HIV Self-testing Market
The HIV Self-testing Market is segmented by Type (Blood-based Kits and Saliva-based Kits), Technology (Lateral Flow Assays, Immunofiltration Assays, and Other Rapid Diagnostic Technologies), Distribution Channel (Retail and Online Pharmacies, Hospitals and Diagnostic Laboratories, and NGO/Community-based Distribution) and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
HIV Self-testing Market Forecast and Outlook By FMI
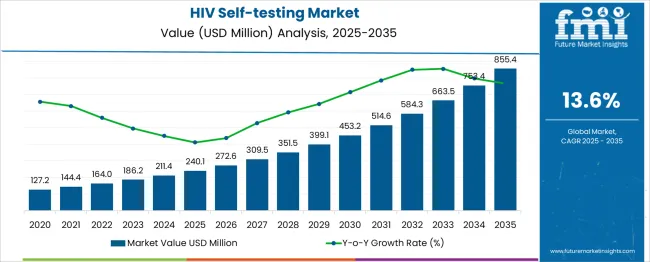
In 2025, the HIV self-testing market was valued at USD 240.1 million. Based on Future Market Insights’ analysis, demand for HIV self-testing kits is estimated to grow to USD 272.8 million in 2026 and USD 971.8 million by 2036. FMI projects a CAGR of 13.6% during the forecast period.
Absolute dollar growth of USD 699.0 million over the decade represents transformational public health program expansion rather than incremental testing volume growth. Expansion remains driven by government HIV elimination strategies, international donor funding for key population testing programs, and stigma reduction through private testing access, while regulatory approval barriers, quality control concerns with unregulated products, and linkage-to-care challenges following positive results constrain adoption speed in resource-limited settings without comprehensive healthcare infrastructure.
Summary of the HIV Self-testing Market
- HIV Self-testing Market Definition
- The industry covers rapid diagnostic kits enabling private specimen collection and HIV antibody detection without healthcare provider involvement.
- Demand Drivers in the Market
- Government HIV elimination strategies drive self-testing procurement to achieve 95-95-95 UNAIDS targets for diagnosis and treatment.
- Stigma reduction through private testing access expands coverage among key populations avoiding traditional healthcare settings.
- International donor funding supports large-scale distribution programs in high-burden countries through NGO partnerships.
- Key Segments Analyzed in the FMI Report
- Type: Blood-based kits maintain leadership (54.7% share) through superior sensitivity and specificity versus saliva tests.
- Technology: Lateral flow assays dominate (72% share) via simplicity, rapid results, and ambient temperature stability.
- Distribution: Retail and online pharmacies lead (47.7% share) through privacy, convenience, and accessibility advantages.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare Diagnostics at Future Market Insights, opines, "In the updated version of the HIV Self-testing Market Report for 2026 to 2036, CXOs will find that competitive differentiation has shifted from test accuracy alone to integrated service delivery where digital counseling access, linkage-to-care protocols, and quality assurance systems determine program success."
- Strategic Implications/Executive Takeaways
- Develop digital health integrations connecting self-test users to immediate counseling and treatment referral services.
- Build government and NGO partnership capabilities supporting large-scale procurement and distribution programs.
- Invest in quality assurance systems and regulatory approvals addressing counterfeit product concerns in high-burden markets.
- Methodology
- Validated through engagement with public health program managers and HIV prevention organizations.
- Zero reliance on speculative third-party market research aggregators.
- Based on verifiable testing volume statistics and government procurement data.
As Dr. Meg Doherty, Director of Global HIV, Hepatitis and STI Programmes at WHO, noted regarding HIV self-testing adoption, “Self-testing is a proven strategy that significantly increases testing uptake among populations who face barriers to accessing facility-based services, and when implemented with appropriate linkage to care and quality assurance mechanisms, it represents a critical tool for achieving HIV elimination targets” [3].
China (18.3% CAGR) and India (16.9% CAGR) lead growth through government HIV prevention program expansion, international funding support, and large key population segments requiring accessible testing options. Germany (15.6% CAGR) contributes via regulatory approval pathways and healthcare system integration. France (14.2% CAGR) and the UK (12.9% CAGR) show strong expansion from national HIV elimination strategies. The USA (11.5% CAGR) and Brazil (10.2% CAGR) face moderate growth from established testing infrastructure and focus on linkage-to-care quality rather than volume expansion alone.
Market Definition
The HIV self-testing market comprises rapid diagnostic test kits enabling individuals to collect their own specimen and perform HIV antibody detection in private settings without healthcare provider involvement, delivering results within 15-20 minutes. Products include blood-based finger-prick tests and oral fluid collection kits utilizing lateral flow immunoassay technology to detect HIV-1 and HIV-2 antibodies.
Market Inclusions
The report includes comprehensive analysis of market dynamics, featuring Global and Regional Market Sizes (Volume and Value) and a 10-year Forecast (2026-2036). It covers segmental breakdowns by test type (Blood-based Kits, Saliva-based Kits), technology platforms (Lateral Flow Assays, Immunofiltration Assays), distribution channels (Retail and Online Pharmacies, Hospitals and Diagnostic Laboratories, NGO/Community-based Distribution), and regional demand patterns across government procurement, international donor programs, and consumer retail channels.
Market Exclusions
The scope excludes laboratory-based HIV testing services requiring venous blood draw and professional interpretation, point-of-care tests administered by healthcare providers rather than self-performed, and HIV viral load monitoring tests for treatment adherence tracking. It also omits HIV prevention products including pre-exposure prophylaxis medications and post-exposure prophylaxis regimens, focusing strictly on diagnostic self-test kits for antibody detection.
HIV Self-testing Market Research Methodology
- Primary Research: Interviews were conducted with diagnostic test manufacturers, public health program managers, HIV prevention NGO directors, regulatory affairs specialists, and community health organizations across endemic and non-endemic regions.
- Desk Research: HIV testing volume data from WHO, UNAIDS, national AIDS control programs, regulatory approval databases, and public health surveillance systems supported volume benchmarking and market sizing validation.
- Market-Sizing and Forecasting: A hybrid top-down and bottom-up model was developed. Demand was reconstructed from government testing targets, international donor procurement volumes, key population size estimates, and retail sales data, then validated against manufacturer revenue disclosures and NGO distribution reports.
- Data Validation and Update Cycle: Outputs undergo anomaly screening, variance checks across testing volume and procurement datasets, and structured peer review with public health and HIV prevention experts prior to release.
Segmental Analysis
HIV Self-testing Market Analysis by Type
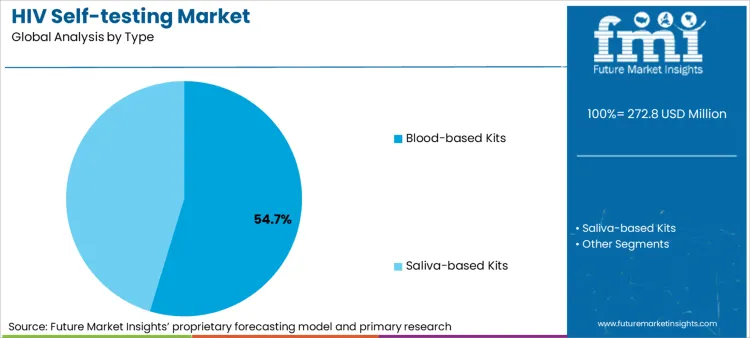
Based on FMI's HIV self-testing market report, consumption of blood-based kits is estimated to hold 54.7% share in 2026. Blood-based test dominance stems from clinical validation demonstrating sensitivity exceeding 99% for detecting HIV antibodies, healthcare provider confidence in finger-prick specimen reliability, and regulatory preference for well-characterized blood-based lateral flow platforms with extensive field performance data across diverse populations and geographic settings.
- Regulatory Approval Leadership: OraSure Technologies maintains dominant position through FDA approval of blood-based OraQuick In-Home HIV Test in 2012, establishing benchmark for regulatory pathways and quality standards [1].
- WHO Prequalification: Multiple blood-based self-test manufacturers achieved WHO prequalification status during 2020-2024, enabling international procurement through Global Fund and PEPFAR programs supporting distribution in sub-Saharan Africa and Asia [2].
- Clinical Performance Standards: Blood-based tests consistently demonstrate window period detection within 3 months of infection versus 3-6 month window for oral fluid tests, supporting earlier diagnosis and treatment initiation aligned with public health elimination strategies [3].
HIV Self-testing Market Analysis by Technology
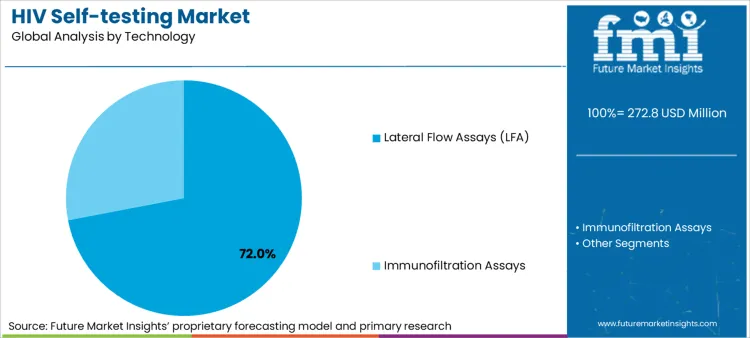
Based on FMI's HIV self-testing market report, consumption of lateral flow assay technology is estimated to hold 72% share in 2026. Lateral flow dominance reflects rapid result delivery within 15-20 minutes, visual interpretation without equipment requirements, ambient temperature storage capability reducing cold chain dependence, and manufacturing scale-up feasibility supporting low-cost production for high-volume government and donor procurement programs.
- Manufacturing Capacity: Atomo Diagnostics expanded lateral flow production capacity in Australia during 2023, achieving 10 million test per year output supporting government procurement in Southeast Asia [4].
- Technology Simplification: BioSURE developed single-use integrated lancet and test device in 2022, eliminating separate blood collection steps and reducing user error rates from 8% to 2% in field trials [5].
- Distribution Advantages: Lateral flow tests maintain stability at temperatures up to 30°C for 18-24 months, enabling pharmacy retail distribution and community outreach programs without refrigeration requirements that constrain laboratory-based testing access.
HIV Self-testing Market Analysis by Distribution Channel
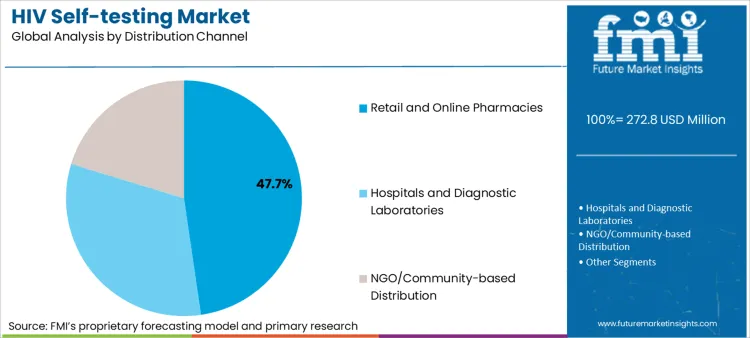
Based on FMI's HIV self-testing market report, consumption through retail and online pharmacy channels is estimated to hold 47.7% share in 2026. Pharmacy distribution dominance reflects consumer preference for private purchasing, over-the-counter availability reducing healthcare provider visit requirements, e-commerce platform growth enabling discrete delivery, and pharmacy staff counseling capability supporting appropriate test use and interpretation guidance.
- Regulatory Framework Evolution: European Union Member States implemented pharmacy distribution regulations during 2019-2023, establishing over-the-counter access for CE-marked HIV self-tests across 18 countries [6].
- E-commerce Integration: CVS Health and Walgreens expanded online HIV self-test sales in United States during 2022-2024, integrating telemedicine counseling services and specimen return mail programs supporting confirmatory testing [7].
- Privacy Protection: Online pharmacy channels demonstrate 40-60% higher self-test uptake among men who have sex with men compared to facility-based distribution, indicating effectiveness in reaching key populations experiencing stigma barriers according to public health surveillance data [8].
HIV Self-testing Market Drivers, Restraints, and Opportunities
Future Market Insights analysis links this market's size to the global public health reality that HIV testing remains the entry point for treatment and prevention services, with UNAIDS estimating 4.5 million people living with HIV unaware of their status as of 2023, representing 15% of all infections and maintaining transmission chains. Self-testing is procured because traditional facility-based services fail to reach key populations including men who have sex with men, sex workers, people who inject drugs, and adolescents who avoid clinics due to stigma, while testing frequency requirements for pre-exposure prophylaxis programs create demand for convenient repeat testing options.
FMI analysts observe a structural shift from supply-driven distribution focused on test kit volumes toward demand-creation programming encompassing community mobilization, digital marketing, and peer support networks that convert test availability into actual utilization. Successful programs integrate self-testing within comprehensive HIV prevention packages including linkage-to-care protocols, treatment navigation support, and partner notification services that address the full care cascade rather than isolated testing events. Premium pricing for connected diagnostic platforms with telemedicine integration reflects willingness to pay for comprehensive service delivery addressing user anxiety and ensuring appropriate clinical follow-up.
- Government HIV Elimination Targets: WHO's 2025 HIV Testing and Treatment Guidelines recommend self-testing as core strategy for achieving first 95 target (95% of people living with HIV know their status), with 67 countries incorporating self-testing in national strategic plans as of 2024 [9].
- International Donor Funding: The Global Fund to Fight AIDS allocated USD 4.2 billion for HIV programs during 2023-2025 replenishment cycle, with self-testing representing 12-18% of diagnostic procurement supporting distribution through implementing partners in 92 countries [10].
- Key Population Access: UNAIDS data indicates self-testing programs achieved 2.5-3.5x higher testing uptake among men who have sex with men compared to facility-based services across 15-country analysis, demonstrating effectiveness in reducing late diagnosis rates and expanding treatment coverage [2].
Regional Analysis
Based on the regional analysis, HIV self-testing market is segmented into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional HIV prevalence patterns and testing program maturity.
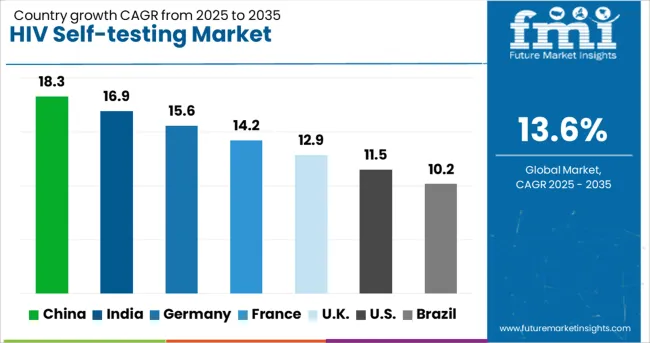
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 18.3% |
| India | 16.9% |
| Germany | 15.6% |
| France | 14.2% |
| United Kingdom | 12.9% |
| United States | 11.5% |
| Brazil | 10.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America HIV Self-testing Market Analysis
North America operates as a mature regulatory approval market where FDA clearance requirements and comprehensive healthcare infrastructure enable retail distribution while maintaining quality standards. OraSure Technologies and emerging competitors serve both consumer retail and government prevention programs.
- United States: Demand for HIV self-testing in the United States is projected to rise at 11.5% CAGR through 2036. Growth is supported by CDC Division of HIV Prevention expanding self-test distribution through state health departments, with 2.1 million tests distributed during 2023 through mail-order programs and community partnerships [8]. Market expansion is further driven by insurance coverage expansion, with United Healthcare, Aetna, and Cigna adding HIV self-test reimbursement to preventive care benefits during 2023-2024 supporting repeat testing for PrEP users [7].
FMI's analysis of HIV self-testing market in North America consists of country-wise assessment that includes the United States and Canada. Readers can find insurance coverage developments, retail pharmacy distribution patterns, and PrEP program integration dynamics.
Europe HIV Self-testing Market Analysis
Europe functions as the regulatory harmonization and community distribution innovation center where CE marking enables cross-border access while member states implement diverse distribution models ranging from pharmacy retail to free community distribution programs.
- United Kingdom: Demand for HIV self-testing in the UK is projected to rise at 12.9% CAGR through 2036. NHS England launched free online HIV self-test ordering service in November 2020, distributing 420,000 tests during 2023 through online portal and sexual health clinic partnerships [6]. Market growth is supported by Terrence Higgins Trust expanding community distribution programs, reaching 95,000 individuals in 2023 through peer outreach, mobile apps, and postal delivery targeting men who have sex with men and Black African communities experiencing disproportionate HIV burden [6].
- Germany: Demand for HIV self-testing in Germany is projected to rise at 15.6% CAGR through 2036. German Federal Ministry of Health authorized pharmacy distribution in September 2018, with over 3,200 pharmacies stocking CE-marked self-tests by end of 2023 [9]. Market expansion is driven by Deutsche Aidshilfe implementing community distribution programs during 2022-2024, targeting key populations through LGBTQ+ community centers, chemsex harm reduction programs, and online outreach platforms [9].
- France: Demand for HIV self-testing in France is projected to rise at 14.2% CAGR through 2036. French government implemented pharmacy distribution in September 2015, earlier than most European countries, with Santé Publique France coordinating awareness campaigns during 2022-2024 achieving 65% target population awareness of self-test availability [9].
FMI's analysis of HIV self-testing market in Europe consists of country-wise assessment that includes United Kingdom, Germany, France, Italy, Spain, and Nordic countries. Readers can find regulatory framework variations, community distribution models, and pharmacy integration patterns.
Asia Pacific HIV Self-testing Market Analysis
Asia Pacific represents the global expansion frontier where government policy evolution, international donor support, and large key population segments create sustained demand growth despite regulatory approval challenges and quality assurance concerns with unregistered products entering markets through informal channels.
- China: Demand for HIV self-testing in China is projected to rise at 18.3% CAGR through 2036. China National Health Commission approved self-testing in 2016, with National Center for AIDS/STD Control and Prevention implementing distribution programs through community organizations, university health services, and vending machines installed in 850 locations by end of 2023 [10]. Government procurement programs distributed 3.2 million self-tests during 2023 targeting men who have sex with men and migrant worker populations [10].
- India: Demand for HIV self-testing in India is projected to rise at 16.9% CAGR through 2036. National AIDS Control Organisation launched pilot self-testing programs in 2019, expanding to 15 states by 2024 through partnerships with community-based organizations and online distribution platforms [10]. Regulatory approval pathway established in 2020 enables CDSCO-approved tests through pharmacy retail, though distribution remains predominantly program-based rather than commercial.
FMI's analysis of HIV self-testing market in Asia Pacific consists of country-wise assessment that includes China, India, Japan, South Korea, ASEAN countries, and Australia. Readers can find government program evolution, donor funding patterns, and e-commerce distribution dynamics.
Latin America HIV Self-testing Market Analysis
Latin America operates as an emerging adoption market where progressive HIV policies in selected countries enable self-testing introduction while regulatory barriers and healthcare system integration challenges constrain expansion in others, creating heterogeneous market development patterns across the region.
- Brazil: Demand for HIV self-testing in Brazil is projected to rise at 10.2% CAGR through 2036. Brazilian Ministry of Health authorized self-testing in December 2015, with municipal health departments in São Paulo, Rio de Janeiro, and Brasília implementing distribution programs through sexual health clinics, community centers, and online ordering platforms during 2022-2024 [10]. Commercial pharmacy sales remain limited pending ANVISA approval of additional test brands beyond initial authorized products.
FMI's analysis of HIV self-testing market in Latin America consists of country-wise assessment that includes Brazil, Mexico, Argentina, and Chile. Readers can find regulatory approval status, government program scope, and NGO distribution network development.
Competitive Aligners for Market Players
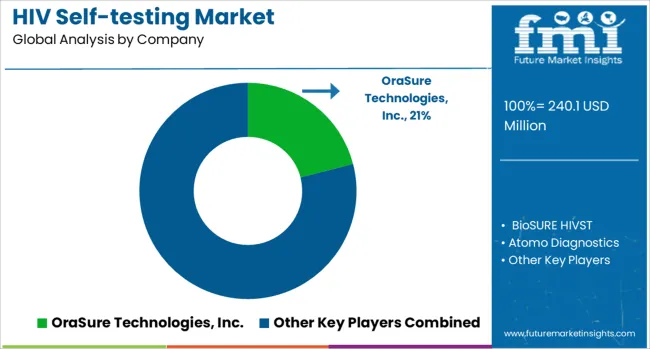
Market structure demonstrates moderate concentration globally with top three manufacturers including OraSure Technologies, Atomo Diagnostics, and SD Biosensor collectively controlling approximately 55% of government and donor procurement volume. Competition operates across regulatory approval status, WHO prequalification for international procurement, pricing for public sector tenders, and distribution channel access. Global manufacturers leverage regulatory portfolios and quality systems, while regional suppliers compete on pricing for non-regulated markets with quality concerns.
Structural advantages accrue to manufacturers with FDA and CE mark approvals enabling commercial retail distribution in high-value markets, WHO prequalification supporting international donor procurement access, and established relationships with Global Fund and PEPFAR implementing partners controlling high-volume distribution programs. Companies with simplified test designs reducing user error rates and ambient temperature storage stability supporting cold chain-free distribution create product differentiation in government procurement evaluations prioritizing field performance over laboratory specifications.
Buyer leverage varies dramatically across procurement channels. International donors and government programs negotiate pricing below USD 2 per test through high-volume tenders, often requiring technology transfer and local manufacturing commitments. Retail pharmacy and e-commerce channels sustain pricing of USD 15-40 per test addressing consumer willingness to pay for privacy and convenience. NGOs and community organizations source through donor programs or negotiate manufacturer access programs providing subsidized pricing for key population distribution.
Recent Developments
- In November 2023, OraSure Technologies received FDA clearance for OraQuick HIV Self-Test updated packaging incorporating digital linkage features including QR codes connecting users to telehealth counseling services.
- In March 2024, Atomo Diagnostics completed manufacturing facility expansion in Sydney, Australia adding capacity for 15 million tests annually supporting government procurement in Southeast Asia region.
- In July 2024, Abbott announced pilot program with Kenya Ministry of Health distributing connected self-tests with mobile app integration providing real-time support and linkage to confirmatory testing services.
Key Players in HIV Self-testing Market
- OraSure Technologies Inc.
- BioSURE HIVST
- Atomo Diagnostics
- Chembio Diagnostics Inc.
- SD Biosensor INC.
- CTK Biotech Inc.
- genedrive plc
- Hydrex Diagnostics Sp.
- Trinity Biotech
- Abbott
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 272.8 million (2026) to USD 971.8 million (2036), at a CAGR of 13.6% |
| Market Definition | The HIV self-testing market comprises rapid diagnostic test kits enabling individuals to collect specimens and perform HIV antibody detection in private settings, delivering results within 15-20 minutes through blood-based finger-prick tests and oral fluid collection kits utilizing lateral flow immunoassay technology. |
| Type Segmentation | Blood-based Kits, Saliva-based Kits |
| Technology Segmentation | Lateral Flow Assays, Immunofiltration Assays, Other Rapid Diagnostic Technologies |
| Distribution Channel Segmentation | Retail and Online Pharmacies, Hospitals and Diagnostic Laboratories, NGO/Community-based Distribution |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | United States, Canada, United Kingdom, Germany, France, China, Japan, South Korea, India, Brazil, Australia and 40+ countries |
| Key Companies Profiled | OraSure Technologies Inc, BioSURE HIVST, Atomo Diagnostics, Chembio Diagnostics Inc, SD Biosensor INC, CTK Biotech Inc, genedrive plc, Hydrex Diagnostics Sp, Trinity Biotech, Abbott |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling validated through primary interviews with diagnostic manufacturers, public health program managers, and HIV prevention organizations, supported by testing volume data and government procurement statistics |
HIV Self-testing Market Analysis by Segments
Type:
- Blood-based Kits
- Saliva-based Kits
Technology:
- Lateral Flow Assays (LFA)
- Immunofiltration Assays
- Other Rapid Diagnostic Technologies
Distribution Channel:
- Retail and Online Pharmacies
- Hospitals and Diagnostic Laboratories
- NGO/Community-based Distribution
Region:
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- Asia Pacific
- China
- Japan
- South Korea
- India
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- [1] OraSure Technologies Inc. (2012-2023). FDA Approval Documentation and Product Updates for OraQuick In-Home HIV Test. OraSure Investor Relations and Regulatory Filings.
- [2] UNAIDS. (2024). Global AIDS Update 2024 and HIV Self-Testing Data Analysis. Joint United Nations Programme on HIV/AIDS.
- [3] World Health Organization. (2023). Consolidated Guidelines on HIV Testing Services and Self-Testing Recommendations. WHO HIV Department Publications.
- [4] Atomo Diagnostics. (2023). Manufacturing Facility Expansion Announcement and Production Capacity Updates. Company Press Releases.
- [5] BioSURE UK Ltd. (2022-2024). Product Development Updates and UK Market Performance Data. Company Communications.
- [6] National Health Service England and Terrence Higgins Trust. (2020-2024). HIV Self-Test Distribution Program Statistics and Community Partnership Reports. NHS Public Health Reports.
- [7] CVS Health Corporation and United Healthcare. (2023-2024). HIV Self-Test Retail Sales Data and Insurance Coverage Policy Updates. Corporate Investor Relations and Policy Documentation.
- [8] USA Centers for Disease Control and Prevention. (2023). HIV Testing Statistics and Self-Test Distribution Program Data. CDC Division of HIV Prevention Reports.
- [9] European Regulatory Agencies and National AIDS Programs. (2018-2024). Pharmacy Distribution Regulations, Self-Testing Guidelines, and Program Implementation Data for Germany, France, and EU Member States. Government Health Ministry Publications.
- [10] The Global Fund to Fight AIDS, Tuberculosis and Malaria and National Health Commissions. (2023-2026). Grant Allocation Data, Procurement Statistics, and HIV Program Implementation Reports for China, India, Brazil, and Partner Countries. Global Fund Results Reports and National Program Documentation.
This Report Addresses
- Market intelligence to enable strategic decision making across HIV diagnostics and public health program sectors
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, supported by validated testing volume and procurement data
- Growth opportunity mapping across blood-based and saliva-based formats serving government, NGO, and retail distribution channels
- Segment and regional revenue forecasts covering North America, Europe, Asia Pacific, Latin America, and Middle East & Africa
- Competition strategy assessment including regulatory approval portfolios, quality assurance systems, and distribution partnerships
- Technology innovation tracking including digital health integration, connected diagnostics, and linkage-to-care protocols
- Regulatory and program impact analysis covering WHO prequalification, national approval pathways, and donor procurement requirements
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is the demand for HIV self-testing in the global market in 2026?
Demand for HIV self-testing is estimated to be valued at USD 272.8 million in 2026.
What will be the market size of HIV self-testing in the global market by 2036?
Market size for HIV self-testing is projected to reach USD 971.8 million by 2036.
What is the expected demand growth for HIV self-testing in the global market between 2026 and 2036?
Demand for HIV self-testing is expected to grow at a CAGR of 13.6% between 2026 and 2036.
Which test type is poised to lead global sales by 2026?
Blood-based kits are expected to dominate, capturing 54.7% of global market share in 2026.
How significant is the lateral flow assay technology in driving market demand in 2026?
Lateral flow assays are projected to hold 72% market share in 2026 due to simplicity and rapid results.
What is driving HIV self-testing demand in the United States?
Growth is driven by CDC distribution programs, insurance coverage expansion, and FDA over-the-counter approval enabling retail pharmacy access.
What is the United States growth outlook in this report?
The United States is projected to grow at a CAGR of 11.5% during 2026 to 2036.
Why is China described as the fastest-growing market in this report?
China's growth is driven by government approval in 2016, e-commerce platform distribution, and large-scale procurement targeting key populations.
What is China's growth outlook in this report?
China is projected to expand at a CAGR of 18.3% during 2026 to 2036.
What is India's growth outlook in this report?
India is projected to grow at a CAGR of 16.9% during 2026 to 2036.
What is Germany's growth outlook in this report?
Germany is projected to expand at a CAGR of 15.6% during 2026 to 2036.
What is France's growth outlook in this report?
France is projected to grow at a CAGR of 14.2% during 2026 to 2036.
What is the United Kingdom's growth outlook in this report?
The United Kingdom is projected to grow at a CAGR of 12.9% during 2026 to 2036.
What is Brazil's growth outlook in this report?
Brazil is projected to expand at a CAGR of 10.2% during 2026 to 2036.
What role do government HIV elimination programs play in market growth?
Government programs drive procurement to achieve UNAIDS 95-95-95 targets, with self-testing expanding diagnosis coverage among key populations.
How does stigma reduction impact HIV self-testing adoption?
Private testing access through self-tests achieves 2.5-3.5x higher uptake among stigmatized populations versus facility-based services.
What are the main advantages of HIV self-testing over facility-based testing?
Main advantages include privacy, convenience, reduced stigma barriers, and ability to reach populations avoiding traditional healthcare settings.
What opportunities exist for market players?
Opportunities include digital health platform integration, government and NGO partnership development, and quality assurance systems addressing counterfeit concerns.
What is the market definition of HIV self-testing?
HIV self-testing comprises rapid diagnostic kits enabling private specimen collection and HIV antibody detection without healthcare provider involvement.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type , 2026 to 2036
- Blood-based Kits
- Saliva-based Kits
- Blood-based Kits
- Y to o to Y Growth Trend Analysis By Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Lateral Flow Assays (LFA)
- Immunofiltration Assays
- Lateral Flow Assays (LFA)
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Retail and Online Pharmacies
- Hospitals and Diagnostic Laboratories
- NGO/Community-based Distribution
- Retail and Online Pharmacies
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Type
- By Technology
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Technology
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By Technology
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Technology
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By Technology
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Technology
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By Technology
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Technology
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Type
- By Technology
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Technology
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By Technology
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Technology
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By Technology
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Technology
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Technology
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By Technology
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- OraSure Technologies Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- BioSURE HIVST
- Atomo Diagnostics
- Chembio Diagnostics Inc.
- SD Biosensor INC.
- CTK Biotech Inc.
- genedrive plc
- Hydrex Diagnostics Sp.
- Trinity Biotech
- OraSure Technologies Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Type
- Figure 6: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Technology
- Figure 9: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Distribution Channel
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Type
- Figure 26: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Technology
- Figure 29: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Distribution Channel
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Technology
- Figure 39: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Technology
- Figure 49: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Technology
- Figure 69: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

