HIV/HBV/HCV Test Kits Market
The HIV/HBV/HCV Test Kits Market is segmented by Kit Type (Single Analyte Test Kits, Multiplex / Combo Test Kits and Others), Sample Type (blood, serum/plasma-based, dried blood spot (DBS), urine, and oral fluid/saliva), Sales Channel (hospitals, clinics, diagnostic laboratories, government organizations and NGOs, Physicians’ Offices & Clinics, Retail Pharmacy Chains, E-Commerce and others), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
HIV/HBV/HCV Test Kits Market Size, Market Forecast and Outlook By FMI
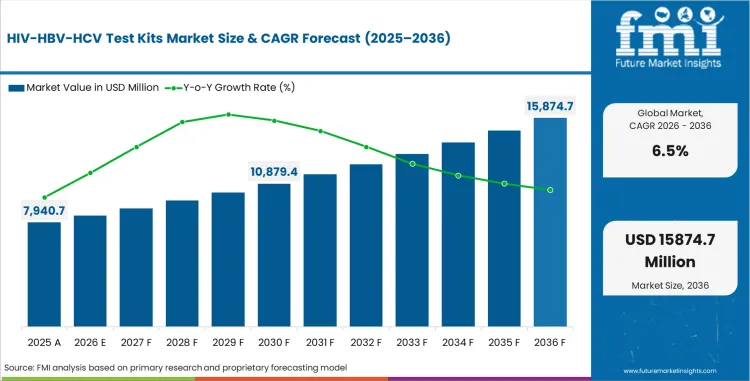
In 2026, the HIV/HBV/HCV test kits market was valued at USD 7,940.7 million. Based on Future Market Insights analysis, demand for HIV/HBV/HCV test kits is estimated to grow to USD 14,905.7 million by 2036. FMI projects a CAGR of 6.5% during the forecast period.
Absolute dollar growth of USD 6,965.1 million over the decade signals strong expansion rather than a demand reset. As per FMI, demand is expected to remain supported by rising infectious disease screening volumes, wider emphasis on early diagnosis, and continued testing demand across hospitals, clinics, laboratories, and public health programs. At the same time, reimbursement variation, procurement discipline, and regulatory oversight are expected to keep market development structured across mature diagnostic systems. “Ensuring rapid access to quality-assured diagnostic tests is essential to protecting the health of vulnerable populations, including pregnant women, this milestone reflects our continued commitment to accelerating the availability of safe, effective and quality health innovations where they are needed most.” said Dr Rogério Gaspar, Director of WHO’s Department of Prequalification and Regulation of Medicines and Health Products. [1]
Summary of HIV/HBV/HCV test kits market
- HIV/HBV/HCV test kits market definition
- The industry covers diagnostic kits used to detect HIV, HBV, and HCV across hospitals, clinics, laboratories, and public health testing environments.
- Demand Drivers in the Market
- Demand is rising by mandatory screening programs for blood-borne and sexually transmitted infections rather than discretionary diagnostic testing growth.
- Broader emphasis on early diagnosis and treatment linkage is driving test adoption across clinical and public health settings.
- Decentralized testing expansion is creating demand for rapid diagnostic kits that can be operated outside the laboratory infrastructure, as reported by FMI.
- Key Segments Analyzed in the FMI Report
- By Kit Type: HIV Test Kits lead at 67.6% share in 2026.
- By Sample Type: Blood Leads at 47.5% share in 2026.
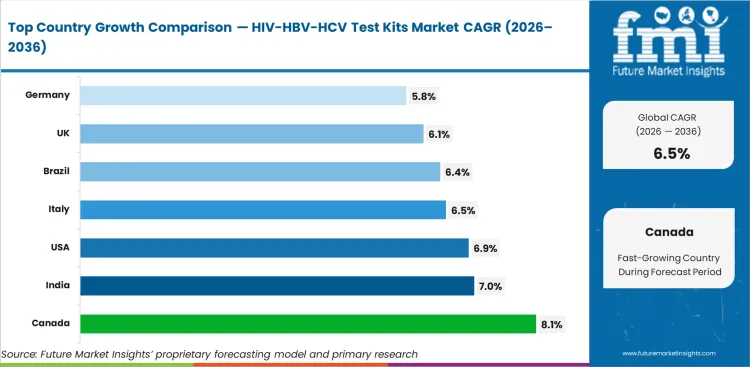
- Geography: Canada and India show the fastest growth at 8.1% and 7.0% CAGR, supported by stronger screening access and broader testing coverage.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant at Future Market Insights, opines, ‘Buyer preference is being shaped by assay confidence, specimen flexibility, and channel compatibility, as testing providers place greater value on dependable diagnosis, program scalability, and patient reach across routine screening pathways.’
- Strategic Implications and Executive Takeaways
- Strengthen buyer contracting with channel-ready product positioning, validation-backed performance claims, and sample-specific workflow support aligned to screening demand.
- Build growth plans around public health and decentralized testing networks where recurring infectious disease screening supports repeat kit procurement.
- Protect approved supplier positions through diagnostic reliability and specimen adaptability, since validated testing solutions are not switched easily once embedded into program workflows.
- Methodology
- As per FMI, sizing and segment splits were reconciled using company primary disclosures and demand mapping by testing channel, disease screening flow, and specimen type.
- Kit type and sample type shares were validated by linking test demand patterns to infectious disease screening volumes across institutional and outreach settings.
- Pricing sensitivity was stress tested under procurement mix and channel-substitution scenarios to confirm revenue response under realistic diagnostic conditions.
Based on FMI’s report, Canada (8.1% CAGR) and India (7.0%) are expected to lead growth due to expanding screening access and stronger public health testing coverage. The U.S. (6.9%), Italy (6.5%), and the U.K. (6.1%) are expected to maintain healthy expansion through continued diagnostic demand and established testing pathways. China (5.3%), Germany (5.8%), Japan (3.3%), and Brazil (6.4%) are expected to contribute through recurring screening demand, with growth shaped by healthcare access, treatment linkage, and testing program intensity.
HIV/HBV/HCV Test Kits Market Definition
In this market, test kits in vitro diagnostic used in detecting and screening of human immunodeficiency virus, hepatitis B virus, and hepatitis C virus are included. Such tests are supplied to hospitals, clinics, diagnostic laboratories, governmental institutions, NGOs, and other health institutions for use in infectious diseases screening, detection, surveillance, and linking to treatment when test-based identification is required in medical practice. The following types of test kits are included: HIV test kits, HBV test kits, HCV test kits, and combination test kits which are used in testing blood, urine, and saliva samples for infectious diseases. Screening volume, public health needs, choice of assay, specimen type, and accessibility determine the demand in such markets. The revenues generated come from sales of test kits.
HIV/HBV/HCV Test Kits Market Inclusions
The report provides global and regional market sizing, as well as a 10-year forecast from 2026 to 2036. The report also provides market sizing by kit type, sample type, and Sales Channel, with country-level CAGR comparisons across key markets. The report also provides analysis on competitive positioning of key suppliers, channel-level testing dynamics, and the influence of diagnostic reliability, sample flexibility, and program-level procurement on supplier choice, as per FMI.
HIV/HBV/HCV Test Kits Market Exclusions
The scope does not include broader molecular diagnostic platforms, central laboratory analyzers, antiviral treatment products, or unrelated infectious disease assays outside HIV, HBV, and HCV testing. Screening services without kit sales, sample collection accessories, and generalized laboratory consumables are also excluded. The scope also does not include non-diagnostic awareness products or treatment-monitoring solutions that do not function as HIV/HBV/HCV test kits, but only test kits used for screening and diagnostic detection.
HIV/HBV/HCV Test Kits Market Research Methodology
- Primary Research: Interviews were carried out with laboratory professionals, infectious disease specialists, procurement managers, and public health testing stakeholders to confirm demand patterns and purchase behavior.
- Desk Research: Public data from regulatory bodies, disease control agencies, company materials, testing guidelines, and diagnostic publications were analyzed to compare usage trends and channel dynamics.
- Market Sizing and Forecasting: A hybrid approach was created based on disease testing demand mapping, specimen mix, and end-user utilization patterns, and then harmonized through volume and price triangulation.
- Data Validation and Update Cycle: Results were filtered for variance, tested for internal consistency, and assessed through structured peer review before distribution.
Segmental Analysis
HIV/HBV/HCV Test Kits Market Analysis by Kit Type
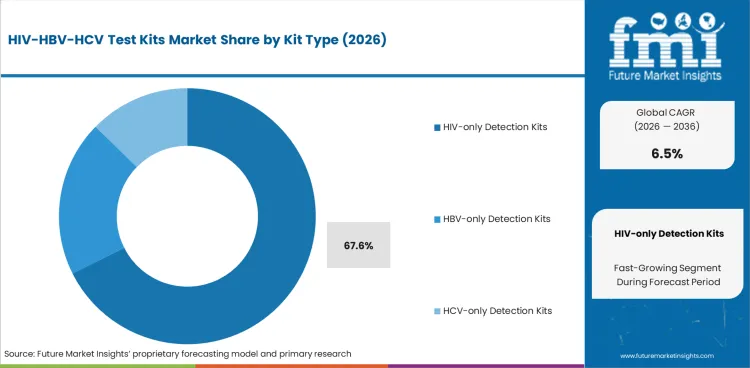
Based on FMI’s report, HIV test kits are estimated to hold 67.6% share in 2026. This lead position is supported by the larger global testing base for HIV screening, broader inclusion of HIV diagnostics in routine public health programs, and stronger continuity of screening across high-risk, antenatal, institutional, and outreach settings. Their larger share is being reinforced by longstanding policy support for HIV detection, recurring testing frequency in select populations, and continued use across both centralized and decentralized care channels.
- Largest Global Testing Base: HIV test kit demand remains highest because the global screening base is exceptionally large. UNAIDS reported 40.8 million people were living with HIV in 2024, with 87% knowing their status, reflecting sustained large-scale testing activity worldwide. [2]
- Routine Program Integration: HIV testing is more deeply embedded in public health delivery than many other blood-borne infections. WHO notes HIV testing spans facility-based, community-based, self-testing, home, mobile, and outreach services, supporting broader and more continuous kit utilization across care settings. [3]
- Decentralized Format Expands Access: HIV test kits hold the highest share because rapid tests and self-tests can be deployed across facility, community, outreach, and home-based settings, allowing HIV screening to reach wider populations through more flexible delivery models than many other blood-borne infection pathways.
HIV/HBV/HCV Test Kits Market Analysis by Sample Type
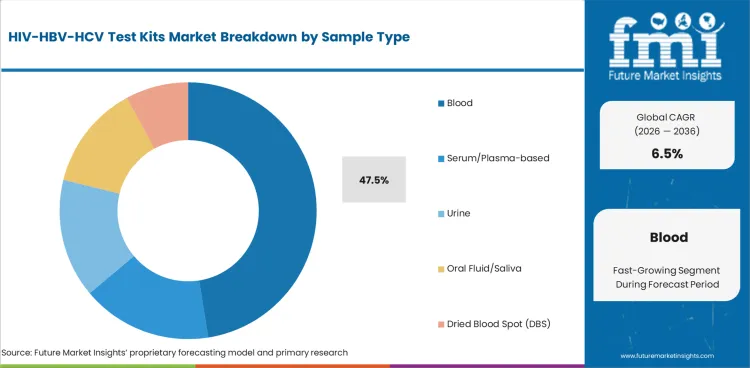
Blood accounts for 47.5% share in 2026, based on FMI’s report, reflecting its role as the most established and clinically trusted specimen type across HIV, HBV, and HCV testing workflows. This leading position is supported by broad compatibility with diagnostic methods, stronger confidence in analytical performance, and continuing use in institutional testing pathways where result reliability and laboratory familiarity remain important. Demand is therefore being concentrated in blood-based testing where accuracy, validation depth, and workflow standardization continue to carry high weight.
- Reference Specimen Across Assays: Blood remains the leading sample type because HIV, HBV, and HCV diagnostic algorithms are still anchored in serum, plasma, venous whole blood, or fingerstick blood, making it the most validated specimen across screening, reflex, and confirmatory pathways. [4]
- Higher Analytical Confidence: Blood-based specimens continue to be preferred where result reliability is critical. NIH’s 2025 HIV testing review notes blood-derived samples are generally favored because they contain higher concentrations of HIV antibodies and support broader follow-on testing needs. [5]
- Most Validated Specimen Pathway: Blood holds the highest share because it remains the most clinically validated and widely accepted specimen type across HIV, HBV, and HCV testing, with strong alignment to routine laboratory protocols, confirmatory workflows, and institutional diagnostic practice.
HIV/HBV/HCV Test Kits Market Drivers, Restraints, and Opportunities
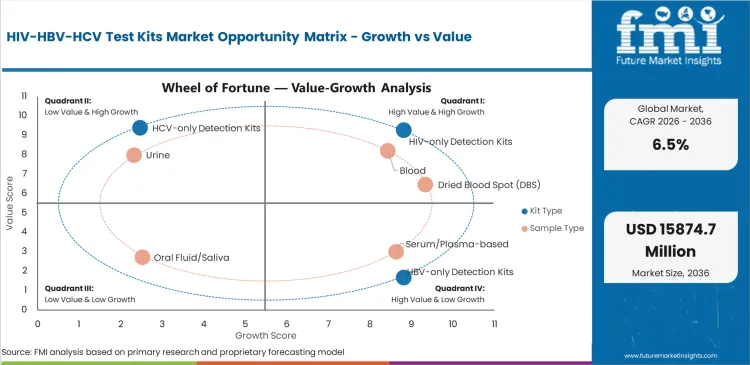
Future Market Insights analysis indicates that historical patterns point to a screening-driven infectious disease diagnostics category where demand is anchored in early detection, transmission control, and treatment linkage across institutional and public health pathways. Estimated valuation in 2026 is being supported by broader awareness of blood-borne infection burden, rising testing intensity, and continued integration of screening into clinical, antenatal, and outreach-based care environments, as per FMI.
While category demand is being restrained by pricing pressure in public procurement, uneven access to confirmatory care, and variability in screening intensity across markets, value is being supported by wider testing access, stronger disease control initiatives, and increasing use of sample-flexible testing solutions. Based on FMI’s report, the forecast trajectory reflects a market where HIV test kits remain the leading kit category, while suppliers are differentiated by assay confidence, channel reach, and program-level fit.
- Screening-Backed Demand: Demand is being supported as providers continue to expand early detection efforts across institutional and public health testing pathways.
- Procurement-Led: Growth is being moderated where government and bulk-buyer purchasing keeps pricing discipline high across standard testing formats.
- Access Expansion: Demand is being supported as more decentralized testing programs are being integrated into broader infectious disease control strategies.
Regional Analysis
Based on the regional analysis, HIV/HBV/HCV test kits market is segmented into North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe, and Middle East & Africa across 40+ countries. Regional performance is assessed using country-level demand signals tied to infectious disease screening intensity, laboratory access, diagnostic infrastructure, and public health procurement, as per FMI. The full report also offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR |
|---|---|
| Canada | 8.1% |
| India | 7.0% |
| United States | 6.9% |
| Brazil | 6.4% |
| Italy | 6.5% |
| United Kingdom | 6.1% |
| Germany | 5.8% |
| China | 5.3% |
| Japan | 3.3% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America HIV/HBV/HCV Test Kits Market Analysis
North America is shaped by structured screening frameworks, laboratory-led diagnosis, and strong integration of infectious disease testing into institutional care pathways. Hoffmann-La Roche Ltd. holds strong placement through assay credibility, broad diagnostic reach, and established use across major testing channels. Bio-Rad Laboratories Inc. and bioMérieux SA compete through laboratory presence, infectious disease testing portfolios, and channel familiarity across hospitals and diagnostic settings. Based on FMI’s report, demand remains anchored in recurring screening, hospital testing, and laboratory-based infectious disease management.
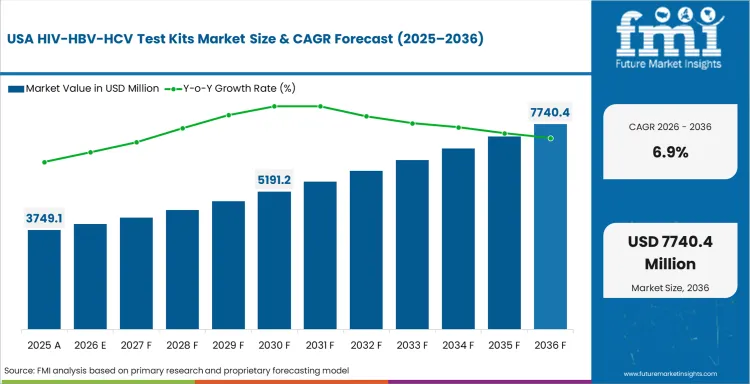
- United States: The rise in the growth rate of HIV/HBV/HCV test kits market in the United States will be on account of a CAGR of 6.9%, owing to higher rates of screening, better access to testing, and prevalence of HIV/HBV/HCV diagnostic procedures in the country.
- Canada: The rise in the growth rate of the market for HIV/HBV/HCV test kits in Canada will benefit from a CAGR of 8.1% till 2036, attributed to higher rates of testing by the public healthcare industry.
FMI’s report includes a detailed analysis of the growth in the North American region, along with a country-wise assessment that includes the United States and Canada. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the North America region.
East Asia HIV/HBV/HCV Test Kits Market Analysis
The East Asia region still maintains its high volume for diagnostics because of screening requirements, lab structure, and increased focus on disease surveillance. The positioning of suppliers in this region will depend on accuracy in the test results, suitability of samples, and whether suppliers can facilitate both organizational and program-based testing. According to the FMI report, the demand for the services will continue to grow due to screening for hepatitis and HIV, increasing awareness about diagnostics, and improved access to diagnostic programs.
- China: Market value-wise, the demand for HIV/HBV/HCV test kits in China is forecasted to witness a CAGR of 5.3% during the forecast period of 2036. This will be facilitated due to the presence of screening requirements on a massive scale, improved availability of tests, and focus on diagnosis of hepatitis and HIV.
- Japan: The growth in demand for HIV/HBV/HCV test kits in Japan is anticipated to record a CAGR of 3.3% during the forecast period of 2036. This will be attributed to the stable testing practices in institutions, along with adherence to standardized laboratory testing, despite the presence of mature screening systems.
The complete report offers an in-depth evaluation of the East Asian market for the HIV/HBV/HCV diagnostic kits from 2021 to 2036. This evaluation covers price trends, drivers of growth, and other relevant factors such as those shaping procurement trends in China, Japan, South Korea, and surrounding regions.
South Asia & Pacific HIV/HBV/HCV Test Kits Market Analysis
South Asia & Pacific is being shaped by expanding screening access, rising awareness of infectious disease burden, and broader integration of testing into public and clinical care pathways. Supplier positioning in the region is influenced by affordability, specimen adaptability, and ability to support decentralized and institutional testing needs at scale. Based on FMI’s report, demand remains supported by increasing program-level screening and stronger emphasis on earlier diagnosis in developing healthcare systems.
- India: The growth rate of demand for HIV/HBV/HCV test kit products in India over the period of 2036 will be estimated at 7.0% CAGR. Increased screening for infectious diseases, increased availability of testing services, and increased use of test kits will drive growth.
Analysis presented in this report covers the global market for HIV, HBV & HCV Testing Kits Market in South Asia & Pacific countries during the forecast period 2021-2036. Pricing, trend analysis and influencing factors like production, consumption, supply, demand and market growth analysis by countries of South Asia & Pacific are covered in this report.
Western Europe HIV/HBV/HCV Test Kits Market Analysis
Western Europe is quality-led and regulation-aware, where diagnostic use is shaped by laboratory discipline, clinical testing pathways, and established public health screening standards. Hoffmann-La Roche Ltd., Siemens AG, and Abbott Laboratories maintain relevance through strong diagnostic portfolios, institutional credibility, and broad testing channel access across the region. Use continues to be influenced by assay trust, laboratory compatibility, and public procurement structure. As per FMI, recurring hospital and laboratory demand continues to shape test kit use across Western Europe.
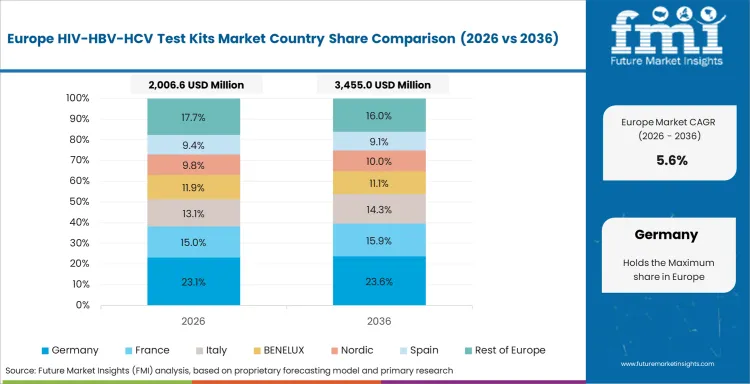
- Germany: The CAGR of HIV/HBV/HCV Test Kits market in Germany is expected to register 5.8% over the forecast period of 2036. The growth is attributed to the existence of systematic testing methods for infectious diseases, advanced lab infrastructure, and consistent use of advanced testing technologies throughout the course of treatment.
- United Kingdom: The demand for HIV/HBV/HCV test kits in the United Kingdom will grow at a CAGR of 6.1% during the forecast period of 2036. The growth will be fueled by consistent demand for screening programs, availability of testing at institutional level, and sustained use of structured diagnostic tests, even though it will pass through procurement efficiency and maturity of testing processes.
- Italy: The demand for HIV/HBV/HCV test kits in Italy will grow at a CAGR of 6.5% during the forecast period of 2036. The growth will be fueled by consistent screening practices, increased use of tests, and enhanced use of test-based screening processes for infectious diseases.
FMI’s analysis of HIV/HBV/HCV test kits market in Western Europe consists of country-wise assessment that includes Germany, the UK, France, Italy, Spain, BENELUX, Nordic Countries, and Rest of Western Europe. Readers can know various regulations and latest trends in the regional market.
Latin America HIV/HBV/HCV Test Kits Market Analysis
Latin America is shaped by rising screening access, mixed public-private diagnostic infrastructure, and continued dependence on institutional and program-led testing for infectious disease detection. Global suppliers retain visibility through distributor-led access, public health participation, and laboratory channel presence across major care settings. According to FMI’s report, demand conversion is being supported by stronger disease awareness, broader testing initiatives, and gradual improvement in organized diagnostic coverage.
- Brazil: The demand for HIV/HBV/HCV test kits in Brazil is expected to grow at a CAGR of 6.4% during the forecast period of 2036. This is driven by expanding screening demand, wider healthcare access, and continued public health focus on HIV and hepatitis detection, although this remains influenced by procurement timing and channel-level funding patterns.
The report includes an analysis of the market in Brazil, Argentina, and Rest of Latin America. The reader can gain information about various factors, such as the pricing analysis and trends, that are currently influencing the market in the Latin America region.
Competitive Aligners for Market Players
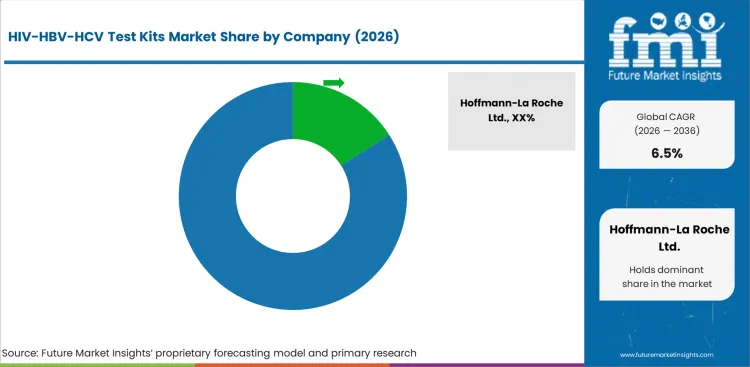
The market structure remains moderately concentrated, with effective competition centered on suppliers capable of supporting assay reliability, specimen-specific usability, and broad channel access across structured infectious disease testing pathways. The data provided shows the presence of a leading participant in the market, with the rest of demand distributed across a focused set of established diagnostic companies. The key competitive factor is diagnostic confidence aligned to screening need, as supplier qualification is filtered through assay performance, workflow compatibility, and suitability across public and clinical testing environments. The product portfolio also reflects this emphasis, as HIV test kits account for the leading share in the supplied segmentation, as indicated by FMI.
Companies with established diagnostic credibility, strong laboratory integration, and broad product support across HIV and hepatitis testing carry structural advantages because buyer qualification is governed by performance trust, channel acceptance, and confidence in recurring screening use. Scale leaders benefit from deeper institutional access, stronger program-level recognition, and the ability to serve multiple specimen pathways under the same testing framework. Portfolio breadth across blood, urine, and saliva-based workflows helps protect account positions when providers standardize around validated suppliers. Suppliers with narrower operational reach tend to compete through selective geographies, niche program participation, or localized channel access, which can weaken their position during structured supplier evaluation.
Customer concentration reinforces buyer leverage. Hospitals, clinics, and diagnostic laboratories account for a large part of recurring demand, while government organizations and NGOs form an important supplementary tier of testing activity. Large procurement programs commonly keep approved diagnostic vendors active, prioritize test kits that align with screening workflows, and favor suppliers with dependable availability across recurring public health demand. Pricing power is therefore moderated for standard testing formats, with measured advantage retained only where assay trust, sample flexibility, or channel fit creates stronger procurement preference, Future Market Insights analysis.
Recent Developments
- In March 2026, Roche announced that the cobas MPX-E assay, a qualitative in-vitro test for the detection and discrimination of Human Immunodeficiency Virus (HIV 1 and 2) and Hepatitis C, B, and E viruses, is now available in countries accepting the CE mark. [6]
- In July 2025, The World Health Organization has prequalified the first triple rapid diagnostic test detecting HIV, hepatitis B, and syphilis, marking a major step toward eliminating mother-to-child transmission. The multiplex test enables faster, integrated antenatal screening, improving early diagnosis and treatment access, particularly in low-resource settings, while supporting global efforts to reduce preventable maternal and infant infections.
- In September 2025, Reszon Diagnostics International Sdn. Bhd., a subsidiary of Hextar Healthcare, has launched the RESZON HCV Rapid Test, a point-of-care diagnostic for hepatitis C detection. Delivering results within 15–20 minutes without specialized equipment, the test enhances decentralized screening across clinics and resource-limited settings, supporting broader access to early diagnosis and infectious disease control programs.
- In June 2024, ELITechGroup announced approval of its BBV Panel for the Japanese market. ELITechGroup offers a fully automated PCR solution to test the BBV panel, which includes quantitative assays for monitoring of Human Immunodeficiency Virus (HIV-1), Human Hepatitis B Virus (HBV), and Human Hepatitis C Virus (HCV). [7]
Key Players in HIV/HBV/HCV Test Kits Market
- Hoffmann-La Roche Ltd.
- Bio-Rad Laboratories Inc.
- bioMérieux SA
- QIAGEN
- Meridian Bioscience, Inc.
- Hologic Inc.
- Siemens AG
- Maternova
- Creative Diagnostics
- Abbott Laboratories
- Bio-Techne
- Becton, Dickinson and Company
- Takara Bio Inc.
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 7,940.7 million (2026) to USD 14,905.7 million (2036), at a CAGR of 6.5% |
| Market Definition | The HIV/HBV/HCV test kits market comprises diagnostic kits used to detect and screen human immunodeficiency virus, hepatitis B virus, and hepatitis C virus across clinical and public health settings, where demand is shaped by screening intensity, assay reliability, specimen suitability, and access across institutional and program-led testing environments. |
| Kit Type Segmentation | Single Analyte Test Kits, Multiplex / Combo Test Kits and Others |
| Sample Type Segmentation | Blood, Serum/Plasma-based, Urine, Oral Fluid/Saliva, Dried Blood Spot (DBS) |
| Sales Channel Segmentation | Hospitals, Clinics, Diagnostic Laboratories, Government Organizations and NGOs, Physicians’ Offices & Clinics, Retail Pharmacy Chains, E-Commerce and Others |
| Regions Covered | North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe, and Middle East & Africa |
| Countries Covered | United States, Canada, United Kingdom, Germany, France, China, Japan, South Korea, Brazil, Australia, India, and 40+ countries |
| Key Companies Profiled | Hoffmann-La Roche Ltd., Bio-Rad Laboratories Inc., bioMérieux SA, QIAGEN, Meridian Bioscience, Inc., Hologic Inc., Siemens AG, Maternova, Creative Diagnostics, Abbott Laboratories, Bio-Techne, Becton, Dickinson and Company and Takara Bio Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with diagnostic and procurement stakeholders, supported by demand triangulation across screening volumes, specimen mix, and test kit adoption patterns, as per FMI. |
HIV/HBV/HCV Test Kits Market Analysis by Segments
Kit Type
- Single Analyte Test Kits
- HIV-only Detection Kits
- HBV-only Detection Kits
- HCV-only Detection Kits
- Multiplex / Combo Test Kits
- HIV + HBV Detection Kits
- HIV + HCV Detection Kits
- HIV + HBV + HCV Triple Test Kits
- HIV/HBV/HCV + Syphilis Combo Panels
- Others
Sample Type
- Blood
- Serum/Plasma-based
- Urine
- Oral Fluid/Saliva
- Dried Blood Spot (DBS)
Sales Channel
- Hospitals
- Clinics
- Diagnostic Laboratories
- Government Organizations and NGOs
- Physicians’ Offices & Clinics
- Retail Pharmacy Chains
- E-Commerce
- Others
Region
- North America
- U.S.
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- East Asia
- China
- Japan
- South Korea
- South Asia & Pacific
- India
- ASEAN Countries
- Australia & New Zealand
- Rest of South Asia
- Western Europe
- Germany
- U.K.
- France
- Italy
- Spain
- BENELUX
- Nordic Countries
- Rest of Western Europe
- Eastern Europe
- Russia
- Hungary
- Poland
- Rest of Eastern Europe
- Middle East & Africa
- Saudi Arabia
- Türkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- [1] World Health Organization (WHO) (2025), WHO prequalifies the first triple diagnostic test for HIV, hepatitis B and syphilis, a milestone toward global disease elimination goals.
- [2] UNAIDS (2025), Global HIV & AIDS statistics — Fact sheet
- [3] World Health Organization (WHO) (2025), Global HIV Programme- HIV testing services
- [4] World Health Organization (WHO) (2024), Consolidated guidelines on differentiated HIV testing services
- [5] NIH-Bookshelf (2025), StatPearls- HIV Testing
- [6] Roche (2026), Roche launches the cobas MPX-E assay, a new 4-in-1 donor screening test to further safeguard the global blood supply.
- [7] ELITechGroup (2024), ELITechGroup Announces Regulatory Approval of its Blood Borne Virus Panel in Japan.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This Report Addresses
- Market intelligence to enable structured strategic decision making across infectious disease screening expansion and diagnostic portfolio planning
- Market size estimation and 10-year revenue forecasts from 2026 to 2036 for HIV/HBV/HCV test kit demand
- Growth opportunity mapping across kit type classes and sample-based testing pools
- Segment and regional revenue forecasts aligned to the stated taxonomy and covered geographies
- Competition strategy assessment including supplier positioning, portfolio fit, and diagnostic channel coverage benchmarking
- Product and compliance tracking focused on assay reliability, specimen standards, and screening-led quality expectations
- Regulatory impact analysis covering test-led diagnosis selection and provider-level protocol adoption in regulated care settings
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is the demand for HIV/HBV/HCV Test Kits in the global market in 2026?
Demand for HIV/HBV/HCV test kits in the global market is estimated to be valued at USD 7,940.7 million in 2026, as per FMI.
What will be the market size of HIV/HBV/HCV Test Kits in the global market by 2036?
Market size for HIV/HBV/HCV test kits is projected to reach USD 14,905.7 million by 2036.
What is the expected demand growth for HIV/HBV/HCV Test Kits in the global market between 2026 and 2036?
Demand for HIV/HBV/HCV test kits in the global market is expected to grow at a CAGR of 6.5% between 2026 and 2036.
Which kit type is poised to lead global sales by 2026?
HIV test kits are expected to be the dominant kit type, capturing 67.6% share in 2026.
How significant is the role of blood in driving sample demand in 2026?
Blood is projected to hold 47.5% share of sample type demand in 2026.
Which country is expected to record the fastest growth during the forecast period?
Canada is projected to grow at a CAGR of 8.1% during 2026 to 2036 among the listed countries.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Kit Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Kit Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Kit Type , 2026 to 2036
- HIV-only Detection Kits
- HBV-only Detection Kits
- HCV-only Detection Kits
- HIV-only Detection Kits
- Y to o to Y Growth Trend Analysis By Kit Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Kit Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sample Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sample Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sample Type, 2026 to 2036
- Blood
- Serum/Plasma-based
- Urine
- Oral Fluid/Saliva
- Dried Blood Spot (DBS)
- Blood
- Y to o to Y Growth Trend Analysis By Sample Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Sample Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Kit Type
- By Sample Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Kit Type
- By Sample Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Kit Type
- By Sample Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Kit Type
- By Sample Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Kit Type
- By Sample Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Kit Type
- By Sample Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Kit Type
- By Sample Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Kit Type
- By Sample Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Kit Type
- By Sample Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Kit Type
- By Sample Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Kit Type
- By Sample Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Kit Type
- By Sample Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Kit Type
- By Sample Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Kit Type
- By Sample Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Kit Type
- By Sample Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Kit Type
- By Sample Type
- Competition Analysis
- Competition Deep Dive
- Hoffmann-La Roche Ltd.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Bio-Rad Laboratories Inc.
- bioMérieux SA
- QIAGEN
- Meridian Bioscience, Inc.
- Hologic Inc.
- Siemens AG
- Maternova
- Creative Diagnostics
- Abbott Laboratories
- Bio-Techne
- Becton, Dickinson and Company
- Takara Bio Inc.
- Hoffmann-La Roche Ltd.
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Kit Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Sample Type, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Kit Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Sample Type, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Kit Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Sample Type, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Kit Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Sample Type, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Kit Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Sample Type, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Kit Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Sample Type, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Kit Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Sample Type, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Kit Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Sample Type, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Kit Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Kit Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Kit Type
- Figure 6: Global Market Value Share and BPS Analysis by Sample Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Sample Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Sample Type
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Kit Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Kit Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Kit Type
- Figure 23: North America Market Value Share and BPS Analysis by Sample Type, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Sample Type, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Sample Type
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Kit Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Kit Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Kit Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Sample Type, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Sample Type, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Sample Type
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Kit Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Kit Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Kit Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Sample Type, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Sample Type, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Sample Type
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Kit Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Kit Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Kit Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Sample Type, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Sample Type, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Sample Type
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Kit Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Kit Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Kit Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Sample Type, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Sample Type, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Sample Type
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Kit Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Kit Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Kit Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Sample Type, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Sample Type, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Sample Type
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Kit Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Kit Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Kit Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Sample Type, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Sample Type, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Sample Type
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

