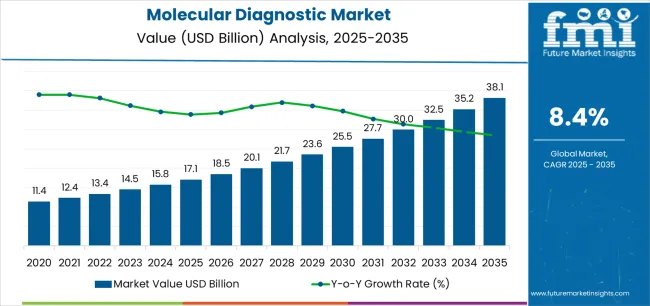
The molecular diagnostics market demonstrated a valuation of USD 17.1 billion in 2025, supported primarily by sustained demand in oncology and essential infectious disease testing. Market expansion is projected to continue, rising to USD 18.5 billion in 2026 and reaching USD 41.5 billion by 2036, reflecting a robust CAGR of 8.4%. As per FMI's projection, growth is no longer solely a function of installed base expansion but is increasingly driven by "compliance gates" and retrofit cycles.
Laboratories are retiring legacy closed systems in favor of high-throughput platforms capable of meeting new regulatory standards, such as the FDA's finalized rule on Laboratory Developed Tests (LDTs) [10]. This regulatory reset forces capital expenditure toward approved, automated platforms to mitigate legal risk, effectively stripping share from manual, in-house assay protocols.
President and CEO of Roche Diagnostics North America, contextualized this technological velocity during a system update: Brad Moore, "This update is significant because it not only allows for new innovation in PCR-testing technology and more choices in omni-channel testing, but it also addresses some key improvements that customers have told us are high priorities for them. All of these updates and improvements can be made to existing 6800/8800 systems through the software upgrade, providing higher throughput to laboratories with the same footprint and delivering more flexibility to physicians and patients.[2]" Rapid sequencing capabilities are compressing the time-to-diagnosis for complex conditions, fundamentally altering how procurement teams evaluate total cost of ownership for genomic hardware.
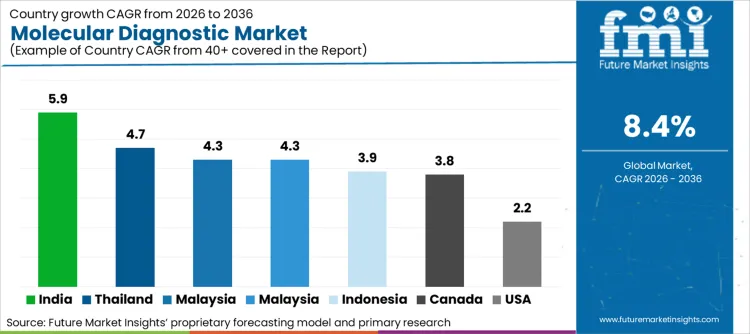
Geographic dispersion of growth highlights a clear divide between mature replacement markets and emerging adopters. High-growth territories include India (5.9% CAGR), Thailand (4.7% CAGR), Malaysia (4.3% CAGR), France (4.3% CAGR), and Indonesia (3.9% CAGR), while established markets like Canada (3.8% CAGR) and the United States (2.2% CAGR) focus on high-value oncology integration.
The molecular diagnostic industry encompasses the development and commercialization of technologies that analyze biological markers in the genome and proteome to detect disease. It primarily involves the detection of specific sequences in DNA or RNA that may be associated with disease, including single nucleotide polymorphisms (SNPs), deletions, rearrangements, insertions, and others.
This market includes all instruments, reagents, kits, and software used for nucleic acid testing (NAT). It covers major technologies such as Polymerase Chain Reaction (PCR), Next-Generation Sequencing (NGS), In Situ Hybridization (ISH), and microarrays used in clinical settings. Applications range from infectious disease screening (COVID-19, HIV, TB) to oncology (liquid biopsy, solid tumor profiling) and genetic testing.
Excluded from this report are traditional immunoassay-based diagnostics, clinical chemistry analyzers, and microbiological culture methods that do not utilize nucleic acid amplification or sequencing. Research-only (RUO) products not intended for clinical diagnostic use, unless they are operating under specific LDT exemptions, are also outside the primary market sizing scope.
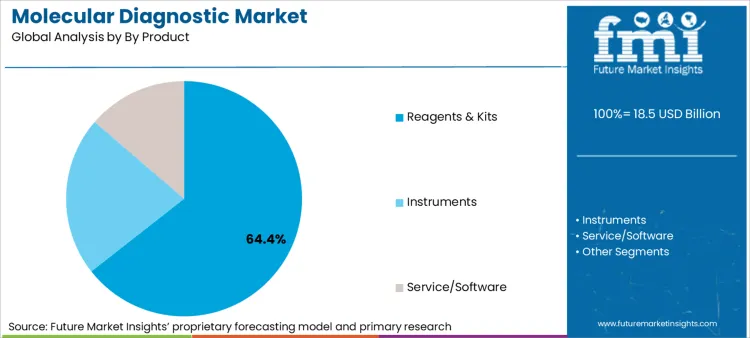
Reagents and kits serve as the recurrent revenue engine for the industry, securing 64.4% of the market share in 2026. This dominance is sustained by the high-frequency consumption of assay-specific chemistries required for both routine infectious disease screening and complex genomic profiling.
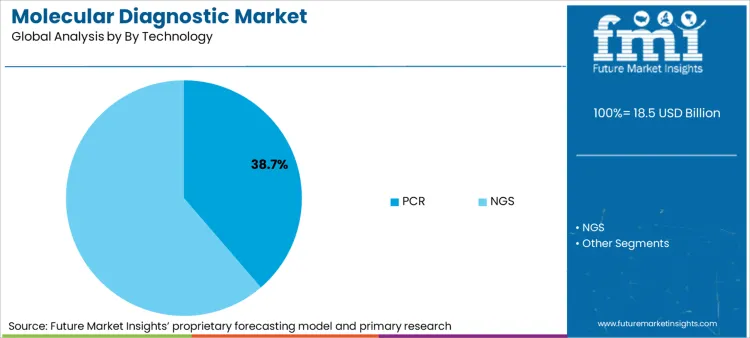
PCR technology remains the workhorse of the sector, holding 38.7% share, while Next-Generation Sequencing (NGS) captures 24.0% by value. The shift is defined by PCR's entrenched role in rapid screening and NGS's indispensability in precision oncology.
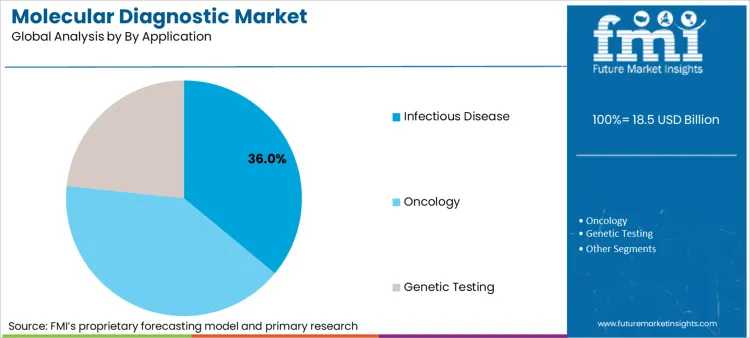
Infectious disease testing accounts for 36% of market volume, but oncology applications are the primary value driver. The disparity arises because infectious disease tests are high-volume/low-margin, whereas oncology based molecular diagnostics command premium pricing due to clinical complexity.
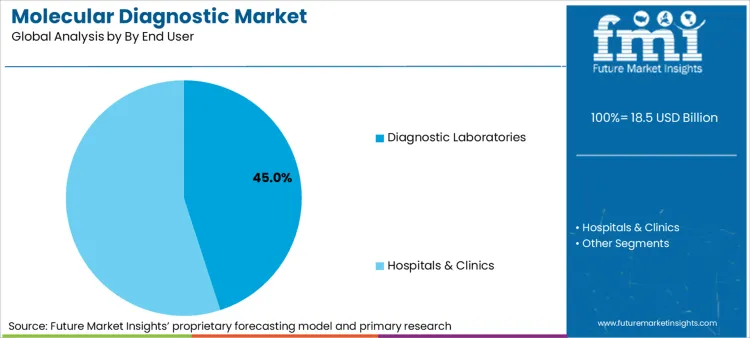
Diagnostic laboratories constitute 45% of the user base, serving as centralized hubs for high-complexity testing. However, the fastest functional growth is occurring in decentralized hospital settings and public health networks.
The primary catalyst for market value expansion is the urgent clinical requirement to close the "screening gap" in oncology. With nearly half of all cancers diagnosed annually having no recommended screening tests, healthcare systems are aggressively pivoting toward liquid biopsy and multi-cancer early detection (MCD) platforms to catch malignancies at treatable stages. This demand is converted into revenue through high-value reimbursement codes and large-scale procurement of NGS platforms. Abbott's strategic acquisition of Exact Sciences in late 2025 underscores this shift, as major players consolidate to capture the nascent but lucrative MCD segment. [1, 9]
A significant restraint involves the tightening regulatory environment for Laboratory Developed Tests (LDTs), specifically in the United States. The FDA's final rule ending enforcement discretion for LDTs imposes Class III medical device requirements on high-complexity in-house tests, creating a massive compliance and cost burden for independent laboratories. This mechanism slows the rollout of novel assays as labs must now invest time and capital into premarket review processes or switch to approved IVD kits. The uncertainty surrounding these regulations forces smaller players to pause innovation or seek acquisition by larger entities capable of navigating the new compliance landscape. [10]
Based on the regional analysis, the molecular diagnostic market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 5.9% |
| Thailand | 4.7% |
| France | 4.3% |
| Malaysia | 4.3% |
| Indonesia | 3.9% |
| Canada | 3.8% |
| United States | 2.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America remains the value capital of the industry, characterized by a transition from infectious disease volume to high-margin oncology diagnostics. The region is seeing a "green recovery" in terms of sustainable lab practices and a heavy emphasis on automation to combat labor shortages.
FMI’s report includes a detailed growth analysis for North America. Including the US and Canada, Mexico offers opportunistic growth in cross-border medical tourism for specialized genetic testing. Buyers in the region should monitor the integration of AI-driven bioinformatics into reimbursement schedules, which will define the profitability of future NGS installations. [10]
Europe is operating as a regulatory pioneer, where stringent IVDR compliance is reshaping the competitive field. The market is driven by localized environmental health initiatives and a strong focus on syndromic testing panels that offer higher diagnostic yields per patient visit.
FMI’s report includes comprehensive coverage of the European landscape. Germany and the UK remain critical markets for automated molecular diagnostics testing system market adoption, driven by centralized lab consolidation. Suppliers should watch for opportunities in the Nordics, where digitized health records are facilitating large-scale population genomics programs similar to France's environmental initiatives. [7]
Asia Pacific is rapidly evolving from a technology importer to a hub of localized innovation and genomic data generation. As per FMI's projection, the region is leveraging pandemic-era investments to build sovereign diagnostic capabilities that address region-specific disease burdens like tuberculosis and dengue.
FMI’s report includes a deep dive into the Asia Pacific opportunity. Vietnam and the Philippines are emerging as key markets for multiplex molecular diagnostics to manage tropical infectious diseases. Strategic players should focus on public-private partnerships for technology transfer, as seen with Indonesia's collaboration with Japanese medical groups to build advanced cardiovascular and diagnostic centers. [4]
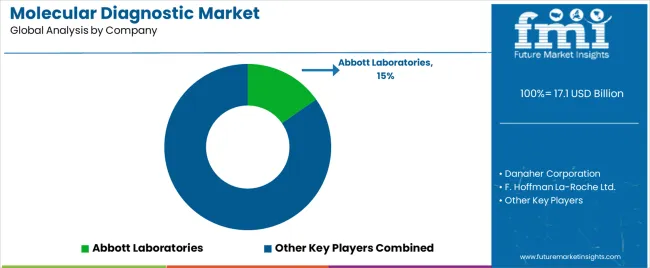
Competition is bifurcated between scale players driving automation and niche innovators focusing on proprietary content. Leading diagnostic giants are leveraging their installed base of instruments to enforce "menu lock-in," ensuring that customers purchasing hardware for one indication (e.g., COVID-19) eventually adopt the same platform for oncology or sexual health testing. This strategy increases switching costs for laboratories and secures long-term reagent revenue streams.
Regulatory agility has become a primary competitive advantage. With the FDA's shift on LDTs and Europe's IVDR rollout, companies that can rapidly secure premarket approval for their assays are displacing smaller competitors who relied on enforcement discretion. Roche’s ability to secure CE marks for updated systems and Abbott’s strategic acquisitions demonstrate how regulatory capital is being used to clear the field of non-compliant rivals. [8, 10]
Innovation in workflow efficiency is differentiating the winners from the commoditized bulk suppliers. Companies introducing technologies that multiply throughput without expanding footprint, such as Roche’s TAGS technology which allows 15 targets in a single well, are winning contracts in space-constrained hospital labs. These technical leaps directly address the "cost-per-reportable-result" metric that dominates tender evaluations in value-conscious markets. [8]
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
| Metric | Value |
|---|---|
| Quantitative Units | USD 18.5 billion (2026) to USD 41.5 billion (2036), at a CAGR of 8.4% |
| Market Definition | The molecular diagnostic industry encompasses the development and commercialization of technologies that analyze biological markers in the genome and proteome to detect disease. |
| Product Segmentation | Reagents & Kits, Instruments, Service & Software |
| Technology Segmentation | PCR, NGS, Others |
| Application Coverage | Infectious Disease, Oncology, Genetic Testing, Others |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, Canada, France, India, Thailand, Malaysia, Indonesia and 40 plus countries |
| Key Companies Profiled | Abbott Laboratories, Danaher (Cepheid), Roche Diagnostics, Siemens Healthineers, BD, Agilent Technologies, bioMérieux |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with resin producers and panel manufacturers, supported by trade data benchmarking and plant level capacity verification |
How large is the demand for Molecular Diagnostic in the global market in 2026?
Demand for Molecular Diagnostic in the global market is estimated to be valued at USD 18.5 billion in 2026.
What will be the market size of Molecular Diagnostic in the global market by 2036?
Market size for Molecular Diagnostic is projected to reach USD 41.5 billion by 2036.
What is the expected demand growth for Molecular Diagnostic in the global market between 2026 and 2036?
Demand for Molecular Diagnostic in the global market is expected to grow at a CAGR of 8.4% between 2026 and 2036.
Which Product is poised to lead global sales by 2026?
Reagents & Kits are expected to be the dominant form, capturing approximately 64.4% of global market share in 2026 due to recurring consumption.
How significant is the role of Infectious Disease in driving Molecular Diagnostic adoption in 2026?
Infectious Disease represents a critical segment, projected to hold a substantial 36.0% share of the total market in 2026.
What is driving demand in India?
Demand is driven by the curation of indigenous genomic databases and a shift toward localized cancer care protocols.
What compliance standards or regulations are referenced for the United States?
The FDA's final rule regarding the oversight of Laboratory Developed Tests (LDTs) is a key regulatory benchmark.
What is the India growth outlook in this report?
India is projected to grow at a CAGR of 5.9% during 2026 to 2036.
Why is Europe described as a priority region in this report?
Europe is prioritizing environmental surveillance and strict IVDR compliance, driving replacement demand for approved systems.
What type of demand dominates in Europe?
Demand is dominated by high-throughput syndromic testing and wastewater surveillance programs.
What is Thailand growth outlook in this report?
Thailand is projected to expand at a CAGR of 4.7% during 2026 to 2036.
Does the report cover Malaysia in its regional analysis?
Yes, Malaysia is included within Asia Pacific under the regional scope of analysis.
What are the sources referred to for analyzing Malaysia?
Official molecular diagnostic test menus from state health departments and hospital procurement lists are cited.
What is the main demand theme linked to Malaysia in its region coverage?
Growth is linked to the expansion of specialized public hospital testing menus for genetic and oncology conditions.
Does the report cover Indonesia in its regional analysis?
Yes, Indonesia is included within Asia Pacific under the regional coverage framework.
What is the main Indonesia related demand theme in its region coverage?
Demand is driven by the maintenance and repurposing of 1,000 PCR laboratories for tuberculosis and endemic disease management.
Which product formats or configurations are strategically important for Asia Pacific supply chains?
Compact, automated PCR systems and localized reagent manufacturing are prioritized to ensure supply security.
What is Molecular Diagnostic and what is it mainly used for?
Molecular diagnostic refers to technologies that analyze biological markers in the genome to detect disease, used for infectious disease, oncology, and genetic testing.
What does Molecular Diagnostic Market mean in this report?
It refers to the global trade of instruments, reagents, and software for nucleic acid testing in clinical settings.
What is included in the scope of this Molecular Diagnostic report?
Scope covers PCR, NGS, and other nucleic acid technologies used in labs, hospitals, and clinics.
What is excluded from the scope of this report?
Traditional immunoassays, clinical chemistry, and research-only (RUO) products without clinical application are excluded.
What does market forecast mean on this page?
Market forecast represents a model based projection built on defined assumptions for strategic planning purposes.
How does FMI build and validate the Molecular Diagnostic forecast?
Forecast is developed using hybrid top down and bottom up modeling validated through trade data, capacity checks, and industry review.
What does zero reliance on speculative third party market research mean here?
Primary interviews and verifiable public datasets are used instead of unverified syndicated market estimates.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.