Temperature Controlled Packaging Solution Market
Temperature Controlled Packaging Solution Market is segmented by Product Type (Passive System, Active System), Payload Capacity (Small (<20 Liters), Medium (20–150 Liters), Large (>150 Liters)), Material Type (Polymer-Based Materials, Glass, Metal, Others), and Region. Forecast for 2026 to 2036.
Temperature Controlled Packaging Solution Market Size, Market Forecast and Outlook By FMI
Summary of the Temperature Controlled Packaging Solution Market
- Demand and Growth Drivers
- Pharmaceutical cold chain expansion driven by biologic drug growth, vaccine distribution logistics, and cell and gene therapy requirements is sustaining demand for validated temperature controlled packaging across the 2 to 8 degree Celsius and ultra-cold temperature ranges.
- Food safety regulation tightening and consumer demand for fresh product quality are driving procurement of cold chain packaging for meat, dairy, seafood, and fresh produce distribution through increasingly complex supply chains.
- E-commerce growth in perishable food and pharmaceutical home delivery is creating new last mile cold chain packaging requirements for smaller shipment sizes, direct to consumer formats, and multi-temperature delivery models.
- Product and Segment View
- Passive System holds 57.0% of the product type segment in 2026, driven by cost effectiveness and reliability of insulated shippers, phase change material systems, and vacuum insulated panels for pharmaceutical and food cold chain logistics.
- Healthcare accounts for 43.6% of the end use segment, reflecting pharmaceutical drug, vaccine, biological sample, and cell therapy cold chain requirements that demand validated temperature maintenance during global distribution.
- Polymer-Based Materials dominate the material type segment, with expanded polystyrene and polyurethane insulation remaining the primary packaging materials despite growing sustainability pressure for recyclable alternatives.
- Geography and Competitive Outlook
- India is expected to register a CAGR of 7.3% through 2036, driven by pharmaceutical manufacturing growth, national vaccine distribution infrastructure development, and cold chain logistics modernization across food and healthcare sectors.
- China at 7.1% reflects pharmaceutical cold chain expansion, food safety regulation enforcement, and e-commerce perishable delivery growth creating new packaging demand categories.
- Sonoco Products Company and Pelican BioThermal lead through comprehensive cold chain packaging portfolios with pharmaceutical validation services and global distribution networks.
- Analyst Opinion
- 'Temperature controlled packaging is evolving from commodity insulation toward intelligent, validated cold chain solutions with integrated monitoring and sustainability features. Companies that combine packaging engineering with real-time temperature tracking, regulatory compliance documentation, and sustainable material alternatives are positioned to capture premium pricing,' says Ismail Sutaria, Managing Director at FMI.
- The market is shifting toward reusable and sustainable packaging solutions as pharmaceutical companies and food distributors face increasing pressure to reduce single use cold chain packaging waste and associated environmental impact.
- Adoption of phase change material systems is increasing due to superior temperature maintenance performance, longer protection duration, and reusability compared to traditional gel pack and dry ice approaches.
- Demand is further shaped by cell and gene therapy logistics requirements that need ultra-cold packaging solutions capable of maintaining temperatures below minus 60 degrees Celsius during global transit, creating a premium product category.
Temperature Controlled Packaging Solution Market Definition
The Temperature Controlled Packaging Solution Market covers insulated containers, phase change materials, refrigerated systems, and associated monitoring devices used to maintain temperature sensitive products within specified ranges during storage and transportation. Products include passive systems (insulated shippers, insulated boxes, phase change material based systems, vacuum insulated panels) and active systems (refrigerated containers, refrigerated trucks and vans, electrically powered units).
Temperature Controlled Packaging Solution Market Inclusions
Market scope includes products classified by product type (passive system, active system), payload capacity (small under 20 liters, medium 20 to 150 liters, large over 150 liters), material type (polymer-based materials including EPS, polyurethane, polypropylene, polyethylene; glass; metal), temperature range (frozen at minus 20 degrees and below, chilled 2 to 8 degrees, ambient 15 to 25 degrees), and end use (healthcare including pharmaceuticals, vaccines, biological samples; food and beverages including fresh produce, meat, poultry, seafood). Revenue coverage spans 2026 to 2036.
Temperature Controlled Packaging Solution Market Exclusions
The scope does not include permanent cold storage warehouse equipment, industrial refrigeration compressor systems, or general purpose insulation materials not designed specifically for packaging and transportation applications.
Temperature Controlled Packaging Solution Market Research Methodology
- Primary Research: FMI analysts conducted interviews with industry participants, technology providers, and end users across key markets.
- Desk Research: Combined data from industry associations, regulatory filings, and manufacturer disclosures.
- Market Sizing and Forecasting: Bottom up aggregation across segments and regions with top down validation against industry benchmarks.
- Data Validation: Cross checked quarterly against industry production data, procurement records, and manufacturer disclosures.
Why is the Temperature Controlled Packaging Solution Market Growing?
- Biologic drug pipeline growth and expanded vaccine distribution programs are creating sustained demand for validated pharmaceutical cold chain packaging that maintains 2 to 8 degree Celsius conditions throughout increasingly complex global distribution networks.
- Last mile cold chain for e-commerce perishable delivery represents a rapidly growing application as direct to consumer food and pharmaceutical delivery models require specialized small format temperature controlled packaging solutions.
Demand for temperature controlled packaging reflects the expanding scope of products requiring cold chain logistics across pharmaceutical, food, and specialty chemical sectors. Pharmaceutical cold chain requirements are growing as biologic drugs, which require refrigerated storage and transport, represent an increasing share of the global pharmaceutical pipeline and approved drug portfolio. Vaccines, blood products, and diagnostic specimens add further volume to healthcare cold chain packaging demand, with seasonal vaccination programs and pandemic preparedness creating variable but significant procurement cycles.
Food cold chain packaging demand is expanding as safety regulations tighten, consumer quality expectations rise, and e-commerce perishable delivery grows. Regulatory requirements for temperature documentation during food transport are driving adoption of validated packaging solutions with integrated temperature monitoring and data logging capabilities. The shift toward online grocery, meal kit delivery, and direct to consumer perishable food distribution creates new packaging requirements for smaller shipment sizes that must maintain cold chain integrity through last mile delivery with variable transit times.
Sustainability pressures are reshaping packaging material and design choices across the cold chain industry. Single use expanded polystyrene containers face regulatory restrictions in several markets and growing customer preference for recyclable or reusable alternatives, driving transition toward returnable container systems, recyclable insulation materials, and phase change material solutions that reduce waste while maintaining or improving temperature performance. These transitions create procurement opportunities for packaging suppliers that can deliver sustainable solutions meeting pharmaceutical and food cold chain validation requirements.
Market Segmentation Analysis
- Passive System holds 57.0% of product type, reflecting the cost effectiveness, simplicity, and reliability of insulated shippers and PCM systems for standard pharmaceutical and food cold chain applications.
- Healthcare accounts for 43.6% of end use, driven by pharmaceutical drug, vaccine, and biological sample cold chain requirements demanding validated packaging with documented temperature maintenance performance.
The Temperature Controlled Packaging Solution Market is segmented by product type, payload capacity, material type, temperature range, and end use. Passive System dominates product demand through cost effective insulated and PCM solutions. Healthcare leads the end use segment through pharmaceutical and vaccine cold chain requirements.
Insights into the Passive System Product Type Segment
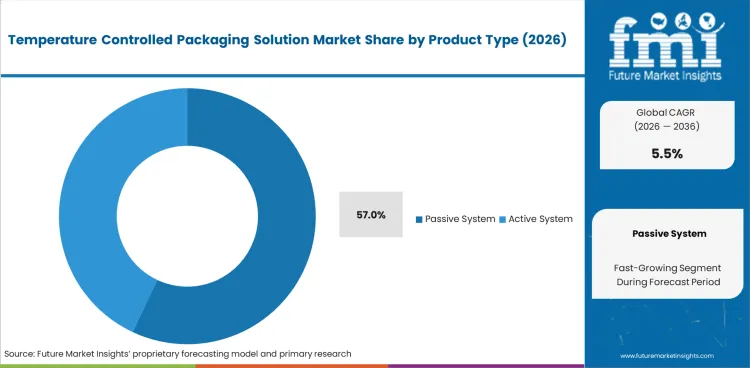
In 2026, Passive System is expected to represent 57.0% of the product type segment. Passive systems maintain temperature without external power using insulation materials and phase change materials that absorb or release thermal energy to keep contents within specified temperature ranges. Products include expanded polystyrene shippers, polyurethane insulated boxes, phase change material pouches and panels, and vacuum insulated panels.
Growth reflects the cost effectiveness and operational simplicity of passive solutions for pharmaceutical and food logistics applications where temperature maintenance periods of 24 to 96 hours are sufficient for standard distribution routes and transit times.
Insights into the Healthcare End Use Segment
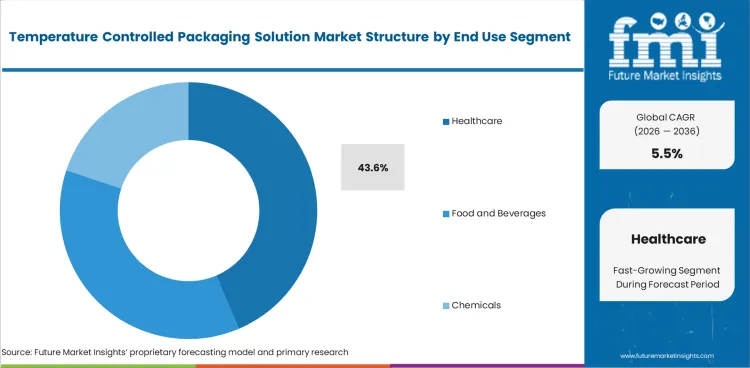
Healthcare accounts for 43.6% of the end use segment in 2026. This category covers cold chain packaging for pharmaceutical drugs including biologics and specialty medications, vaccines for routine immunization and pandemic programs, biological samples for clinical laboratory transport, and cell and gene therapy products requiring ultra-cold or cryogenic conditions during transit.
Growth reflects the expanding biologic drug pipeline, broadening vaccine distribution programs, increasing clinical sample transport volumes, and the emergence of cell and gene therapy logistics as a high value, technically demanding cold chain application category.
Temperature Controlled Packaging Solution Market Drivers, Restraints, and Opportunities
- Pharmaceutical cold chain growth and food safety regulation enforcement sustain demand for validated temperature controlled packaging solutions across healthcare and food distribution applications.
- Sustainability regulations restricting single use packaging materials and customer preference for environmental responsibility create transition costs, design challenges, and R and D investment requirements.
- Cell and gene therapy logistics create new ultra-cold and cryogenic packaging requirements with premium pricing potential and specialized engineering demands.
The Temperature Controlled Packaging Solution Market is shaped by pharmaceutical cold chain expansion, food safety regulation enforcement, and sustainability mandates. Growth is supported by biologic drug pipeline development, while packaging waste regulations and material sustainability requirements create transition challenges and innovation opportunities.
Pharmaceutical Cold Chain Expansion
Demand is shaped by biologic drug pipeline growth, expanded vaccine distribution programs, and increasing clinical sample transport volumes that create growing pharmaceutical cold chain packaging procurement. Validated packaging with integrated temperature monitoring and regulatory compliance documentation is increasingly required by pharmaceutical companies, regulators, and logistics providers.
Sustainability Transition Requirements
Growth margins and product development priorities are affected by regulatory restrictions on single use polystyrene packaging and customer demand for sustainable alternatives. Transition to reusable container systems, recyclable insulation materials, and bio-based phase change materials requires R and D investment, supply chain reconfiguration, and customer qualification testing.
Ultra-Cold Logistics Innovation
Adoption of specialized ultra-cold and cryogenic packaging is expanding with cell and gene therapy commercialization that requires maintenance of minus 60 to minus 80 degree Celsius conditions during global transit. This premium product category commands significantly higher pricing than standard cold chain packaging and requires specialized engineering and validation capabilities.
Analysis of Temperature Controlled Packaging Solution Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| India | 7.3% |
| China | 7.1% |
| USA | 6.2% |
| Germany | 5.5% |
| UK | 4.3% |
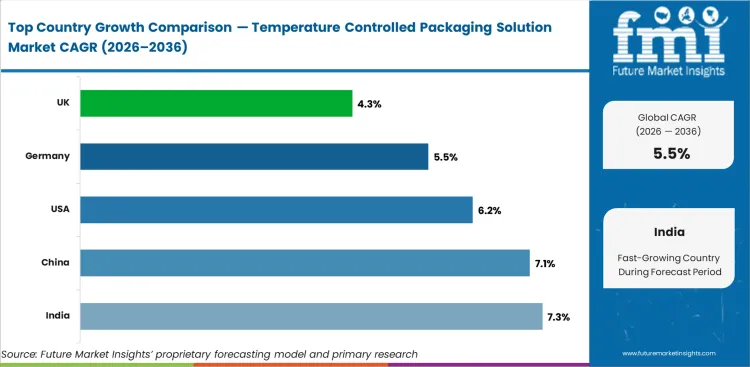
- India leads with a 7.3% CAGR through 2036, driven by pharmaceutical manufacturing growth, national vaccine infrastructure development, and cold chain modernization across food and healthcare sectors.
- China at 7.1% reflects pharmaceutical cold chain expansion, food safety regulation enforcement, and e-commerce perishable delivery growth creating new demand categories.
- The USA (6.2%) and Germany (5.5%) show steady growth driven by healthcare cold chain requirements, sustainability transitions, and cell therapy logistics development.
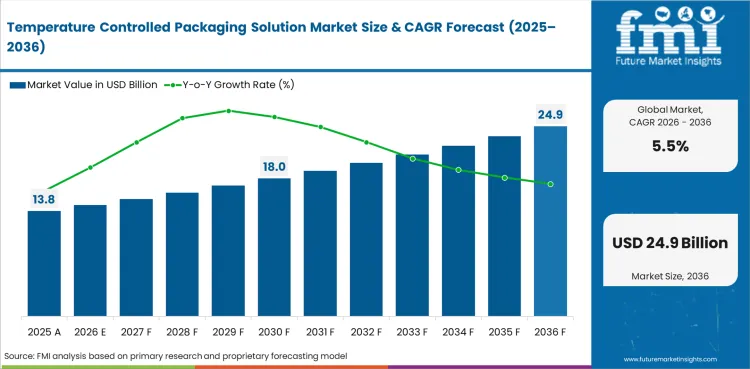
The global Temperature Controlled Packaging Solution Market is projected to grow at a CAGR of 5.5% from 2026 to 2036. The analysis covers more than 30 countries, with the primary markets listed below.
Demand Outlook for Temperature Controlled Packaging Solution Market in India
India is expected to grow at 7.3% through 2036, reflecting pharmaceutical manufacturing growth positioning India as a global drug export hub, national vaccine distribution infrastructure development, and cold chain logistics modernization across food and healthcare sectors.
- Pharmaceutical manufacturing growth drives cold chain packaging demand for drug export logistics across global markets.
- National vaccine distribution programs sustain insulated shipper procurement for immunization supply chains.
- Cold chain logistics modernization creates adoption of validated packaging across food and healthcare distribution.
Future Outlook for Temperature Controlled Packaging Solution Market in China
China is expected to grow at 7.1% through 2036, driven by pharmaceutical cold chain expansion, food safety regulation enforcement, and e-commerce perishable delivery growth.
- Pharmaceutical cold chain expansion drives validated packaging demand as biologic drug production and distribution grow.
- Food safety regulation enforcement creates structured cold chain compliance requirements across distribution networks.
- E-commerce perishable delivery growth creates last mile packaging demand for direct to consumer food and pharmaceutical products.
Opportunity Analysis of Temperature Controlled Packaging Solution Market in the United States
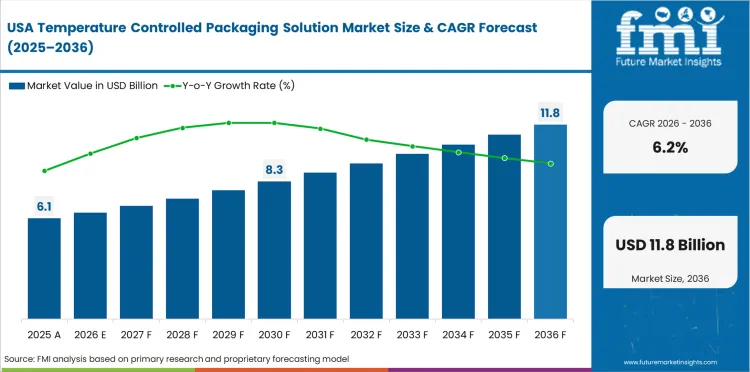
The USA is expected to grow at 6.2% through 2036, reflecting pharmaceutical distribution logistics, food safety compliance, cell therapy cold chain requirements, and sustainability driven packaging transitions.
- Biologic drug distribution sustains pharmaceutical cold chain packaging demand across hospital and specialty pharmacy supply chains.
- Cell and gene therapy logistics create ultra-cold packaging procurement for this rapidly growing pharmaceutical category.
- Sustainability pressure drives transition from EPS to recyclable and reusable cold chain packaging alternatives.
In-depth Analysis of Temperature Controlled Packaging Solution Market in Germany
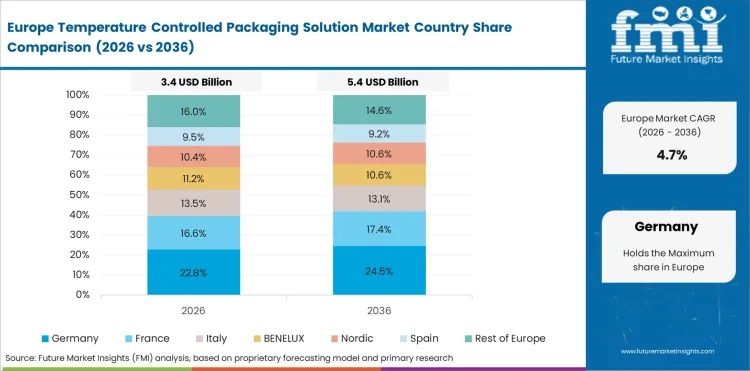
Germany is expected to grow at 5.5% through 2036, reflecting pharmaceutical manufacturing concentration, EU food safety regulation compliance, and sustainability driven packaging innovation leadership.
- Pharmaceutical manufacturing concentration in Germany drives cold chain packaging demand for European and global drug distribution.
- EU food safety regulations create compliance driven procurement for validated cold chain packaging solutions.
- Sustainability leadership drives development and adoption of recyclable and reusable packaging systems.
Sales Analysis of Temperature Controlled Packaging Solution Market in the United Kingdom
The UK is expected to grow at 4.3% through 2036, supported by pharmaceutical logistics requirements, food supply chain cold chain compliance, and packaging sustainability mandate compliance.
- Pharmaceutical logistics sustain cold chain packaging procurement across NHS and private healthcare supply chains.
- Food supply chain cold chain requirements drive validated packaging adoption for temperature sensitive product distribution.
- Packaging sustainability mandates accelerate transition from single use to reusable and recyclable cold chain materials.
Competitive Landscape and Strategic Positioning
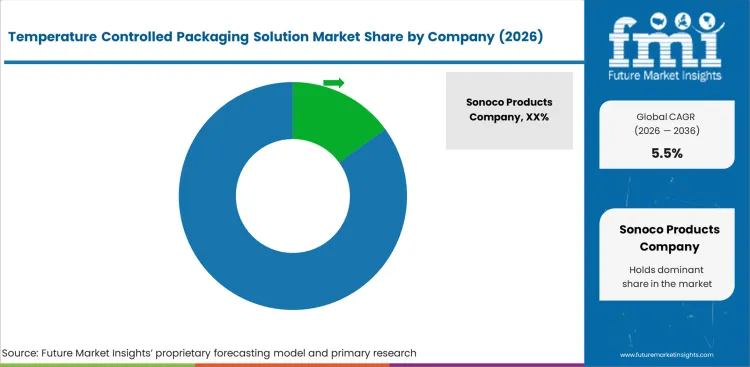
- Sonoco Products Company leads through its ThermoSafe brand providing comprehensive cold chain packaging for pharmaceutical and food logistics with established validation services and global manufacturing.
- Pelican BioThermal and Cold Chain Technologies maintain strong positions through specialized pharmaceutical cold chain packaging with reusable container programs and thermal engineering expertise.
- Emerging players target sustainable packaging innovation, smart monitoring integration, ultra-cold logistics solutions, and last mile delivery packaging for e-commerce applications.
Sonoco Products Company maintains a leading position through its ThermoSafe brand providing comprehensive cold chain packaging solutions for pharmaceutical and food logistics with established thermal engineering, validation services, and global manufacturing infrastructure. Pelican BioThermal delivers reusable cold chain containers for pharmaceutical distribution through its Crēdo product line with a global container management network.
Cold Chain Technologies provides validated pharmaceutical cold chain packaging systems with design engineering and qualification testing services. Cryopak Industries offers insulated packaging and temperature monitoring solutions for healthcare and food applications. Sofrigam SA provides European-focused cold chain packaging solutions with particular strength in pharmaceutical applications.
va-Q-tec AG delivers vacuum insulated panel technology for high performance thermal protection in pharmaceutical and industrial cold chain applications. GEBHARDT Logistic Solutions provides automated cold chain logistics systems.
Barriers to entry include pharmaceutical validation requirements and the associated testing infrastructure, established distribution and customer relationships with pharmaceutical and food logistics companies, cold chain thermal engineering expertise, and the capital investment required for reusable container fleet management programs. Strategic priorities include developing sustainable packaging alternatives, expanding ultra-cold and cryogenic capabilities for cell therapy logistics, integrating smart temperature monitoring technology, and building last mile delivery packaging for e-commerce cold chain applications.
Key Companies in the Temperature Controlled Packaging Solution Market
Key global companies leading the temperature controlled packaging solution market include:
- Sonoco Products Company, Pelican BioThermal LLC, Snyder Industries Inc. maintain leadership through established market presence, broad product portfolios, and global manufacturing and distribution infrastructure.
- Cold Chain Technologies, Cryopak Industries Inc., Sofrigam SA Ltd., va-Q-tec AG have established strong positions through specialized capabilities and focused customer relationships.
- GEBHARDT Logistic Solutions GmbH, ACH Foam Technologies, TKT GmbH, Saeplast Americas Inc. are among the emerging players gaining traction through specialized technology and focused innovation.
Competitive Benchmarking: Temperature Controlled Packaging Solution Market
| Company | Packaging Portfolio | Pharma Validation | Sustainability Innovation | Geographic Coverage |
|---|---|---|---|---|
| Sonoco Products (ThermoSafe) | High | Strong | Medium | Global |
| Pelican BioThermal | High | Strong | High | Global |
| Cold Chain Technologies | Medium | Strong | Medium | N. America |
| Cryopak Industries | Medium | Moderate | Medium | N. America |
| Sofrigam SA | Medium | Strong | Medium | Europe |
| va-Q-tec AG | Medium | Strong | High | Europe, Global |
| GEBHARDT Logistic | Low | Moderate | Medium | Europe |
| ACH Foam Technologies | Low | Moderate | Low | N. America |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Temperature Controlled Packaging Solution Market
- In 2025, Sonoco Products launched a fully recyclable insulated shipper replacing expanded polystyrene for pharmaceutical 2 to 8 degree cold chain applications, achieving equivalent thermal performance with curbside recyclable materials.
- In 2025, Pelican BioThermal expanded its reusable container fleet with an ultra-cold model supporting cell and gene therapy logistics at minus 80 degrees Celsius with 120 hour temperature maintenance duration.
Key Players in the Temperature Controlled Packaging Solution Market
Major Global Players
- Sonoco Products Company
- Pelican BioThermal LLC
- Snyder Industries Inc.
- Cold Chain Technologies
- Cryopak Industries Inc.
- Sofrigam SA Ltd.
- va-Q-tec AG
Emerging Players/Startups
- GEBHARDT Logistic Solutions GmbH
- ACH Foam Technologies
- TKT GmbH
- Saeplast Americas Inc.
- Inmark LLC
Report Scope and Coverage

| Metric | Value |
|---|---|
| Quantitative Units | USD 14.56 billion to USD 24.87 billion, at a CAGR of 5.5% |
| Market Definition | The market covers insulated containers, phase change materials, refrigerated systems, and monitoring devices for maintaining temperature sensitive products during storage and transportation across healthcare and food supply chains. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, UK, India, China, Germany, Japan, France, Brazil, 30 plus countries |
| Key Companies Profiled | Sonoco Products Company, Pelican BioThermal, Cold Chain Technologies, Cryopak Industries, Sofrigam SA, va-Q-tec AG, GEBHARDT Logistic Solutions, ACH Foam Technologies, TKT GmbH, Saeplast Americas |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom up and top down methodology starting with verified transaction data, projecting adoption velocity across segments and regions. |
Segmentation
Temperature Controlled Packaging Solution Market Segmented by Product Type:
- Passive System
- Insulated Shippers
- Insulated Boxes
- Phase-Change Material Based Systems
- Vacuum Insulated Panels
- Active System
- Refrigerated Containers
- Refrigerated Trucks and Vans
- Electrically Powered Units
Temperature Controlled Packaging Solution Market Segmented by Payload Capacity:
- Small (<20 Liters)
- Medium (20–150 Liters)
- Large (>150 Liters)
Temperature Controlled Packaging Solution Market Segmented by Material Type:
- Polymer-Based Materials
- Expanded Polystyrene
- Polyurethane
- Polypropylene
- Polyethylene
- Glass
- Metal
- Others
Temperature Controlled Packaging Solution Market Segmented by Temperature Range:
- Frozen (-20°C and Below)
- Chilled (2°C to 8°C)
- Ambient (15°C to 25°C)
Temperature Controlled Packaging Solution Market Segmented by End Use:
- Healthcare
- Pharmaceuticals
- Vaccines
- Biological Samples
- Food and Beverages
- Fresh Produce
- Meat
- Poultry
- Seafood
- Dairy Products
- Processed Foods
- Chemicals
- Specialty Chemicals
- Industrial Chemicals
- Others
- Cosmetics
- Flowers and Plants
Temperature Controlled Packaging Solution Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Research Sources and Bibliography
- 1. World Health Organization. (2025). WHO Vaccine Cold Chain Management Guidelines and Standards. WHO.
- 2. USA Food and Drug Administration. (2025). FDA Guidance on Temperature Controlled Drug Distribution Practices. FDA.
- 3. European Commission. (2024). EU Good Distribution Practice for Medicinal Products. EC.
- 4. International Safe Transit Association. (2025). ISTA Cold Chain Packaging Testing Protocols and Standards. ISTA.
- 5. International Air Transport Association. (2025). IATA Temperature Control Regulations for Pharmaceutical Air Transport. IATA.
- This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with publication dates, URLs, and supporting data for all cited works.
This Report Answers
- Estimating market size and revenue projections from 2026 to 2036.
- Segmentation by product type, payload capacity, material type, temperature range, and end use.
- Regional insights covering more than 30 markets.
- Analysis of passive and active cold chain packaging systems across pharmaceutical and food applications.
- Competitive landscape assessment including validation capability and sustainability innovation benchmarking.
- Growth opportunities in pharmaceutical cold chain, ultra-cold therapy logistics, sustainable packaging, and e-commerce last mile delivery.
- Supply chain, material sourcing, and container logistics evaluation.
- Data delivery in PDF and Excel formats.
Frequently Asked Questions
What is the global market demand for Temperature Controlled Packaging Solutions in 2026?
In 2026, the global Temperature Controlled Packaging Solution Market is expected to be worth USD 14.56 billion, reflecting sustained pharmaceutical cold chain growth driven by biologic drugs and vaccines, food safety regulation enforcement, and emerging e-commerce perishable delivery requirements.
How big will the Temperature Controlled Packaging Solution Market be in 2036?
By 2036, the market is expected to reach USD 24.87 billion, supported by continued biologic drug pipeline expansion, cell and gene therapy ultra-cold logistics development, global vaccine distribution infrastructure growth, food safety regulation enforcement, and sustainability driven packaging innovation.
How much is demand expected to grow between 2026 and 2036?
Between 2026 and 2036, demand is expected to grow at a CAGR of 5.5%, driven by pharmaceutical cold chain volume growth, food cold chain regulation tightening, e-commerce perishable delivery expansion, and packaging material transitions toward sustainable alternatives that maintain cold chain performance.
Which product type segment is expected to lead in 2026?
Passive System is expected to hold 57.0% of the product type segment in 2026, reflecting the cost effectiveness and operational reliability of insulated shippers, phase change material systems, and vacuum insulated panels for pharmaceutical and food cold chain logistics where standard transit times of 24 to 96 hours are sufficient.
What is driving demand in India?
India is expected to grow at 7.3% through 2036, driven by pharmaceutical manufacturing growth positioning India as a global drug export hub, national vaccine distribution infrastructure development for population scale immunization, and cold chain logistics modernization across food and healthcare distribution networks.
What is driving demand in China?
China is expected to grow at 7.1% through 2036, reflecting pharmaceutical cold chain expansion as domestic biologic drug production grows, food safety regulation enforcement creating structured compliance requirements, and e-commerce perishable delivery growth creating new last mile cold chain packaging demand.
What does this report mean by Temperature Controlled Packaging Solution Market definition?
The market covers insulated containers, phase change materials, refrigerated transport systems, and associated temperature monitoring devices used to maintain pharmaceuticals, vaccines, biological samples, food products, and other temperature sensitive goods within specified temperature ranges during storage, transportation, and last mile delivery.
How does FMI validate the forecast?
Forecasting models use a hybrid bottom up and top down approach, starting with verified pharmaceutical distribution volume data, food cold chain spending, and packaging procurement records, validating against global pharmaceutical production statistics, cold chain logistics industry reports, and packaging manufacturer disclosures.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type, 2026 to 2036
- Passive System
- Insulated Shippers
- Insulated Boxes
- Phase-Change Material Based Systems
- Vacuum Insulated Panels
- Active System
- Refrigerated Containers
- Refrigerated Trucks and Vans
- Electrically Powered Value (USD Million)s
- Passive System
- Y to o to Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Payload Capacity
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Payload Capacity, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Payload Capacity, 2026 to 2036
- Small (<20 Liters)
- Medium (20–150 Liters)
- Large (>150 Liters)
- Small (<20 Liters)
- Y to o to Y Growth Trend Analysis By Payload Capacity, 2021 to 2025
- Absolute $ Opportunity Analysis By Payload Capacity, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Material Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Material Type, 2026 to 2036
- Polymer-Based Materials
- Expanded Polystyrene
- Polyurethane
- Polypropylene
- Polyethylene
- Glass
- Metal
- Others
- Polymer-Based Materials
- Y to o to Y Growth Trend Analysis By Material Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Material Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Temperature Range
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Temperature Range, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Temperature Range, 2026 to 2036
- Frozen (-20°C and Below)
- Chilled (2°C to 8°C)
- Ambient (15°C to 25°C)
- Frozen (-20°C and Below)
- Y to o to Y Growth Trend Analysis By Temperature Range, 2021 to 2025
- Absolute $ Opportunity Analysis By Temperature Range, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Healthcare
- Pharmaceuticals
- Vaccines
- Biological Samples
- Food and Beverages
- Fresh Produce
- Meat
- Poultry
- Seafood
- Dairy Products
- Chemicals
- Specialty Chemicals
- Industrial Chemicals
- Others
- Cosmetics
- Flowers and Plants
- Healthcare
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Payload Capacity
- By Material Type
- By Temperature Range
- By End Use
- Competition Analysis
- Competition Deep Dive
- Sonoco Products Company
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Pelican BioThermal LLC
- Snyder Industries Inc.
- GEBHARDT Logistic Solutions GmbH
- ACH Foam Technologies, LLC
- TKT GmbH; Cold Chain Technologies
- Cryopak Industries Inc.
- Sofrigam SA Ltd.
- va-Q-tec AG
- Saeplast Americas Inc.
- Inmark, LLC
- Tempack Packaging Solutions S.L.
- EcoCool GmbH
- Exeltainer SL
- American Aerogel Corporation
- Sonoco Products Company
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Payload Capacity, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Temperature Range, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Payload Capacity, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Temperature Range, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Payload Capacity, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Temperature Range, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Payload Capacity, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Temperature Range, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Payload Capacity, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Temperature Range, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Payload Capacity, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Temperature Range, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Payload Capacity, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Temperature Range, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Payload Capacity, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Temperature Range, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Payload Capacity, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Payload Capacity, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Payload Capacity
- Figure 9: Global Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Material Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Material Type
- Figure 12: Global Market Value Share and BPS Analysis by Temperature Range, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Temperature Range, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Temperature Range
- Figure 15: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by End Use
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Product Type
- Figure 32: North America Market Value Share and BPS Analysis by Payload Capacity, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Payload Capacity, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Payload Capacity
- Figure 35: North America Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Material Type, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Material Type
- Figure 38: North America Market Value Share and BPS Analysis by Temperature Range, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Temperature Range, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Temperature Range
- Figure 41: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by End Use
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Product Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Payload Capacity, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Payload Capacity, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Payload Capacity
- Figure 51: Latin America Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Material Type, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Material Type
- Figure 54: Latin America Market Value Share and BPS Analysis by Temperature Range, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Temperature Range, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Temperature Range
- Figure 57: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by End Use
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Payload Capacity, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Payload Capacity, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Payload Capacity
- Figure 67: Western Europe Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Material Type, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Material Type
- Figure 70: Western Europe Market Value Share and BPS Analysis by Temperature Range, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Temperature Range, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Temperature Range
- Figure 73: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by End Use
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Payload Capacity, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Payload Capacity, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Payload Capacity
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Material Type, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Material Type
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Temperature Range, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Temperature Range, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Temperature Range
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Product Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Payload Capacity, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Payload Capacity, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Payload Capacity
- Figure 99: East Asia Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Material Type, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Material Type
- Figure 102: East Asia Market Value Share and BPS Analysis by Temperature Range, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Temperature Range, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Temperature Range
- Figure 105: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by End Use
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Payload Capacity, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Payload Capacity, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Payload Capacity
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Material Type, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Material Type
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Temperature Range, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Temperature Range, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Temperature Range
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Payload Capacity, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Payload Capacity, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Payload Capacity
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Material Type, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Material Type
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Temperature Range, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Temperature Range, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Temperature Range
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

