Respiratory Inhaler Devices encompass three dominant architectures with fundamentally different economic profiles. Pressurised metered dose inhalers combine a metallic canister, metering valve, actuator and propellant system. The valve represents the most technically demanding component, requiring precision engineering to deliver consistent doses across hundreds of actuations. Manufacturing involves propellant filling under pressure, valve crimping and leak testing. Bill of materials cost for pressurised devices includes aluminium canisters, elastomeric seals, valve components and propellant, with the valve assembly accounting for a substantial portion of device cost.
Dry powder inhalers eliminate propellant dependence through mechanical dose delivery mechanisms. These devices range from single dose capsule systems to multi dose reservoir designs with integrated dose counters. Complexity varies significantly across architectures. Simple capsule-based devices require minimal tooling investment but demand precise airflow resistance characteristics. Advanced multi dose systems incorporate gear trains, dose indexing mechanisms and counter displays that increase component count and assembly complexity. Tooling investment for injection molded components and assembly fixtures scales with device sophistication.
Soft mist inhalers represent the most mechanically complex category, using spring mechanisms or compressed gas to generate fine aerosol clouds without traditional propellants. These devices require extensive development to achieve consistent plume characteristics and dose delivery. Manufacturing involves multi step assembly with tight tolerances for mechanical components. The precision engineering required elevates both development cost and unit manufacturing cost compared to simpler dry powder designs.
Cost structures influence pricing behaviour across categories. Pressurised devices benefit from established manufacturing processes but face propellant transition costs. Dry powder devices demonstrate wider cost variation based on mechanical complexity. Soft mist devices command premium positioning through differentiated delivery characteristics despite higher production costs.
Legacy pressurised inhalers relied on chlorofluorocarbon propellants until environmental concerns triggered global phase out. The subsequent transition to hydrofluoroalkane propellants required comprehensive reformulation as drug solubility and stability profiles differ substantially between propellant systems. This transition is now being repeated as regulatory focus shifts to low global warming potential alternatives.
Propellant reformulation extends beyond simple substitution. Drug manufacturers must reoptimize excipient selection, adjust drug concentration and modify suspension or solution formulations to maintain therapeutic equivalence. Each formulation change necessitates stability studies, compatibility testing and manufacturing process validation. The development timeline for propellant transition typically spans three to five years before regulatory submission.
Valve technology requires concurrent redesign because seal materials and metering chambers must accommodate different propellant properties. Propellant vapour pressure, solvency characteristics and material compatibility dictate valve component selection. Valve suppliers invest in new elastomer formulations and component geometries to ensure dose accuracy and seal integrity across product shelf life.
Manufacturing infrastructure demands significant capital investment during propellant transitions. Filling lines require modification or replacement to handle new propellant systems safely and efficiently. Pressure vessels, filling heads and quality control equipment must meet updated specifications. Environmental controls in filling areas need enhancement to manage different propellant handling requirements.
Regulatory revalidation imposes substantial compliance costs. Authorities require bioequivalence studies demonstrating that reformulated products deliver equivalent clinical performance to predecessor versions. Human factors studies confirm that patients can use modified devices correctly. Post approval manufacturing changes trigger regulatory notifications and potential site inspections. The cumulative regulatory burden adds millions in development cost per product.
Respiratory Inhaler Devices undergo drug device combination product evaluation, a regulatory pathway more demanding than standalone drug or device approval. Regulators assess device performance, drug formulation and the integrated product system simultaneously. This combination approach requires extensive documentation covering device design controls, manufacturing processes and quality systems alongside traditional drug development data.
Human factors engineering studies represent a critical and resource intensive approval requirement. These studies evaluate whether intended users can correctly operate the device without excessive training. Protocol design involves identifying use errors, testing with representative patient populations and implementing design modifications based on findings. Comprehensive human factors validation typically requires multiple study rounds with diverse patient demographics, adding twelve to eighteen months to development timelines.
Bioequivalence testing for generic or reformulated inhalers presents unique challenges compared to oral solid dose forms. In vitro characterization includes aerodynamic particle size distribution, plume geometry, dose content uniformity and delivered dose consistency. These tests require specialized cascade impactor equipment and validated analytical methods. Clinical bioequivalence studies must demonstrate comparable lung deposition and systemic exposure, often requiring sophisticated pharmacokinetic sampling and imaging techniques.
Device modifications trigger regulatory scrutiny even for established products. Changes to valve components, actuator design or manufacturing processes require regulatory assessment of potential impact on product performance. Significant changes may necessitate bridging studies or full bioequivalence demonstrations. This regulatory sensitivity limits manufacturing flexibility and increases the cost of continuous improvement initiatives.
Post approval surveillance obligations extend regulatory burden beyond launch. Manufacturers must monitor device complaints, track malfunction rates and report adverse events. Annual reporting compiles device performance data for regulatory review. Any signal of performance degradation or safety concern triggers investigation requirements and potential corrective actions. These ongoing obligations represent permanent compliance costs that factor into product pricing strategies.
Manufacturing scale fundamentally shapes unit economics for Respiratory Inhaler Devices through fixed cost absorption and process optimization opportunities. High volume production enables investment in dedicated automation that reduces labour content and improves consistency. Capital intensity varies significantly across device types, with pressurised inhalers requiring specialized filling equipment and dry powder devices demanding precision assembly automation.
Automated valve assembly and crimping systems represent critical competitive advantages for pressurised device manufacturers. These systems position valve stems, apply crimping force with precise control and verify crimp integrity through vision systems or mechanical testing. Automation eliminates operator variability in valve seating, a critical parameter affecting dose accuracy and leak rates. High speed lines achieve production rates exceeding several hundred units per minute, dramatically reducing per unit labour cost compared to manual or semi automated processes.
Dose testing automation enables 100 percent inline inspection without creating production bottlenecks. Automated testing systems actuate devices, collect aerosol output and measure delivered dose using weight based or optical methods. Real time feedback allows immediate process adjustment when dose uniformity trends toward specification limits. This continuous monitoring improves yield by catching process drift before significant quantities of out of specification product accumulate.
Yield management directly impacts manufacturing cost competitiveness. Each rejected unit represents wasted material, labour and overhead allocation. Suppliers achieving 95 percent yield versus 85 percent yield demonstrate 10 percent lower unit costs assuming equivalent material and labour inputs. Yield optimization requires statistical process control, root cause analysis capability and continuous improvement culture. Leading manufacturers invest in advanced analytics to identify yield detractors and prioritize corrective actions.
Scale also influences supplier negotiating power with component vendors. High volume purchasers secure preferential pricing on aluminium canisters, valve components and propellants. These material cost advantages compound across millions of units, creating sustainable cost positions that smaller competitors cannot match. Vertical integration decisions, such as in house valve manufacturing versus external sourcing, further differentiate cost structures among device suppliers.
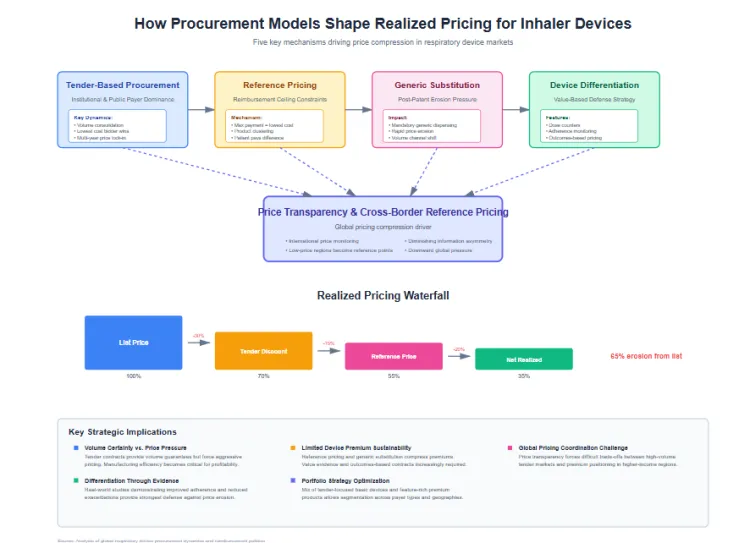
Tender based procurement dominates institutional and public payer purchasing for Respiratory Inhaler Devices in most regions. Hospital systems and national health authorities consolidate volume to negotiate aggressive pricing. Tender processes typically award business to the lowest cost bidder meeting technical specifications, creating intense price competition among suppliers. Multi year contracts lock in pricing but provide volume certainty that justifies capital investment in manufacturing capacity.
Reference pricing mechanisms constrain reimbursement levels by establishing maximum payment amounts based on the lowest cost therapeutically equivalent alternative. Payers group similar inhaler products into clusters and reimburse only up to the reference price. Manufacturers pricing above reference levels force patients to pay out of pocket differences, effectively limiting demand for premium priced products. This policy structure pressures suppliers to align pricing with generic or lower cost alternatives regardless of device differentiation.
Generic substitution pressure intensifies as patents expire on branded inhaler products. Pharmacy benefit managers and national formularies mandate generic dispensing when available, channeling volume to lower priced alternatives. Device differentiation provides limited protection against generic erosion for inhalers compared to oral solids because bioequivalence demonstration requirements create barriers for generic entry. However, once generic inhalers achieve approval, price erosion proceeds rapidly.
Device features offer potential pricing defense through demonstrated value beyond drug delivery. Dose counters, adherence monitoring capability and ergonomic advantages support premium positioning when payers recognize these attributes improve patient outcomes. Evidence generation through real world studies showing improved adherence or reduced exacerbations strengthens value arguments. However, payers increasingly demand outcomes based pricing or risk sharing arrangements rather than accepting feature based price premiums.
Price transparency initiatives and cross border reference pricing compress international price variation. Payers monitor pricing in other countries and demand comparable levels. Manufacturers face difficult decisions regarding pricing strategies when low prices in high volume tender regions become reference points for higher income regions. This dynamic creates downward pricing pressure globally as information asymmetry diminishes.
Simple dry powder inhalers using capsule based delivery achieve the lowest unit manufacturing cost at high volume. These devices eliminate propellant filling and complex valve assembly while using inexpensive injection molded plastic components. However, multi dose dry powder inhalers with integrated dose counters approach or exceed pressurised device costs due to mechanical complexity.
Propellant transitions extend development timelines by three to five years and add millions in reformulation, validation and regulatory costs per product. Manufacturers typically maintain higher pricing during transition periods to recover these investments, but tender pressure and generic competition often prevent full cost recovery through pricing.
Inhalers deliver drugs directly to lungs through aerosolization, making device performance critical to therapeutic effect. Small variations in particle size, plume characteristics or dose consistency significantly impact efficacy and safety. This performance sensitivity combined with drug device combination complexity justifies enhanced regulatory oversight compared to oral solid dose forms.
Device specific training requirements, bioequivalence demonstration needs and patient familiarity create switching barriers. Healthcare providers must educate patients on proper technique for each device type. Payers face adherence risks if patients struggle with unfamiliar devices. These factors provide some insulation from pure price based competition despite tender pressures.
Progressive payers assess total cost of therapy including adherence rates, exacerbation frequency and healthcare utilization. Devices demonstrating superior adherence through ease of use or reminder features potentially reduce overall costs despite higher acquisition prices. Real world evidence showing reduced hospitalizations strengthens value propositions, though adoption of value based assessment remains inconsistent across regions.
The Respiratory Inhaler Devices Market Is Segmented By Product (Dry Powder Inhaler, Metered Dose Inhaler, Nebulizer, Soft Mist Inhaler), Drug Class (Short-Acting Beta2 Agonist Inhalers, Short-Acting Muscarinic Antagonist Inhalers, Long-Acting Beta2 Agonist Inhalers, Long-Acting Muscarinic Antagonist Inhalers, Corticosteroid Inhalers, Combination LABA/LAMA Inhalers, Combination ICS/LABA Inhalers), Technology (Manual Inhalers, Digital Inhalers), Disease Indication (Asthma, Chronic Obstructive Pulmonary Disease, Pulmonary Arterial Hypertension, Acute Respiratory Distress, Pulmonary Fibrosis), Sales Channel (Hospitals, Specialty Clinics, Retail Pharmacy Chains, Online Pharmacies or Mail-Order Pharmacies), And Region. Forecast For 2026 To 2036.
Understanding Market Share Trends in Respiratory Inhaler Devices
Respiratory Measurement Devices Market Size and Share Forecast Outlook 2025 to 2035
Japan Respiratory Inhaler Devices Market Size and Share Forecast Outlook 2025 to 2035
Therapeutic Respiratory Devices Market Overview - Trends & Forecast 2025 to 2035