Hillrom emphasizes reliability, service continuity, and uptime in tender documentation and product positioning rather than competing solely on purchase price. Sales narratives focus on years of uninterrupted diagnostic capacity instead of technical specifications alone. Marketing materials highlight service response times, parts availability across global networks, and calibration stability that extends intervals between mandatory recertification. This framing reorients procurement conversations away from unit cost comparisons toward operational assurance.
Tender language often includes guarantees around mean time between failures, warranty terms that cover both parts and labor, and commitments to software updates that maintain regulatory compliance as standards evolve. By positioning spirometers as critical infrastructure comparable to imaging equipment or patient monitors, Hillrom justifies pricing tiers that reflect long-term performance rather than immediate hardware costs. Procurement officers in large hospital systems respond to this messaging because equipment downtime creates cascading problems: rescheduled appointments, delayed diagnoses, frustrated clinicians, and lost billable procedures. Hillrom leverages this operational anxiety to defend premium pricing as risk mitigation rather than luxury spending.
Spare parts availability establishes a foundational pricing floor because hospitals cannot afford extended waits for critical components. Hillrom maintains regional distribution centers and service depots that stock sensors, flow heads, calibration syringes, and circuit boards. This logistics infrastructure carries fixed costs that must be recovered through device pricing and service contracts. Competitors offering lower upfront prices often lack comparable parts networks, forcing buyers to accept longer repair cycles or source components through third-party suppliers with uncertain quality.
Calibration stability determines how often devices require verification testing to maintain regulatory compliance and diagnostic accuracy. Spirometers with superior sensor design and temperature compensation maintain calibration longer, reducing the frequency of mandatory checks and associated labor costs. Durable sensors that resist wear from repeated patient use extend replacement intervals, lowering consumables expenses over the device lifespan. Post-sale service responsiveness, including remote diagnostics, on-site technician visits, and loaner equipment during repairs, protects hospital operations from revenue loss. These factors collectively establish a pricing floor below which Hillrom cannot profitably deliver the reliability that justifies its premium positioning.
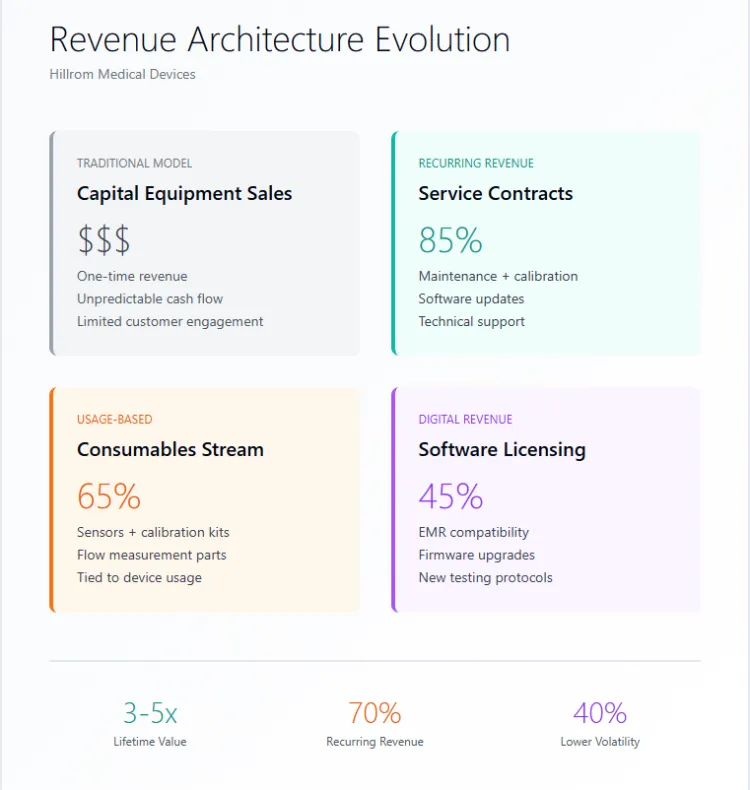
Service contracts convert one-time capital expenditures into recurring operational expenses, improving Hillrom's revenue predictability while reducing buyer cost risk. Annual maintenance agreements bundle calibration verification, preventive maintenance visits, software updates, and technical support into fixed fees. This model allows hospitals to budget with certainty rather than facing unpredictable repair costs that strain departmental finances. For Hillrom, recurring revenue smooths cash flow volatility and increases customer lifetime value beyond initial device sales.
Consumables including calibration kits, disposable sensors, and flow-measurement components create ongoing revenue streams tied directly to device usage. High-volume clinics require frequent sensor replacements, generating proportional consumables income. Software support subscriptions ensure compatibility with evolving electronic medical record systems and regulatory requirements, creating upgrade revenue opportunities as healthcare IT infrastructure advances. Firmware updates that enhance functionality or add new testing protocols justify additional licensing fees. This revenue architecture improves supplier economics by diversifying income sources and strengthening customer relationships through continuous engagement rather than transactional sales cycles.
Clinics value uninterrupted throughput because spirometer failures impose costs that exceed device savings. When a spirometer becomes unavailable, scheduled lung-function tests must be cancelled or rescheduled, disrupting patient care pathways and delaying clinical decisions. Cancelled appointments represent lost billable procedures, reducing departmental revenue and worsening cost-recovery metrics that hospital administrators track closely. Rescheduling patients consumes administrative labor, frustrates referring physicians, and erodes patient satisfaction scores that increasingly influence hospital reimbursement rates.
For busy pulmonology departments or primary-care clinics conducting routine respiratory assessments, each day of spirometer downtime translates to dozens of missed tests. If a device failure persists for a week while awaiting parts or technician visits, the accumulated revenue loss can approach or exceed the price difference between premium and budget spirometry equipment. Procurement teams increasingly perform break-even analyses that quantify downtime risk: if a lower-cost spirometer fails twice as often as a Hillrom device, the savings disappear within the first service interruption. Hillrom leverages this economic reality to defend premium pricing, positioning reliability as the ultimate cost-control measure rather than an expensive luxury.
Large hospital systems with complex clinical governance structures prioritize high reliability because equipment failures ripple across multiple departments and affect hundreds of patients. Integrated healthcare networks that operate under capitated payment models or value-based reimbursement contracts cannot afford operational inefficiencies that undermine care coordination and quality metrics. These organizations employ procurement specialists who conduct sophisticated total cost of ownership analyses, evaluating failure rates, service response times, and consumables costs over projected lifespans. They recognize that lifecycle value determines true cost, not purchase price alone.
Regulated facilities including teaching hospitals, research institutions, and specialized respiratory centers face stringent quality assurance requirements that demand equipment with documented calibration stability and regulatory compliance. These buyers pay premium prices willingly because non-compliant devices create accreditation risks and threaten institutional reputation. Geographic regions with mature healthcare infrastructure and established service networks, particularly North America and Western Europe, demonstrate higher receptivity to lifecycle-value pricing because buyers trust that post-sale support commitments will be honored.
Pricing flexibility varies significantly in low-resource settings where upfront capital constraints dominate procurement decisions regardless of lifecycle economics. Hospitals in emerging economies may lack budgetary mechanisms to evaluate total cost of ownership or lack confidence that service networks will remain accessible over device lifespans. In these environments, Hillrom faces pressure to offer entry-level products or flexible financing arrangements that reduce initial barriers while preserving some lifecycle revenue through consumables and service contracts.

Spirometer failures disrupt patient scheduling, delay diagnoses, and eliminate billable test procedures. The revenue lost during even brief downtime episodes often exceeds the savings from purchasing cheaper devices, making reliability the primary cost-control factor for high-volume facilities.
Total cost includes purchase price, annual maintenance contracts, calibration verification, consumable sensors and flow heads, software licensing, technician labor for repairs, and opportunity cost of cancelled tests during downtime. Consumables and service contracts typically represent 40 to 60 percent of total ownership cost over a device's useful life.
Contracts guarantee scheduled calibration that maintains regulatory compliance and diagnostic accuracy, preventing sudden device decertification that would halt testing. Preventive maintenance identifies component wear before failures occur, reducing emergency repair costs and unplanned downtime that disrupts clinical schedules.
Each spirometer failure cancels an average of 10 to 30 scheduled tests depending on repair duration and clinic volume. At typical reimbursement rates of $50 to $150 per spirometry test, a single week-long failure costs $2,500 to $15,000 in lost revenue, easily exceeding price differences between premium and budget devices.
Hospitals can negotiate multi-year service contracts for volume discounts, standardize spirometer models across departments to reduce parts inventory, train additional staff on basic troubleshooting to reduce service calls, and implement preventive maintenance schedules that extend component life and reduce emergency repairs.
The edge computing enclosure environmental stress test equipment market is segmented by Stress Type (Temperature Cycling, Temperature Humidity, Thermal Shock, Corrosion, Dust Ingress, Rain Spray, Solar Loading, Combined Stress), Equipment Format (Walk-in Chambers, Reach-in Chambers, Modular Rigs, Remote Conditioners, HALT Systems, Portable Rigs), Test Standard (IEC 60068, GR-63, NEBS, MIL-STD, ETSI EMC, ASTM Methods, IP Ratings), Enclosure Type (Outdoor Cabinets, Micro Data Centers, Wall Cabinets, Rack Cabinets, Shelter Enclosures, Pole Enclosures), Deployment Environment (Telecom Edge, Industrial Edge, Retail Edge, Healthcare Edge, Smart City Edge, Defense Edge), Component Tested (Cooling Systems, Power Systems, Cabinet Shells, Door Seals, Filters, Sensors, Control Units), End User (Telecom Operators, Edge OEMs, Test Labs, Colocation Firms, Industrial Integrators, Defense Programs, Enterprise IT Teams), Sales Channel (Direct Sales, Channel Partners, System Integrators, Third-Party Labs, Rental Services), and Region. Forecast for 2026 to 2036
The AI inference hardware performance benchmarking test systems market is segmented by Benchmark Type (Standard Benchmark Suites, Application-Specific Benchmarks, Power and Performance Profiling, Latency and SLA Validation Harnesses), Hardware Under Test (GPUs, CPUs, NPUs and TPUs, FPGAs, Custom ASIC Accelerators), Metric Focus (Throughput, Latency, Power Efficiency, Accuracy Retention, Cost per Inference, Thermal Stability), Deployment Environment (Hyperscaler Cloud Benchmarking, On-Prem Enterprise Benchmarking, Edge Inference Benchmarking, Embedded and Industrial Benchmarking), End User (Cloud Providers, Chip Vendors, Server and OEM Vendors, Large Enterprises, Research and Validation Labs), Sales Channel (Direct Sales, System Integrators, Software Subscriptions, Managed Benchmarking Services, Validation Contracts), and Region. Forecast for 2026 to 2036.
The data center power quality and harmonics monitoring test systems market is segmented by Product Type (Power Meters, Power Analyzers, Monitoring Software, Portable Testers), Monitoring Scope (Harmonics, Sags and Swells, Transients, Unbalance, Frequency, Flicker), Installation Point (UPS Input, UPS Output, PDU Panels, Switchgear, Generator Bus, Busway), Facility Type (Hyperscale, Colocation, Enterprise, Edge), Voltage Class (Low Voltage, Medium Voltage, Mixed Voltage), Deployment Mode (Fixed Systems, Portable Systems, Hybrid Systems). Forecast for 2026 to 2036.
The vehicle cabin air quality and filtration efficiency test systems market is segmented by Test Type (Filtration Efficiency, Gas and VOC Testing, Particulate Exposure Testing, Sensor Calibration), Vehicle Type (Passenger Cars, Light Commercial Vehicles, Heavy Commercial Vehicles, Buses), Technology (Chamber Systems, Particle Analyzers, Gas Analyzers, Integrated Benches), Deployment (OEM Laboratories, Third-party Laboratories, Engineering Service Providers), Sales Channel (Direct Sales, Distributors, System Integrators, Service Contracts), and Region. Forecast for 2026 to 2036.
The liquid immersion cooling fluid compatibility test equipment market is segmented by Test Type (Material Compatibility, Corrosion Testing, Dielectric Testing, Thermal Aging, Contamination Analysis, Oxidation Stability), Fluid Chemistry (Synthetic Hydrocarbons, Fluorinated Fluids, Esters, Silicone Fluids, Mineral Oils), Material Under Test (Elastomers, Plastics, Metals, Coatings, PCB Assemblies, Storage Devices), Equipment Format (Benchtop Systems, Modular Rigs, Pilot Systems, Integrated Labs), Automation Level (Manual Systems, Semi-automated Systems, Fully Automated Systems), Application Stage (R&D Screening, Qualification and Acceptance, Pre-deployment Validation, Failure Analysis, Periodic Monitoring), End Use (Fluid Manufacturers, Hardware OEMs, Immersion System Suppliers, Hyperscale Operators, Third-party Labs, Research Institutes), and Region. Forecast for 2026 to 2036.