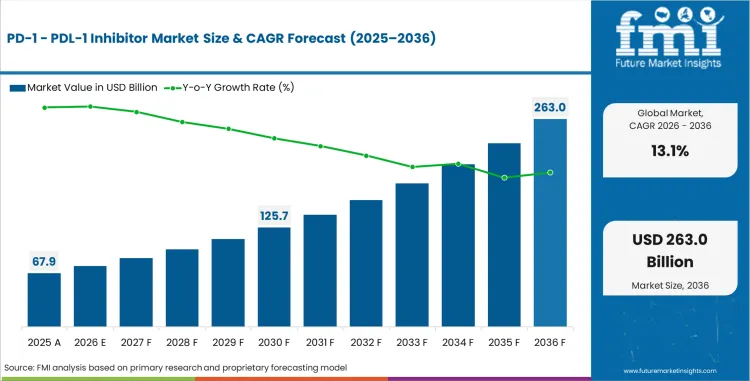
In 2026, the PD-1/PDL-1 Inhibitor market is valued at USD 67,996.6 million. Based on the provided market model, demand is estimated to grow to USD 232,839.8 million by 2036. The market is projected to expand at a CAGR of 13.1% during the forecast period.
Absolute dollar growth of USD 164,843.2 million over the decade points to an oncology category that is still expanding through label breadth, earlier-line movement, and combination therapy adoption rather than simple patient-pool replacement. The revenue build is structurally distinct from a traditional branded oncology market. It is easier to put the clamps on a PD-1 or PD-L1 in monopoly position when big tumors like NSCLC, melanoma, bladder cancer and RCC are up for grabs and growth pumps through example expansion via regimen expansion, adjuvant/perioperative migration, longer treatment in selected populations and more comfort among oncologists with checkpoint blockade across tumor boards/panel-driven care. This pattern is well illustrated by the FDA records and official labels for pembrolizumab and nivolumab, including approvals that cover melanoma, NSCLC, HNSCC, RCC, urothelial carcinoma and Merkel cell carcinoma among others.
As Dr. Marjorie Green, senior vice president and head of oncology, global clinical development at Merck Research Laboratories, stated while discussing KEYTRUDA data presented at ESMO 2025, “Our data at ESMO this year affirm the continued impact and potential of KEYTRUDA in certain types of cancer and serve as a testament to the progress we’re making in our rapidly advancing pipeline,” reflecting how major suppliers continue to frame checkpoint inhibitors as long-duration growth engines across both established and newer tumor settings. [1]
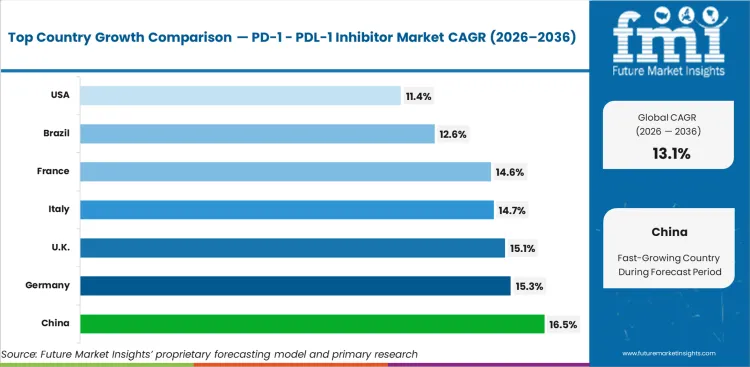
China (16.5% CAGR) is expected to lead the tracked markets, followed by South Korea (15.6%), Germany (15.3%), Japan (15.3%), and the UK (15.1%), where oncology infrastructure, broader immunotherapy access, and deeper integration into treatment algorithms continue to widen treated patient pools. France (14.6%) and Italy (14.7%) form the next tier, while Brazil (12.6%) and the USA (11.4%) remain commercially significant because of their large oncology burden and established checkpoint inhibitor use, even though the USA growth rate is being measured against a far more mature installed base.
The market covers monoclonal antibody therapies that inhibit programmed cell death protein 1 (PD-1) or programmed death-ligand 1 (PD-L1) and are used in cancer immunotherapy to restore anti-tumor immune response. It includes nivolumab, pembrolizumab, atezolizumab, avelumab, and durvalumab across approved and commercially relevant oncology settings. Revenue is generated through branded checkpoint inhibitor sales delivered primarily in institutional oncology pathways.
Commercial demand spans melanoma, non-small cell lung cancer, renal cell carcinoma, head and neck squamous cell carcinoma, bladder cancer, Merkel cell carcinoma, and a wider group of solid tumors and hematologic settings captured under “others.” This is not a retail-led oncology category. Oncologists, biomarker testing, line-of-therapy positioning, institutional reimbursement, and regulatory label strength control treatment selection.
Report consist of sizing of the market on a global and regional basis along with market forecast for 2026 to 2036. Segment-level sizing and forecasts across different product types, indication and distribution channel, country-level CAGRs for the top markets. Includes business through approved PD-1/PD-L1 agents, dispensed in institutional oncology settings (i.e., hospitals/pharmacy) and other regulatory routes: revenue generated through each cancer treatment.
The scope additionally illustrates the influence of label breadth, tumor-type coverage, combination regimen role, biomarker-directed, reimbursement access and physician familiarity across oncology practice on product preference. It is clear from company disclosures and official drug information that competition here is not confined to a single tumor type.
The scope does not consider CTLA-4 inhibitors, CAR-T therapies, antibody-drug conjugates that are obtained through targeted kinase inhibitors, chemotherapy agents, biosimilar spillover outside the defined checkpoint products. It also excludes supportive oncology care drugs that do not belong to the PD-1/PD-L1 inhibitor class. It further excludes non-commercial investigational molecules that have no market-stage revenue relevance to the forecast framework.
This separation matters, because the logic of prescription writing for checkpoint inhibitors is different from that in the larger oncology drug universe. A hospital might have multiple systemic modalities in the same tumor setting, but PD-1/PD-L1 inhibitor selection is decided by evidence for immune checkpoint engagement as well as line-specific label status, biomarker requirements and established protocol placement not merely general oncology purchasing.
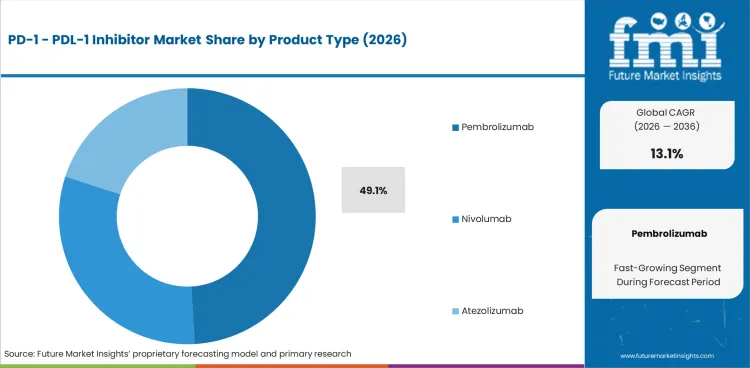
Based on the provided market model, Pembrolizumab is estimated to hold 49.1% share in 2026, followed by Nivolumab at 26.1%. Pembrolizumab leads because it has the broadest and most commercially entrenched indication footprint in the checkpoint inhibitor class. It is not simply a melanoma or NSCLC product anymore. FDA labeling spans melanoma, NSCLC, HNSCC, urothelial carcinoma, RCC, Merkel cell carcinoma, and a wider set of tumors, which gives it repeated exposure across tumor boards, infusion protocols, and reimbursement pathways. Merck’s own oncology communications continue to position KEYTRUDA as a cornerstone asset across both established and newer cancer settings.
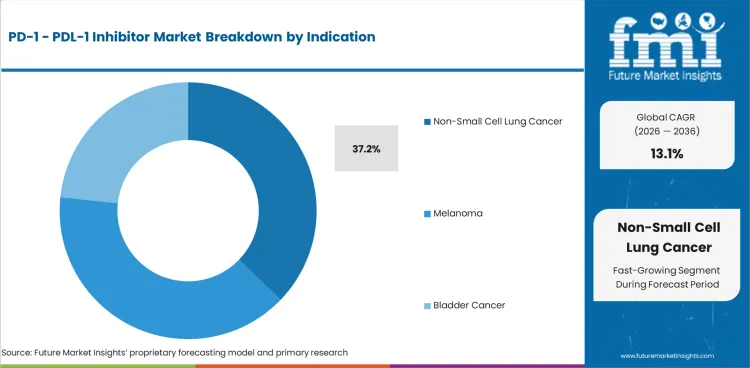
Non-Small Cell Lung Cancer accounts for 37.2% share in 2026, followed by Bladder Cancer at 23.6%. NSCLC leads because it combines a very large treated population with repeated immunotherapy use across metastatic, perioperative, and combination regimens. The checkpoint class has become deeply embedded in lung-cancer management, and recent FDA actions continue to reinforce that role in earlier-stage disease as well as advanced settings. Nivolumab’s 2024 FDA approval in neoadjuvant/adjuvant resectable NSCLC and long-standing pembrolizumab use in metastatic NSCLC both show how this indication remains central to class economics.
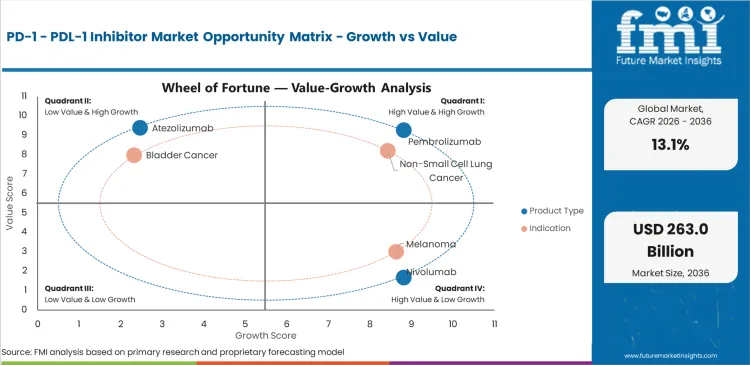
Future Market Insights analysis indicates that the industry benefits from a strong clinical migration toward immunotherapy-backed cancer management, but growth is not detached from treatment architecture. Revenue increases once checkpoint inhibitors enter into larger tumor pools, earlier stages or better defined biomarker or combination settings. Hence, commercial leadership is being constructed via depth and regulatory reach of evidence over minimum promotional thrust. FDA oncology license announcements across 2024-2026 are also suggesting that checkpoint inhibitors will reach new settings in NSCLC, urothelial carcinoma (UC), HNSCC and more.
The primary limitation is not clinical relevance; it is the required complexity of treatment and access. These agents continue to be expensive, highly protocol-driven, and very much dependent on institutional reimbursement, biomarker testing, infusion capacity, and oncologist comfort with sequencing. Competition is sharpening, too, as big incumbents protect pathway position and regional and China-based developers expand into the broader checkpoint area. The strongest opportunity lies in expanding earlier-line use, defending hospital access, and sustaining physician trust through survival data and tumor-specific differentiation rather than broad class-level claims.
Based on the regional analysis, the PD-1/PDL-1 inhibitor market is segmented into North America, Latin America, Western Europe, East Asia, and other major oncology geographies across high-value cancer-treatment markets. Regional performance is assessed using country-level demand signals tied to immunotherapy access, oncology infrastructure, regulatory adoption, hospital infusion capacity, and the speed at which checkpoint blockade is incorporated into treatment pathways.
.webp)
| Country | CAGR |
|---|---|
| US | 11.4% |
| Brazil | 12.6% |
| Germany | 15.3% |
| UK | 15.1% |
| France | 14.6% |
| Italy | 14.7% |
| China | 16.5% |
| South Korea | 15.6% |
| Japan | 15.3% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
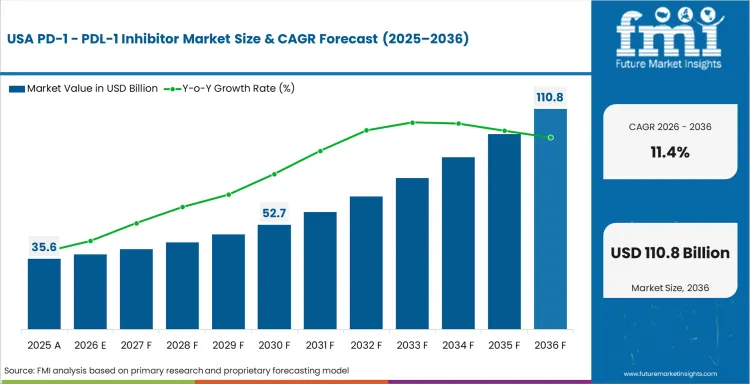
North America holds majority of the value pool because the USA has the deepest checkpoint inhibitor installed base, strong reimbursement mechanics for major oncology regimens, and extensive physician familiarity across lung cancer, melanoma, RCC, and bladder cancer. Growth is slower than in newer access-expansion markets, but this is still where pathway position matters most. Commercial success here depends on maintaining hospital and oncology-network relevance rather than creating awareness from scratch.
FMI’s report includes a detailed analysis of North America, along with a country-wise assessment that includes the USA, Canada, and Mexico. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the North America region.
East Asia is where commercial momentum and pipeline ambition are interacting most visibly. China, South Korea, and Japan are not only growth markets for imported checkpoint inhibitors, but also increasingly relevant for regional developer participation and local immuno-oncology competition. Product access, domestic innovation, and expanding oncology treatment infrastructure are all pulling in the same direction. Roche’s pipeline materials continue to frame Tecentriq as a core anti-PD-L1 asset, while Asia-based companies such as BeiGene and Akeso sit inside the broader competitive set provided for this market.
The full report analyzes the PD-1/PDL-1 Inhibitor market across East and South Asia from 2026 to 2036, covering pricing, technology adoption, and growth drivers in China, Japan, India, and adjacent regional markets.
Latin America remains more selective in access than the USA or Western Europe, yet Brazil stands out as a meaningful growth market because oncology modernization and broader uptake of innovative cancer drugs are widening the treated base. Growth is tied to reimbursement reach, institutional concentration, and the availability of specialist oncology channels that can absorb high-cost immunotherapy.
The report consists of a detailed analysis for the market in Brazil, Argentina, and Rest of Latin America. Readers can find detailed information about pricing analysis and regional trends affecting growth in the Latin America region.
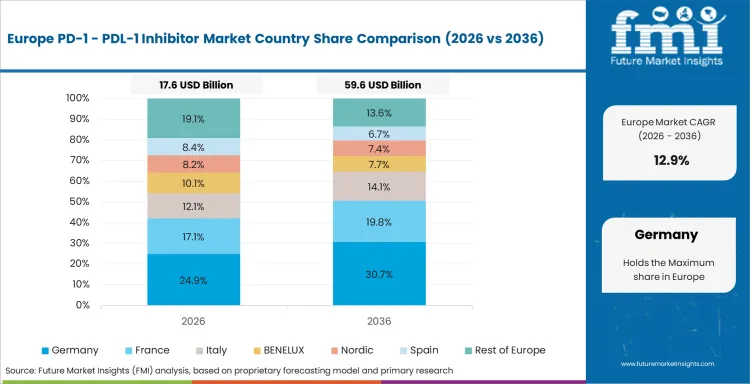
Europe remains commercially important because strong oncology infrastructure, formal treatment pathways, and wide use of immunotherapy in large solid tumors are shaping uptake. Germany, the UK, France, and Italy all show elevated growth in the provided market model, reflecting both treatment expansion and continued pathway deepening. The region is also strategically relevant for supplier milestones. Intuitive device news is irrelevant here, but in oncology AstraZeneca, Roche, BMS, and Merck all continue to treat Europe as a key market for checkpoint growth and line extension.
FMI’s analysis of the PD-1/PDL-1 inhibitor market in Europe consists of country-wise assessment that includes Germany, Italy, France, the United Kingdom, Spain, Nordic countries, Benelux, and Rest of Europe. Readers can know various regulations and latest trends in the regional market.
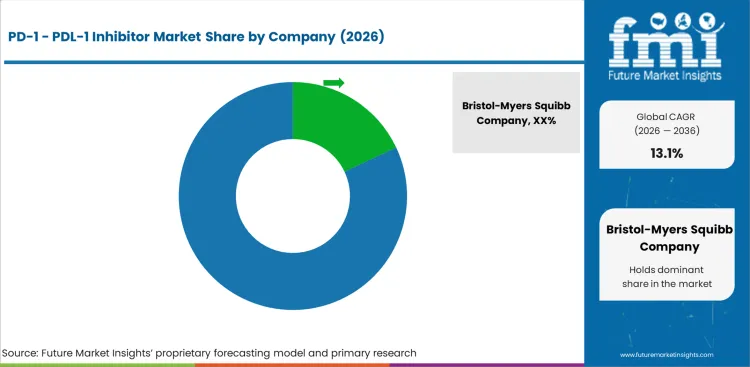
Competition appears broad at first glance, but commercial power is concentrated around a relatively small group of companies that have already secured major tumor positions and built durable hospital-oncology presence. Majority companies focuses on label breadth, survival data, combination regimen relevance, and deep familiarity among oncologists who treat NSCLC, melanoma, RCC, bladder cancer, and HNSCC every week.
Portfolio depth creates structural advantage because oncologists and hospital committees often prefer brands that can stay relevant across multiple tumors and lines rather than molecules with a narrower role. That reduces switching incentives. If one supplier already covers frontline NSCLC, perioperative lung cancer, melanoma, RCC, and selected head and neck or urothelial settings, the commercial relationship becomes much harder to dislodge. This is why pembrolizumab’s lead position in the supplied data is commercially coherent. It reflects not only sales scale, but also repeated clinical visibility across tumor boards, formularies, and national oncology discussions.
Buyer power still matters, especially because hospital pharmacies account for the largest share in the supplied model and because oncology formularies increasingly examine regimen cost, sequencing, biomarker restrictions, and treatment setting. Yet price is only one trigger. In this category, share loss usually comes when a rival secures broader label reach, better positioning in a higher-volume line of therapy, or a clearer survival advantage in a major tumor. Companies named in the current market universe include Bristol-Myers Squibb Company, Eli Lilly and Company, F. Hoffmann-La Roche Ltd., AstraZeneca Plc., Pfizer Inc., Merck KGaA, Sanofi S.A., Amgen Inc., Ono Pharmaceutical Co., Ltd., Glenmark Pharmaceuticals, Akeso Biopharma Co., Ltd., Jiangsu Alphamab Biopharmaceuticals Co., Ltd., BeiGene Ltd., Sino Biopharmaceutical Limited, and Gilead Sciences, Inc.
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:

| Metric | Value |
|---|---|
| Quantitative units | US$ 15,287.2 million (2026) to US$ 232,839.8 million (2036), at a CAGR of 13.1% |
| Market definition | The market covers monoclonal antibody therapies that inhibit programmed cell death protein 1 (PD-1) or programmed death-ligand 1 (PD-L1) and are used in cancer immunotherapy to restore anti-tumor immune response. It includes nivolumab, pembrolizumab, atezolizumab, avelumab, and durvalumab across approved and commercially relevant oncology settings. Revenue is generated through branded checkpoint inhibitor sales delivered primarily in institutional oncology pathways. Commercial demand spans melanoma, non-small cell lung cancer, renal cell carcinoma, head and neck squamous cell carcinoma, bladder cancer, Merkel cell carcinoma, and a wider group of solid tumors and hematologic settings captured under “others.” |
| Product Type Segmentation | Nivolumab, Pembrolizumab, Atezolizumab, Avelumab & Durvalumab |
| Indication Segmentation | Melanoma, Non-Small Cell Lung Cancer (NSCLC), Renal Cell Carcinoma (RCC), Head and Neck Squamous Cell Cancer (HNSCC), Bladder Cancer, Merkel Cell Carcinoma (MCC) & Others |
| Distribution Channel Coverage | Hospital Pharmacies, Retail Pharmacies, Online Pharmacies |
| Regions covered | North America, Latin America, East Asia, South Asia, Western Europe, Eastern Europe, Middle East and Africa |
| Countries covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia and 40 plus countries |
| Key companies profiled | Bristol-Myers Squibb Company, Eli Lilly and Company, F. Hoffmann-La Roche Ltd., AstraZeneca Plc., Pfizer Inc., Merck KGaA, Sanofi S.A., Amgen Inc., Ono Pharmaceutical Co., Ltd., Glenmark Pharmaceuticals, Akeso Biopharma Co., Ltd., Jiangsu Alphamab Biopharmaceuticals Co.,Ltd., BeiGene LTD, Sino Biopharmaceutical Limited, Gilead Sciences, Inc. & Others |
| Forecast period | 2026 to 2036 |
| Approach | A hybrid top-down and bottom-up market modeling approach was used, validated through primary interviews with medical oncologists, hospital pharmacists, cancer center procurement teams, and immuno-oncology manufacturers, and further supported by product portfolio mapping, indication-level treatment assessment, and distribution channel benchmarking, as per FMI. |
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
How large is the demand for PD-1/PDL-1 Inhibitor in the global market in 2026?
Demand for PD-1/PDL-1 Inhibitor in the global market is estimated to be valued at US$ 67,996.6 Mn in 2026, as per FMI.
What will be the market size of PD-1/PDL-1 Inhibitor in the global market by 2036?
Market size for PD-1/PDL-1 Inhibitor is projected to reach US$ 232,839.8 Mn by 2036.
What is the expected demand growth for PD-1/PDL-1 Inhibitor in the global market between 2026 and 2036?
Demand for PD-1/PDL-1 Inhibitor in the global market is expected to grow at a CAGR of 13.1% between 2026 and 2036.
Which product type is poised to lead global demand by 2026?
Pembrolizumab is expected to be the dominant product, capturing 29.4% share in 2026.
Which indication is expected to account for the largest share in 2026?
Non-Small Cell Lung Cancer (NSCLC) is expected to hold the highest share at 37.2% in 2026.
Which distribution channel is expected to dominate the mix in 2026?
Hospital Pharmacies is expected to lead material use with 72.6% share in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.