Radiopharmacies Market
Radiopharmacies Market Analysis Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Radiopharmacies Market Forecast and Outlook By FMI
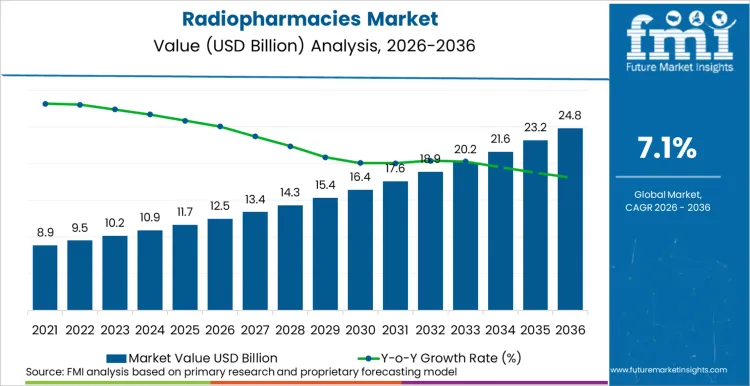
Future Market Insights anticipates the global radiopharmacies market to expand from USD 12.5 billion in 2026 to USD 24.8 billion by 2036, registering a CAGR of 7.1% over the forecast period. This expansion is being supported by steady growth in PET procedure volumes, wider clinical penetration of targeted radiotracers, and increasing institutional dependence on specialized radiopharmacy operations to maintain dose integrity within narrow time windows. According to FMI’s analysis, PET radiopharmacy continues to represent the largest radiopharmacy type, reflecting the procedural backbone created by PET imaging demand across oncology, neurology, and cardiology workflows.
FMI emphasizes centralization and compliance-based operating models as the essential structural shift anticipating 2036. Commercialized, centralized service models have continued as the mainstream backbone for distributing services, sustained by scale efficiency, standardized cGMP measures, and enhanced geographic footprint. At the same time, the tracer blend is adding a growing dimension of complexity as the mix shifts beyond the core 18F-FDG base toward PSMA, neuro (Amyloid, Tau), and DOTATATE/NET, raising scheduling, batch management, and regulatory documentation considerations. The regulatory sphere is raising the competitive bar as well, where commercialized cGMP operations have remained the essential backbone for scalable delivery, while hospital-based nuclear pharmacy and investigational clinical trial handling have played a critical role for niche footprint and first-to-market initiatives.
Summary of Radiopharmacies Market
- The radiopharmacies market is projected to expand at a CAGR of 7.1% from 2026 to 2036.
- Market value is expected to increase from USD 12.5 Billion in 2026 to USD 24.8 Billion by 2036.
- Growth is driven by sustained PET and SPECT procedure volumes, rising theranostics-linked radiotracer demand, and stricter delivery-time expectations supporting local production and distribution networks.
- FMI analysis is based on historical market size, radiopharmacy type mix, radiotracer utilization by scan volume, service-model penetration, and country-level nuclear medicine capacity indicators.
- Market evolution is shaped by centralization economics, compliance intensity, batch quality standardization, and on-time delivery performance.
- PET expansion and broader radiotracer portfolios-including PSMA, neuro tracers, and NET tracers-are strengthening routine clinical workflows beyond traditional FDG demand.
- The United States holds the largest share of the global radiopharmacies market due to high PET utilization, strong imaging center density, and well-established commercial radiopharmacy networks.
- Country-level assessment included nuclear medicine procedure volumes, PET infrastructure depth, commercial radiopharmacy footprint, and regulatory handling practices.
- By 2036, the global radiopharmacies market is projected to reach USD 24.8 Billion.
- Compliance-driven growth is being reinforced by tightening cGMP requirements, audit-ready documentation practices, centralized production scaling, and increasing hospital reliance on external radiopharmacy supply networks.
Recent developments from leading industry players underscore these shifts. In March 2025, Curium announced the completion of its acquisition of Eczacıbaşı-Monrol Nuclear Products Co. (Monrol), positioning the deal as a step-change in Lu-177 manufacturing scale and an expansion of PET and SPECT geographic coverage. Reflecting this direction, Curium’s CEO of International Markets Mr. Chaitanya Tatineni, said, “We are delighted to welcome close to 400 highly-skilled and dedicated new colleagues to Curium Group. The coming together of Curium and Monrol will transform our scale, capabilities and reach across several critical areas, in particular Lu-177, and create multiple new opportunities to enhance our offering of life-changing diagnostic and therapeutic solutions to patients around the world.”
Future Market Insights highlights that service differentiation is being driven by the ability to execute reliable, compliant, and time-sensitive supply at scale across hospitals and imaging centers, while supporting an expanding tracer mix and stricter chain-of-custody expectations. With hospitals and imaging centers jointly accounting for the majority of demand, providers that combine centralized cGMP rigor with responsive distribution models are expected to strengthen positioning through 2036, particularly as theranostics and specialty PET tracers continue to widen the operational requirements of routine supply.
Radiopharmacies Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 12.5 Billion |
| Industry Value (2036) | USD 24.8 Billion |
| CAGR (2026 to 2036) | 7.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Why is the Demand for Radiopharmacies Market Growing?
Demand for radiopharmacies is growing as nuclear medicine workflows continue to scale-up around time-sensitive tracer availability, compliance-led batch release discipline, and expanding clinical utilization driven by PET. Demand is being anchored within PET radiopharmacy, reflecting the sustained dominance of PET within routine imaging pathways, while SPECT radiopharmacy continues to hold a durable role within established diagnostic practice. Commercial centralized service models operational pull is being supported by predictable production economics and multisite distribution needs with incremental dependence upon hub-and-spoke networks in balancing reach with timing constraints. Utilization concentrated within 18F-FDG is being broadened by PSMA, neuro tracers, and DOTATATE/NET being integrated into routine ordering patterns. Commercial cGMP compliance dominates the handling requirements, reinforcing standardization in production and documentation. Demand from end-users is being led by hospitals and imaging centers, reflecting procedural concentration and repeat ordering cadence in sustaining radiopharmacy throughput.
How is the Radiopharmacies Market Segmented?
The radiopharmacies market is segmented by radiopharmacy type, service model, radiotracer class, regulatory handling, and end user. By radiopharmacy type, the market includes PET radiopharmacy, SPECT radiopharmacy, integrated cyclotron + radiopharmacy, and hospital in-house. Based on service model, demand is categorized into commercial centralized, hub-and-spoke network, hospital-based, and research / academic models. By radiotracer class, the market spans 18F-FDG, PSMA, neuro (amyloid / tau), DOTATATE / NET, cardiac perfusion, and other. By regulatory handling, operations are segmented into cGMP commercial, hospital nuclear, and clinical trial investigational. By end user, demand is segmented into hospitals, imaging centers, and research institutes.
How Do PET Radiopharmacy Secure Market Leadership?
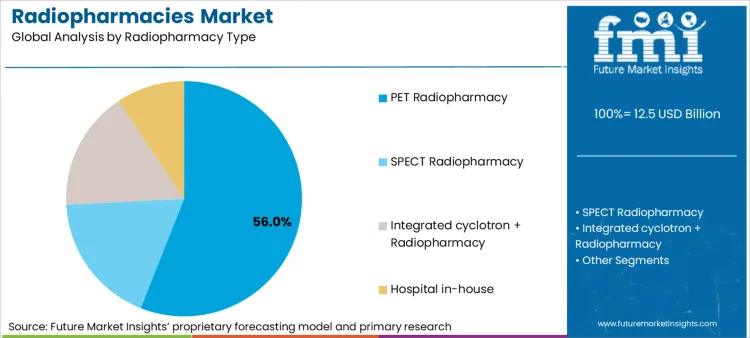
As per Future Market Insights, PET Radiopharmacy holds the largest market share in the global radiopharmacies market, mainly because of the ADE measures linked to the throughput intensity and time-sensitive dispatch demands generally required for PET radiotracers. As estimated, PET Radiopharmacy holds a total radiopharmacy type demand share of 56.0%, and it is mainly driven by daily routine cycles, quality release, and high utilization rates for oncology, cardiology, and neurology examinations. PET services are usually centered on standard synthesis, quality release, and scheduled dispatch windows, which may facilitate multi-sited servicing.
Another factor is ongoing dominance with respect to infrastructural investment. In contrast, PET radiopharmacy is often developed from a perspective of spatial centralization or networking wherein access to cyclotrons, cleanroom validation, and radiochemist skill sets can be consolidated. As PET procedures continue to grow exponentially, the demand for PET radiopharmacy continues to remain the driving factor for radiopharmacy, both at the commercial level and other institutional levels.
How Does 18F-FDG Lead Radiopharmacy Demand?
As per FMI, 18F-FDG as the leading radiotracer class, accounting for 34.0% of overall radiotracer demand handled through radiopharmacy operations. 18F-FDG has remained the backbone radiotracer for routine PET imaging, creating stable baseline production volume and predictable scheduling across daily distribution routes. Its widespread clinical utility drives consistent ordering patterns, supporting operational scale advantages for sites running multiple synthesis and dispensing cycles.
Demand leadership is also supported by workflow standardization. The 18F-FDG production and release processes are highly systematized and allow repeatable quality control with distribution in a reliable manner within the restrictions imposed by the short half-life. During this time, 18F-FDG has remained the principal volume anchor shaping radiopharmacy utilization and capacity planning in the continuous prioritization of high-throughput PET services in hospitals and imaging centers.
How Are Regulatory Frameworks Influencing Safer Radiopharmacy Operations?
The expectations surrounding aseptic procedures, batch release documents, and radiation safety are increasingly influencing the operating models within radiopharmacies. This is, in turn, raising the profile of cGMP commercial handling, where validated cleanroom procedures and batch release processes can be routinely maintained. At the same time, hospital-based nuclear operations are being challenged to improve their overall governance models, especially when dealing with high volume patients and multiple tracers. Furthermore, as the issue of compliance continues to raise its profile, procurement models for nuclear reactors are being influenced by audit, deviation, and traceability rather than demonstrable production capabilities.
Why Is Continuous Manufacturing Reshaping Demand Patterns?
This growing trend of automation-oriented, enumerable cycles of productive processes is again changing the dimension of the demands for radiopharmacy chains. The windows of delivery, along with the constraints on short half-lives, have forced radiopharmacies to prefer daily cycles of tight schedules, rather than preparing these by a batch-based, site-specific approach. The need for centralization of capacities, utilizing the hub-and-spoke approach, has formed a critical dimension of this trend. Compatibility with rapid QC release, standardized synthesis modules, and closed-system dispensing has become a key selection factor when new capacity is added. As a result, service demand is increasingly being allocated toward operators that can run sustained daily output with predictable release timing and minimized batch variability.
How Are Digital Tools and Advanced Radiotracers Driving Innovation?
Digital tools are increasingly being used to improve scheduling discipline and end-to-end visibility across radiopharmacy operations. Dose forecasting, site-wise allocation, and release-to-delivery tracking are being integrated to reduce wastage and protect on-time delivery performance, particularly for multi-site imaging networks. At the same time, portfolio expansion in advanced radiotracers is changing operating requirements. Growth in PSMA, neuro and workflows is increasing the need for flexible synthesis capability, tracer-specific QC pathways, and controlled handling practices that can be repeated reliably across batches. As tracer diversity rises, innovation is being pulled toward digital orchestration and modular production systems that can maintain compliance while scaling multi-tracer output.
How Will Radiopharmacies Market Expansion Unfold Across Key Global Regions?
Future Market Insights identifies the United States (CAGR ~7.6%), Germany (~6.1%), the United Kingdom (~6.4%), France (~6.0%), Japan (~5.8%), China (~8.9%), and South Korea (~7.1%) as the core demand drivers shaping radiopharmacy expansion globally. The United States leads through high PET utilization, dense imaging infrastructure, and a mature commercial centralized radiopharmacy model that supports rapid, compliant distribution to hospitals and imaging centers. Again, Germany, the UK, and France keep European demand anchored due to strong nuclear medicine adoption, regulated operational frameworks where cGMP-compliant handling and traceability requirements are being stressed upon. Japan's demand is being undergirded by stable nuclear medicine volumes, disciplined clinical use of PET and SPECT tracers, and high compliance expectations in hospital-linked radiopharmacy operations. It is seen that rapid build-out of PET capability is accelerating growth in China, increasing uptake of oncology imaging and expanding installation of cyclotrons and PET/CT systems is supportive of localized radiotracer supply. That, coupled with strong nuclear medicine capability, expanding PET radiopharmacy capacity, and continued investment in compliant production and distribution systems, acts as a propellant for South Korea. FMI stresses the point that these countries represent a combination of imaging capacity expansion and cGMP-driven operational controls, giving rise to structurally resilient radiopharmacy demand through 2036.
Radiopharmacies Market Regional CAGR (2026 to 2036)
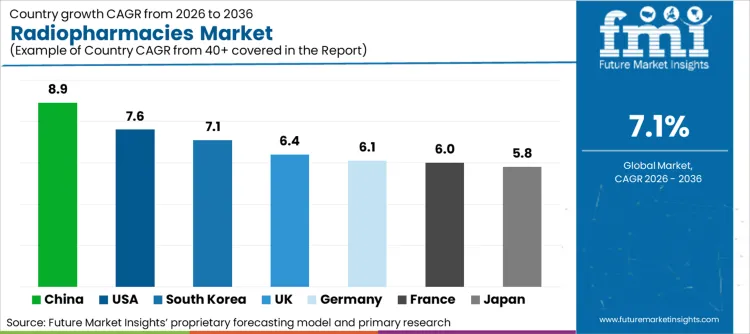
| Country | CAGR (2026 to 2036) |
|---|---|
| United States | 7.6% |
| Germany | 6.1% |
| United Kingdom | 6.4% |
| France | 6.0% |
| Japan | 5.8% |
| China | 8.9% |
| South Korea | 7.1% |
Is Commercial Centralization Redefining Radiopharmacy Scaling in the United States?
The United States market for radiopharmacies is expected to grow at a 7.6% CAGR, and the main factors that are catalyzing this market’s growth include high volumes of procedures carried out involving PET, and the maturity of commercialized radiopharmacy operational forms that can serve a widely expanded geography. The need for these services is being fueled by more dependence on scheduled and validated supply of radiotracers, particularly within the hospital and imaging sector, instead of production mainly for short-half-life PET tracers, on account of logistical necessity. According to FMI, the market is being propelled by increased application of PET in cancer and cardiovascular diagnostics, and increasing preference for cGMP handling and traceability. The market is expected to be supported over a sustained period due to increasing diversification of radiologists and growing operational reliability.
Does Germany’s Regulatory Rigor Sustain High-Value Radiopharmacy Demand?
According to Future Market Insights, Germany’s radiopharmacies market is projected to expand at a 6.1% CAGR, shaped by strong nuclear medicine adoption and rigorous operational expectations around quality systems and compliant handling. Demand is being reinforced as radiotracer preparation and distribution increasingly require standardized documentation, validated processes, and consistent product integrity across hospital and commercial networks. FMI analysts observe that growth is being supported by steady PET and SPECT utilization and by disciplined investment in compliant production environments. Expansion is expected to remain stable, driven more by value and compliance intensity than by rapid volume escalation.
How Is the United Kingdom’s Networked Distribution Model Influencing Market Growth?
The United Kingdom radiopharmacies market is expected to grow at rate of 6.4% CAGR. The use of PET and SPECT diagnostics is expected to show continued strong support. The benefits of a well-established and strong hub and spoke distribution model are also expected to support growth. FMI analysts explain that the radiopharmacies market in the United Kingdom is driven by the need for operational efficiency and the increased demand for a standard approach to the production of radiopharmacicals.
Is France’s Compliance-Led Operational Structure Supporting Stable Radiopharmacy Expansion?
The French radiopharmacies market is projected to grow at a CAGR of 6.0 %, by the stability of nuclear medicine usage and the overall structure of regulatory requirements for the correct handling of radiopharmaceuticals. The demand for radiopharmacies is additionally underpinned in the French market through the hospital and imaging center demand for continuous supplies of tracers within a system of validated preparation and documentation. Analysts from FMI drew attention to the reality of the French radiopharmacies market growth, citing the support of continued growth of PET radiopharmacies and the relative increase in the number of centralized or integrated collection models.
Does Japan’s Discipline in Nuclear Medicine Practice Sustain Long-Term Demand?
According to a study by Future Market Insights, the Japanese market is expected to grow at a rate of 5.8% CAGR, influenced by nuclear medicine volumes, a tightly regulated clinical environment, and a high degree of institutionalization of quality and reliability. The market is reportedly boosted by the continuation of hospital-based radiopharmacies associated practices and their importance to trace and distribute nuclear medicine, especially when operational consistency is a growing imperative. The analysts at FMI have outlined that the market is growing because of consistent practices of PET and SPECT used for cancer and neurologic conditions, and a tightly managed progression of the variety of nuclear medicine used. The rate of expansion is steady.
How Is China’s PET Infrastructure Build-Out Accelerating Radiopharmacy Demand?
According to Future Market Insights, China’s radiopharmacies market is projected to expand at an 8.9% CAGR, underpinned by rapid expansion of PET imaging capability and rising uptake of oncology-focused nuclear diagnostics. Demand is being reinforced as new radiopharmacy capacity is being added to support localized production and timely distribution, especially for short half-life tracers that require precise delivery coordination. FMI analysts observe that growth is being supported by expanding installation of cyclotron and PET/CT infrastructure and increasing attention to operational compliance and standardized handling. Market expansion is expected to remain structurally supported as imaging access widens and radiotracer supply networks scale through 2036.
Is South Korea’s Advanced Nuclear Medicine Capability Supporting Sustained Market Momentum?
Future Market Insights analysts note that South Korea’s radiopharmacies market is expected to grow at a 7.1% CAGR, driven by strong nuclear medicine capability and continued investment in PET radiopharmacy infrastructure. Demand is being reinforced as hospitals and imaging centers expand tracer utilization and require dependable, compliant supply with high operational precision. FMI highlights that growth is being supported by expanding PET radiopharmacy capacity and increasing emphasis on controlled production environments and distribution reliability. Expansion is expected to remain steady as radiotracer portfolios evolve and as compliant supply systems deepen across advanced nuclear medicine networks.
What Strategic Moves Are Defining Competitive Landscape?
As per FMI, the level of competition in the radiopharmacies space is increasingly driven by the solutions offered in regulatory handling, density, and radiotracer portfolio, rather than the solutions offered merely from a dispensing standpoint. The top players are investing in the construction of PET radiopharmacy infrastructure adjacent to high-throughput imaging routes in an effort to decrease losses owing to radioactive decay and enhance traceability and timeliness. This trend is also supporting the viability of commercialized centralized operations.
Another key move in defining a company strategy is the scaling of service networks through commercial centralized and hub-and-spoke-type network strategies. Cardinal Health and Curium, are using their distribution footprints and operational standardization as differentiating elements for competition. Meanwhile, hospital-based and hospital in-house models have been maintained by these companies in certain systems, where scheduling, dosing, and investigational supply need to remain in control.
FMI also emphasizes strategic expansion throughout the radiotracer types and managerial regulation levels. Though emphasis is being placed on the reliability of 18F-FDG, the breadth of the PSMA, neuro, and DOTATATE/NET types is still growing for oncology and neurology growth. Determining competitive positions will be driven by capabilities to manage under cGMP commercial, hospital nuclear, and clinical trial investigational models, where documentation, QA release discipline, and validated cold chain and radiation safety processes are managed as procurement criteria. This is changing the dynamic of competition to a compliant service systems structure, helping those players who have considerable network capabilities, regulation discipline, and partnership potential for production capabilities through 2036.
Recent Developments:
- In June 2025, PharmaLogic Holdings Corp., a leading radiopharmaceutical CDMO, has completed its acquisition of a majority stake in Agilera Pharma AS from Norway’s Institute for Energy Technology. The deal strengthens PharmaLogic’s capabilities in development, manufacturing, and global distribution of therapeutic radiopharmaceuticals, expanding its international footprint. With Agilera’s commercial supply to over 30 countries, the acquisition advances PharmaLogic’s vision to build the first fully integrated global CDMO platform dedicated to radiopharmaceutical therapeutics.
- In September 2025, DHL Supply Chain has agreed to acquire SDS Rx, a USA-based provider of specialized final-mile healthcare delivery, strengthening its life sciences and healthcare capabilities under DHL Health Logistics. The deal expands DHL’s same-day, expedited, and specialty pharmacy logistics network across 200+ locations. Following its 2025 acquisition of CryoPDP, the move accelerates DHL’s Strategy 2030 growth agenda, enhancing cold chain, specialty pharma, and patient-centric logistics solutions across North America.
- In January 2025, Telix Pharmaceuticals Limited has completed its US$230 million acquisition of RLS (USA) Inc., America’s only Joint Commission-accredited radiopharmacy network. The deal adds 30+ USA radiopharmacies and strengthens Telix’s integrated radiopharmaceutical ecosystem, expanding in-house manufacturing, cyclotron capabilities, and nationwide distribution. RLS will operate independently within Telix Manufacturing Solutions, accelerating access to innovative diagnostic and therapeutic radiopharmaceuticals across the USA.
Key Players in Radiopharmacies Market
- Cardinal Health
- Curium
- Jubilant Radiopharma
- Siemens Healthineers
- Lantheus
- GE HealthCare
- Eckert & Ziegler
- Bracco Imaging
- SOFIE
- IBA
Market Definition
The radiopharmacies market refers to the global industry involved in the production, preparation, quality release, distribution, and dispensing of radiopharmaceutical doses used in diagnostic imaging and radiotheranostic workflows. Radiopharmacies play a critical role in supplying time-sensitive radiotracers to hospitals and imaging centers while maintaining radiation safety controls, validated handling procedures, and regulated quality systems. The market size is measured in USD billion and analyzed over the 2026 to 2036 forecast period.
The market includes radiopharmacy operations segmented by Radiopharmacy Type, Service Model, Radiotracer Class, Regulatory Handling, and End User. Radiopharmacy types encompassed within the market include PET radiopharmacy, SPECT radiopharmacy, integrated cyclotron + radiopharmacy, and hospital in-house models. Service delivery is provided through commercial centralized, hub-and-spoke network, hospital-based, and research / academic models. Radiotracer classes included in the market comprise 18F-FDG, PSMA, neuro (amyloid tau), DOTATATE / NET, cardiac perfusion, and other tracer categories. Regulatory handling levels covered include cGMP commercial, hospital nuclear, and clinical trial investigational operations. End users include hospitals, imaging centers, and research institutes.
The market excludes radiopharmaceutical API manufacturing not associated with radiopharmacy dose preparation and dispensing, nuclear imaging equipment sales, cyclotron hardware sold independently of radiopharmacy services, non-medical industrial radioisotope handling, and laboratory radioassay activities unrelated to patient-ready radiotracer dose supply. Also excluded are purely therapeutic radiopharmaceutical administrations delivered without radiopharmacy distribution service revenue attribution.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 24.8 Billion |
| Radiopharmacy Type | PET radiopharmacy, SPECT radiopharmacy, Integrated cyclotron + radiopharmacy, Hospital in-house |
| Service Model | Commercial centralized, Hub-and-spoke network, Hospital-based, Research / academic |
| Radiotracer Class | 18F-FDG, PSMA, Neuro (amyloid tau), DOTATATE / NET, Cardiac perfusion, Other |
| Regulatory Handling | cGMP commercial, Hospital nuclear, Clinical trial investigational |
| End User | Hospitals, Imaging centers, Research institutes |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | USA, Germany, UK, France, Japan, China, South Korea, and 40+ countries |
| Key Companies Profiled | Cardinal Health, Curium, Jubilant Radiopharma, Siemens Healthineers, Lantheus, GE HealthCare, Eckert & Ziegler |
| Additional Attributes | Revenue analysis by radiopharmacy type and radiotracer class, assessment of service model economics, evaluation of regulatory handling capability as a procurement gate, competitive positioning based on network reach and supply reliability |
Radiopharmacies Market by Segments
By Radiopharmacy Type:
- PET Radiopharmacy
- SPECT Radiopharmacy
- Integrated cyclotron + Radiopharmacy
- Hospital in-house
By Service Model:
- Commercial centralized
- Hub-and-Spoke Network
- Hospital-based
- Research / academic
By Regulatory Handling:
- cGMP commercial
- Hospital nuclear pharmacy
- Clinical trial / investigational
By End User:
- Hospitals
- Diagnostic Imaging Centers
- Research institutes
By Region:
- North America
- Europe
- East Asia
- South Asia
- Latin America
- Middle East & Africa
Bibliography
- International Atomic Energy Agency. (2024). Radiopharmacy Operations and Quality Assurance for PET and SPECT Radiopharmaceuticals. IAEA.
- USA Food and Drug Administration. (2024). cGMP Requirements for PET Drugs and Radiopharmaceutical Manufacturing and Handling. FDA.
- European Medicines Agency. (2024). Guidance on Radiopharmaceutical Quality, Manufacture, and Control for Clinical Use. EMA.
- European Directorate for the Quality of Medicines & HealthCare. (2024). European Pharmacopoeia: Monographs and General Chapters for Radiopharmaceutical Preparations. EDQM.
- World Health Organization. (2023). WHO Technical Guidance on Safe Handling, Preparation, and Distribution of Radiopharmaceuticals. WHO.
- Society of Nuclear Medicine and Molecular Imaging. (2024). Practice Standards for PET/SPECT Radiopharmaceutical Preparation, Dispensing, and Administration. SNMMI.
Frequently Asked Questions
What is the current global market size for radiopharmacies?
The global radiopharmacies market is valued at USD 12.5 billion in 2026, reflecting expanding PET and SPECT throughput and increasing reliance on specialized preparation and distribution infrastructure.
What is the projected CAGR for the market over the next 10 years?
The market is projected to grow at a 7.1% CAGR from 2026 to 2036, supported by wider radiotracer utilization in oncology, cardiology, and neuroimaging workflows.
Which regions are experiencing the fastest expansion?
China is experiencing the fastest expansion at 8.9%, supported by accelerating molecular imaging installations, expanding radiopharmacy licensing, and increased clinical adoption of PET radiopharmacy models.
What are the primary market drivers?
Growth is being driven by rising PET radiopharmacy dominance, increasing centralized commercial models, expanding use of 18F-FDG and next-generation tracers, and stronger regulatory emphasis on cGMP-aligned handling.
Who are the leading providers in the industry?
Cardinal Health, Curium, and Jubilant Radiopharma are among leading providers, differentiated through large distribution footprints, cold-chain and time-critical logistics capability, and compliance-led operational controls.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Radiopharmacy Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Radiopharmacy Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Radiopharmacy Type , 2026 to 2036
- PET Radiopharmacy
- SPECT Radiopharmacy
- Integrated cyclotron + Radiopharmacy
- Hospital in-house
- PET Radiopharmacy
- Y to o to Y Growth Trend Analysis By Radiopharmacy Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Radiopharmacy Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Radiopharmacy Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Radiopharmacy Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Radiopharmacy Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Radiopharmacy Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Radiopharmacy Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Radiopharmacy Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Radiopharmacy Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Radiopharmacy Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Radiopharmacy Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Radiopharmacy Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Radiopharmacy Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Radiopharmacy Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Radiopharmacy Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Radiopharmacy Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Radiopharmacy Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Radiopharmacy Type
- Competition Analysis
- Competition Deep Dive
- Cardinal Health
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Curium
- Jubilant Radiopharma
- Siemens Healthineers
- Lantheus
- GE HealthCare
- Eckert & Ziegler
- Bracco Imaging
- SOFIE
- Cardinal Health
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Radiopharmacy Type , 2021 to 2036
- Table 3: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Radiopharmacy Type , 2021 to 2036
- Table 5: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: Latin America Market Value (USD Million) Forecast by Radiopharmacy Type , 2021 to 2036
- Table 7: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Western Europe Market Value (USD Million) Forecast by Radiopharmacy Type , 2021 to 2036
- Table 9: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Eastern Europe Market Value (USD Million) Forecast by Radiopharmacy Type , 2021 to 2036
- Table 11: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: East Asia Market Value (USD Million) Forecast by Radiopharmacy Type , 2021 to 2036
- Table 13: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: South Asia and Pacific Market Value (USD Million) Forecast by Radiopharmacy Type , 2021 to 2036
- Table 15: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Middle East & Africa Market Value (USD Million) Forecast by Radiopharmacy Type , 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Radiopharmacy Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Radiopharmacy Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Radiopharmacy Type
- Figure 6: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Region
- Figure 9: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 10: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 11: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 12: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 17: North America Market Value Share and BPS Analysis by Radiopharmacy Type , 2026 and 2036
- Figure 18: North America Market Y-o-Y Growth Comparison by Radiopharmacy Type , 2026-2036
- Figure 19: North America Market Attractiveness Analysis by Radiopharmacy Type
- Figure 20: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 21: Latin America Market Value Share and BPS Analysis by Radiopharmacy Type , 2026 and 2036
- Figure 22: Latin America Market Y-o-Y Growth Comparison by Radiopharmacy Type , 2026-2036
- Figure 23: Latin America Market Attractiveness Analysis by Radiopharmacy Type
- Figure 24: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 25: Western Europe Market Value Share and BPS Analysis by Radiopharmacy Type , 2026 and 2036
- Figure 26: Western Europe Market Y-o-Y Growth Comparison by Radiopharmacy Type , 2026-2036
- Figure 27: Western Europe Market Attractiveness Analysis by Radiopharmacy Type
- Figure 28: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: Eastern Europe Market Value Share and BPS Analysis by Radiopharmacy Type , 2026 and 2036
- Figure 30: Eastern Europe Market Y-o-Y Growth Comparison by Radiopharmacy Type , 2026-2036
- Figure 31: Eastern Europe Market Attractiveness Analysis by Radiopharmacy Type
- Figure 32: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: East Asia Market Value Share and BPS Analysis by Radiopharmacy Type , 2026 and 2036
- Figure 34: East Asia Market Y-o-Y Growth Comparison by Radiopharmacy Type , 2026-2036
- Figure 35: East Asia Market Attractiveness Analysis by Radiopharmacy Type
- Figure 36: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 37: South Asia and Pacific Market Value Share and BPS Analysis by Radiopharmacy Type , 2026 and 2036
- Figure 38: South Asia and Pacific Market Y-o-Y Growth Comparison by Radiopharmacy Type , 2026-2036
- Figure 39: South Asia and Pacific Market Attractiveness Analysis by Radiopharmacy Type
- Figure 40: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Middle East & Africa Market Value Share and BPS Analysis by Radiopharmacy Type , 2026 and 2036
- Figure 42: Middle East & Africa Market Y-o-Y Growth Comparison by Radiopharmacy Type , 2026-2036
- Figure 43: Middle East & Africa Market Attractiveness Analysis by Radiopharmacy Type
- Figure 44: Global Market - Tier Structure Analysis
- Figure 45: Global Market - Company Share Analysis

