Clinical Diagnostics Market
The Clinical Diagnostics Market is segmented by Product Type (Instruments, Reagents and Consumables, Software and Services), Technology (Immunoassay, Clinical Chemistry, Molecular Diagnostics, Haematology, Urinalysis, and Others), End Use (Hospitals, Reference Laboratories, Physician Offices, and Others), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Clinical Diagnostics Industry Size and Share Forecast Outlook By FMI
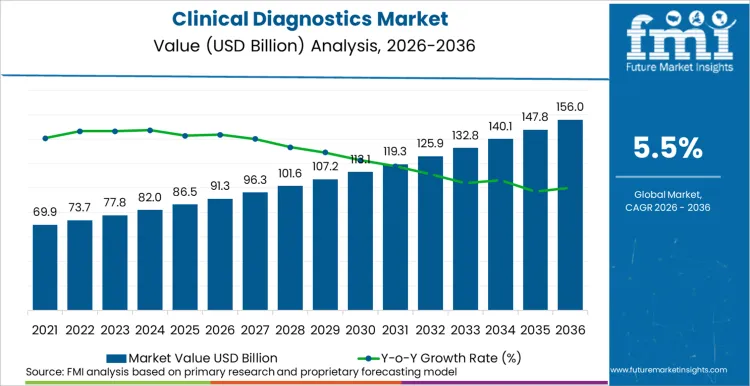
In 2025, the clinical diagnostics market was valued at USD 86.5 billion. Based on Future Market Insights' analysis, demand is estimated to grow to USD 91.3 billion in 2026 and USD 155.9 billion by 2036. FMI projects a CAGR of 5.5% during the forecast period.
Absolute dollar growth of USD 64.6 billion over the decade reflects sustained volume expansion driven by ageing populations, chronic disease prevalence growth, and the integration of molecular diagnostics and liquid biopsy into routine clinical pathways. Roche's announcement that its NGS technology can decode an entire human genome in under four hours is compressing diagnostic turnaround and expanding the addressable test menu. Thermo Fisher's FDA approval of the Ion Torrent Oncomine Dx Target Test for liquid biopsy pan-cancer profiling further accelerates molecular diagnostics adoption beyond specialty oncology centres into reference laboratory networks.
Summary of Clinical Diagnostics Market
- Value Chain Power Concentration
- Power concentrated among four global IVD manufacturers:
- Roche
- Abbott
- Siemens Healthineers
- Danaher (Beckman Coulter)
- These firms control the installed base of high-throughput immunoassay and clinical chemistry analysers in hospital core laboratories.
- Reagent rental models create multi-year revenue lock-in.
- Reference laboratory power concentrated with Quest Diagnostics and Labcorp.
- High test volumes give reference labs pricing leverage in reagent procurement negotiations.
- Power concentrated among four global IVD manufacturers:
- Sourcing Fragility
- Dependency on specialty reagent raw materials and biological reference standards.
- Molecular platforms rely on enzyme and nucleotide supply chains with limited qualified sources.
- Semiconductor components required for analyser electronics.
- EU IVDR transition requires reagent re-validation against new performance standards.
- Brand & Compliance Risks
- EU IVDR implementation timelines and notified body backlog.
- Risk of product withdrawal in Europe due to regulatory delays.
- FDA laboratory-developed test (LDT) regulation impacts U.S. market dynamics.
- Early IVDR-compliant manufacturers positioned to capture market share.
- Geographic Leverage
- United States: Reference lab consolidation and molecular reimbursement leadership.
- China: Volume growth via hospital lab expansion and domestic IVD scaling.
- Europe: IVDR-driven compliance restructuring.
- India: Hospital infrastructure expansion and rural point-of-care testing growth.
- Japan: Highest per-capita diagnostic testing volume.
- Strategic Imperatives Through 2036
- Expand molecular diagnostics menus on installed analyser platforms.
- Achieve earliest IVDR compliance to secure European market share.
- Develop AI-assisted diagnostic interpretation capabilities.
- Position diagnostics as clinical decision support rather than commodity sample processing.
As Jim Davis, CEO of Quest Diagnostics, stated regarding 2025 financial performance, 'Our performance demonstrates continued execution of our strategy to deliver category-defining clinical innovations that fulfill customers' needs.' [1]
The United States (6.0% CAGR) leads through Medicare population growth, reference laboratory consolidation, and molecular test reimbursement expansion. China (7.2% CAGR) is the fastest-growing market through hospital laboratory modernization and domestic IVD manufacturer scaling. Germany (5.0% CAGR) grows through IVD Regulation (IVDR) compliance investment. India (6.8% CAGR) benefits from hospital infrastructure expansion and point-of-care testing deployment. Japan (4.5% CAGR) contributes through high per-capita testing rates and automation investment.
Clinical Diagnostics Market Definition
The clinical diagnostics market covers the manufacture, sale, and servicing of in vitro diagnostic instruments, reagents, consumables, and software used in hospitals, reference laboratories, and physician offices for the analysis of blood, urine, tissue, and other human specimens. Technologies include immunoassay, clinical chemistry, molecular diagnostics, haematology, and urinalysis.
Clinical Diagnostics Market Inclusions
The report covers global and regional market sizes by revenue for the 2026 to 2036 forecast period. It includes segmental breakdowns by product type, technology, and end use. Reagent rental model economics, laboratory automation tracking, and molecular diagnostics test menu expansion are included.
Clinical Diagnostics Market Exclusions
The scope excludes imaging diagnostics, pathology slide scanners, and radiology equipment. Research-use-only (RUO) products not cleared for clinical use are outside scope. Over-the-counter consumer self-test kits (home pregnancy tests, OTC COVID tests) are excluded.
Clinical Diagnostics Market Research Methodology
- Primary Research: Interviews were conducted with laboratory directors, hospital procurement managers, IVD instrument manufacturers, molecular diagnostics platform developers, and reference laboratory executives across the United States, Europe, China, and India.
- Desk Research: FDA 510(k) and PMA clearance databases, CE-IVDR registration records, Medicare reimbursement code publications, and hospital laboratory capital expenditure surveys supported demand benchmarking.
- Market-Sizing and Forecasting: A hybrid model combined top-down healthcare expenditure allocation for diagnostics with bottom-up instrument installed base, reagent consumption per test, and molecular diagnostics test volume data, validated against IVD manufacturer annual reports.
- Data Validation and Update Cycle: Outputs undergo anomaly screening, variance checks across test volume and IVD revenue datasets, and structured peer review prior to release.
Clinical Diagnostics Industry Analysis by Top Investment Segments
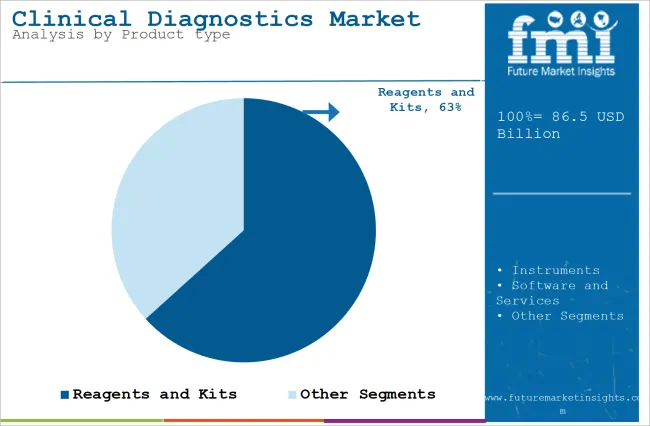
The section contains information about the leading segments in the industry. Based on product type, the reagents and kits segment is expected to account for 63.3% of the global share in 2026.
Reagents and Kits segment will dominate the Clinical Diagnostics Market Owing to their Essential Components of Diagnostic Testing in the Market
| By Product type | Reagents and Kits |
|---|---|
| Value Share (2026) | 63.3% |
The reagents and kits products segment is projected to be a dominating segment in terms of revenue, accounting for almost 63.3% of the market share in 2026.
Reagents and kits form the backbone of numerous clinical diagnostic tests. Biochemical reagents, immunoassays, molecular reagents, and enzymes are used to detect and quantify many biomarkers, pathogens, genetic material, or diseases in patient samples (e.g., blood, urine, or saliva).
These reagents are engineered to bind with biological material to allow the detection of a specific marker related to a disease or condition.
As there is hardly any diagnostic procedure (from clinical chemistry to molecular diagnostics) that does not involve reagents and kits, their higher consumption in various diagnostic platforms consequently makes them a leader in the market.
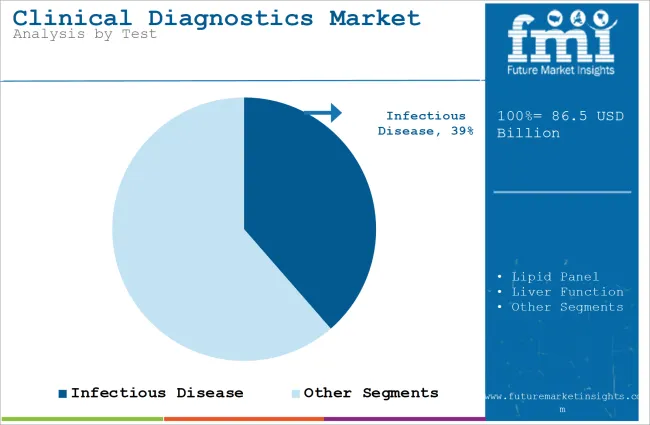
Infectious Disease is expected to dominate the Clinical Diagnostics Market owing to its Increasing Prevalence
| By Test | Infectious Disease |
|---|---|
| Value Share (2026) | 38.6% |
Infectious diseases continue to be a significant threat globally and therefore constitute the first concern of diagnostic products. Diseases caused by tuberculosis, hepatitis, HIV/AIDS, malaria, and the new entrant COVID-19 still present enormous morbidity and mortality worldwide.
Global epidemiological burdens of such diseases, especially in developing and low-middle income countries, contribute to an enormous need for more precise and quick diagnosis for better control and management and to halt their transmission.
The new infectious diseases evolving, such as novel viruses (e.g., SARS-CoV-2), antimicrobial-resistant pathogens, and zoonotic diseases, are other approaches in continuing to challenge global health.
The hazard of pandemics and epidemics continues to pose a threat; therefore, strong and reliable diagnostic solutions, leading to fast identification of the infection, prevent outbreaks and help in timely intervention.
Key Clinical Diagnostics Industry Trends
Increasing Prevalence of Chronic Diseases and Aging Populations is driving the Clinical Diagnostics Market Growth
A rise in the chronic diseases, combined with a world aging population, creates a big thrust in clinical diagnostics. Increasing incidences of chronic conditions, like diabetes, cardiovascular diseases, cancer, and respiratory disorders, mainly as a result of lifestyle and environmental changes and due to demographic ageing, make their continuous monitoring and early detection critical for controlling the symptoms and the long-term consequences.
For example, blood glucose testing should be conducted at regular intervals by patients with diabetes, and diagnostic procedures for cancer also involve biomarker tests and genetic examinations to establish whether a tumor exists and which intervention would be appropriate. As such, chronic illnesses require proper detection tools that should help in their prevention, diagnosis, and even follow-up and monitoring.
The world population is aging faster than ever in human history; projections show that, by 2050, people aged 60 years and above will nearly double. Age, as a natural part of life, goes with it increased vulnerability to developing chronic diseases or conditions such as Alzheimer's, arthritis, or hypertension. Such increases enhance the demand for clinical diagnostics.
Old-aged patients usually demand regular health checks, early diagnosis of diseases, and treatments accordingly, leading to the demand for diagnostic equipment that can identify diseases at an initial stage. Such changes are pressurizing the health systems around the globe to upgrade and provide better and efficient diagnostic technology.
Increased burden of chronic diseases and an aging population are fast giving growth to the clinical diagnostics market, as healthcare service providers now strive hard to meet the increasing demand for more sophisticated testing methods that improve patient care.
Technological Advancements and Innovation in Diagnostic Tools is driving Revenue Growth for Clinical Diagnostics
The continuous introduction of innovations in diagnostic tools and techniques is majorly driving the market of Clinical Diagnostics. However, over the last few years, considerable shifts have been introduced in the present landscape of diagnostic, which involved molecular diagnostics, next-generation sequencing (NGS), point-of-care (POC) testing, and artificial intelligence (AI) diagnostic solutions.
Such advances enhance the pace, accuracy, and cost-effectiveness of diagnostics and make early disease detection more accessible, therefore, allowing more informed clinical decision-making. For example, molecular diagnostics makes it possible to conduct specific genetic testing, including identification of predispositions, infections, and cancer at the molecular level.
Comprehensive genome analysis via technologies such as NGS facilitates identification and is most crucial for personalizing medicine and targeted therapies with enhanced treatment outcome.
Laboratory automation has improved throughput, minimized human error, and enhanced consistency in testing. As diagnostic tools begin to be cheaper in price, the patient begins to opt for home-based testing and remote monitoring platforms, driving market growth.
Further advancements of technology will enhance the development of high-quality diagnostics with increasing accuracy, resulting in improved detection at an earlier stage of the disease, offering personal care for individuals, and improving the effective interventions in various treatments of disease.
Growth of Personalized Medicine and Point-of-Care Testing is Creating Opportunities in the Market
One potential growth opportunity lies in the upcoming personalized medicine and point-of-care (POC) testing opportunity in the Clinical Diagnostics market. Personalized medicine is targeted at patients where treatment plans for patients are matched to genetic makeup, lifestyle, and environment in order to obtain the best-fit therapies, where precise and timely diagnostic data help ensure that they receive the appropriate therapies.
With the rising demand for precision health care, more and more will be needed diagnostic tools that can deliver personalized insights. Advances in genetic testing, biomarker discovery, and liquid biopsy technologies enable clinicians to identify the best therapeutic options for patients, especially for oncology where treatments can often be highly individualized based on their tumor's genetic profiles.
POC diagnostic instruments provide fast test capabilities outside a standard laboratory and report results within hours, enabling a quicker turnaround on decision-making and treatment. The escalating adoption of mobile technologies and telemedicine increases demand for POC testing, enabling patients and care providers to monitor and manage health conditions from remote locations.
High Costs and Regulatory Barriers may Restrict Market Growth
Despite the growth potential in the clinical diagnostics market, one of the biggest drawbacks is that advanced diagnostic equipment is costly and has intricate regulatory issues that often lead to a lengthy delay in new product launches. It would make these technologies prohibitively expensive for smaller health care providers and patients, mainly in low resource settings.
With many diagnostic innovations promising better accuracy and speed, a high initial investment in procuring, implementing, and maintaining them is a reason for limited widespread adoption. For example, complex molecular diagnostic instruments require highly specialized training for their operators and involve significant capital investments, thus limiting their use to large, well-funded healthcare facilities.
Moreover, regulatory complexities in various locations, such as the USA and Europe, can further hamper the diagnostic devices' access to the marketplace, especially on newer technologies and AI-based products for diagnostics. Regulatory bodies are stringent in making their guidelines such as the USA Food and Drug Administration (FDA), the European Medicines Agency of Europe, require extensive testing methods before a product makes it to market.
Comparison of Historical vs Future Outlook on Changing Consumer Trends for Clinical Diagnostics
The global clinical diagnostics industry recorded a CAGR of 6.3% during the historical period between 2020 and 2024. The growth of clinical diagnostics industry was positive as it reached a value of USD 82.6 billion in 2024 from USD 313.7 billion in 2020.
Historically, the clinical diagnostics market was primarily centered around traditional laboratory tests that involved manual processes and relatively slow turnaround times. In the early stages, diagnostic technologies relied heavily on microscopic analysis and biochemical assays to detect diseases. The most common diagnostic tests involved blood tests, urine tests, and imaging methods.
These were not only limited in scope but also in precision. This innovation increased testing efficiency, reduced human error, and laid the ground for the more sophisticated systems that followed.
Presently, the market of clinical diagnostics involves a host of advanced technologies and methodologies applied within different health facilities. The foundations of clinical diagnostics revolve around three major diagnostic principles: molecular diagnostics, point-of-care (POC) testing, and immunoassays. Such tests are generally rapid, highly accurate, and invasive.
Some such diseases are genetic disorders, cancer, and infections. Such technologies promise earlier detection and the possibility of more proper treatment plans. This is increasingly important as health care evolves toward preventive and personalized care.
The clinical diagnostics market is headed toward further expansion as several new trends are likely to be seen impacting it in the coming years. An emerging trend currently, which stands to significantly revolutionize the clinic, is personalization in medical treatments.
Increased implementation of precision medicine in the health sector implies more genetic testing and analysis of potential biomarkers coupled with companion diagnostics, which inform a patient that can be more customized according to individual characteristics to treatment.
The potential for significant growth in the oncology diagnostics market will come through revolutionizing oncology diagnostics through liquid biopsy technology, a technology that would help diagnose and monitor cancer without requiring invasive procedures.
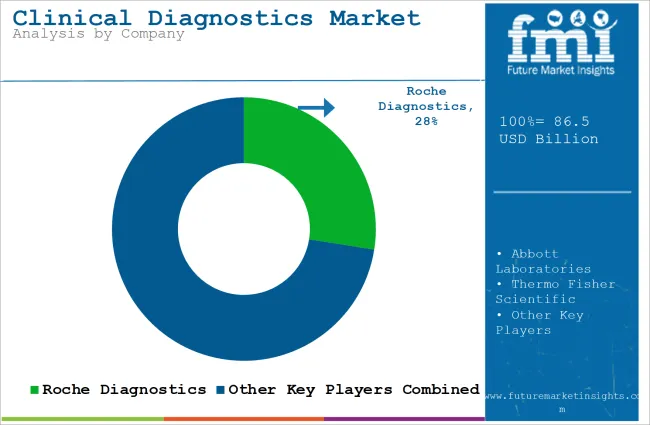
Key Players in the Clinical Diagnostics Industry by Share
Tier 1 companies are the industry leaders with 55.4% of the global industry. These companies stand out for having a large product portfolio and a high production capacity. These industry leaders also stand out for having a wide geographic reach, a strong customer base, and substantial experience in manufacturing and having enough financial resources, which enables them to enhance their research and development efforts and expand into new industries.
The companies within tier 1 have a good reputation and high brand value. Prominent companies within tier 1 include Roche Diagnostics, Abbott Laboratories, Thermo Fisher Scientific, and Siemens Healthineers.
Tier 2 companies are relatively smaller as compared with tier 1 players. The tier 2 companies hold a market share of 27.2% worldwide. These firms may not have cutting-edge technology or a broad global reach, but they do ensure regulatory compliance and have good technology. The players are more competitive when it comes to pricing and target niche markets.
Key Companies under this category include Danaher Corporation, Bio-Rad Laboratories, Becton, Dickinson and Company, Quest Diagnostics, bioMérieux among others.
Compared to Tiers 1 and 2, Tier 3 companies offer clinical diagnostics, but with smaller revenue spouts and less influence. These companies mostly operate in one or two countries and have limited customer base. They specialize in specific products and cater to niche markets, adding diversity to the industry.
Analysis of Top Countries Using Clinical Diagnostic Tools
The market analysis for clinical diagnostics in various nations is covered in the section below. An analysis of important nations in North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, and Middle East & Africa of the world has been mentioned below.
It is projected that the United States will maintain its leading position in North America through 2036, holding a value share of 91.2%. By 2036, China is expected to experience a CAGR of 5.6% in the Asia-Pacific region.
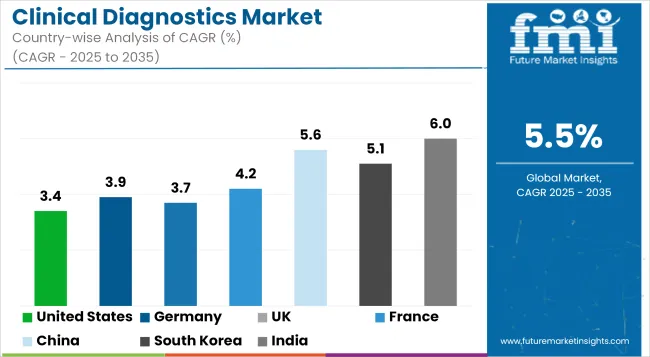
| Countries | Value CAGR (2026 to 2036) |
|---|---|
| United States | 3.4% |
| Germany | 3.9% |
| UK | 3.7% |
| France | 4.2% |
| China | 5.6% |
| South Korea | 5.1% |
| India | 6.0% |
Strong Healthcare Infrastructure and Investment in Medical Technology is Fueling Growth is driving growth on Germany
Germany’s clinical diagnostics market is poised to exhibit a CAGR of 3.9% between 2026 and 2036. The Germany holds highest market share in European market.
Germany is one of the most developed and complete healthcare systems in Europe, thereby directly contributing to the growth of the clinical diagnostics market. This is due to a well-structured hospital infrastructure along with networks of diagnostic centers and research institutes, which have high-tech diagnostic technologies.
In such a set-up, various modern diagnostic technologies, such as molecular diagnostics, genetic testing, and NGS, are likely to be embraced across the market.A high commitment to standards, coupled with good reimbursement, from Germany drives a demand for new diagnostic equipment that is not inferior. Germany also spends heavily on medical research and development (R&D), which influences diagnostic technology.
The government and private enterprise have been particularly active in furthering biotechnology, biomarker discovery, and precision medicine-all requiring sophisticated platforms for testing.Significant investment by the country in medical technology companies and clinical trials fuels innovation and leads to the creation of novel diagnostic tools that quickly emerge within the healthcare system.
Increasing Investment in Research and Development is a growth factor in the United States
United States is anticipated to show a CAGR of 3.4% between 2026 and 2036.
Robust healthcare infrastructure coupled with major research and development spending remains an important force of the United States Clinical Diagnostics market. No other healthcare system is quite at par to what exists within the United States: cutting-edge hospitals, a lot of in-country research centers and diagnostic labs across the world.
The United States hosts some of the biggest players in healthcare provider companies and diagnostics firms, heavily investing in diagnostic innovation. Such investment allows for innovative development and deployment of top-notch tools that range from molecular diagnostics, genetic testing, to NGS and AI-based systems for diagnosis.
Besides funding for research, the USA private sector also greatly contributes to the expansion of clinical diagnostics with the advancement.
The market of the country contains leading diagnostic companies such as Thermo Fisher Scientific, Abbott Laboratories, Roche Diagnostics, and Quest Diagnostics, who advance their diagnostic technology and add on to their existing product portfolios regularly.
Rising Prevalence of Chronic Diseases and Aging Population drives the Market in India
India is anticipated to show a CAGR of 6.0% between 2026 and 2036.
India is experiencing a monumental shift in its health care system that has been characterized with an ever-expanding burden of non-communicable diseases as well as an ageing population.Chronic diseases such as diabetes, cardiovascular diseases, hypertension, chronic respiratory diseases and cancer have increased globally over time, attributed largely to wide urbanization, lifestyle, unhealthy diet and inactivity.
India has second highest diabetic population in the world, with more than 77 million bad patient suffering from it. Since they are chronic in nature and require frequent monitoring and management, regular diagnostic testing plays a crucial role in improving patient outcomes and decreasing related complications.More importantly, and perhaps more alarmingly, there is a vast ageing population in India that is only going to grow; it is projected to expand exponentially for the older populations those over 60 years of age in the coming decades.
Neurodegenerative disorders (Alzheimer's and Parkinson's disease), cancers, and musculoskeletal disorders are abundant in the older group of women and men.As population ages, there is a growing need for diagnostic technologies that can identify disease at the earliest stages, and monitor its progression.
Leading Clinical Diagnostics Market Players
In terms of this market, the companies involved are resorting to multiple strategies to retain a competitive position. Strategic partnerships and collaborations with research institutions and healthcare providers are being utilized to broaden their product portfolio.
Geographical expansion into the emerging markets, particularly United States and Asia Pacific countries, has been another strategic priority for these companies, where growth in the healthcare infrastructure and awareness is strong.
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the report:
- In January 2026, Thermo Fisher Scientific received FDA approval for the Ion Torrent Oncomine Dx Target Test, expanding liquid biopsy use for pan-cancer profiling. [1]
- In January 2026, Roche announced its next-generation NGS technology can decode an entire human genome in under four hours. [2]
- In February 2026, Quest Diagnostics reported record 2025 financial results and increased its quarterly dividend 7.5%, reflecting category-defining innovation execution. [3]
- In November 2025, Siemens Healthineers expanded its Atellica analyser platform with new immunoassay menu additions for autoimmune and infectious disease testing. [4]
- In 2025, Abbott launched its next-generation Alinity hq haematology analyser with AI-powered cell classification for improved diagnostic accuracy. [5]
Top Clinical Diagnostics Suppliers
- Roche Diagnostics
- Abbott Laboratories
- Thermo Fisher Scientific
- Siemens Healthineers
- Danaher Corporation
- Bio-Rad Laboratories
- Becton, Dickinson and Company
- Quest Diagnostics
- bioMérieux
- Others
Scope of the Report
| Attribute | Details |
|---|---|
| Quantitative Units | USD 91.3 billion (2026) to USD 155.9 billion (2036), at a CAGR of 5.5% |
| Market Definition | Covers manufacture, sale, and servicing of IVD instruments, reagents, consumables, and software for clinical specimen analysis in hospitals, reference laboratories, and physician offices. |
| Product Type Segmentation | Instruments, Reagents and Consumables, Software and Services |
| Technology Segmentation | Immunoassay, Clinical Chemistry, Molecular Diagnostics, Haematology, Urinalysis, Others |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, China, Germany, Japan, India, United Kingdom, France, South Korea, Brazil, Australia and 40 plus countries |
| Key Companies Profiled | Roche Diagnostics, Abbott Diagnostics, Siemens Healthineers, Danaher (Beckman Coulter), Thermo Fisher Scientific, bioMerieux, Sysmex, Ortho Clinical Diagnostics, Quest Diagnostics, Labcorp |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up modeling validated through primary interviews with laboratory directors, IVD manufacturers, and reference laboratory executives |
Top Segments Studied in the Clinical Diagnostics Industry Research Report
By Product type:
In terms of product type, the industry is divided into instruments, reagents and kits, software and services.
By Test:
In terms of test, the industry is segregated into lipid panel, liver function, kidney function, complete blood count, infectious disease, thyroid function, glucose monitoring, and cancer diagnostics among others.
By End User:
In terms of end user, the industry is divided into hospitals, specialty clinics, diagnostic laboratories, point-of-care settings, research institutes and academic centers.
By Region:
Key countries of North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe and Middle East and Africa (MEA) have been covered in the report.
Bibliography
- [1] Thermo Fisher Scientific. (2026). Ion Torrent Oncomine Dx Target Test FDA Approval. January 2026.
- [2] Roche. (2026). Next-Generation NGS Genome Sequencing Announcement. January 2026. https://www.roche.com
- [3] Quest Diagnostics. (2026). Q4 and Full-Year 2025 Financial Results. February 2026.2025-Financial-Results-Provides-Guidance-for-Full-Year-2026-Increases-Quarterly-Dividend-7-5-to-0-86-Per-Share
- [4] Siemens Healthineers. (2025). Atellica Analyser Platform Menu Expansion. November 2025.
- [5] Abbott. (2025). Alinity hq Haematology Analyser Launch.
- [6] FDA. (2025). In Vitro Diagnostics Device Approvals.
- [7] European Commission. (2025). EU IVDR Implementation Status.
- [8] WHO. (2025). Essential Diagnostics List.
- [9] CMS. (2025). Medicare Clinical Laboratory Fee Schedule.
- [10] OECD. (2025). Health Spending Indicators.
- [11] Future Market Insights. (2026). Clinical Diagnostics Market Report.
- [12] Future Market Insights. (2025). In Vitro Diagnostics Market.
This Report Addresses
- Market intelligence for IVD procurement, hospital laboratory planning, and reference laboratory strategy
- Market size estimation and 10-year revenue forecasts from 2026 to 2036
- Growth opportunity mapping across immunoassay, clinical chemistry, molecular diagnostics, and haematology
- Segment and regional revenue forecasts covering hospitals, reference laboratories, and physician offices
- Competition strategy assessment including reagent rental model economics and installed base leverage
- Technology tracking including liquid biopsy, rapid genome sequencing, and AI-assisted interpretation
- Regulatory impact analysis covering EU IVDR transition, FDA LDT regulation, and Medicare reimbursement
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats
Frequently Asked Questions
How large is the clinical diagnostics market in 2026?
The global market is estimated at USD 91.3 billion in 2026.
What will the market size be by 2036?
The market is projected to reach USD 155.9 billion by 2036.
What is the expected CAGR?
Demand is expected to grow at a CAGR of 5.5% between 2026 and 2036.
Which technology segment is growing fastest?
Molecular diagnostics is the fastest-growing technology segment, driven by liquid biopsy adoption and NGS test menu expansion.
Which country is growing fastest?
China at 7.2% CAGR through hospital laboratory modernization and domestic IVD manufacturer scaling.
What product type generates the most revenue?
Reagents and consumables generate the largest share of market revenue through multi-year reagent rental contracts.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product type , 2026 to 2036
- Reagents and Kits
- Instruments
- Software and Services
- Reagents and Kits
- Y to o to Y Growth Trend Analysis By Product type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Test
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test, 2026 to 2036
- Infectious Disease
- Lipid Panel
- Liver Function
- Kidney Function
- Infectious Disease
- Y to o to Y Growth Trend Analysis By Test, 2021 to 2025
- Absolute $ Opportunity Analysis By Test, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product type
- By Test
- By Country
- Market Attractiveness Analysis
- By Country
- By Product type
- By Test
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product type
- By Test
- By Country
- Market Attractiveness Analysis
- By Country
- By Product type
- By Test
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product type
- By Test
- By Country
- Market Attractiveness Analysis
- By Country
- By Product type
- By Test
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product type
- By Test
- By Country
- Market Attractiveness Analysis
- By Country
- By Product type
- By Test
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product type
- By Test
- By Country
- Market Attractiveness Analysis
- By Country
- By Product type
- By Test
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product type
- By Test
- By Country
- Market Attractiveness Analysis
- By Country
- By Product type
- By Test
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product type
- By Test
- By Country
- Market Attractiveness Analysis
- By Country
- By Product type
- By Test
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product type
- By Test
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product type
- By Test
- Competition Analysis
- Competition Deep Dive
- Roche Diagnostics
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Abbott Laboratories
- Thermo Fisher Scientific
- Siemens Healthineers
- Danaher Corporation
- Bio-Rad Laboratories
- Roche Diagnostics
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Test, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product type
- Figure 6: Global Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Test, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Test
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product type
- Figure 23: North America Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Test, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Test
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product type
- Figure 30: Latin America Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Test, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Test
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Test, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Test
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Test, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Test
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product type
- Figure 51: East Asia Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Test, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Test
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Test, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Test
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Test, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Test
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

