Cancer Diagnostics Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
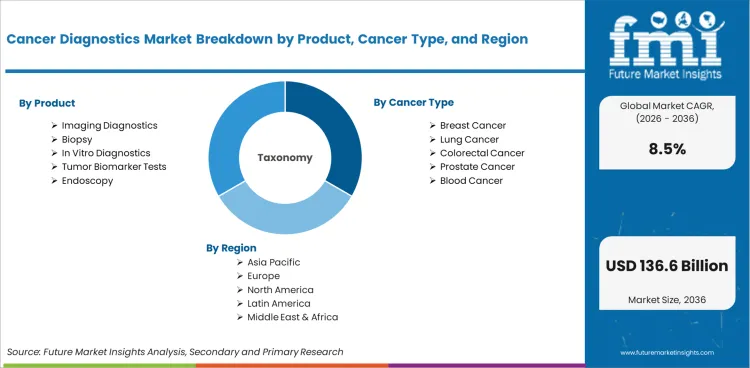
The Cancer Diagnostics Market is segmented by Product (Imaging Diagnostics, Biopsy, In Vitro Diagnostics, Tumor Biomarker Tests, Endoscopy), Cancer Type (Breast Cancer, Lung Cancer, Colorectal Cancer, Prostate Cancer, Blood Cancer), End User, Technology, Sample, and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Cancer Diagnostics Market Size, Market Forecast and Outlook By FMI
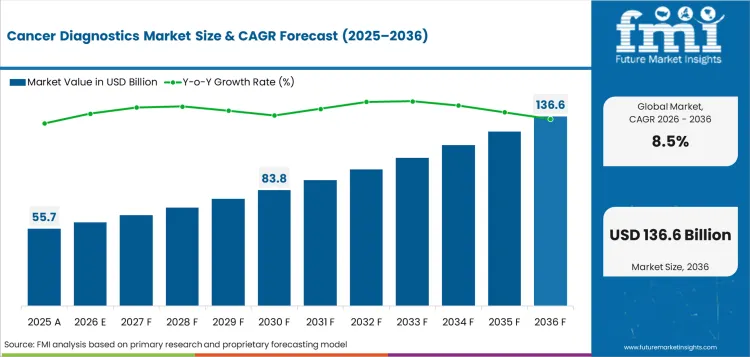
Cancer diagnostics market was valued at USD 55.7 billion in 2025 and is expected to reach USD 60.4 billion in 2026. Market value is forecast to reach USD 136.6 billion by 2036 at 8.5% CAGR during 2026 to 2036. Hospitals are anticipated to account for 48.0% of end user demand in 2026. Blood samples are expected to represent 41.0% of sample demand in 2026.
Summary of the Cancer Diagnostics Market
- Demand and Growth Drivers
- Screening programs bring more people into cancer testing before symptoms become serious. Early testing gives doctors more time to confirm disease and choose the right care route.
- Precision oncology is changing how doctors select treatment. Tumor marker tests help match patients with therapies suited to their cancer type.
- Blood-based testing is gaining use in follow-up care. Patients can give samples more easily, so doctors can track disease response with less procedure burden.
- Product and Segment View
- Imaging diagnostics helps doctors find suspicious areas before a biopsy is done. MRI and CT scan use stays important because treatment planning starts with clear disease location.
- Molecular diagnostics supports treatment decisions after cancer is confirmed. Doctors use mutation results to understand which therapy may work better for a patient.
- Tissue testing keeps clinical value in most cancer pathways. Pathologists need tissue evidence to confirm tumor type and guide the next step in care.
- Geography and Competitive Outlook
- India and South Korea have better readiness as hospitals and screening access expand. More patients are entering formal diagnosis, so labs need faster and reliable testing workflows.
- China supports high testing volume due to its large cancer care system. Hospitals need tools that can handle many samples without slowing treatment decisions.
- Established suppliers stand out through approved assays and hospital service reach. Companies with easy lab workflows are better placed because hospitals want fewer delays in daily testing.
- Analyst Opinion
- FMI Principal Consultant Sabyasachi Ghosh, says, “Cancer diagnostics is becoming a clinical choice. Oncology centers need faster confirmation and tests tied to treatment labels. Hospitals are likely to favor suppliers with approved assays and platforms able to support tissue and blood workflows. Cost pressure is likely to stay visible in public programs, so suppliers need simple workflows and reliable reagent access.”
- Cancer Diagnostics Market Value Analysis
- Cancer diagnostics market is moving from basic disease confirmation toward regular support for treatment planning.
- Adoption is being shaped by wider screening access and hospital-based oncology testing.
- Market value is supported by molecular diagnostics because doctors use test results to match patients with suitable therapies.
- Blood-based testing is creating value in follow-up care because repeat sample collection is easier for patients.
- Imaging and tissue testing keep the diagnosis pathway clear by helping doctors locate tumors and confirm cancer type.
Cancer diagnostics are witnessing spike in adoption for screening programs and oncology labs as test developers move from single-site procedures toward accessible blood and genomic testing routes. In July 2024, FDA approved Guardant Health’s Shield blood test for colorectal cancer screening in average-risk adults aged 45 years or older. The test gives providers another route to reach adults who avoid colonoscopy or stool-based screening. In October 2024, Exact Sciences said FDA approved Cologuard Plus with 95% colorectal cancer sensitivity and 94% specificity across nearly 19,000 average-risk participants. The result gives health systems a clearer benchmark for non-invasive colorectal screening performance. In August 2024, Illumina received FDA approval for TruSight Oncology Comprehensive as a distributable cancer biomarker test across 517 genes. The kit gives qualified labs a stronger route to run solid tumor profiling closer to oncology care. These approvals show that cancer diagnostics suppliers are building value around access and faster treatment routing.
Cancer Diagnostics Market Definition
Cancer diagnostics refers to products, tests, instruments, imaging tools, and sample-based workflows used to detect cancer, confirm tumor type, support staging, guide therapy selection, and monitor treatment response across clinical oncology care.
Cancer Diagnostics Market Inclusions
Scope includes oncology imaging, tissue biopsy, liquid biopsy, in vitro diagnostics, tumor biomarker tests, diagnostic endoscopy, screening endoscopy, pathology testing, histopathology, cytopathology, PCR-based testing, next generation sequencing, and sample handling for blood specimens.
Scope includes imaging diagnostics, biopsy, in vitro diagnostics, tumor biomarker tests, endoscopy, molecular diagnostics, pathology testing, blood samples, tissue samples, urine samples, and cancer testing workflows used in hospitals, diagnostic laboratories, oncology clinics, and research centers. Key stakeholders include hospitals, diagnostic labs, oncology clinics, cancer research institutes, diagnostic product manufacturers, imaging equipment suppliers, assay developers, laboratory service providers, healthcare regulators, oncologists, pathologists, radiologists, and healthcare payers.
Cancer Diagnostics Market Exclusions
Scope excludes cancer treatment drugs, chemotherapy products, radiotherapy equipment sold only for treatment, surgical oncology devices without diagnostic use, general wellness tests without validated cancer use, research-only tools without clinical diagnostic use, basic laboratory consumables sold outside cancer workflows, hospital IT systems without diagnostic interpretation use, general imaging equipment without oncology positioning, and non-cancer screening products.
Cancer Diagnostics Market Research Methodology
- Primary Research: FMI analysts reviewed oncology diagnostic workflows and hospital testing needs. Supplier portfolios and clinical lab buying patterns were checked.
- Desk Research: Public data from cancer agencies and regulators shaped market checks. Official company releases were used only for company proof.
- Market Sizing and Forecasting: Sizing used supplied market anchors and cancer test intensity. Screening activity and assay mix shaped the forecast model.
- Data Validation: Forecast checks compared cancer burden data and companion diagnostic approvals. Screening participation and supplier portfolio activity were reviewed.
Why is the Cancer Diagnostics Market Expanding?
- Cancer testing is becoming part of care at an earlier stage. Doctors need test results before they decide the right treatment path.
- Screening programs are bringing more people into follow-up testing. A positive screening result often needs imaging or lab confirmation.
- Precision oncology is raising the value of tumor marker tests. Doctors use these tests to match treatment with cancer type.
- Blood-based testing is making follow-up easier for selected patients. It can reduce the need for repeated tissue procedures.
Cancer diagnostics are expanding because doctors need clearer answers before treatment starts. In the United States, 2,041,910 new cancer cases are expected in 2025. Large patient volume puts pressure on hospitals to confirm disease faster. Imaging helps doctors locate suspicious areas. Lab tests help confirm cancer type. These tools help hospitals move patients from suspicion to care planning with fewer delays.
Treatment planning is making diagnosis more valuable. Many cancer drugs now need a test result before use. For example, FDA companion diagnostic listings include in vitro and imaging tools tied to specific therapies. Wider use of Molecular Diagnostics supports this shift because doctors need tumor marker evidence before selecting therapy. Liquid Biopsy adds value as blood samples can support repeat monitoring.
Market Segmentation Analysis
- In 2026, Imaging diagnostics is expected to account for 32.0% product demand.
- Breast cancer demand is forecast at 24.0% share in 2026.
- Hospitals are anticipated to represent 48.0% end user share in 2026.
- In 2026, molecular diagnostics is expected to hold 36.0% technology share.
- Blood samples are forecast to represent 41.0% sample demand in 2026.
Cancer diagnostics market is segmented by product, cancer type, end user, technology, sample, and region. Product includes imaging diagnostics, biopsy, in vitro diagnostics, tumor biomarker tests, and endoscopy. Cancer type includes breast cancer and lung cancer. Colorectal cancer, prostate cancer, and blood cancer complete the cancer type scope.
Insights into Cancer Diagnostics Market by Product
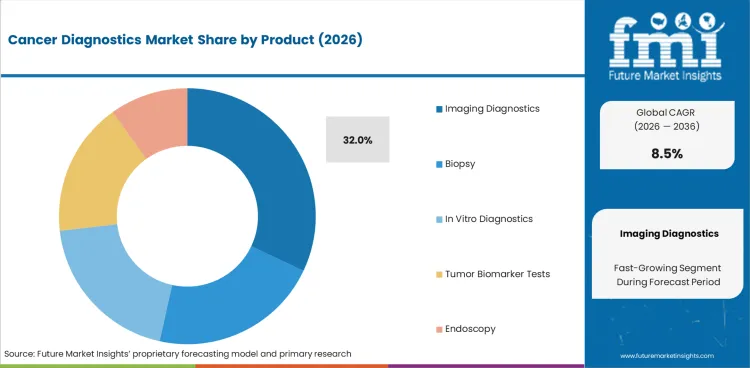
- Imaging diagnostics is expected to hold 32.0% share in 2026. Hospitals use MRI and CT scan to find the tumor location before biopsy or treatment planning begins. Demand for precision cancer imaging is expected to rise as doctors need clearer images to plan the next step in care.
- Biopsy continues to support cancer confirmation after imaging finds a suspicious area. In vitro diagnostics and tumor biomarker tests add value because many treatment decisions now need lab-based proof before therapy starts.
Insights into Cancer Diagnostics Market by Cancer Type
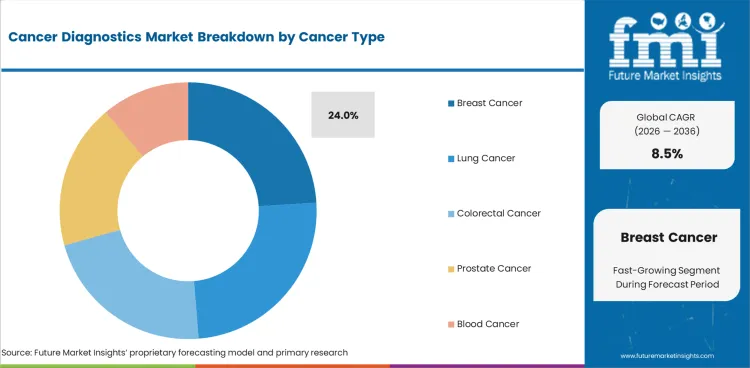
- Breast cancer is forecast to account for 24.0% share in 2026. Screening programs create steady demand for imaging and pathology confirmation after mammography. Breast cancer diagnostics use is expected to grow as doctors need faster follow-up after abnormal screening results.
- Lung cancer testing depends on imaging first and tissue confirmation next. Mutation testing is also important because treatment choice often depends on the cancer profile. Colorectal cancer testing supports regular lab demand through screening follow-up and treatment planning.
Insights into Cancer Diagnostics Market by End User

- Hospitals are anticipated to represent 48.0% share in 2026. Cancer diagnosis often needs imaging rooms and pathology access in one care route. Hospital networks prefer diagnostic platforms that help doctors move patients from testing to treatment planning faster.
- Diagnostic laboratories handle higher test volumes for molecular panels and routine pathology. Cancer research institutes use specialized assays to study cancer behavior and support early clinical validation.
Insights into Cancer Diagnostics Market by Technology
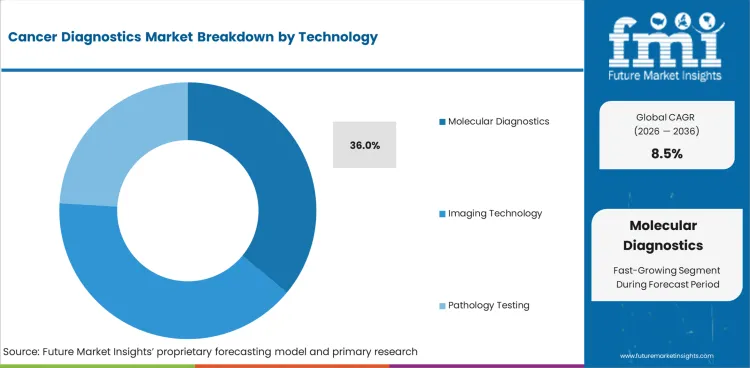
- Molecular diagnostics is expected to hold 36.0% share in 2026. Doctors use mutation and biomarker results to choose targeted therapy or immunotherapy. Molecular diagnostic platforms are becoming more important as treatment labels depend more on test results.
- Imaging technology supports staging before treatment starts. Pathology testing keeps clinical value because tissue review helps confirm tumor type and disease pattern.
Insights into Cancer Diagnostics Market by Sample
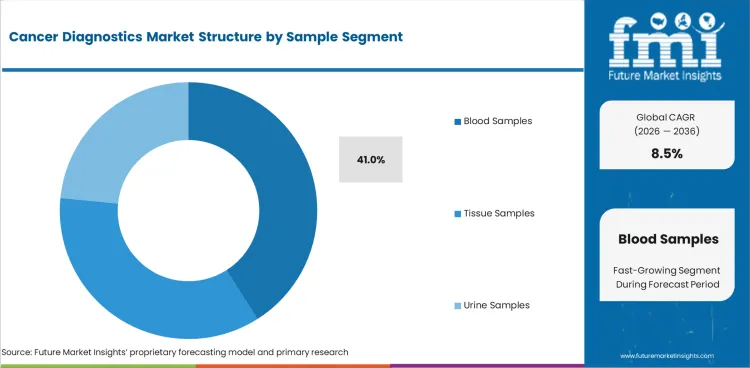
- Blood samples are forecast to represent 41.0% share in 2026. Blood collection is easier for patients and can support repeat testing during follow-up care. Liquid biopsy use is expected to expand in selected cancer pathways where doctors need monitoring without repeated tissue procedures.
- Tissue samples keep high clinical value because pathologists need tissue evidence to confirm cancer type. Urine samples support selected testing needs and can make routine follow-up easier for some patients.
Cancer Diagnostics Market Drivers, Restraints, and Opportunities
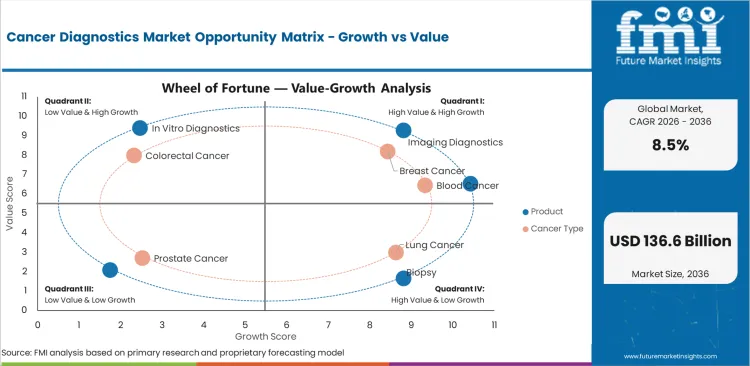
- Earlier screening is a main driver for cancer diagnostics. More screening follow-up means more need for imaging and pathology support.
- Precision oncology is raising demand for tests tied to treatment choice. These tests help doctors decide which therapy may work better.
- High test cost is a restraint for smaller hospitals. Wider use needs clearer payment support and simpler lab workflows.
- Blood-based testing creates an opportunity in follow-up care. Easier sample collection can make repeat testing less difficult for patients.
Screening Access Supports Diagnostic Flow
Screening programs help doctors find possible cancer cases before symptoms become severe. A patient with an abnormal result still needs confirmation before treatment starts. This is where imaging and pathology become important. The market is expected to witness expansion as hospitals can connect screening with follow-up testing. A faster route from screening to diagnosis helps doctors begin care planning earlier.
Precision Oncology Raises Test Value
Cancer care is becoming more specific to each patient. Doctors often need biomarker results before choosing targeted therapy. Approved assays carry more value when test results help define the right patient group. Hospitals value tests that guide therapy choice and fit regular lab work.
Analysis of Cancer Diagnostics Market by Key Countries
.webp)
| Country | CAGR |
|---|---|
| India | 6.3% |
| South Korea | 5.4% |
| China | 5.2% |
| Australia | 4.2% |
| Spain | 4.0% |
| Germany | 3.9% |
| United States | 3.6% |
Source: Future Market Insights, 2026.
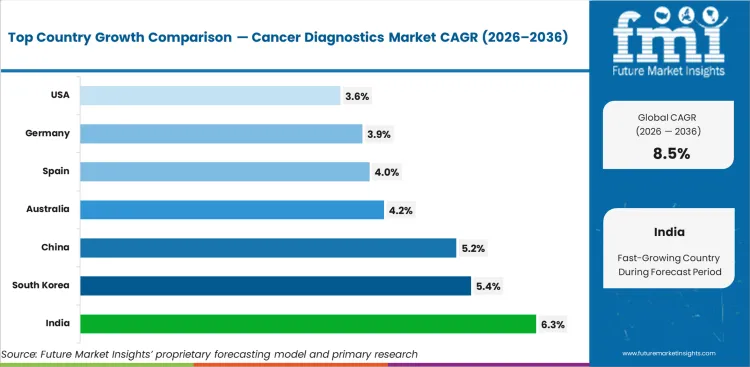
Cancer Diagnostics Market CAGR Analysis by Country
- India is expected to record 6.3% CAGR through 2036.
- South Korea is forecast at 5.4% CAGR in the market between 2026 and 2036.
- China’s cancer diagnostics market is projected to expand at 5.2% CAGR through 2036.
- During the forecast period, Australia is anticipated to expand at 4.2% CAGR.
- Spain is expected to advance at 4.0% CAGR from 2026 to 2036.
- Over the same period, Germany is forecast to register 3.9% CAGR in the market.
- Industry outlook in the United States points to 3.6% CAGR through 2036.
Demand Outlook for Cancer Diagnostics Market in India
India is projected to observe steady cancer diagnostics outlook due to rise in patients opting screening and follow-up care. India is expected to record 6.3% CAGR by 2036 as testing reaches more patients beyond major cities. India’s health ministry cited Indian Council of Medical Research estimates of over 14 lakh cancer cases in 2023. Public hospitals and private oncology centers are expanding access to imaging and pathology services. Affordable workflows are likely to be important because high patient volume can place pressure on lab capacity.
- Large hospital networks are adding imaging and biopsy services so doctors can confirm cancer earlier.
- Hospitals in Delhi and Mumbai are using more molecular tests to guide treatment choices.
- Regional labs need simple sample handling so patients outside metro cities can access testing at lower cost.
Sales Analysis of Cancer Diagnostics Market in South Korea
South Korea has an established cancer care system supported by screening programs and specialist hospitals. Public cancer registry research expected 304,754 new cancer cases and 84,019 cancer deaths in 2025. Hospitals are using more imaging and molecular review before treatment decisions. South Korea is forecast at 5.4% CAGR in the sector by 2036. Faster movement from screening to diagnosis supports demand for reliable hospital-based testing.
- Seoul-based hospitals need multi-cancer tests because they handle complex oncology cases.
- National screening access keeps follow-up testing active for breast cancer and stomach cancer.
- Local diagnostic firms benefit from hospital systems that can use test results quickly in care planning.
Demand Outlook for Cancer Diagnostics Market in China
The market in China is expected to expand at 5.2% CAGR between 2026 and 2036. China has a large cancer testing base because patient volume is high across major hospital systems. International Agency for Research on Cancer GLOBOCAN 2022 China fact sheet reported 4,824,703 new cancer cases and 2,574,176 cancer deaths in China in 2022. Hospitals are adding imaging and molecular testing capacity to manage this caseload. Local suppliers need tests that can handle large sample volumes without delaying results.
- Large hospitals in Beijing and Shanghai support wider use of genetic testing panels.
- Provincial screening programs raise follow-up demand for lung cancer and colorectal cancer tests.
- Domestic reagent makers support laboratories by reducing waiting time for routine test results.
Future Outlook for Cancer Diagnostics Market in Australia
Australia has steady cancer diagnostics demand because screening programs are well organized. By 2036, the industry in Australia is forecast to expand at 4.2% CAGR. Around 170,000 cancer cases are expected to be diagnosed in the country by 2025. Screening follow-up keeps imaging and pathology services active across public health systems. Regional access gaps make sample transport and remote review more important for patients outside major cities.
- Breast screening programs keep imaging demand active across public health services.
- Bowel screening creates regular need for colonoscopy follow-up and pathology review.
- Remote care networks need digital pathology support so patients can get diagnosis closer to home.
Opportunity Analysis of Cancer Diagnostics Market in Spain
Spain’s cancer diagnostics demand is shaped by public oncology services and regional screening programs. OECD cancer profile data identifies breast cancer and colorectal cancer as major incidence areas in Spain. Spain is expected to advance at 4.0% CAGR through 2036. Follow-up testing is needed after abnormal screening results. Public hospitals will need steady pathology and endoscopy capacity as screening access improves.
- Public hospitals need pathology services to confirm cancer after abnormal screening results.
- Regional screening programs move more patients into follow-up imaging and lab testing.
- Madrid and Barcelona hospital groups support molecular testing because treatment planning often needs biomarker results.
Sales Analysis of Cancer Diagnostics Market in Germany
Germany is forecast to register 3.9% CAGR in the market by 2036. Germany has a mature cancer care system with high standards for diagnosis. OECD profile data expected 529,955 new cancer cases in Germany in 2022. University hospitals and certified cancer centers use molecular panels and digital pathology tools in treatment planning. Stable test volume and strict quality needs keep supplier qualification important.
- University hospitals support sequencing and pathology use across major cancer pathways.
- Screening systems create regular imaging and endoscopy referrals for follow-up diagnosis.
- German laboratories prefer validated assays because cancer test results guide treatment decisions.
Demand Outlook for Cancer Diagnostics Market in the United States
United States demand reflects mature test access and wider use of companion diagnostics. Federal cancer statistics reported that 613,349 people died of cancer in the United States in 2023. The figure keeps payment and test-menu decisions tied to real disease burden in a mature diagnostics market. Hospital networks are adding tests with clear treatment value. Industry in the United States is expected to rise at 3.6% CAGR during the forecast period. Payment review is likely to keep test menus focused on clinical proof.
- Cancer centers use genomic profiling because many therapies need biomarker evidence before use.
- FDA companion diagnostic review gives hospitals more confidence in approved oncology tests.
- Private payers favor tests that clearly help doctors select the right patient group.
Competitive Landscape and Strategic Positioning
- Competition in cancer diagnostics centers on assay menu breadth and regulatory clearance.
- Suppliers stand out by combining tissue testing with blood testing. Imaging support adds another route into oncology departments.
- Global diagnostic firms hold advantage through service networks and companion diagnostic partnerships.
- Entry barriers include clinical evidence and reimbursement access. Sample quality control adds another qualification hurdle.
Key players such as Roche and Thermo Fisher Scientific have broad assay portfolios and established laboratory platforms. Siemens Healthineers and GE HealthCare support imaging needs before biopsy and treatment planning. Illumina and QIAGEN add value in genomic testing, especially for cancers that need mutation results before treatment.
Digital pathology and companion diagnostics are becoming stronger points of separation. Suppliers compete through assay clearance and pathology workflow fit. Hospitals prefer platforms that help move confirmed results into oncology review with fewer workflow delays.
Payment access can shape how fast labs add new oncology tests. Clinical laboratories need clear coding and coverage routes before adding complex genomic profiling to routine menus. Tests with easier payment administration are better positioned for hospital adoption.
Key Companies in the Cancer Diagnostics Market
- Global In Vitro Diagnostic Leaders: F. Hoffmann-La Roche Ltd and Abbott Laboratories hold broad assay and instrument portfolios. Thermo Fisher Scientific Inc. and bioMérieux SA support molecular and laboratory workflows.
- Genomics and Biomarker Specialists: Illumina Inc. and Myriad Genetics Inc. support sequencing and hereditary cancer test demand. QIAGEN and Agilent Technologies Inc. add biomarker and pathology depth.
- Imaging and Screening Firms: Siemens Healthineers and GE HealthCare support oncology imaging workflows. Hologic Inc. and Becton, Dickinson and Company support screening and laboratory workflows.
Competitive Benchmarking: Cancer Diagnostics Market
| Company | Assay Menu Depth | Oncology Workflow Fit | Sample Coverage | Geographic Footprint |
|---|---|---|---|---|
| F. Hoffmann-La Roche Ltd | High | High | Tissue and blood | Global |
| Thermo Fisher Scientific Inc. | High | High | Tissue and blood | Global |
| Abbott Laboratories | Medium | Medium | Blood and tissue | Global |
| Illumina Inc. | High | Medium | Tissue and blood | Global |
| bioMérieux SA | Medium | Medium | Blood and tissue | Global |
| Siemens Healthineers | Medium | High | Imaging and radiotracer | Global |
| GE HealthCare | Medium | High | Imaging | Global |
| Hologic Inc. | Medium | High | Breast screening | North America and Europe |
| Agilent Technologies Inc. | High | High | Tissue pathology | Global |
| Becton, Dickinson and Company | Medium | Medium | Cell and lab samples | Global |
| Myriad Genetics Inc. | Medium | Medium | Blood and tissue | United States centered |
| QIAGEN | High | Medium | Tissue and blood | Global |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Cancer Diagnostics Market
- In January 2025, Roche received FDA approval for the PATHWAY HER2 test to identify HER2-ultralow metastatic breast cancer patients eligible for ENHERTU.
- In January 2026, Illumina secured Centers for Medicare and Medicaid Services reimbursement for TruSight Oncology Comprehensive at USD 2,989.55 per test.
Key Players in the Cancer Diagnostics Market
Major Global Players
- F. Hoffmann-La Roche Ltd
- Thermo Fisher Scientific Inc.
- Abbott Laboratories
- Siemens Healthineers
- GE HealthCare
Genomics and Pathology Specialists
- Illumina Inc.
- Agilent Technologies Inc.
- QIAGEN
- Myriad Genetics Inc.
- bioMérieux SA
Screening and Laboratory Workflow Firms
- Hologic Inc.
- Becton, Dickinson and Company
- Exact Sciences Corporation
- Danaher Corporation
Report Scope and Coverage
| Parameter | Details |
|---|---|
| Quantitative Units | USD 60.4 billion to USD 136.6 billion at 8.5% CAGR |
| Market Definition | Diagnostic products and workflows used for cancer detection, confirmation, staging, therapy selection, and monitoring |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | India, South Korea, China, Australia, Spain, Germany, United States, and other countries |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd, Thermo Fisher Scientific Inc., Abbott Laboratories, Illumina Inc., bioMérieux SA, Siemens Healthineers, GE HealthCare, Hologic Inc., Agilent Technologies Inc., BD, Myriad Genetics Inc., and QIAGEN |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up method using supplied market anchors, cancer burden, screening activity, assay mix, and supplier portfolio checks |
Cancer Diagnostics Market Breakdown by Product, Cancer Type, End User, Technology, Sample, and Region
Cancer Diagnostics Market Segmented by Product
- Imaging Diagnostics
- Biopsy
- In Vitro Diagnostics
- Tumor Biomarker Tests
- Endoscopy
Cancer Diagnostics Market Segmented by Cancer Type
- Breast Cancer
- Lung Cancer
- Colorectal Cancer
- Prostate Cancer
- Blood Cancer
Cancer Diagnostics Market Segmented by End User
- Hospitals
- Diagnostic Laboratories
- Cancer Research Institutes
- Specialty Clinics
Cancer Diagnostics Market Segmented by Technology
- Molecular Diagnostics
- Imaging Technology
- Pathology Testing
Cancer Diagnostics Market Segmented by Sample
- Blood Samples
- Tissue Samples
- Urine Samples
Cancer Diagnostics Market by Region
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East and Africa
Research Sources and Bibliography
- Australian Institute of Health and Welfare. (2025, October 8). Cancer data in Australia. Australian Institute of Health and Welfare.
- Exact Sciences. (2024, October 4). FDA approves Exact Sciences’ Cologuard Plus test, setting a new benchmark in non-invasive colorectal cancer screening. Exact Sciences.
- USA Food and Drug Administration. (2024, August 21). Summary of Safety and Effectiveness Data: TruSight Oncology Comprehensive, P230011. USA Food and Drug Administration.
- Illumina, Inc. (2024, August 27). FDA approves Illumina cancer biomarker test with two companion diagnostics to rapidly match patients to targeted therapies. Illumina.
- Illumina, Inc. (2026, January 20). Illumina secures CMS reimbursement for TruSight Oncology Comprehensive, expanding access to precision oncology. Illumina.
- International Agency for Research on Cancer. (2024, February 8). China: Globocan 2022. Global Cancer Observatory.
- Jung, K.-W., Kang, M. J., Park, E.-H., Hwa, Y. E., Kim, H.-J., Kim, J.-E., Kong, H.-J., Choi, K. S., & Yang, H.-K. (2025, March 11). Prediction of cancer incidence and mortality in Korea, 2025. Cancer Research and Treatment, 57(2), 331-338.
- National Cancer Institute. (2025, May 7). Cancer statistics. National Cancer Institute.
- Centers for Disease Control and Prevention. (2026, January 13). Cancer data and statistics. Centers for Disease Control and Prevention.
- OECD & European Commission. (2025, February). EU country cancer profile: Germany 2025. OECD Publishing.
- OECD & European Commission. (2025, February). EU country cancer profile: Spain 2025. OECD Publishing.
- Press Information Bureau. (2025, February 13). Towards a cancer-free India. Government of India, Ministry of Health and Family Welfare.
- Roche. (2025, January 31). Roche receives FDA approval for the first companion diagnostic to identify patients with HER2-ultralow metastatic breast cancer eligible for ENHERTU. Roche.
- USA Food and Drug Administration. (2024, July 26). Premarket approval: Shield, P230009. USA Food and Drug Administration.
- USA Food and Drug Administration. (2026, February 5). List of cleared or approved companion diagnostic devices. USA Food and Drug Administration.
This Report Answers
- What is the current and future value outlook for Cancer Diagnostics Market from 2026 to 2036?
- What CAGR is expected for Cancer Diagnostics Market during the 2026 to 2036 forecast period?
- Which product segment is expected to lead Cancer Diagnostics Market demand in 2026?
- Which end user segment is expected to create the most demand for cancer diagnostic products?
- How do screening programs and companion diagnostics influence Cancer Diagnostics Market expansion?
- Which countries are expected to record faster Cancer Diagnostics Market CAGR values through 2036?
- Which companies shape competitive structure across assays and imaging?
- What products and workflows are included within Cancer Diagnostics Market scope?
- How does FMI build the Cancer Diagnostics Market forecast using supplied values and external checks?
Frequently Asked Questions
What is the global market demand for Cancer Diagnostics Market in 2026?
In 2026, Cancer Diagnostics Market is expected to be worth USD 60.4 billion as hospitals expand imaging and molecular testing workflows.
What is the expected value of Cancer Diagnostics Market by 2036?
Cancer Diagnostics Market is forecast to reach USD 136.6 billion by 2036 as precision oncology and screening follow-up raise test volume.
What CAGR is expected for Cancer Diagnostics Market from 2026 to 2036?
Cancer Diagnostics Market is expected to rise at 8.5% CAGR during 2026 to 2036 with wider molecular and imaging use.
Which product segment leads Cancer Diagnostics Market?
Imaging diagnostics is expected to lead product demand with 32.0% share in 2026 as MRI and CT scans support staging decisions.
Which end user segment leads Cancer Diagnostics Market?
Hospitals are anticipated to represent 48.0% end user share in 2026 as diagnosis and treatment planning meet in oncology centers.
Why does India record faster Cancer Diagnostics Market expansion?
India is expected to record 6.3% CAGR through 2036 as cancer caseload and diagnostic network expansion raise access.
What is included in Cancer Diagnostics Market?
Scope includes imaging diagnostics, biopsy, in vitro diagnostics, tumor biomarker tests, endoscopy, molecular technology, pathology testing, and sample workflows.
How is Cancer Diagnostics Market forecast built?
Forecasting uses supplied market values, cancer burden checks, screening activity, assay mix, country outlook, and company portfolio review.
How do hospitals choose cancer diagnostic suppliers?
Hospitals usually compare clinical accuracy and assay clearance before adding a diagnostic platform to oncology workflows.
Why are blood samples important in cancer diagnostics?
Blood samples support repeat monitoring and liquid biopsy testing, which can reduce reliance on invasive tissue collection for selected oncology workflows.
How do companion diagnostics affect cancer treatment?
Companion diagnostics identify patients suited for targeted therapies, so oncologists can match a drug to a tumor biomarker before treatment starts.
Which companies are important in Cancer Diagnostics Market?
Roche, Thermo Fisher Scientific, Illumina, Abbott, Siemens Healthineers, GE HealthCare, Agilent, Hologic, Myriad Genetics, QIAGEN, and BD are major suppliers.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Hardware
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Hardware , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Hardware , 2026 to 2036
- Quarter-turn
- Captive screw
- Tool-less latch
- Hybrid latch
- Quarter-turn
- Y to o to Y Growth Trend Analysis By Hardware , 2021 to 2025
- Absolute $ Opportunity Analysis By Hardware , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Panels
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Panels, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Panels, 2026 to 2036
- Sidewall panels
- Ceiling panels
- PSU panels
- Cargo panels
- Sidewall panels
- Y to o to Y Growth Trend Analysis By Panels, 2021 to 2025
- Absolute $ Opportunity Analysis By Panels, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Aircraft Class
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Aircraft Class, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Aircraft Class, 2026 to 2036
- Narrowbody
- Widebody
- Regional jets
- Business jets
- Narrowbody
- Y to o to Y Growth Trend Analysis By Aircraft Class, 2021 to 2025
- Absolute $ Opportunity Analysis By Aircraft Class, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sales Route
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sales Route, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sales Route, 2026 to 2036
- Retrofit kits
- Line-fit modules
- Spare panels
- Retrofit kits
- Y to o to Y Growth Trend Analysis By Sales Route, 2021 to 2025
- Absolute $ Opportunity Analysis By Sales Route, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material System
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Material System, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Material System, 2026 to 2036
- Honeycomb composite
- Thermoplastic panel
- Aluminum panel
- Hybrid laminate
- Honeycomb composite
- Y to o to Y Growth Trend Analysis By Material System, 2021 to 2025
- Absolute $ Opportunity Analysis By Material System, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- By Country
- Market Attractiveness Analysis
- By Country
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- By Country
- Market Attractiveness Analysis
- By Country
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- By Country
- Market Attractiveness Analysis
- By Country
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- By Country
- Market Attractiveness Analysis
- By Country
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- By Country
- Market Attractiveness Analysis
- By Country
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- By Country
- Market Attractiveness Analysis
- By Country
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- By Country
- Market Attractiveness Analysis
- By Country
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Hardware
- By Panels
- By Aircraft Class
- By Sales Route
- By Material System
- Competition Analysis
- Competition Deep Dive
- Collins Aerospace
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Safran Cabin
- Diehl Aviation
- FACC
- Jamco Corporation
- AVIC Cabin Systems
- The Gill Corporation
- SEKISUI KYDEX
- Collins Aerospace
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Hardware , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Panels, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Aircraft Class, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Sales Route, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Material System, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Hardware , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Panels, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Aircraft Class, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Sales Route, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Material System, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Hardware , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Panels, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Aircraft Class, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Sales Route, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Material System, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Hardware , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Panels, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Aircraft Class, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Sales Route, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Material System, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Hardware , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Panels, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Aircraft Class, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Sales Route, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Material System, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Hardware , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Panels, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Aircraft Class, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Sales Route, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Material System, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Hardware , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Panels, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Aircraft Class, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Sales Route, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Material System, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Hardware , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Panels, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Aircraft Class, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Sales Route, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Material System, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Hardware , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Hardware , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Hardware
- Figure 6: Global Market Value Share and BPS Analysis by Panels, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Panels, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Panels
- Figure 9: Global Market Value Share and BPS Analysis by Aircraft Class, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Aircraft Class, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Aircraft Class
- Figure 12: Global Market Value Share and BPS Analysis by Sales Route, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Sales Route, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Sales Route
- Figure 15: Global Market Value Share and BPS Analysis by Material System, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Material System, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Material System
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Hardware , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Hardware , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Hardware
- Figure 32: North America Market Value Share and BPS Analysis by Panels, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Panels, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Panels
- Figure 35: North America Market Value Share and BPS Analysis by Aircraft Class, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Aircraft Class, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Aircraft Class
- Figure 38: North America Market Value Share and BPS Analysis by Sales Route, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Sales Route, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Sales Route
- Figure 41: North America Market Value Share and BPS Analysis by Material System, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Material System, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Material System
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Hardware , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Hardware , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Hardware
- Figure 48: Latin America Market Value Share and BPS Analysis by Panels, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Panels, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Panels
- Figure 51: Latin America Market Value Share and BPS Analysis by Aircraft Class, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Aircraft Class, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Aircraft Class
- Figure 54: Latin America Market Value Share and BPS Analysis by Sales Route, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Sales Route, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Sales Route
- Figure 57: Latin America Market Value Share and BPS Analysis by Material System, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Material System, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Material System
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Hardware , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Hardware , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Hardware
- Figure 64: Western Europe Market Value Share and BPS Analysis by Panels, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Panels, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Panels
- Figure 67: Western Europe Market Value Share and BPS Analysis by Aircraft Class, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Aircraft Class, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Aircraft Class
- Figure 70: Western Europe Market Value Share and BPS Analysis by Sales Route, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Sales Route, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Sales Route
- Figure 73: Western Europe Market Value Share and BPS Analysis by Material System, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Material System, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Material System
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Hardware , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Hardware , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Hardware
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Panels, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Panels, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Panels
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Aircraft Class, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Aircraft Class, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Aircraft Class
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Sales Route, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Sales Route, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Sales Route
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Material System, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Material System, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Material System
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Hardware , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Hardware , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Hardware
- Figure 96: East Asia Market Value Share and BPS Analysis by Panels, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Panels, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Panels
- Figure 99: East Asia Market Value Share and BPS Analysis by Aircraft Class, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Aircraft Class, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Aircraft Class
- Figure 102: East Asia Market Value Share and BPS Analysis by Sales Route, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Sales Route, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Sales Route
- Figure 105: East Asia Market Value Share and BPS Analysis by Material System, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Material System, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Material System
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Hardware , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Hardware , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Hardware
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Panels, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Panels, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Panels
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Aircraft Class, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Aircraft Class, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Aircraft Class
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Sales Route, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Sales Route, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Sales Route
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Material System, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Material System, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Material System
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Hardware , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Hardware , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Hardware
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Panels, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Panels, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Panels
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Aircraft Class, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Aircraft Class, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Aircraft Class
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Sales Route, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Sales Route, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Sales Route
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Material System, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Material System, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Material System
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

