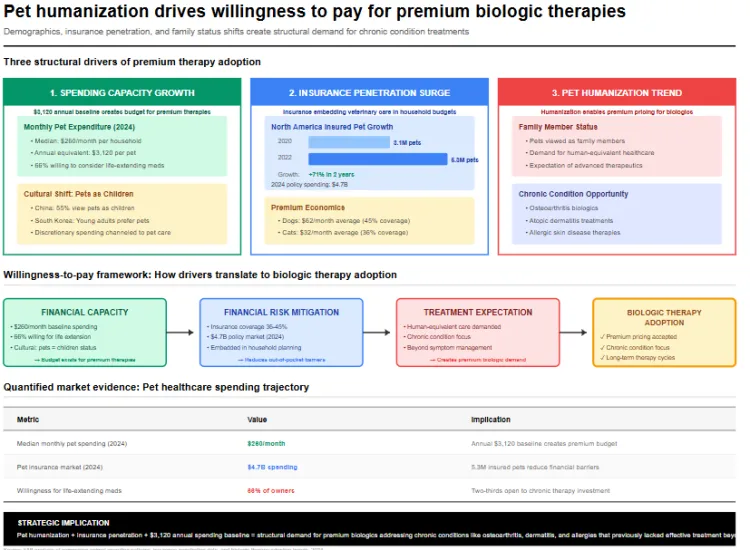
Demographics and cultural shifts drive unprecedented spending on companion animal healthcare. Median monthly pet expenditure reached 260 dollars in 2024, with 66 percent of owners willing to consider life extending medicines for their dogs and cats. Chinese surveys indicate 55 percent of pet owners now view pets as children, while younger adults in South Korea increasingly prefer pets over human offspring, channeling discretionary funds into premium care.
Pet insurance penetration amplifies this willingness to pay. The number of insured pets in North America rose from 3.1 million in 2020 to 5.3 million in 2022, with consumer spending on pet insurance policies exceeding 4.7 billion dollars in 2024. Accident and illness coverage premiums average 62 dollars monthly for dogs and 32 dollars for cats, yet these costs increased more slowly than broader pet services inflation. Insurance holders demonstrate 45 percent coverage among dog parents and 36 percent among cat parents, embedding veterinary care into household financial planning.
Humanization of pets transforms treatment decisions. Pet owners increasingly demand the same level of healthcare access for animals as for themselves, viewing pets as family members deserving advanced therapeutics. This shift enabled biologic therapies targeting chronic conditions like osteoarthritis, atopic dermatitis, and allergic skin disease that previously lacked effective treatments beyond symptom management with corticosteroids or antibiotics.
Monoclonal antibodies provide species specific targeting that conventional drugs cannot match. Lokivetmab for canine atopic dermatitis directly binds and neutralizes canine interleukin 31, the cytokine associated with chronic itching, offering relief without the systemic side effects of corticosteroids like Cushing syndrome, diabetes, or liver damage. Bedinvetmab and frunevetmab target nerve growth factor overproduction in osteoarthritis, controlling pain in dogs and cats for four weeks or longer from a single injection.
Duration of effect differentiates biologics from small molecule drugs requiring daily administration. Anti nerve growth factor antibodies maintain therapeutic concentrations for 28 to 30 days through subcutaneous injection, eliminating compliance challenges associated with oral medications. European field studies demonstrated 43.5 percent of dogs treated with bedinvetmab showed pain score improvements compared with 16.9 percent receiving placebo, while United States trials achieved treatment success in 47.4 percent versus 36.6 percent for placebo.
Mechanism specificity reduces adverse events. Unlike broad spectrum pharmacotherapy affecting multiple biological pathways, monoclonal antibodies target single proteins involved in disease progression. Clinical trials for veterinary monoclonal antibodies report transient adverse events primarily limited to lethargy, inappetence, or gastrointestinal upset, contrasting sharply with corticosteroid complications or non steroidal anti inflammatory drug risks including kidney damage in cats with chronic kidney failure.
Cell culture and purification processes require specialized facilities meeting good manufacturing practice standards. Monoclonal antibody production uses mammalian cell lines that demand precise environmental control, nutrient delivery, and contamination prevention throughout multi week culture periods. Veterinary biologics manufacturers must achieve yields sufficient to support commercial volumes while maintaining consistency across production batches subject to USDA Center for Veterinary Biologics purity, potency, safety, and efficacy testing.
Fill finish operations present distinct challenges for veterinary biologics. Products must be formulated for single use vials or prefilled syringes compatible with veterinary practice workflows, maintaining stability throughout distribution and storage. Cold chain requirements for most veterinary monoclonal antibodies demand refrigeration between 2 and 8 degrees Celsius from manufacturing through clinic administration, constraining distribution networks and increasing logistics costs in regions lacking reliable temperature controlled supply chains.
Manufacturing scale determines pricing feasibility and product availability. Veterinary biologics command price premiums reflecting development costs, regulatory expenses, and production complexity, with monoclonal antibody therapies costing 100 to 300 dollars per dose depending on animal size and indication. Limited production capacity at qualified facilities creates supply constraints during peak demand periods, particularly as new indications expand addressable populations from seasonal allergy sufferers to year round chronic disease management.
USDA Center for Veterinary Biologics oversees products targeting immune system mechanisms under the Virus Serum Toxin Act. Lokivetmab received the first USDA license for monoclonal antibody therapy in 2016, establishing precedent for veterinary biologics evaluation criteria. Applicants must demonstrate that products are pure, safe, potent, and efficacious through standardized testing protocols before receiving establishment licenses and individual product licenses required for commercial distribution.
FDA Center for Veterinary Medicine regulates monoclonal antibodies with non immune targets. Frunevetmab for feline osteoarthritis received FDA approval in January 2022 as the first monoclonal antibody under FDA jurisdiction, followed by bedinvetmab for canine osteoarthritis in May 2023. The jurisdictional determination depends on whether a product works primarily through immune processes versus other biological mechanisms, with a joint USDA CVB and FDA CVM committee coordinating regulatory responsibility.
Conditional approvals and provisional licenses accelerate launch timelines when products address significant unmet needs. Elanco received conditional USDA approval for canine parvovirus monoclonal antibody in May 2023, enabling commercial use while additional efficacy data accumulates. Post approval surveillance requirements mandate ongoing safety monitoring through adverse event reporting systems, with manufacturers obligated to investigate suspected legal violations and consumer complaints throughout a product lifecycle.
Predictable dosing intervals create recurring revenue opportunities. Monthly administration schedules for lokivetmab, bedinvetmab, and frunevetmab establish routine clinic visits that strengthen veterinarian client relationships while generating consistent practice income. Veterinary clinics can structure wellness programs incorporating biologic therapies alongside preventive care, diagnostic monitoring, and nutritional counseling to increase client retention and lifetime value.
Subscription models reduce price sensitivity for biologics. Pet owners paying monthly premiums for comprehensive wellness plans perceive incremental costs differently than lump sum treatment expenses. Clinics offering subscription packages report improved compliance with therapeutic protocols as financial barriers decrease and convenience increases through scheduled appointments and automated reminder systems.
Long term chronic disease management aligns biologic characteristics with subscription economics. Osteoarthritis requires continuous pain control throughout an animal remaining lifespan, potentially spanning 5 to 10 years for medium and large breed dogs. At 12 treatments annually, a subscription approach generates 1200 to 3600 dollars in recurring revenue per patient while delivering consistent quality of life improvements that sustain owner satisfaction and referrals.

Veterinary monoclonal antibody therapeutics and development
Pet insurance spending and premiumisation trends
USDA regulatory framework for veterinary biologics
FDA veterinary medicine oversight
Veterinary practice and clinical implementation
Osteoarthritis, atopic dermatitis, allergic skin disease, and certain cancers demonstrate strong clinical responses to monoclonal antibodies. Conditions involving immune dysregulation, inflammatory cascades, or specific growth factor overproduction represent ideal targets. Ongoing research focuses on renal disease, cardiac conditions, and additional immune mediated disorders.
Pet owners demonstrate willingness to pay 3 to 5 times traditional medication costs when biologics offer superior efficacy, convenience, or safety profiles. Insurance coverage and subscription models reduce perceived price barriers. Premium segment households prioritize pet quality of life over treatment costs, particularly for chronic conditions affecting daily comfort.
Monoclonal antibodies typically require refrigeration between 2 and 8 degrees Celsius throughout distribution and storage. Products must not freeze, with temperature excursions potentially compromising efficacy. Clinics need dedicated refrigeration capacity with temperature monitoring and backup systems. Some formulations under development target ambient stability to expand accessibility.
USDA CVB and FDA CVM evaluate purity through analytical testing, safety via target species trials, potency using validated assays, and efficacy through controlled clinical studies. Immunogenicity assessment examines antibody responses against the therapeutic protein itself. Post marketing surveillance tracks adverse events and long term safety signals across diverse patient populations.
Wellness subscription plans bundle preventive care, routine diagnostics, and chronic disease management including biologic therapies. Monthly payment structures reduce financial barriers while generating predictable revenue. Digital platforms enable automated appointment scheduling, inventory management, and client communications. Partnerships between pharmaceutical companies and veterinary chains support adoption.
Animal Healthcare Packaging Market Size and Share Forecast Outlook 2025 to 2035
Companion Animal Healthcare Market Size and Share Forecast Outlook 2025 to 2035
Demand for Animal Healthcare in UK Size and Share Forecast Outlook 2026 to 2036
Demand for Animal Healthcare in South Korea Size and Share Forecast Outlook 2026 to 2036
Demand for Animal Healthcare in Japan Size and Share Forecast Outlook 2025 to 2035