Anatomic Pathology Market
The Anatomic Pathology Market Is Segmented By Product (Instruments, Consumables, Services), Application (Disease Diagnosis, Drug Discovery and Development, Others), Distribution Channel (Hospitals, Diagnostic Laboratories, Research Laboratories, Others), Technology (Manual Systems, Semi-Automated Systems, Automated Systems, Digital Platforms), Disease Area (Oncology, Gastrointestinal Disease, Dermatopathology, Others), And Workflow Stage (Sample Preparation, Tissue Processing, Staining and Slide Preparation, Interpretation and Reporting). Forecast For 2026 To 2036.
Anatomic Pathology Market Size, Market Forecast and Outlook By FMI
Summary of the Anatomic Pathology Market
- Demand and Growth Drivers
- Rising biopsy volumes and cancer case load are supporting steady use of pathology products across hospital labs and diagnostic networks.
- Daily reagent and sample handling drive recurring costs throughout staining and tissue review.
- Laboratory upgrades which involves digital pathology and imaging systems are raising demand for faster review and tighter workflow control.
- Product and Segment View
- Consumables are likely to stay the leading product group as each pathology case needs repeat use of stains along with antibodies along with probes, and slides.
- Disease diagnosis is expected to remain the leading application as biopsy confirmation still guides treatment selection across oncology and other disease pathways.
- Tissue processing is projected to lead workflow stage demand as every specimen passes through fixation along with embedding or sectioning and preparation before review.
- Geography and Competitive Outlook
- India is expected to rise quickly as private diagnostic chains and hospital expansion add more tissue testing capacity across major cities.
- China is set for rapid expansion as hospital modernization and cancer screening follow-up influences pathology case volume growth.
- F. Hoffmann-La Roche Ltd., Danaher Corporation, Agilent Technologies Inc. and Labcorp are some of the key players in the industry active across instruments or consumables along with digital workflow tools and service support.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant for Healthcare at FMI, opines, "Laboratories are moving toward pathology systems that keep workflow stable and support levels steady. Suppliers that maintain sample quality and daily consumable supply are likely to strengthen their standing over the forecast period."
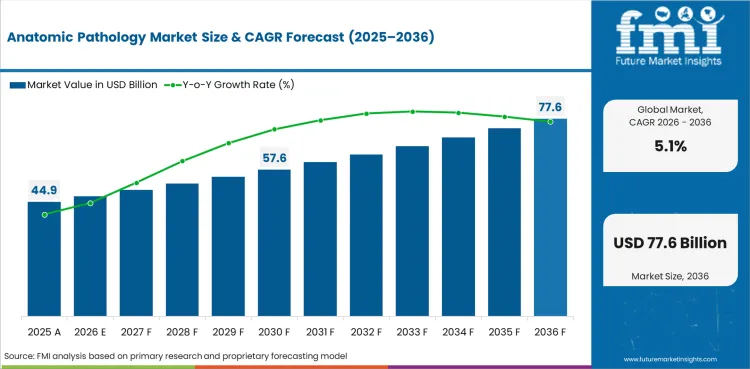
- Anatomic Pathology Market Value Analysis
- The anatomic pathology industry is rising at a steady expansion paceas tissue diagnosis stays a crucial aspect to routine care and oncology work.
- Consumables support a large share of revenue because every specimen needs repeat material use before slide review is complete.
- Instrument upgrades support value creation as laboratories depends on faster processing with stronger image quality and lower manual burden.
- Service demand promotes market expansion ashospital labs and independent diagnostic networks seek maintenance training and workflow support across installed systems.
Anatomic Pathology Market Definition
The anatomic pathology market covers instruments, consumables, and services used for tissue and cell examination in clinical and research settings. It includes products, and services used for histopathology, cytopathology, tissue processing, staining, slide preparation, and related review tasks. The study tracks revenue tied to disease diagnosis, drug development, and other tissue-based assessment activity.
Anatomic Pathology Market Inclusions
Scope for the market includes pathology, instruments, consumables, and services, used across hospital labs, diagnostic laboratories, and research laboratories. The study includes tissue processing systems, staining systems, slide, scanners, reagents, antibodies, probes, kits, slides, and service support. Use linked to disease diagnosis, and drug discovery activity is included.
Anatomic Pathology Market Exclusions
Clinical chemistry systems, hematology analyzers, radiology equipment, and unrelated in vitro diagnostics are outside the market scope. Standalone molecular tests that are sold outside pathology workflow positioning are excluded. General hospital supplies that do not support tissue-based assessment are not counted.
Anatomic Pathology Market Research Methodology
- Primary Research: FMI analysts reviewed input from pathologists along with laboratory directors, and supplier representatives to confirm demand patterns and product use.
- Desk Research: The study used public material from regulatory bodies, pathology publications, company releases, and laboratory operations references to compare product adoption and service activity.
- Market Sizing and Forecasting: Baseline values were aligned to testing intensity along with product mix, and channel use across the supplied study scope before forecast extension.
- Data Validation and Update Cycle: Results were checked for internal consistency and peer reviewed segmentation were built from workflow use patterns inside the supplied study.
Why is the Anatomic Pathology Market Growing?
- Tissue-based diagnosis stays essential in cancer care and surgical pathology.
- Recurring reagent use supports stable revenue flow across daily case work.
- Automation and digital review tools open room for faster turnaround and tighter quality control.
The industry expansion is predicted to witness steady growth rate because anatomic pathology supports diagnosis each day across hospitals and independent laboratories. Interest in cellular pathology and immunohistochemistry is lifting consumable use as biomarker testing becomes more routine.
Market Segmentation Analysis
- Consumables are estimated to account for 37.9% in 2026 as each pathology case needs repeat use of stains reagents and slides.
- Disease diagnosis is expected to account for 48.0% in 2026 as biopsy interpretation is central to treatment planning.
- Hospitals are projected to hold 42.0% share of distribution channelin 2026 because they manage the largest surgical and inpatient tissue testing load.
- Oncology is likely to account for 46.0% in 2026 since cancer biopsy review remains the largest knowledge base for pathology teams.
Insights into the Consumables Product Segment
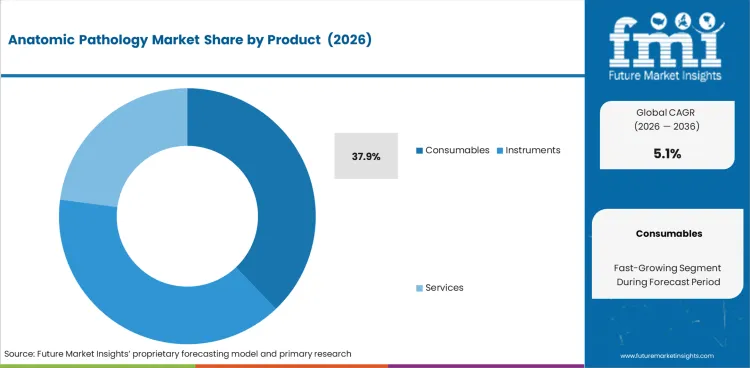
- Consumables are expected to account for 37.9% in 2026 because of regular use in laboratories for staining and specimen preparation work.
- Repeat purchasing stays much higher for consumables than for instruments across most pathology laboratories.
- Each pathology case needs fresh reagents or slides. That requirement keeps consumables in the lead.
Insights into the Services Product Segment
- Services are expected to rise steadily through 2036 because laboratories need maintenance training and workflow help after system installation.
- Scanner deployment and software use are increasing demand for field support across pathology sites.
- Service quality influences renewal decisions in laboratories that use complex pathology systems.
Insights into the Disease Diagnosis Application Segment
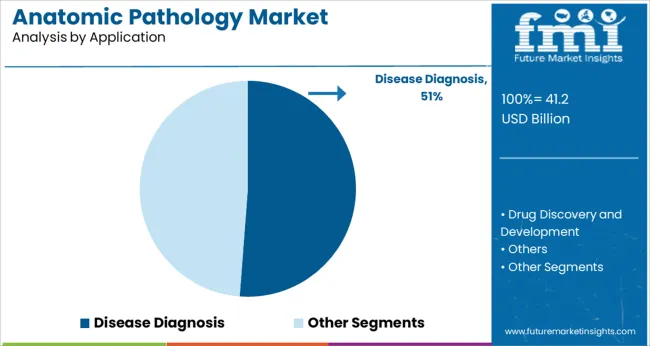
- Disease diagnosis is expected to account for 48.0% of the application segment in 2026 as pathology review supports disease confirmation grading and treatment planning across routine clinical work.
- Biopsy interpretation stays central to daily pathology demand across hospitals and diagnostic laboratories.
Insights into the Drug Discovery and Development Application Segment
- Drug discovery and development is expected to rise steadily through 2036 as tissue review and biomarker interpretation support translational research and therapy assessment.
- Research teams use pathology tools to study tissue response during preclinical and clinical work.
- This application segment benefits from the growing use of tissue-based evidence in development programs.
Insights into the Hospitals Distribution Channel Segment
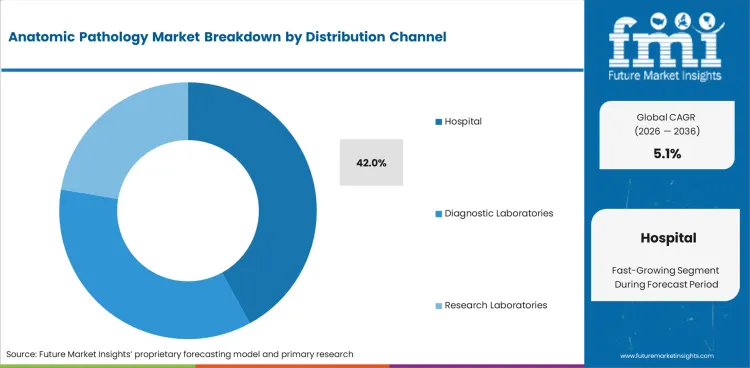
- Hospitals are expected to account for 42.0% of the distribution channel segment in 2026 because inpatient testing and surgical biopsy flow keep hospital pathology units under steady volume pressure.
- Large case-loads support regular use of instruments with consumables and service contracts inside hospital laboratories.
- Approved supplier lists can help hospitals keep long vendor relationships once systems are in place.
Insights into the Diagnostic Laboratories Distribution Channel Segment
- Diagnostic laboratories are expected to advance at a healthy pace through 2036 due to outsourced tissue testing volume rising across multi-site laboratory networks.
- Private laboratory groups are adding capacity in urban centers. That trend supports new system demand.
- Standardized workflow remains important in diagnostic laboratories operating at scale.
Insights into the Automated Systems Technology Segment
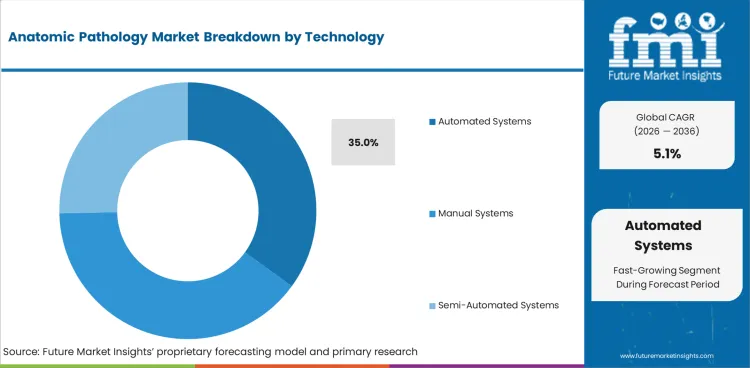
- Automated systems are expected to account for 35.0% of the technology segment in 2026 as laboratories want steadier processing quality with less hands-on work.
- Automation supports faster slide handling and more consistent staining across routine laboratory workflows. Higher volume sites are leading this move as they need stable output every day.
Insights into the Digital Platforms Technology Segment
- Digital platforms are expected to rise at a significant rate through 2036 because image sharing remote review and software-led quality checks are entering more pathology workflows.
- New scanner launches and software clearances are supporting digital adoption across pathology laboratories. Remote review tools are helping pathologists manage complex cases with stronger access to second opinions.
Insights into the Oncology Disease Area Segment
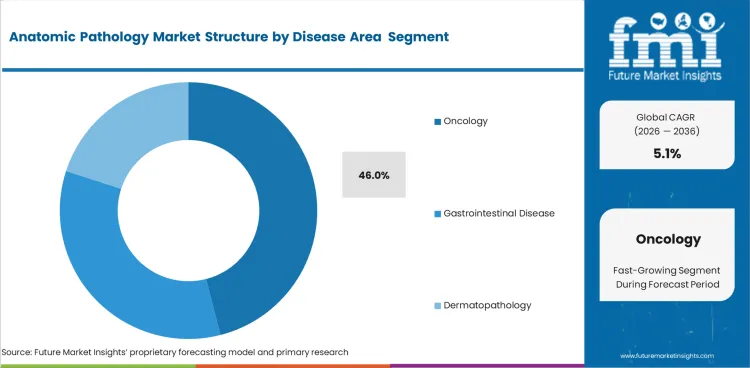
- Oncology is expected to account for 46.0% of the disease area segment in 2026 as cancer diagnosis drives a large share of biopsy review immunostaining and tissue interpretation.
- Rising cancer case load keeps oncology tissue analysis ahead of other disease-focused pathology work.
- Biomarker-linked treatment plans are adding to pathology workload in oncology settings.
Insights into the Dermatopathology Disease Area Segment
- Dermatopathology is expected to record steady growth through 2036 because skin lesion assessment and melanoma review support stable investigation volume in specialty practice.
- Specialist referrals help sustain testing activity in this disease area.
- Hospital and specialty laboratories both support demand for dermatopathology review.
Insights into the Tissue Processing Workflow Stage Segment
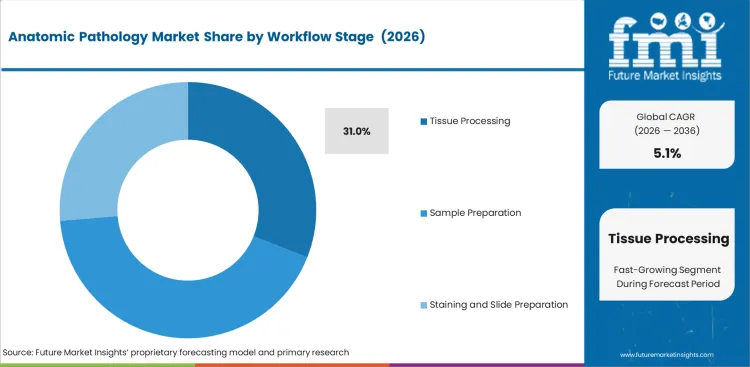
- Tissue processing is expected to account for 31.0% of the workflow stage segment in 2026 because specimens must move through fixation, embedding, and section preparation before interpretation can begin.
- This stage influences stable product use because it is linked directly to each case entering the laboratory.
- Daily sample flow keeps processing demand ahead of later workflow stages on a revenue basis.
Insights into the Interpretation and Reporting Workflow Stage Segment
- Interpretation and reporting are expected to rise steadily through 2036 because scanner use, image management tools, and specialist review networks help pathologists handle higher case complexity.
- Workflow software is improving access to second opinions across pathology teams.
- Remote collaboration is supporting stronger use of digital review in interpretation and reporting.
Anatomic Pathology Market Drivers and Restraints along with Opportunities
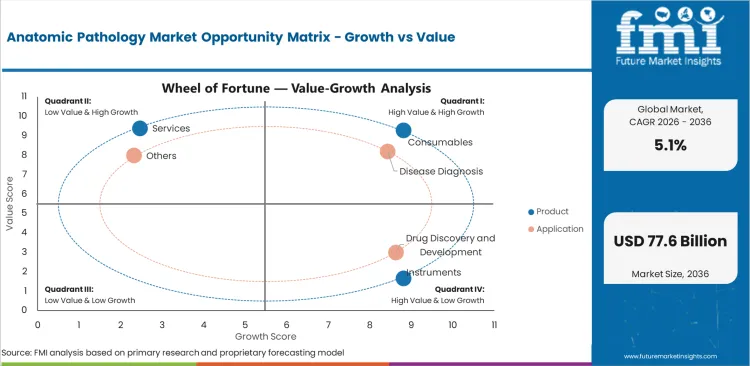
- Rising cancer testing and biopsy review support steady demand across pathology products and services.
- Budget pressure and staffing gaps can slow new instrument buying in some laboratories.
- Automation, digital review, and laboratory standardization create room for product upgrades and service expansion.
Demand remains steady because pathology stays central to tissue-based diagnosis across cancer care and routine hospital practice. Daily case work keeps consumables moving and supports service demand around installed systems.
Growth stays measured in mature systems because reimbursement pressure, labor shortages, and capital approval cycles slow large equipment replacement. Modernization projects still expand in sites that need stronger quality control and faster case handling.
Regional Market Analysis
.webp)
| Country | CAGR |
|---|---|
| India | 6.5% |
| China | 6.1% |
| Japan | 5.9% |
| France | 5.9% |
| South Africa | 5.9% |
| The United Kingdom | 5.3% |
| The United States | 4.1% |
Source: Future Market Insights analysis based on the supplied study inputs and published pathology references.
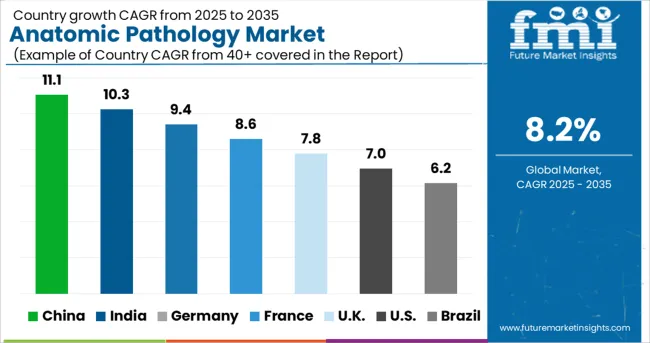
Anatomic Pathology Market CAGR Analysis by Country
- India is expected to lead the country growth ranking with a 6.5% CAGR through 2036. This growth is anticipated because the country is expanding from a lower installed base, while private diagnostic chains and hospital networks continue to add histopathology capacity. Wider access to biopsy‑based diagnosis and increased laboratory buildout across major cities are strengthening daily pathology workloads.
- China is anticipated to follow closely with a 6.1% CAGR. Market expansion is expected as hospital modernization programs and rising cancer screening follow‑up increase tissue testing volumes. The adoption of structured pathology workflows is supporting higher demand for instruments, consumables, and processing systems.
- Japan and France are each expected to grow at a 5.9% CAGR. In both markets, steady disease diagnosis demand is anticipated to sustain routine tissue examination activity. Growth remains balanced as mature laboratory infrastructures focus on consistent workflow performance rather than rapid capacity expansion.
The global anatomic pathology market is expected to expand at a CAGR of 5.1% from 2026 to 2036. The study covers multiple developed and emerging markets, with growth differences primarily influenced by laboratory maturity and the pace of pathology workflow modernization.
Demand Outlook for Anatomic Pathology Market in the United States
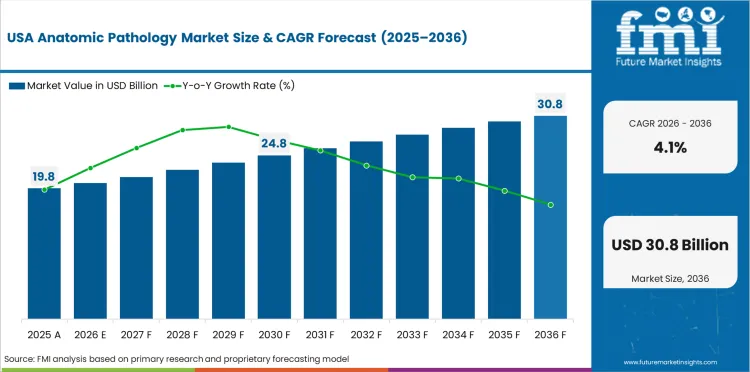
The United States demand for the sector is expected to grow at a 4.1% CAGR through 2036 as routine biopsy-based diagnosis keeps pathology laboratories productive across hospital systems and diagnostic networks. Tissue review is aligned to regular patient management and this keeps demand steady for instruments consumables and service support. Replacement demand is also set to propel industry outlook as mature laboratories refine processing quality and reporting speed.
- Biopsy-led diagnosis keeps daily pathology use steady.
- Hospital laboratory activity sustains recurring product demand.
- Replacement demand supports measured system upgrades.
Sales Analysis of Anatomic Pathology Market in China
Industry in China are projected to record a 6.1% CAGR through 2036 as pathology laboratory expansion and rising biopsy use lift demand across hospital-based tissue testing. Hospital modernization is improving access to structured pathology workflows and that is helping slide processing and interpretation volumes move upward.
- Pathology laboratory expansion lifts national demand.
- Hospital modernization improves workflow adoption.
- Biopsy use keeps tissue testing volumes rising.
Opportunity Analysis of Anatomic Pathology Market in India
Adoption in India is expected to expand at a 6.5% CAGR through 2036 because pathology laboratory infrastructure is improving and biopsy-linked diagnostic demand is rising across hospital and diagnostic networks. Tissue-based workflows are entering more care settings and this is raising demand for core pathology products.
- Expanding laboratory infrastructure improves service reach.
- Biopsy-linked demand lifts daily pathology activity.
- Growing access helps more specimens enter formal testing.
In-Depth Analysis of Anatomic Pathology Market in Germany
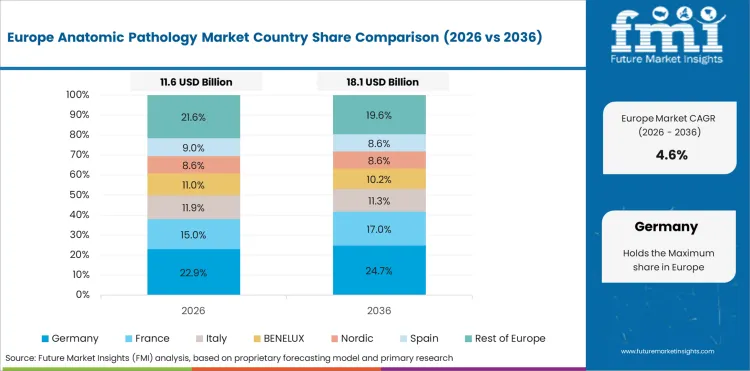
Germany’s market is likely to post a 4.5% CAGR through 2036 as stable pathology laboratory demand keeps tissue-based workflows active across structured clinical settings. Disease diagnosis is consistent leading laboratories to align with routine specimen handling and interpretation work. Mature operations keep growth steady, as pathology demand remains strong. Germany therefore reflects steady replacement and workflow optimization demand.
- Stable laboratory demand keeps testing activity firm.
- Routine tissue workflows support repeat product use.
- Moderate growth reflects a mature installed base.
In-Depth Analysis of Anatomic Pathology Market in Japan
I Japan is forecast to grow at a 5.9% CAGR through 2036 as steady pathology test demand supports tissue examination across established diagnostic practice. Disease diagnosis keeps specimen review active and this helps maintain recurring use of stains reagents and related workflow products.
- Steady test demand supports recurring pathology use.
- Tissue examination stays active in routine diagnosis.
- Balanced laboratory demand keeps growth healthy.
Anatomic Pathology Market Analysis in South Korea
Demand in South Korea is expected to register a 5.6% CAGR through 2036 as laboratory work is intensifying and biopsy use in diagnosis continues to rise. Hospitals and diagnostic laboratories are processing more tissue-based cases and this keeps demand active for pathology workflow products. Growth for the sector is healthy because the market is already more developed than several rising Asian peers.
- Intensifying laboratory work raises daily testing need.
- Biopsy use keeps tissue workflows active.
- Healthy growth reflects a developed diagnostic base.
Anatomic Pathology Market Analysis in France
The industry in France is projected to record a 5.9% CAGR through 2036 as steady disease diagnosis demand keeps pathology workflow intensity firm across tissue-based testing. Laboratories continue to process specimens at a stable pace and this supports regular use of pathology instruments consumables and related services. Widespread tissue testing keeps the market on a dependable path with less volatility than faster moving countries.
- Steady disease diagnosis keeps workflow demand firm.
- Widespread tissue testing supports regular product use.
- Stable laboratory activity keeps the country near the upper growth band.
Competitive Landscape and Strategic Positioning
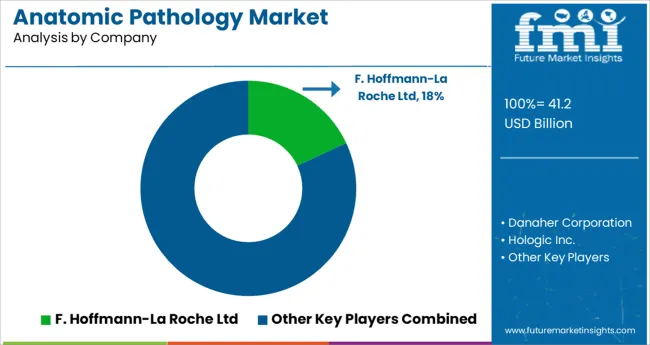
- Competition is moderately concentrated across integrated instrument groups, laboratory service firms, and digital workflow specialists.
- Large suppliers compete through installed base strength and field service reach.
- Newer firms compete through scanners and workflow tools tied to digital pathology.
Competition in anatomic pathology is shaped by installed instruments and service performance after placement. Suppliers with strong stain systems and workflow support can hold accounts for longer periods because pathology teams are cautious about changing approved systems.
National laboratory service firms and digital pathology specialists create extra pressure in segments tied to outsourced testing and remote review. Laboratories also look for uptime, training and software support that can keep case flow steady.
Interest in digital pathology and imaging systems is increasing the role of image management and workflow software in new deals. That trend can help digital-based firms expand alongside larger instrument suppliers.
Key Companies in the Anatomic Pathology Market
Key companies in the anatomic pathology industry include integrated instrument suppliers, laboratory service firms, and digital workflow specialists.
- Global Leaders: F. Hoffmann-La Roche Ltd, Danaher Corporation, Hologic Inc., Agilent Technologies Inc., Cardinal Health Inc.
- Service and Network Operators: NeoGenomics, Laboratory Corporation of America Holdings.
- Digital and Workflow Specialists: Gestalt Diagnostics, Inspirata Inc., Nichirei Bioscience, BioGenex.
Key Developments for the Anatomic Pathology Market
- In July 2025, PathAI launched the Precision Pathology Network to expand access to AI-powered pathology tools and collaborative research.
- In February 2026, Labcorp expanded its collaboration with PathAI to deploy the AISight Dx1 digital pathology platform across its network.
- In March 2026, Clarapath launched the latest SectionStar platform for clinical histology workflows.
Competitive Benchmarking: Anatomic Pathology Market
| Company | Product Breadth | Workflow Coverage | Service Support | Footprint |
|---|---|---|---|---|
| F. Hoffmann-La Roche Ltd | High | Strong | Strong | Global |
| Danaher Corporation | High | Strong | Strong | Global |
| Hologic Inc. | Moderate | Moderate | Moderate | North America and Europe |
| Agilent Technologies Inc. | High | Strong | Moderate | Global |
| NeoGenomics | Moderate | Moderate | Strong | North America |
| Laboratory Corporation of America Holdings | Moderate | Strong | Strong | North America |
| Gestalt Diagnostics | Low | Moderate | Moderate | North America |
Source: Future Market Insights analysis based on the supplied study inputs and company positioning references.
Key Players in the Anatomic Pathology Market
Major Companies
- F. Hoffmann-La Roche Ltd
- Danaher Corporation
- Hologic Inc.
- Agilent Technologies, Inc.
- NeoGenomics
- Cardinal Health, Inc.
Regional Participants
- PHC Holdings Corporation
- Gestalt Diagnostics
- Inspirata Inc
- Laboratory Corporation of America Holdings
- Nichirei Bioscience (Nichirei Corporation)
Report Scope and Coverage for Anatomic Pathology Market
| Item | Value |
|---|---|
| Market | Anatomic Pathology Market |
| Estimated Value in 2025 | USD 42.7 billion |
| Expected Value in 2026 | USD 44.9 billion |
| Forecast Value in 2036 | USD 73.8 billion |
| CAGR 2026 to 2036 | 5.1% |
| Quantitative Units | Revenue in USD million |
| Parent Segments Covered | Product, Application, Distribution Channel, Technology, Disease Area, Workflow Stage |
| Regions Covered | North America, Latin America, East Asia, South Asia and Pacific, Western Europe, Eastern Europe, Middle East and Africa |
| Countries Covered | United States, Canada, ,Brazil, China, Japan, South Korea, India, South Africa, Germany, United Kingdom, France |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd, Danaher Corporation, Hologic Inc., Agilent Technologies Inc., NeoGenomics, Labcorp, Cardinal Health Inc. |
| Forecast Period | 2026 to 2036 |
| Historical Reference | 2025 |
Source: Future Market Insights analysis based on the supplied study inputs and 2026 framing.
Anatomic Pathology Market Analysis by Segments
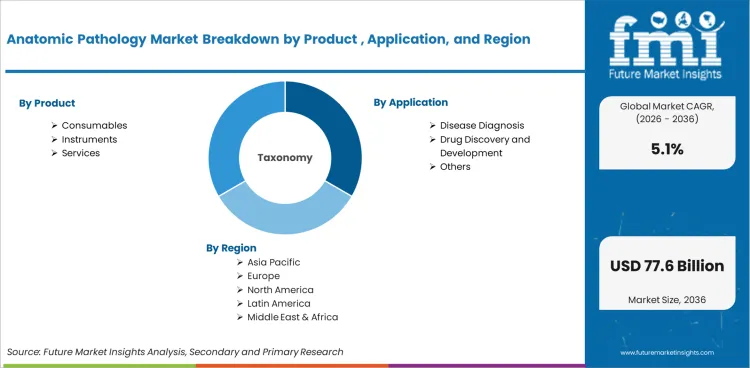
Product
- Instruments
- Consumables
- Services
Application
- Disease Diagnosis
- Drug Discovery and Development
- Others
Distribution Channel
- Hospitals
- Diagnostic Laboratories
- Research Laboratories
- Others
Technology
- Manual Systems
- Semi-Automated Systems
- Automated Systems
- Digital Platforms
Disease Area
- Oncology
- Gastrointestinal Disease
- Dermatopathology
- Others
Workflow Stage
- Sample Preparation
- Tissue Processing
- Staining and Slide Preparation
- Interpretation and Reporting
Research Sources and Bibliography
- Leica Biosystems. (2026, March 23). Leica Biosystems solidifies leadership in digital pathology with launch of next-generation Aperio GT Elite scanner and Aperio iQC software.
- College of American Pathologists. (2024, February 23). Principles of analytic validation of immunohistochemical assays: Guideline update.
- World Health Organization. (2024, February 1). Global cancer burden growing, amidst mounting need for services.
- Puckett, Y., Wilson, A. M., Farci, F., et al. (2024, July 2). Melanoma pathology. StatPearls.
- College of American Pathologists. (2026). Cancer protocol templates.
- Clarapath. (2026, March 10). Clarapath launches its latest SectionStar platform for clinical histology workflows.
- Labcorp. (2026, February 23). Labcorp expands collaboration with PathAI to deploy FDA-cleared digital pathology platform nationwide.
- PathAI. (2025, July 8). PathAI launches Precision Pathology Network to advance AI-powered pathology.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary research documentation.
This Report Answers
- What size is the anatomic pathology market in 2026 and how large can it reach by 2036?
- How fast is anatomic pathology demand expected to expand between 2026 and 2036?
- Which product application distribution channel technology disease area and workflow stage segments lead 2026 demand?
- Why are consumables and disease diagnosis holding the largest share of 2026 demand?
- How are automation and digital slide review changing laboratory upgrade plans?
- Which countries are expected to post the fastest growth through 2036?
- Which companies stay most active across instruments consumables services and digital pathology tools?
Frequently Asked Questions
How large is the anatomic pathology market in 2026?
The anatomic pathology market is expected to reach USD 44.9 billion in 2026 as hospital labs and diagnostic networks keep tissue testing volumes steady.
How large can the anatomic pathology market reach by 2036?
The market is projected to reach USD 73.8 billion by 2036 as cancer testing and laboratory modernization support steady revenue expansion.
What growth rate is expected for the anatomic pathology market through 2036?
Industry is expected to expand at a 5.1% CAGR from 2026 to 2036 with growth led by routine tissue diagnostics.
Which product leads the anatomic pathology market by 2026?
Consumables lead the product mix with an expected 37.9% share in 2026 as every case needs repeat reagent use.
Which application leads the anatomic pathology market by 2026?
Disease diagnosis leads the anatomic pathology market with a projected 48.0% share in 2026 because biopsy review stays central to treatment choice.
Which country grows fastest in the anatomic pathology market study?
India records the fastest growth for the sector at 6.5% CAGR through 2036 as private labs and hospital chains keep adding histology capacity.
Why do hospitals stay central in the anatomic pathology market?
Hospitals are witnessing stable demandin the anatomic pathology market because surgical case flow and inpatient biopsy review keep daily pathology workloads higher than other settings.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Consumables
- Instruments
- Services
- Consumables
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Disease Diagnosis
- Drug Discovery and Development
- Others
- Disease Diagnosis
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Hospital
- Diagnostic Laboratories
- Research Laboratories
- Hospital
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Automated Systems
- Manual Systems
- Semi-Automated Systems
- Automated Systems
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Disease Area
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Disease Area, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Disease Area, 2026 to 2036
- Oncology
- Gastrointestinal Disease
- Dermatopathology
- Oncology
- Y to o to Y Growth Trend Analysis By Disease Area, 2021 to 2025
- Absolute $ Opportunity Analysis By Disease Area, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Workflow Stage
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Workflow Stage, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Workflow Stage, 2026 to 2036
- Tissue Processing
- Sample Preparation
- Staining and Slide Preparation
- Tissue Processing
- Y to o to Y Growth Trend Analysis By Workflow Stage, 2021 to 2025
- Absolute $ Opportunity Analysis By Workflow Stage, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Application
- By Distribution Channel
- By Technology
- By Disease Area
- By Workflow Stage
- Competition Analysis
- Competition Deep Dive
- F. Hoffmann-La Roche Ltd
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Danaher Corporation
- Hologic Inc.
- Agilent Technologies, Inc.
- NeoGenomics
- Cardinal Health, Inc.
- F. Hoffmann-La Roche Ltd
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Disease Area, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Disease Area, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Disease Area, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Disease Area, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 31: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Disease Area, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 38: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 39: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 40: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Disease Area, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 43: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 45: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 46: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 47: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 48: South Asia and Pacific Market Value (USD Million) Forecast by Disease Area, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 50: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 51: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 52: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 53: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 54: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 55: Middle East & Africa Market Value (USD Million) Forecast by Disease Area, 2021 to 2036
- Table 56: Middle East & Africa Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Distribution Channel
- Figure 12: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Technology
- Figure 15: Global Market Value Share and BPS Analysis by Disease Area, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Disease Area, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Disease Area
- Figure 18: Global Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Workflow Stage
- Figure 21: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 23: Global Market Attractiveness Analysis by Region
- Figure 24: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 29: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 30: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 31: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 32: North America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Product
- Figure 35: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Application
- Figure 38: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Distribution Channel
- Figure 41: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Technology
- Figure 44: North America Market Value Share and BPS Analysis by Disease Area, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Disease Area, 2026-2036
- Figure 46: North America Market Attractiveness Analysis by Disease Area
- Figure 47: North America Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 49: North America Market Attractiveness Analysis by Workflow Stage
- Figure 50: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 51: Latin America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Product
- Figure 54: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Application
- Figure 57: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 60: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 62: Latin America Market Attractiveness Analysis by Technology
- Figure 63: Latin America Market Value Share and BPS Analysis by Disease Area, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Disease Area, 2026-2036
- Figure 65: Latin America Market Attractiveness Analysis by Disease Area
- Figure 66: Latin America Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 68: Latin America Market Attractiveness Analysis by Workflow Stage
- Figure 69: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 70: Western Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Product
- Figure 73: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Application
- Figure 76: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 77: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 78: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 79: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 81: Western Europe Market Attractiveness Analysis by Technology
- Figure 82: Western Europe Market Value Share and BPS Analysis by Disease Area, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Disease Area, 2026-2036
- Figure 84: Western Europe Market Attractiveness Analysis by Disease Area
- Figure 85: Western Europe Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 87: Western Europe Market Attractiveness Analysis by Workflow Stage
- Figure 88: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Product
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 94: Eastern Europe Market Attractiveness Analysis by Application
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 97: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 100: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Disease Area, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Disease Area, 2026-2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Disease Area
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Workflow Stage
- Figure 107: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 108: East Asia Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 109: East Asia Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 110: East Asia Market Attractiveness Analysis by Product
- Figure 111: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 112: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 113: East Asia Market Attractiveness Analysis by Application
- Figure 114: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 115: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 116: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 117: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 118: East Asia Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 119: East Asia Market Attractiveness Analysis by Technology
- Figure 120: East Asia Market Value Share and BPS Analysis by Disease Area, 2026 and 2036
- Figure 121: East Asia Market Y-o-Y Growth Comparison by Disease Area, 2026-2036
- Figure 122: East Asia Market Attractiveness Analysis by Disease Area
- Figure 123: East Asia Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 125: East Asia Market Attractiveness Analysis by Workflow Stage
- Figure 126: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 128: South Asia and Pacific Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 129: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 130: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 131: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 132: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 133: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 134: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 135: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 136: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 137: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 138: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 139: South Asia and Pacific Market Value Share and BPS Analysis by Disease Area, 2026 and 2036
- Figure 140: South Asia and Pacific Market Y-o-Y Growth Comparison by Disease Area, 2026-2036
- Figure 141: South Asia and Pacific Market Attractiveness Analysis by Disease Area
- Figure 142: South Asia and Pacific Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 143: South Asia and Pacific Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 144: South Asia and Pacific Market Attractiveness Analysis by Workflow Stage
- Figure 145: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 147: Middle East & Africa Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 148: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 149: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 150: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 151: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 152: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 153: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 154: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 155: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 156: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 157: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 158: Middle East & Africa Market Value Share and BPS Analysis by Disease Area, 2026 and 2036
- Figure 159: Middle East & Africa Market Y-o-Y Growth Comparison by Disease Area, 2026-2036
- Figure 160: Middle East & Africa Market Attractiveness Analysis by Disease Area
- Figure 161: Middle East & Africa Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 162: Middle East & Africa Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 163: Middle East & Africa Market Attractiveness Analysis by Workflow Stage
- Figure 164: Global Market - Tier Structure Analysis
- Figure 165: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

