Digital stethoscope market value is projected to reach USD 347.2 million in 2026 and expand at a 6.2% CAGR through 2036, taking total revenue to USD 636.8 million by the end of the period. Growth is being supported by higher telemedicine utilization, rising remote patient monitoring adoption, and increasing demand for connected diagnostic tools across hospitals, clinics, and home care settings. Product demand is shifting toward devices that offer stronger acoustic processing, consistent signal quality, and seamless data capture for use in digital clinical workflows.
Manufacturers are prioritizing software-driven differentiation through advanced signal processing and algorithm-enabled interpretation features, while wireless connectivity and cloud integration are becoming more important for data sharing, documentation, and longitudinal monitoring. The market is moving beyond basic sound amplification toward platforms positioned for improved diagnostic consistency and workflow efficiency, particularly in distributed care models where clinicians require reliable auscultation data outside traditional exam rooms.
Demand is also being reinforced by growing use cases in cardiac and respiratory monitoring, where repeat assessments and monitoring-friendly device formats increase utilization. Infection control priorities are supporting adoption in high-throughput clinical environments by encouraging solutions that reduce shared-contact risk and enable easier device hygiene management. Government digitization programs and broader investment in remote care delivery are contributing to adoption in underserved and rural areas. Home healthcare is expected to remain a key growth area, supported by aging demographics, chronic disease management needs, and wider availability of clinician-supported remote monitoring pathways.
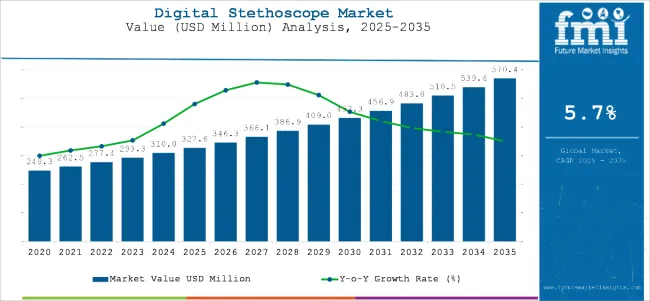
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 347.2 Million |
| Industry Value (2036) | USD 636.8 Million |
| CAGR (2026 to 2036) | 6.2% |
Source: Future Market Insights’ proprietary forecasting model and primary research
Telemedicine growth is increasing demand for digital stethoscopes that transmit auscultation audio to clinicians during virtual visits. Improved wireless connectivity is enabling reliable real time sharing, which strengthens clinical value and supports purchasing by hospitals, clinics, and telehealth providers. Smartphone compatible models are widening use by letting patients and caregivers capture and upload recordings from home or remote sites. Training and protocol uptake around digital sound capture is improving consistency and documentation, which further supports adoption. Vendors are expanding cloud connected offerings that simplify storage, playback, and collaboration across care teams. Integration with electronic health records is becoming a standard requirement because it streamlines workflow, supports audit trails, and improves continuity across repeated consultations, in large national remote monitoring programs.
Global landscape is intricately segmented to address diverse requirements of the medical diagnostic industry, categorizing sector by technology integration, product functionality, end-user application, and connectivity features. Structural division allows stakeholders to identify specific diagnostic targets, such as rising demand for AI-powered analysis in cardiology applications or preference for wireless connectivity in emergency medicine. By analyzing these segments, manufacturers can tailor product development to meet distinct needs of hospital systems versus home healthcare installations. As per FMI's projection, segmentation also highlights shift from basic sound amplification approaches to integrated diagnostic intelligence solutions. As industry matures, detailed segmentation becomes essential for understanding clinical workflow requirements and regulatory compliance, ensuring product development aligns with evolving healthcare technology standards.

Integrated chest piece systems are expected to hold 49.4% of the technology share in 2026 because they keep the standard exam format clinicians are trained on while adding digital performance benefits. Demand is supported by ergonomic designs that fit routine bedside use, along with embedded microphone improvements and compact electronics that enhance sound capture without changing established workflows. Better battery performance and dependable daily usability also reinforce adoption in high-use clinical settings.

Hospitals are projected to represent 23.8% of end-user share in 2026 because purchases are often driven by structured equipment upgrades and the need to standardize tools across departments. Central procurement supports multi-unit rollouts, while leadership prioritizes consistent diagnostic quality and repeatable staff training. High use in emergency care and intensive care settings also increases demand for durable devices and reliable performance.
Wireless transmission is expected to account for 34.6% of connectivity share in 2026 because it supports audio sharing, documentation, and remote collaboration across telehealth and in-person workflows. As remote care programs expand, demand rises for stethoscopes that connect with mobile devices and clinical platforms for recording and secure transfer. Suppliers are competing on secure connectivity, stability, and ease of integration into digital care systems.
Rising use of AI-enabled clinical decision support is expected to shift demand toward digital stethoscopes that offer built-in analytics and software-driven upgrades. As pattern recognition tools improve, buyers are placing more weight on devices that can support more consistent interpretation, structured documentation, and early risk flagging for cardiac and respiratory signals. This is also expanding the role of cloud-based analytics and subscription software, which can lift recurring revenue alongside device sales. Suppliers that do not offer AI-ready products may see weaker adoption among hospitals and health systems that prioritize connected diagnostics and measurable workflow gains. Automated anomaly detection is also becoming a key differentiation point as vendors compete on algorithm performance and integration into digital care pathways.
Growth in home-based care is expected to expand distribution beyond hospitals and clinics toward direct-to-patient and partner-led channels. Digital stethoscopes that integrate with home monitoring tools and telehealth platforms are gaining relevance for elderly care and chronic disease management, especially when caregivers need simple workflows and reliable data sharing. Telehealth providers are supporting demand for user-friendly, app-connected devices that can capture and transmit recordings during virtual visits. As remote monitoring programs scale, vendors are likely to grow through ecommerce, home healthcare providers, and device bundles tied to subscription care services.
Regulatory tightening is likely to raise evidence requirements while improving confidence in device performance. Stronger clinical validation expectations can increase development cost and time to market, but they also help differentiated products secure procurement approval and wider clinical use. Clearances and standards that support claims around diagnostic accuracy and workflow benefit can strengthen competitive positioning. Greater alignment of device standards across regions may also support international rollout for manufacturers that meet documentation, security, and performance expectations, while rising outcomes-evidence needs will keep investment high in clinical studies and real-world validation.
Global landscape for medical diagnostic devices is characterized by diverse healthcare policies, influenced by digital transformation strategies and clinical practice standards. Established markets prioritize innovation-based solutions and premium diagnostic capabilities, whereas emerging economies focus on accessible technology and basic healthcare delivery. As per FMI's estimates, Asia-Pacific is emerging as rapid growth hub due to increasing healthcare digitization and expanding medical infrastructure development. Conversely, European sectors pivot towards AI-integrated diagnostics and telehealth optimization protocols. Government initiatives in developed nations supporting healthcare technology advancement ensure sustained demand for sophisticated digital stethoscopes across all medical segments.
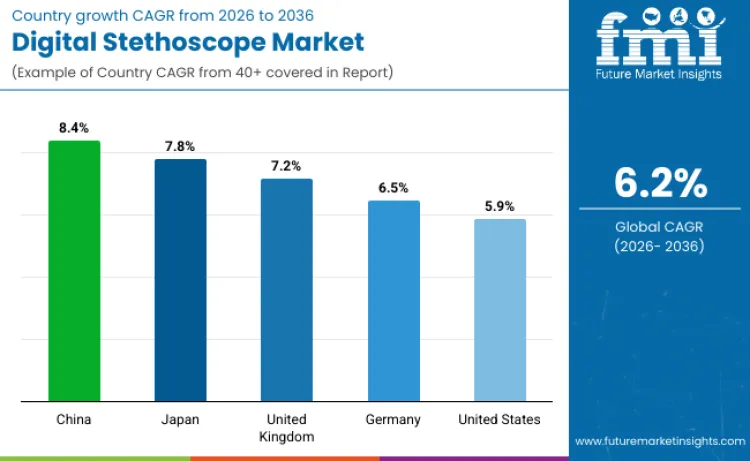
| Country | CAGR (2026 to 2036) |
|---|---|
| Japan | 7.8% |
| United States | 5.9% |
| Germany | 6.5% |
| United Kingdom | 7.2% |
| China | 8.4% |
Source: Future Market Insights - analysis driven by proprietary forecasting models and primary research
Japan is expected to see digital stethoscope sales expand at a 7.8% CAGR through 2036, supported by rising demand from an aging population and continued modernization of diagnostic equipment across care settings. Government-backed digital health efforts are increasing uptake of remote monitoring and telemedicine-enabled tools, which supports demand in hospitals and elderly care facilities. Higher use of advanced diagnostic devices per patient, combined with strong domestic electronics and device manufacturing capabilities, is improving product availability and accelerating innovation cycles. Wider integration of digital diagnostics into precision-focused care pathways is also reinforcing Japan’s position as a major growth market.
The United States market is projected to grow at a 5.9% CAGR, driven by value-based care models that reward better outcomes and greater monitoring continuity. Increasing healthcare investment is strengthening distribution for connected diagnostic tools, while ongoing telemedicine usage, including for rural access, is lifting demand for devices that support remote examination and data sharing. Policy support for healthcare technology innovation and strong commercial partnerships between manufacturers and providers are also improving adoption across large medical centers and multi-site health systems.
Germany is forecast to expand at a 6.5% CAGR, supported by higher emphasis on prevention and earlier detection workflows that increase routine diagnostic utilization. National digitization programs and institutional investment in validated medical technology are strengthening demand for connected tools that improve documentation and clinical consistency. Partnerships between device suppliers and providers are improving availability and integration into care workflows. A rigorous regulatory and quality environment supports confidence in device accuracy and safety, which helps sustain adoption.
The United Kingdom is expected to grow at a 7.2% CAGR, supported by NHS digital transformation priorities and rising deployment of remote patient monitoring. Digital health programs and clinical pathway optimization are increasing demand for tools that connect with telehealth services and support structured data capture. High expectations for evidence and assessment are pushing suppliers toward validated offerings, while focus on integrated care delivery and patient safety continues to support wider institutional uptake.
China is projected to grow at an 8.4% CAGR, supported by healthcare infrastructure upgrades and expanding smart hospital programs that increase adoption of connected diagnostics. Greater emphasis on early detection is driving higher testing volumes, while competitive domestic manufacturing is improving affordability and access. Ongoing investment in healthcare AI and diagnostic automation is also supporting demand for devices that integrate into digital hospital workflows, reinforcing China as a high-opportunity market for digital medical devices.
Competition in the digital stethoscope market is increasingly shaped by technology-led differentiation, especially in sound quality, software capability, and workflow integration. Leading brands such as 3M Littmann and Eko Health are investing in clinical validation with hospitals and research institutions to strengthen evidence and support adoption in regulated care settings. A clear trend is the development of platform-style offerings that can be configured for different specialties and patient groups, helping suppliers expand use cases while keeping device workflows consistent.
Partnerships with healthcare software vendors are also becoming more important as buyers prioritize integration with remote monitoring and digital documentation systems. Innovation remains a key lever, with companies advancing sensor design and signal processing to improve acoustic capture and reliability across environments. Consolidation is steady, with device firms acquiring software capabilities to speed integration, lower development costs, and compete more effectively with large medical equipment manufacturers. 3M Littmann remains a major participant, supported by its global reach and ongoing investment in acoustic technology.
| Items | Values |
|---|---|
| Quantitative Units | USD Million |
| Technology Segments | Integrated Chest-Piece Systems; Wireless Transmission Systems; Digital Signal Processing; AI-Powered Analytics; Others |
| Product Categories | Amplifying Stethoscopes; Recording Stethoscopes; Bluetooth-Enabled Stethoscopes; Smartphone-Compatible Stethoscopes; Others |
| End-User Types | Hospitals; Clinics; Home Healthcare; Academic Medical Centers; Emergency Medical Services; Others |
| Application Classifications | Cardiology; Pulmonology; General Medicine; Pediatrics; Veterinary Medicine; Others |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Key Countries | Japan, United States, Germany, United Kingdom, China |
| Key Companies Profiled | 3M Littmann Electronic Stethoscopes; Eko Health Inc.; Thinklabs Medical LLC; Contec Medical Systems Co.; American Diagnostic Corporation; Hill-Rom Holdings; Welch Allyn (Baxter International); eKuore Mobile Health Devices; Cardionics Inc.; SMART SOUND LLC |
| Additional Attributes | Dollar sales measured for digital stethoscopes used in clinical diagnosis and patient monitoring applications, specified by technology type (integrated chest-piece, wireless transmission, AI-powered), product functionality, end-user requirements, clinical application focus (cardiology, pulmonology, general medicine), connectivity specifications, distribution model (direct vs. distributor), and compliance alignment with evolving medical device regulations and clinical practice standards. |
How big is the global digital stethoscope market?
The global digital stethoscope market is valued at USD 347.2 million in 2026.
What is the growth outlook over the next 10 years?
The market is projected to grow at a CAGR of 6.2% from 2026 to 2036, reaching USD 636.8 million by 2036.
Which applications or care settings drive demand?
Demand is primarily driven by hospital-based diagnostics and telemedicine-supported care, with hospitals accounting for 23.8% of end-user adoption and strong use in cardiology and pulmonology.
How does adoption differ by region or healthcare system?
Adoption is highest in North America and Europe due to advanced telehealth integration, while Asia Pacific is expanding rapidly through healthcare digitization and infrastructure development.
Who are the leading manufacturers, and how is leadership defined?
Leadership is defined by acoustic technology expertise, AI integration capability, and global clinical adoption, with 3M Littmann, Eko Health, Thinklabs Medical, and Contec Medical Systems leading the market.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.