Clinical Trials Market
Clinical Trials Market Analysis Size and Share Forecast Outlook 2026 to 2036
Clinical Trials Market Forecast and Outlook 2026 to 2036
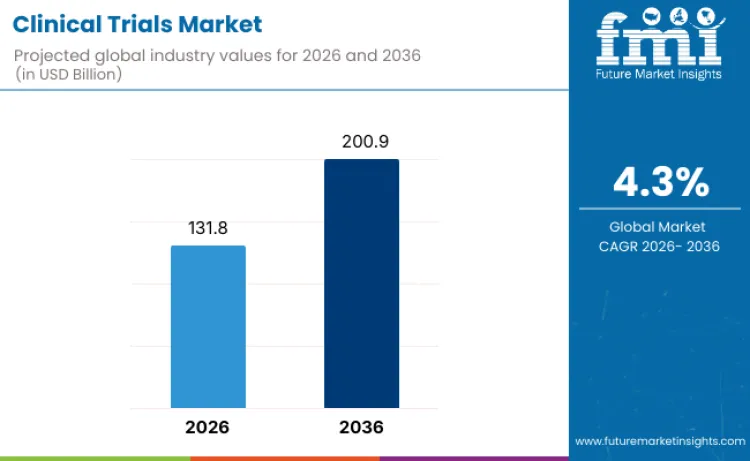
The clinical trials market is valued at USD 131.8 billion in 2026 and is projected to reach USD 200.9 billion by 2036, expanding at a CAGR of 4.3% during the forecast period. Growth reflects the increasing complexity of drug development and the steady rise in research activity across pharmaceuticals, biotechnology, and medical devices. Sponsors continue to scale trial volumes as pipelines move toward targeted therapies, biologics, and advanced treatment approaches that demand extensive clinical validation.
Demand is shaped by the growing burden of chronic diseases, oncology indications, and rare disorders that require long duration studies and multi phase evaluation. Pharmaceutical companies depend on structured trial programs to meet regulatory expectations while managing development timelines and investment risk. Biotechnology firms add strong momentum as innovation accelerates in gene therapies, cell based treatments, and personalized medicine. Medical device manufacturers also contribute through trials focused on safety assessment, performance validation, and post approval studies.
Quick Stats for Clinical Trials Market
- Market Value (2026): USD 131.8 billion
- Market Forecast Value (2036): USD 200.9 billion
- Forecast CAGR (2026 to 2036): 4.3%
- Dominant Trial Phase by Revenue: Phase 3 clinical trials due to large patient volumes and regulatory requirements
- Fastest Growing Trial Phase: Phase 4 trials supported by post approval surveillance demand
- Core Demand Drivers: Oncology pipelines, rare disease trials, biologics development, post marketing evidence needs
- Key Growth Regions: United States, China, Western Europe, India
- Leading Industry Participants: IQVIA, ICON plc, Charles River Laboratories, PPD, Syneos Health
Regional demand remains concentrated in North America and Europe, supported by mature regulatory systems, experienced investigators, and well developed research infrastructure. Asia Pacific continues to gain attention as sponsors access large patient populations, faster enrollment cycles, and operational efficiencies across China and India. Contract research organizations remain central to execution, supporting trial design, site management, data collection, and regulatory coordination.
Operational complexity remains a defining feature of the industry. Patient recruitment timelines, protocol design, and compliance requirements influence cost structures and execution strategies. Sponsors invest in digital platforms, decentralized trial models, and advanced analytics to improve study efficiency, patient participation, and data reliability across development programs.
Clinical Trials Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 131.8 billion |
| Market Forecast Value (2036) | USD 200.9 billion |
| Forecast CAGR (2026 to 2036) | 4.3% |
How Are the Segments Classified in the Clinical Trials Market?
The clinical trials market is structured by trial phase and region to reflect differences in study complexity, regulatory oversight, and geographic execution capacity. By phase, the industry includes Phase 1, Phase 2, Phase 3, and Phase 4 trials, each serving a distinct role from early safety validation to post approval evidence generation. Phase based segmentation mirrors the drug development lifecycle and associated investment intensity. Regionally, activity spans North America, Latin America, Western Europe, Eastern Europe, South Asia and Pacific, East Asia, and the Middle East and Africa. Mature regions lead in large scale late phase trials, while emerging regions contribute through faster recruitment, cost efficiency, and expanding clinical infrastructure. This structure highlights how sponsors balance scientific rigor, regulatory timelines, patient access, and operational scale across global programs.
Which Trial Phase Accounts for the Largest Share of Market Demand?
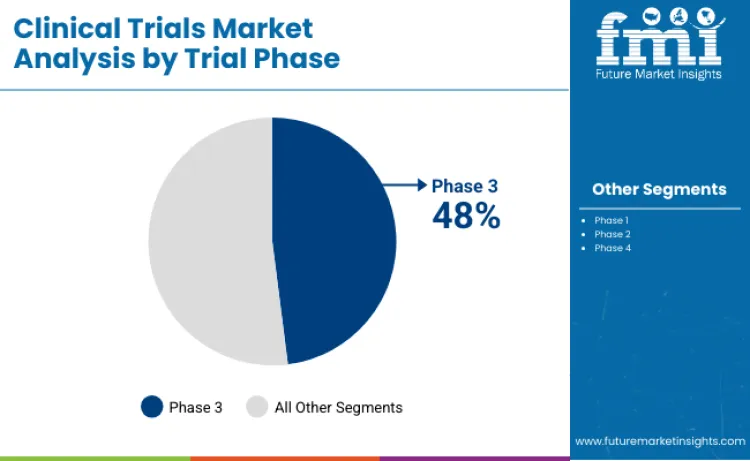
Phase 3 trials account for approximately 48% of total global clinical trials spending in 2026, making them the most resource intensive phase across the development lifecycle. These trials involve large patient cohorts, longer study durations, and rigorous endpoint validation to satisfy regulatory approval requirements. Budgets are highest at this stage due to multicenter coordination, complex protocol management, and extensive data monitoring obligations. Oncology, rare disease therapies, and advanced biologics continue to drive Phase 3 activity as regulators require strong comparative and safety evidence before market authorization. While Phase 1 and Phase 2 trials remain critical for early validation, Phase 3 dominance reflects its position as the decisive commercialization checkpoint, where sponsors focus heavily on execution reliability, regulatory confidence, and risk mitigation.
Which Phase Is Expanding at the Fastest Rate?
Phase 4 trials are growing at the fastest pace, advancing at around 5% CAGR from 2026 to 2036. Growth is driven by rising regulatory emphasis on post marketing surveillance, real world evidence generation, and long term safety validation. Payers increasingly require effectiveness data beyond controlled trial settings, pushing sponsors to invest in observational studies and digital follow up models. Lower per patient monitoring costs, wider cohort inclusion, and integration with electronic health records support scalable Phase 4 execution. This shift reflects how evidence generation now extends well beyond approval, influencing reimbursement, labeling updates, and lifecycle management strategies.
What Are the Key Dynamics Shaping the Clinical Trials Market?
Market momentum is shaped by expanding oncology and vaccine pipelines, growth in rare disease research, and the shift toward decentralized and patient centric trial models. Digital consent, remote monitoring, and wearable data collection reduce recruitment friction and improve retention. Collaboration between academic centers and contract research organizations expands access to diverse patient pools. Data privacy rules, cybersecurity requirements, and diversity mandates add complexity to trial design. The industry advances by balancing speed, compliance, data quality, and patient engagement across increasingly global and technology enabled trial ecosystems.
How Are Digital Tools and Decentralized Models Influencing Trial Execution?
Wearables, smart sensors, and remote data capture tools are reshaping how trials are conducted. Continuous monitoring provides richer clinical insight than episodic site visits, improving safety detection and endpoint accuracy. Machine learning supports adaptive designs, dropout prediction, and subgroup identification during ongoing studies. Decentralized models reduce site burden, expand geographic reach, and improve patient participation. These changes shorten timelines and control costs while maintaining data integrity. Adoption reflects sponsor demand for operational resilience, faster decision making, and flexible trial execution across therapeutic areas.
How Do Regulatory Requirements and Operational Constraints Affect Market Scalability?
Regulatory oversight strongly influences scalability across the clinical trials market. Requirements around patient safety, data integrity, diversity representation, and digital oversight increase compliance costs and extend study setup timelines. Regional differences in data protection laws and ethics review processes complicate global trial harmonization. Operational constraints include investigator shortages, site capacity limits, and rising cybersecurity investment needs. While decentralized models ease some pressures, long term scalability depends on standardized digital infrastructure, trained clinical staff, and regulatory alignment across regions. Sponsors scale by investing in compliant technology platforms, diversified site networks, and adaptive trial strategies that balance speed with oversight.
How Is the Clinical Trials Market Evolving Across Leading Countries?
The clinical trials market continues to expand as drug developers accelerate innovation across oncology rare diseases cell therapies and digital health solutions. Growth patterns differ widely by country depending on regulatory efficiency patient access data infrastructure and site capacity. Mature markets focus on protocol complexity real world evidence generation and post approval studies, while emerging hubs emphasize faster approvals cost efficiency and large patient pools. Decentralized trial models data security requirements and workforce availability increasingly shape country level performance. From established research centers such as the United States and Germany to fast scaling hubs including China and India, clinical trial activity reflects a balance between speed quality and compliance that defines competitive positioning across global development programs.
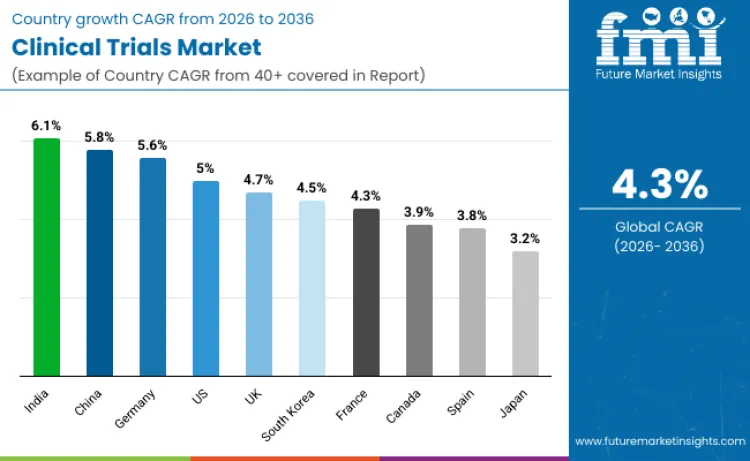
| Country | CAGR |
|---|---|
| United States | 5.0 |
| China | 5.8 |
| Japan | 3.2 |
| Germany | 5.6 |
| United Kingdom | 4.7 |
| France | 4.3 |
| Spain | 3.8 |
| Canada | 3.9 |
| South Korea | 4.5 |
| India | 6.1 |
Why Does the United States Remain the Anchor of the Clinical Trials Market?
The United States clinical trials market grows at a CAGR of 5% supported by its mature biotech ecosystem and strong dominance in oncology and rare disease research. Extensive real world data access and advanced site infrastructure sustain large scale Phase 3 and post approval studies. Regulatory acceptance of decentralized trial models supports tele visits home delivery of study drugs and digital consent adoption. Diversity focused enrollment planning increases investment in community based trial sites and data analytics. Cell and gene therapy pipelines continue to expand study volume despite pricing pressure in select indications. Strong funding for early phase research and advanced feasibility tools keeps the country positioned as the global reference point for complex and high value clinical development.
What Is Powering China’s Rapid Expansion in Clinical Trials Activity?
China records a CAGR of 5.8% driven by faster trial approvals and rising domestic innovation. Accelerated IND timelines allow quicker study activation across biologics oncology and cell therapies. Venture funding supports early phase research while decentralized trial reach now extends into secondary cities. Data compliance investment strengthens local cloud infrastructure and improves sponsor confidence. Mandatory population diversity improves dataset relevance for global filings. Intellectual property protection enhancements further attract multinational sponsors. Late stage demand remains strong across metabolic liver and oncology studies. Execution pressure persists around data export approvals and cybersecurity review processes. Even with operational hurdles China continues to narrow the innovation gap through scale speed and growing regulatory maturity.
How Is Japan Sustaining Clinical Trials Growth Despite Structural Constraints?
Japan advances at a CAGR of 3.2% shaped by quality focused execution and aging population driven research demand. Oncology and neurological trials benefit from large patient pools and strong reimbursement stability. National standardization of electronic source data and remote monitoring reduces site workload and improves study efficiency. Privacy rules limit overseas genomic data sharing which increases demand for domestic analytics capability. Workforce shortages at academic hospitals tighten investigator availability. Public funding now supports AI based protocol design to improve feasibility accuracy. Confirmatory studies and post marketing safety data remain central to payer decision making. Controlled cost management and selective decentralization remain essential to sustain competitive positioning.
Why Does Germany Attract High Value Clinical Trial Programs?
Germany grows at a CAGR of 5.6% supported by regulatory harmonization and strong CRO concentration. Centralized submission systems simplify multinational trial execution. Digital health reimbursement pathways expand post approval evidence budgets. Large urban hospital networks support efficient patient enrollment across cardiometabolic and neurodegenerative studies. Workforce shortages among research nurses increase pressure on timelines. Enhanced cybersecurity regulation raises infrastructure investment requirements. Public research grants and hospital collaborations continue to draw multinational sponsors seeking reliable data quality. Operational discipline and shared staffing models help protect delivery speed. Germany remains a preferred destination for sponsors prioritizing regulatory clarity robust datasets and predictable execution.
How Does the United Kingdom Compete in the Clinical Trials Landscape?
The United Kingdom records a CAGR of 4.7% supported by faster regulatory review and strong real world evidence access. Risk based assessment models shorten approval cycles for innovative therapies. Federated health records enable long term outcome tracking across large populations. Workforce shortages reduce site capacity though mobile services and virtual assessments stabilize enrollment flow. Updated data protection rules raise compliance thresholds. Tax incentives offset rising labor costs. Investment in decentralized infrastructure strengthens trial resilience. The country continues to attract studies focused on health economics and post launch evaluation. Speed of setup combined with data depth sustains its strategic relevance within Europe.
What Sustains France’s Role in Multicountry Clinical Trial Programs?
France grows at a CAGR of 4.3% supported by predictable regulatory timelines and expanding national health data access. Oncology research clusters attract international sponsors seeking specialized expertise. Labor rigidity and complex budget negotiations increase startup costs. Electronic consent pilots improve enrollment efficiency across major hospitals. Generous tax credits help offset staffing expenses. Decentralized monitoring reduces operational pressure. Public research collaboration strengthens feasibility reliability. Language localization and early engagement with authorities remain essential for timeline control. France continues to serve as a core contributor to pan European studies requiring strong clinical rigor and structured patient follow up.
Why Does Spain Deliver Enrollment Speed Advantages?
Spain advances at a CAGR of 3.8% driven by streamlined approvals and strong investigator engagement. Centralized ethics coordination shortens multicenter startup timelines. High smartphone adoption supports remote consent and monitoring models. Strong performance appears in metabolic pediatric and chronic disease studies. Investigator workload remains high placing strain on site capacity. Government funding programs target research nurse shortages. Competitive site costs preserve sponsor interest. Data governance reforms enable broader real world evidence use with consent safeguards. Spain remains attractive for sponsors prioritizing enrollment speed and cost efficiency while maintaining regulatory discipline.
How Is Canada Strengthening Its Position in Global Clinical Research?
Canada records a CAGR of 3.9% supported by flexible licensing pathways and public co funding. Rolling submissions reduce administrative burden for innovative therapies. National funding offsets trial overhead costs. Indigenous engagement frameworks add dataset depth while extending feasibility planning timelines. New AI governance rules raise compliance requirements for digital platforms. Strong cold chain logistics attract cross border studies. Favorable currency dynamics increase sponsor interest. Expansion of trusted research environments remains a priority. Workforce capacity development will determine scalability. Canada continues to function as a strategic extension market for North American and global clinical programs.
What Drives South Korea’s Competitive Clinical Trials Environment?
South Korea grows at a CAGR of 4.5% supported by universal electronic medical records and lower operational costs. Hybrid monitoring models reduce site visits and improve efficiency. Digital therapeutic trials benefit from regulatory sandbox programs. Data export restrictions increase reliance on domestic analytics partnerships. Tax incentives and rare disease fast tracks sustain pipeline activity. Approval timelines remain short though investigator overcommitment presents scheduling risk. Advanced connectivity infrastructure supports remote oversight. Local language digital tools remain critical for patient engagement. South Korea continues to attract multinational trials seeking speed cost control and high data integrity.
Why Is India Emerging as a High Growth Clinical Trials Hub?
India leads expansion with a CAGR of 6.1% driven by regulatory reform cost advantage and vast patient availability. Faster approvals and mandatory public trial registration improve transparency. National electronic consent systems reduce onboarding time. Oncology diabetes and chronic disease trials benefit from rapid recruitment. Data localization requirements push investment into domestic cloud infrastructure. Ethics committee capacity varies across regions prompting national training initiatives. Centralized review and telemedicine expansion support broader site participation. Multilingual digital tools remain critical for quality execution. India’s ability to balance scale speed and compliance will define its long term leadership potential.
How Are Major Players Competing in the Clinical Trials Market?
The clinical trials market is shaped by a small group of large contract research organizations that handle a significant share of global study activity. These companies stand out because of their global reach therapeutic depth and ability to manage complex trials across multiple countries. Drug developers increasingly rely on partners that can shorten timelines improve patient enrollment and maintain strict regulatory standards throughout the trial lifecycle.
IQVIA holds the strongest position in the industry with a share in the mid teens. Its strength lies in combining clinical execution with deep data assets that support better site selection patient matching and trial oversight. This breadth makes it a preferred partner for large pharmaceutical companies running late stage and global programs. ICON plc follows with a solid double digit presence supported by its expanded scale after integrating PRA Health Sciences. The company has built a strong reputation in hybrid and decentralized trials which help reach broader patient groups particularly in oncology and vaccines.
Charles River Laboratories maintains a meaningful role through its close link between early research and clinical development. Many biotech firms value its ability to support projects from preclinical stages into first human studies. Pharmaceutical Product Development under Thermo Fisher Scientific benefits from access to extensive laboratory and manufacturing infrastructure. This positions it well for complex programs such as biosimilars and advanced therapies.
Syneos Health approaches competition through its combined focus on clinical development and commercialization planning. This model appeals to sponsors aiming for faster market entry. Smaller providers continue to play an important role by supporting rare disease and regional studies where flexibility and local knowledge remain critical.
Key Players
- IQVIA
- ICON plc
- Charles River Laboratories
- Pharmaceutical Product Development (PPD)
- Syneos Health
Scope of the Report
| Attribute | Details |
|---|---|
| Market Value (2026) | USD 131.8 billion |
| Market Forecast Value (2036) | USD 200.9 billion |
| Forecast CAGR (2026 to 2036) | 4.3% |
| Report Parameter | USD billion |
| By Phase | Phase 1, Phase 2, Phase 3, and Phase 4 clinical trials across drug and biologics development |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, South Asia and Pacific, East Asia, Middle East and Africa |
| Countries Covered | United States, China, Japan, Germany, United Kingdom, France, Spain, Canada, South Korea, India |
| Key Companies Profiled | Pharmaceutical Product Development Inc., ICON plc, Charles River Laboratories International Inc., IQVIA, Syneos Health, SGS SA, Chiltern International Ltd., Clinipace |
| Additional Attributes | Analysis includes dollar sales by trial phase and region, sponsor and CRO participation trends, country level clinical activity assessment, outsourcing patterns, regulatory environment impact, pipeline driven trial volume shifts, operational complexity trends, and competitive positioning across global clinical research service providers |
Clinical Trials Market by Segment
By Trial Phase:
- Phase 1
- Phase 2
- Phase 3
- Phase 4
By Trial Focus:
- Early stage safety and dosage evaluation
- Efficacy and proof of concept studies
- Large scale confirmatory trials
- Post marketing surveillance and real world evidence
By Therapeutic Area:
- Oncology
- Rare diseases
- Cardiovascular
- Neurology
- Infectious diseases
- Metabolic disorders
- Vaccines and immunology
- Other therapeutic areas
By Study Design:
- Interventional trials
- Observational studies
- Randomized controlled trials
- Decentralized and hybrid trials
By Region:
- North America
- United States
- Canada
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Western Europe
- United Kingdom
- Germany
- France
- Spain
- Italy
- Rest of Western Europe
- Eastern Europe
- Poland
- Czech Republic
- Hungary
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Rest of South Asia and Pacific
- Middle East and Africa
- GCC
- South Africa
- Rest of Middle East and Africa
Frequently Asked Questions
How big is the clinical trials market in 2026?
The clinical trials market is valued at USD 131.8 billion in 2026.
What will be the size of the clinical trials market in 2036?
The industry is projected to reach USD 200.9 billion by 2036.
How fast is the clinical trials market expected to grow between 2026 and 2036?
The clinical trials market is expected to grow at a CAGR of 4.3% from 2026 to 2036.
Which trial phases account for the majority of clinical research activity?
Phase 3 trials account for the largest share of global clinical research activity.
Which regions dominate global clinical trials demand?
North America and Europe dominate global demand due to strong research infrastructure and sponsor presence.
Who are the key participants shaping the clinical trials market?
Leading contract research organizations such as IQVIA, ICON plc, PPD, and Syneos Health drive market activity.
Table of Content
- Executive Summary
- Global Clinical Trials Industry Snapshot
- Key Demand Trends and Growth Drivers
- Trial Phase Performance Overview
- Regional Demand Highlights
- Competitive Landscape Snapshot
- Market Overview
- Clinical Trials Industry Definition and Scope
- Market Size Analysis 2026
- Market Forecast Outlook 2026 to 2036
- Value Chain Overview
- Industry Stakeholder Ecosystem
- Market Background
- Evolution of Global Clinical Research
- Drug Development Lifecycle and Trial Phases
- Role of CROs and Sponsors
- Regulatory Framework Overview
- Technology Adoption in Clinical Trials
- USA Clinical Trials Market Analysis
- USA Market Size and Forecast 2026 to 2036
- USA Market Growth Drivers
- USA Market Restraints and Challenges
- USA Trial Phase Wise Analysis
- USA Therapeutic Area Wise Analysis
- USA Study Design Wise Analysis
- USA Competitive Landscape Overview
- Market Pricing Analysis
- Trial Cost Structure by Phase
- Pricing Variation by Therapeutic Area
- Regional Cost Comparison
- Impact of Regulatory Compliance on Pricing
- Market Structure Analysis
- Trial Phase Based Market Structure
- Study Design Based Market Structure
- Therapeutic Area Based Market Structure
- Regional Market Structure
- Competition Analysis
- Global Competitive Landscape Overview
- Market Share Analysis of Leading CROs
- Company Positioning and Strategy Mapping
- Mergers Acquisitions and Partnerships
- Technology and Service Differentiation
- Assumptions and Acronyms Used
- Key Market Assumptions
- Research Limitations
- List of Acronyms and Definitions
- Research Methodology
- Data Collection Approach
- Primary Research Methodology
- Secondary Research Sources
- Market Estimation Techniques
- Forecast Modeling Approach
- Data Validation and Triangulation
List of Tables
- Table 1: Global Clinical Trials Industry Value Share by Region 2026
- Table 2: Global Clinical Trials Industry Value Forecast by Region 2026 to 2036
- Table 3: Global Clinical Trials Industry CAGR by Region 2026 to 2036
- Table 4: USA Clinical Trials Industry Size 2026 to 2036
- Table 5: USA Clinical Trials Industry Value Share by Trial Phase 2026
- Table 6: USA Clinical Trials Industry Value Forecast by Trial Phase 2026 to 2036
- Table 7: USA Clinical Trials Industry Value Share by Trial Focus 2026
- Table 8: USA Clinical Trials Industry Value Forecast by Trial Focus 2026 to 2036
- Table 9: USA Clinical Trials Industry Value Share by Therapeutic Area 2026
- Table 10: USA Clinical Trials Industry Value Forecast by Therapeutic Area 2026 to 2036
- Table 11: North America Clinical Trials Industry Size 2026 to 2036
- Table 12: Europe Clinical Trials Industry Size 2026 to 2036
- Table 13: East Asia Clinical Trials Industry Size 2026 to 2036
- Table 14: South Asia and Pacific Clinical Trials Industry Size 2026 to 2036
- Table 15: Latin America Clinical Trials Industry Size 2026 to 2036
- Table 16: Middle East and Africa Clinical Trials Industry Size 2026 to 2036
- Table 17: United States Clinical Trials Industry CAGR 2026 to 2036
- Table 18: China Clinical Trials Industry CAGR 2026 to 2036
- Table 19: India Clinical Trials Industry CAGR 2026 to 2036
- Table 20: Germany Clinical Trials Industry CAGR 2026 to 2036
- Table 21: United Kingdom Clinical Trials Industry CAGR 2026 to 2036
- Table 22: Japan Clinical Trials Industry CAGR 2026 to 2036
- Table 23: South Korea Clinical Trials Industry CAGR 2026 to 2036
- Table 24: Phase Wise Cost Structure in Clinical Trials Execution
- Table 25: Sponsor Versus CRO Participation Share
- Table 26: Trial Outsourcing Trends by Phase
- Table 27: Competitive Market Share of Leading CROs 2026
- Table 28: Service Portfolio Mapping of Key Clinical Research Providers
- Table 29: Decentralized Trial Adoption by Region
- Table 30: Regulatory Approval Timelines by Country
- Table 31: Ethics Committee Review Duration Comparison
- Table 32: Investment and R and D Spending Trends in Clinical Research
- Table 33: Strategic Partnerships and Mergers in Clinical Trials Services
- Table 34: Key Assumptions Used for Market Estimation
- Table 35: List of Acronyms and Abbreviations
List of Figures
- Figure 1: Global Clinical Trials Industry Value Share by Region 2026
- Figure 2: Global Clinical Trials Industry Growth Rate by Region 2026 to 2036
- Figure 3: Global Clinical Trials Industry Value Forecast 2026 to 2036
- Figure 4: USA Clinical Trials Industry Value Trend 2026 to 2036
- Figure 5: USA Clinical Trials Industry Value Share by Trial Phase 2026
- Figure 6: USA Clinical Trials Industry Value Share by Therapeutic Area 2026
- Figure 7: Phase Wise Distribution of Clinical Trial Spending
- Figure 8: Trial Volume Distribution by Study Design
- Figure 9: North America Clinical Trials Industry Growth Outlook
- Figure 10: Europe Clinical Trials Industry Growth Outlook
- Figure 11: East Asia Clinical Trials Industry Growth Outlook
- Figure 12: South Asia and Pacific Clinical Trials Industry Growth Outlook
- Figure 13: Latin America Clinical Trials Industry Growth Outlook
- Figure 14: Middle East and Africa Clinical Trials Industry Growth Outlook
- Figure 15: Country Level CAGR Comparison 2026 to 2036
- Figure 16: Phase 3 Trial Dominance Across Development Pipelines
- Figure 17: Phase 4 Trial Growth Trend and Post Approval Focus
- Figure 18: Cost Drivers in Global Clinical Trial Execution
- Figure 19: Impact of Regulatory Requirements on Trial Timelines
- Figure 20: Competitive Landscape Mapping of Leading CROs
- Figure 21: Market Share Distribution of Key Clinical Research Providers
- Figure 22: Service Capability Comparison of Major CROs
- Figure 23: Pipeline Driven Trial Activity Distribution
- Figure 24: Adoption Trend of Decentralized and Hybrid Trials
- Figure 25: Digital Tools Integration in Clinical Trial Management
- Figure 26: Regulatory Approval Flow for Clinical Studies
- Figure 27: Patient Recruitment and Retention Journey
- Figure 28: Site Selection and Activation Timeline
- Figure 29: Data Management and Monitoring Framework
- Figure 30: Long Term Evidence Generation Lifecycle
- Figure 31: Key Growth Drivers Impact Assessment
- Figure 32: Industry Challenges and Execution Risks
- Figure 33: Opportunity Mapping Across Trial Phases
- Figure 34: Future Clinical Research Execution Landscape
- Figure 35: Summary of Key Industry Insights
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

