid diagnostic innovations offer indirect therapeutic value by enabling targeted antibiotic selection and reducing inappropriate broad-spectrum use. Point-of-care PCR panels identifying common uropathogens and resistance markers within hours rather than days of culture allow earlier appropriate therapy escalation or de-escalation. These diagnostics typically add USD 50 to 150 per test compared to standard culture, creating cost-effectiveness debates in different care settings. Clinical governance frameworks from the WHO, Infectious Diseases Society of America and European Association of Urology increasingly emphasize diagnostic stewardship alongside antimicrobial stewardship, potentially expanding diagnostic adoption at the expense of empiric broad-spectrum therapy volumes.
Global trade in UTI antibiotics reflects the broader pharmaceutical API supply chain, with finished-dose product manufacturing increasingly concentrated in emerging economies and API production dominated by India and China. Import reliance creates vulnerabilities, as demonstrated by periodic shortage notifications affecting multiple jurisdictions simultaneously. The COVID-19 pandemic highlighted these dependencies when API exports from India faced temporary restrictions, contributing to antibiotic supply concerns globally.
Regulatory controls on fluoroquinolones illustrate how policy can override traditional economic dynamics. The EMA's 2019 safety warnings and prescribing restrictions reduced ciprofloxacin and levofloxacin UTI prescriptions by an estimated 30 to 40 percent in affected jurisdictions within two years. This policy-driven demand destruction occurred despite continued generic availability and low pricing, demonstrating that clinical governance can supersede economic factors in essential medicine sectors.
Pricing ceilings and reimbursement constraints vary substantially by jurisdiction. Germany's reference pricing system establishes maximum reimbursement amounts for therapeutically equivalent antibiotics, forcing manufacturers to match the lowest generic price to maintain access. France's healthcare system negotiates prices directly for reimbursed medications, with nitrofurantoin reimbursement rates documented below EUR 5 per treatment course. The United States lacks such centralized pricing controls, allowing wider price dispersion, though pharmacy benefit manager negotiations and generic competition typically compress retail prices to USD 10 to 30 for standard regimens.
Emerging economies face distinct access challenges. While API imports from India and China enable local finished-dose manufacturing in countries like Brazil, South Africa and Indonesia, import tariffs, currency fluctuations and local regulatory requirements create pricing variability. WHO prequalification programs for essential antibiotics attempt to standardize quality while facilitating procurement, but coverage remains incomplete for newer agents like fosfomycin.

Source
Community-acquired infections in women drive the majority of prescriptions, accounting for approximately 80 percent of uncomplicated UTI episodes treated in ambulatory settings.
Retail pharmacy generics hold highest penetration with regulated pricing in most jurisdictions, while hospital formularies emphasize tender-based procurement driving margin compression below 15 percent for standard agents.
Nitrofurantoin, fosfomycin, fluoroquinolones and cephalosporins dominate treatment protocols where antimicrobial resistance policy permits their use, with nitrofurantoin maintaining first-line status in over 60 countries.
New anti-infectives targeting resistant uropathogens and unique mechanism therapies achieve average selling prices two to five times higher than generic comparators, with single-dose regimens commanding the highest premiums.
Reliance on India and China API imports introduces exposure to shortage signals, with over 70 percent of global antibiotic API production concentrated in these regions and periodic supply disruptions documented by regulatory agencies.
Catheter Associated Urinary Tract Infections (UTI) Treatment Market - Demand & Forecast 2025 to 2035
Complicated Urinary Tract Infections Treatment Market is segmented by Drug Class (Cephalosporin, Penicillin and Combinations, Quinolones, Azoles and Amphotericin B, Nitro Furans), Distribution Channel (Hospital Pharmacies, Gynecology and Urology Clinics, Drug Stores, Retail Pharmacies), Mechanism Of Action (Broad-Spectrum Antibiotics, Beta-Lactamase Inhibitor Combinations, Carbapenems, Aminoglycosides), and Region. Forecast for 2026 to 2036.
Uncomplicated Urinary Tract Infection Treatment Market Analysis by Penicillin, Quinolones, Sulfonamide, Nitrofuran, and Others Through 2035
Market Share Breakdown of Uncomplicated Urinary Tract Infection Treatment
Demand for Uncomplicated Urinary Tract Infection Treatment in Japan Size and Share Forecast Outlook 2025 to 2035