Digital Pathology Market
The Digital Pathology Market is segmented by Product Type (Equipments, Softwares, Services), Application (Clinical Application and Research Application), End User (Hospitals, Diagnostic Laboratories, Pharmaceutical & Biotechnology Companies, Forensic Laboratories, Research Institutes, Contract Research Organizations (CROs), Clinics) and Region. Forecast for 2026 to 2036.
Digital Pathology Market size, market forecast and outlook By FMI
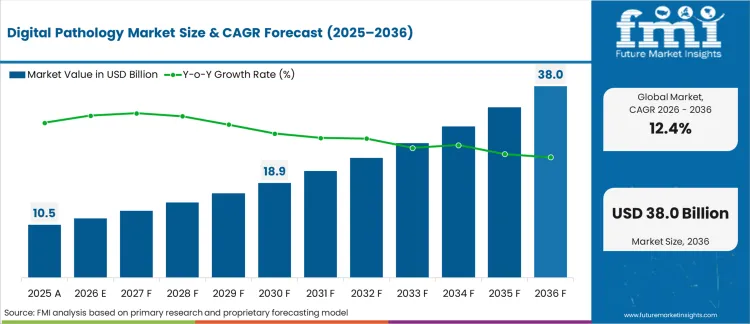
The digital pathology market is expected to expand from USD 11,817.9 million in 2026 to USD 38,036.8 million by 2036. The market is likely to register a 12.4% CAGR during the forecast period. Equipment is anticipated to lead product type with a 57.1% share in 2026. Hospitals are projected to account for 30.9% of end-user demand in 2026.
Summary of the Digital Pathology Market
- Demand and Growth Drivers
- Hospital labs are digitizing slide review to reduce physical handling and delays.
- Diagnostic networks are increasing scan capacity to manage higher case volumes.
- Pharma and CRO users are adopting digital workflows for standardized research review.
- Product and Segment View
- Equipment leads because scanners form the base of digital pathology workflows.
- Services gain importance as labs need validation support and workflow integration.
- Clinical pathology remains central as digital review supports faster case sign-out.
- Geography and Competitive Outlook
- Asia Pacific is likely to lead growth due to lab network expansion.
- North America shows steady demand through hospital digitization and reference labs.
- Europe gains from tender-based modernization and multi-site pathology networks.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant at FMI, suggests “The digital pathology market is moving toward scanner reliability and workflow-linked adoption. Suppliers with strong service coverage and LIS integration support are likely to gain advantage as hospitals scale digital slide review across daily case volumes.”
- Digital Pathology Market Value Analysis
- The digital pathology market is moving from slide digitization into connected lab workflow systems.
- Demand is likely to grow as hospitals prioritize faster review and traceable diagnosis.
- Equipment uptake is improving through high-throughput scanners and image management platforms.
- Investment is set to favor validation services and secure pathology data infrastructure.
Digital Pathology Market definition
The market involves systems and services for digitizing and managing pathology slides. The market involves whole-slide imaging scanners and equipment, software for managing and viewing images, and services for installation, workflow configuration, validation support, and maintenance. The customers for the market involve hospitals, diagnostic labs, research institutes, pharmaceutical and biotech companies, and CROs that employ digital pathology slides for case review, consultation, archiving, and analysis. The market is driven by workload volume, turnaround time requirements, and readiness for infrastructure. The revenue for the market is generated through equipment sales, software licensing, and service contracts.
Digital Pathology Market inclusions
The report includes global and regional market sizes and a 10-year forecast for 2026 to 2036. It provides segment-level sizing by product type, application, and end user, with country-level CAGR comparisons across major markets. Competitive positioning is assessed for key suppliers, alongside analysis of procurement drivers such as interoperability, validation effort, installed-base support, and service intensity across hospital and laboratory settings, based on FMI’s report.
Digital Pathology Market exclusions
The scope excludes conventional optical microscopes without digital scanning capability, general hospital IT systems not used for pathology imaging, and stand-alone AI software sold outside digital pathology workflows. It also omits revenue from pathology testing services, diagnosis fees, and lab consumables such as slides, stains, and reagents. Imaging modalities outside slide-based pathology are excluded. The focus remains on digital pathology platforms and enabling software and services aligned to the stated segments.
Digital Pathology Market research methodology
- Primary research: Interviews were held with pathology lab directors, hospital IT managers, procurement managers, CRO workflow owners, and supplier commercial teams to confirm adoption drivers and implementation barriers.
- Desk research: Public data from regulators, standards organizations, health system publications, annual company reports, investor presentations, and published literature was analyzed to compare validation and workflow needs.
- Market sizing and forecasting: A hybrid approach was employed, combining equipment installed base and replacement logic with software and services revenue build-up, and then combining with segment shares based on application and end-user type.
- Data validation and update cycle: Results were validated by internal consistency screening for segment totals and country growth rates, with variance checks prior to finalization.
Segmental Analysis
Digital Pathology Market Analysis by product type
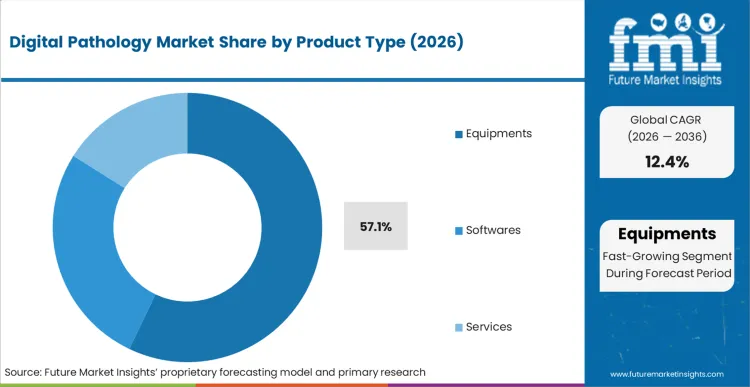
Based on FMI’s report, equipment is estimated to hold 57.1% share in 2026, followed by services at 28.9% share. Equipment leadership is supported by the need for scanner installations as the first step in digitization, which creates upfront capital purchasing before broader software and workflow expansion. Services hold the second position as implementations are being executed at enterprise scale, with validation, integration, training, and ongoing support being purchased to keep scan-to-sign-out performance stable.
- Scanner throughput: Roche’s FDA-cleared Digital Pathology Dx expansion shows this scanner-led entry clearly, the VENTANA DP 600 adds 240-slide capacity and high-resolution, consistent image quality for primary diagnosis. That combination pushes sites to prioritize capital spend on scanners before scaling services such as integration, validation, training, and support.
- Compact scanning: Leica launched the Aperio CS5 as a compact, low to medium volume whole slide imaging system delivering exceptional image quality, supporting 1x3 and 2x3 inch slides, and outputting SVS or DICOM formats with automatic or manual scanning for workflow integration.
- Hardware primacy: Equipment leads because slide scanners are the required first step, delivering clinical-grade whole slide images with high throughput, consistent image quality, and standard outputs (SVS, DICOM). Without this hardware, software, AI analysis, and workflows cannot run.
Digital Pathology Market Analysis by end user
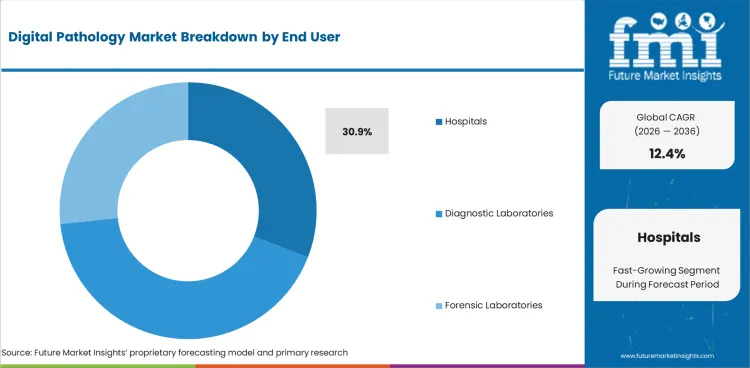
Hospitals account for 30.9% share in 2026, based on FMI’s report, followed by diagnostic laboratories at 18.7% share. Hospital dominance is supported by integrated care pathways where pathology sign-out affects treatment decisions, which increases willingness to invest in digitized review and remote consultation. Diagnostic laboratories follow due to high sample throughput and centralized operations, where scanning and workflow standardization support productivity and quality tracking across large case volumes.
- Networked care: National platforms like NPIC are being built to support clinical sign-out at scale, enabling faster second opinions, improved turnaround time, and a national worklist to distribute cases across up to 30 NHS hospitals through a secure central whole slide image repository.
- Care decisions: Pathology sign-out directly drives treatment choices and MDT discussions, so they invest first in scanners and secure platforms to speed diagnosis, enable second opinions, and support remote consultation.
- Network scale: Health systems run multi-site networks. Digital pathology enables shared worklists, workload balancing across specialties, and centralized image repositories, improving turnaround time and workforce efficiency across facilities.
Digital Pathology Market Drivers, Restraints, and Opportunities
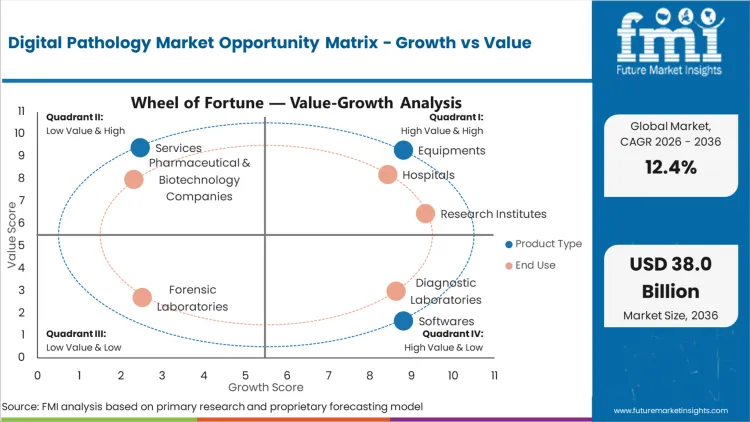
Future Market Insights analysis that historical patterns point at a transitioning diagnostics workflow category where digitization is being adopted to reduce physical slide handling, enable remote sign-out, and standardize review quality across multi-site pathology networks. Adoption is being driven by workload pressure, turnaround-time targets, and the need for traceable case review under accredited laboratory operations, as per FMI.
While rollout pace is being restrained by LIS integration effort, validation workload, and IT storage planning that can stretch implementation across budget cycles, value expansion is being supported by enterprise-scale deployments that bundle scanners, software, and ongoing services into long-cycle operating models. Based on FMI’s report, demand is increasingly being filtered by uptime reliability, workflow integration, and support responsiveness, since downtime and connectivity failures directly disrupt daily case throughput.
- Compliance-led digitization: Growth is shifting toward workflows with documented traceability, controlled access, and audit-ready review practices aligned to CLIA and CAP requirements.
- Integration drag restraint: LIS connectivity, storage architecture, and validation requirements slow go-live timelines, delaying full utilization after installation.
- Network deployment mechanism: Adoption is being accelerated where hospital systems and lab chains use remote review to balance workload and improve turnaround consistency.
Regional Analysis
.webp)
Based on the regional analysis, digital pathology market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. Regional performance is assessed using country-level demand signals linked to lab digitisation pace, scanner deployment, image management adoption, and enterprise procurement in hospitals and diagnostic laboratories, as per FMI. The full report also offers market attractiveness analysis based on regional trends.
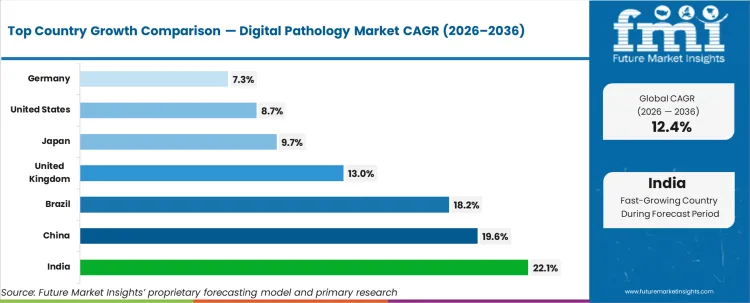
| Country | CAGR |
|---|---|
| USA | 8.7% |
| India | 22.1% |
| Germany | 7.3% |
| UK | 13.0% |
| Japan | 9.7% |
| China | 19.6% |
| Brazil | 18.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Digital Pathology Market Analysis
North America is characterised by enterprise-scale lab digitisation, where adoption is anchored in multi-site hospital systems and large reference laboratories seeking standardised workflows and remote read capability. Key players in North America include Danaher Corporation through Leica Biosystems, with strong scanner and workflow access, F. Hoffmann-La Roche AG with broad pathology informatics placement, and Huron Technologies International, Inc. with focused digital slide scanning footprint. FMI analysts note that demand is being reinforced by clinical pathology use cases and research throughput needs.
- United States: The demand for digital pathology in the United States is expected to grow at 8.7% CAGR until 2036. The demand is driven by the constant installation of scanners in hospitals and diagnostic labs, with growth linked to the volume of slides and workflow standardization.
FMI’s report includes a detailed analysis of the growth in the North American region, along with a country-wise assessment that includes the United States. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the North America region.
Asia Pacific Digital Pathology Market Analysis
Asia Pacific remains the fastest scaling adoption region, supported by rapid laboratory capacity expansion, higher oncology testing demand, and rising spend on digitised pathology workflows in major metros. Olympus Corporation and Nikon Corporation hold strong relevance through imaging and optics capability aligned to pathology workflows, while Hamamatsu Photonics K.K. has an established presence in whole slide imaging systems. F. Hoffmann-La Roche AG and Danaher Corporation sustain placements through regional distribution and large-account procurement. Future Market Insights analysis links growth to equipment uptake and increasing use in drug development and molecular diagnostics pathways.
- China: The demand is expected to grow at a CAGR of 19.6% until 2036, driven by rapid lab installations and hospital digitization initiatives. The purchase of slide scanners is being focused on tier-1 city health infrastructure and research institutions that are expanding their slide scanning capacity.
- India: The demand is expected to grow at a CAGR of 22.1% until 2036, driven by the development of organized diagnostic networks and the adoption of slide scanners in tertiary care hospitals. The demand is being focused on new scanner sales and service-enabled slide scanner adoption to stabilize high-volume lab operations.
- Japan: The market is expected to register a CAGR of 9.7% until 2036, as the modernization of hospital pathology and research institutions is a steady process. The adoption of slide scanners is being driven by integration and validation requirements, and buying decisions remain anchored in established and reliable workflows.
The full report analyzes the digital pathology market across East and South Asia from 2021-2036, covering pricing, trends, and growth drivers in China, Japan, South Korea, India, Indonesia, Malaysia, and Thailand. The assessment highlights trends that dictate regional demand and procurement behaviour.
Latin America Digital Pathology Market Analysis
Latin America is expanding through mixed public and private laboratory investment, where adoption is influenced by import dependence, distributor service coverage, and prioritisation of high-burden specialties such as oncology. Danaher Corporation and F. Hoffmann-La Roche AG maintain visibility through distributor-backed placements, while Koninklijke Philips N.V. supports deployments tied to imaging informatics and workflow integration. FMI opines that demand is being driven by private diagnostic laboratories and larger hospital groups that can justify enterprise platforms
- Brazil: Demand for digital pathology in Brazil is projected to rise at 18.2% CAGR through 2036. Growth is supported by private diagnostic laboratory expansion and rising hospital digitisation activity in major cities, with procurement concentrated in higher throughput sites that can sustain scanner utilisation.
The report consists of a detailed analysis for the market in Brazil, Mexico, Chile, Argentina, Peru and Rest of Latin America. Readers can find detailed information about several factors, such as the pricing analysis and regional trends, which are impacting growth in the Latin America region.
Europe Digital Pathology Market Analysis
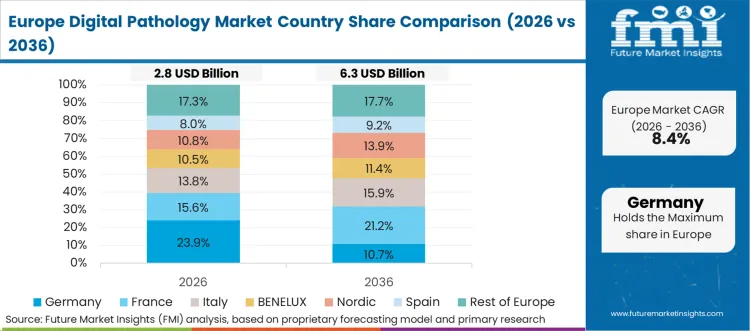
Europe is characterised by tender-led adoption and structured lab modernisation, where procurement is shaped by enterprise documentation needs and multi-site standardisation across hospital groups and diagnostic laboratories. Carl Zeiss AG and Koninklijke Philips N.V. remain important participants through imaging and workflow capabilities, while F. Hoffmann-La Roche AG sustains a strong footprint across pathology informatics and platform deployments. Danaher Corporation maintains placements through Leica Biosystems portfolios across hospital and research settings. Based on FMI’s report, demand is being supported by clinical pathology use and drug development workflows.
- Germany: Demand for digital pathology in Germany is projected to rise at 7.3% CAGR through 2036. Growth is supported by steady lab digitisation programmes and uptake in university hospitals and diagnostic laboratories, with demand weighted toward equipment replacement and workflow standardisation.
- United Kingdom: Demand for digital pathology in the United Kingdom is projected to rise at 13.0% CAGR through 2036. Growth is being driven by hospital network digitisation and diagnostic laboratory modernisation, with adoption supported by system-wide workflow consolidation and rising use in research-linked pathology.
FMI’s analysis of digital pathology market in Europe consists of country-wise assessment that includes the Germany, Italy, France, UK, Spain, Russia, Nordic, Benelux and Rest of Europe. Readers can know various regulations and latest trends in the regional market.
Competitive aligners for market players
Market structure is still fragmented, but effective competition is driven by a short list of suppliers who have the ability to provide certified scanner and software performance, support regulated clinical workflows, and maintain enterprise installations within multi-site hospital systems and large laboratories. Competition is driven less by headline product attributes and more by the pain of adoption within IT, quality, and regulatory domains. LIS and image management layer interoperability, cybersecurity posture, and service responsiveness drive short lists, while total cost of ownership drives final awards within large contracts, as reported by FMI.
Firms with structural advantages often leverage hardware portfolios and software ecosystems with installed base reach that decrease the frequency of switching. Suppliers with a footprint spanning scanners, image management, and workflow software can facilitate standardization across departments, which decreases training costs and increases serviceability. Scale-based firms leverage global service infrastructure, enhanced regulatory and quality documentation, and larger partner ecosystems, which assists in facilitating phased adoption from research use to clinical use environments. Specialized software vendors can effectively compete when installations are kept modular, but are challenged by scanner compatibility and hospital IT gating, which may impede conversion in more conservative healthcare environments, as reported in FMI’s report.
Buyer behavior solidifies the impact of leverage. Many hospitals and labs simultaneously prequalify multiple vendors, engage in side-by-side validations, and stage procurement to prevent vendor lock-in with respect to a single platform. Vendor contracts include service level agreements related to availability, help desk response times, and software migration assistance, ensuring that vendors are held accountable after delivery. Pharmaceutical and biotech customers are more project-oriented and have the ability to switch modules between studies, making it simpler to switch platforms for research-related workloads. This buying behavior compresses the pricing power of core equipment and standard software modules, while strategic premiums are maintained largely where clinical implementation, workflow, and regulatory considerations increase switching costs, Future Market Insights analysis.
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
- In January 2026, Leica Biosystems, a Danaher company and global leader in anatomic and digital pathology solutions, announces the launch of the Aperio CS5 scanner, a compact device designed to support digital transformation in research pathology.
- In January 2026, Agilent Technologies Inc. announced the launch of the Agilent S540MD Slide Scanner System, a whole slide imaging (WSI) digital scanner, available for sale in key European markets. The launch reflects Agilent’s continued investment in digital pathology, expanding its portfolio to meet the growing demand for high-throughput solutions in clinical laboratories.
- In June 2025, Barco launched SlideRightQA, a vendor-neutral, AI-powered digital pathology QA tool that runs 24/7 to detect scan issues such as out-of-focus areas and missed tissue, helping labs triage faster and saving reported 11.5–24 hours of scan-tech time per day.
Key players in Digital pathology market
- Danaher Corporation
- F. Hoffmann-La Roche AG
- Huron Technologies International, Inc.
- Koninklijke Philips N.V.
- Olympus Corporation
- Hamamatsu Photonics K.K.
- Carl Zeiss AG
- Hologic, Inc.
- Nikon Corporation
- PerkinElmer, Inc.
Scope of the report
| Metric | Value |
|---|---|
| Quantitative units | USD 11,817.9 million (2026) to USD 38,036.8 million (2036), at a CAGR of 12.4% |
| Market definition | The digital pathology market comprises global production and trade of whole slide imaging systems, related software, and supporting services used to digitise glass slides for clinical review, consultation, archiving, and research workflows, where adoption is shaped by laboratory throughput needs, validation requirements, and integration with LIS and image management systems. |
| Product segmentation | Equipments, Softwares, Services |
| Application coverage | Clinical Pathology, Molecular Diagnostics, Basic & Applied Research, Drug Development, Others |
| End user coverage | Hospitals, Diagnostic Laboratories, Pharmaceutical & Biotechnology Companies, Forensic Laboratories, Research Institutes, Contract Research Organizations (CROs), Clinics |
| Regions covered | North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe, and Middle East & Africa. |
| Countries covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia and 40+ countries |
| Key companies profiled | Danaher Corporation, F. Hoffmann-La Roche AG, Huron Technologies International, Inc., Koninklijke Philips N.V., Olympus Corporation, Hamamatsu Photonics K.K., Carl Zeiss AG, Hologic, Inc., Nikon Corporation, PerkinElmer, Inc. |
| Forecast period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with pathology laboratories, hospital stakeholders, and solution providers, supported by installed base triangulation and workflow adoption benchmarking, as per FMI. |
Digital pathology market analysis by segments
Product type
- Equipment
- Whole Slide Scanners
- Brightfield Slide Scanners
- Fluorescence Slide Scanners
- Combination Slide Scanners
- Digital Microscopes
- Confocal Microscope
- Brightfield Microscope
- Fluroscent Microscope
- Others
- Tissue Microarray Consumables
- Whole Slide Scanners
Software
- Image Viewing and Analysis Software
- On-premise
- Cloud-based
- Digital Pathology Information Systems
- On-premise
- Cloud-based
- Services
- Installation and Integration Services
- Consulting Services
- Maintenance and Validation Services
Application
- Clinical Applications
- Cancer Diagnosis
- Neurological Disorder Diagnosis
- Cardiovascular Disease Diagnosis
- Infectious Disease Diagnosis
- Others
- Research Applications
- Drug Discovery and Development
- Biomarker Discovery and Validation
- Molecular Pathology Research
- Tissue Engineering
- Others
End user
- Hospitals
- Diagnostic Laboratories
- Pharmaceutical & Biotechnology Companies
- Forensic Laboratories
- Research Institutes
- Contract Research Organizations (CROs)
- Clinics
Region
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- East Asia
- China
- Japan
- South Korea
- South Asia & Pacific
- India
- ASEAN Countries
- Australia & New Zealand
- Rest of South Asia
- Western Europe
- Germany
- UK
- France
- Italy
- Spain
- BENELUX
- Nordic Countries
- Rest of Western Europe
- Eastern Europe
- Russia
- Hungary
- Poland
- Rest of Eastern Europe
- Middle East & Africa
- Saudi Arabia
- Türkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- [1] 2025, Roche’s momentum in digital pathology continues with FDA clearance on its high-volume slide scanner.
- [2] January 2026, Leica Biosystems Expands Research Digital Pathology Portfolio with Launch of the compact Aperio CS5 Scanner.
- [3] National Pathology Imaging Co-operative, Clinical Digital Pathology.
- [4] 2026, Agilent Accelerates Digital Pathology with the Launch of the Agilent S540MD Slide Scanner System
- [5] 2025, Cut costs, not corners with SlideRightQA, our brand-new quality assurance solution for digital pathology.
This report addresses
- Market intelligence to enable structured strategic decision making across pathology digitisation programs and laboratory capacity planning
- Market size estimation and 10-year revenue forecasts from 2026 to 2036 for digital pathology demand
- Growth opportunity mapping across product type, application, and end user purchasing pools
- Segment and regional revenue forecasts aligned to the stated taxonomy and covered geographies
- Competition strategy assessment including portfolio positioning, integration readiness, and channel coverage benchmarking
- Product and compliance tracking focused on validation expectations, documentation requirements, and laboratory quality system alignment
- Regulatory impact analysis covering device performance expectations and laboratory use requirements affecting adoption planning
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is the demand for Digital pathology in the global market in 2026?
Demand for Digital pathology in the global market is estimated to be valued at USD 11,817.9 million in 2026, as per FMI.
What will be the market size of Digital pathology in the global market by 2036?
Market size for Digital pathology is projected to reach USD 38,036.8 million by 2036.
What is the expected demand growth for Digital pathology in the global market between 2026 and 2036?
Demand for Digital pathology in the global market is expected to grow at a CAGR of 12.4% between 2026 and 2036.
Which product type is poised to lead global sales by 2026?
Equipments are expected to be the dominant product type, capturing 57.1% share in 2026.
Which application is expected to account for the largest share in 2026?
Clinical Pathology is expected to hold the highest share at 25.1% in 2026.
How significant is the role of hospitals in driving Digital pathology adoption in 2026?
Hospitals are projected to hold 30.9% share of end user demand in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Thermal Wristbands
- Laser Wristbands
- RFID Wristbands
- Thermal Wristbands
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Printing Method
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Printing Method, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Printing Method, 2026 to 2036
- Thermal Printing
- Laser Printing
- Inkjet Printing
- Thermal Printing
- Y to o to Y Growth Trend Analysis By Printing Method, 2021 to 2025
- Absolute $ Opportunity Analysis By Printing Method, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Printing Method
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Printing Method
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Printing Method
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Printing Method
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Printing Method
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Printing Method
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Printing Method
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Printing Method
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Printing Method
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Printing Method
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Printing Method
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Printing Method
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Printing Method
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Printing Method
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Printing Method
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Printing Method
- Competition Analysis
- Competition Deep Dive
- Brady Worldwide, Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Ahearn & Soper
- Identiplus
- Nang Medical Limited
- Typenex Medical, LLC.
- Capsa Healthcare
- SATO Europe GmbH
- Brady Worldwide, Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Printing Method, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Printing Method, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Printing Method, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Printing Method, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Printing Method, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Printing Method, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Printing Method, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Printing Method, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Printing Method, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Printing Method, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Printing Method
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by Printing Method, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Printing Method, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Printing Method
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Printing Method, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Printing Method, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Printing Method
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Printing Method, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Printing Method, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Printing Method
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Printing Method, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Printing Method, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Printing Method
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Printing Method, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Printing Method, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Printing Method
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Printing Method, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Printing Method, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Printing Method
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Printing Method, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Printing Method, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Printing Method
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

