Regulatory frameworks governing PVDC coated films reflect fundamentally different approaches to environmental policy, chemical risk assessment, and waste management infrastructure. European authorities operate under precautionary principles embedded in circular economy legislation, where material recyclability and system compatibility drive policy decisions. This creates inherent tension with multilayer structures containing chlorinated polymers, regardless of direct toxicity evidence. The emphasis on design for recycling and extended producer responsibility schemes prioritizes mono-material solutions that can be processed through existing mechanical recycling infrastructure.
North American regulatory systems employ risk-based frameworks that evaluate materials based on specific use applications and exposure pathways. The FDA and Health Canada assess PVDC coatings primarily through food contact safety criteria, migration studies, and functional performance validation. Environmental considerations focus on disposal pathway safety rather than upstream material selection, with less emphasis on recyclability mandates compared to European directives.
Asian regulatory environments vary significantly but generally prioritize food safety, shelf life extension, and export compliance over material composition restrictions. Countries with developing cold chain infrastructure view high barrier materials as essential food waste prevention tools. Regulatory capacity focuses on ensuring packaging performs its protective function rather than imposing material preference hierarchies. China and India maintain approval frameworks that accommodate PVDC where functional justification exists, particularly for pharmaceutical and high value food applications.
European policy creates a constrained rather than prohibited environment for PVDC coated films. The EU Packaging and Packaging Waste Directive establishes recyclability targets that disadvantage multilayer structures, while member state interpretations vary in stringency. Germany applies particularly aggressive extended producer responsibility fees that economically penalize non-recyclable formats. France has implemented bonus-malus systems within eco-contributions that make PVDC containing packages financially less attractive.
Retail consolidation amplifies regulatory pressure through private standards that exceed legal requirements. Major European grocery chains have established voluntary phase-out commitments, creating de facto access barriers even where PVDC remains technically permissible. This retail-driven constraint affects brand owner material choices more directly than formal regulation in many cases.
The European Chemicals Agency maintains PVDC and its additives under continuous review through REACH provisions, creating regulatory uncertainty that discourages long-term investment in PVDC-based solutions. While no outright bans exist for packaging applications, the compliance burden and reputational considerations push converters toward alternative barrier technologies. However, pharmaceutical blister packaging and certain processed meat applications retain PVDC usage where no equivalent performing alternative has achieved regulatory approval and validation.
United States and Canadian regulatory frameworks permit PVDC coated films across food and pharmaceutical applications based on demonstrated safety and performance criteria. The FDA maintains food contact substance approvals for PVDC formulations meeting migration thresholds and compositional specifications. This creates a stable regulatory environment where material selection decisions rest primarily on technical and economic factors rather than policy-driven substitution pressure.
Recycling policy in North America emphasizes infrastructure development and collection system optimization rather than material bans. The focus on improving sortation technology and developing advanced recycling pathways creates a longer timeline for material transitions. Industry groups argue that flexible packaging using PVDC prevents more food waste than the packaging itself represents, a position that receives regulatory consideration in risk-benefit assessments.
Pharmaceutical packaging regulations strongly favor established barrier materials with extensive validation histories. PVDC coated films for blister packs and unit dose packaging benefit from this regulatory conservatism, where changing materials requires expensive stability studies and regulatory submissions. Generic drug manufacturers particularly rely on PVDC platforms that match innovator packaging specifications to support bioequivalence claims.
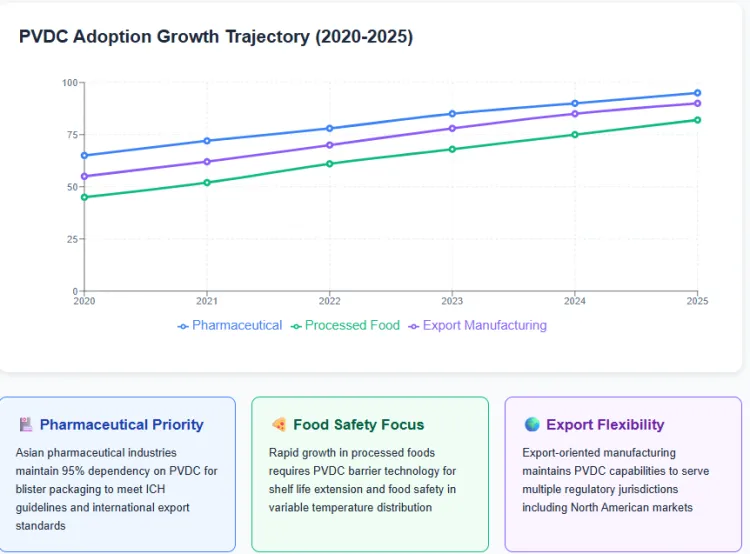
Asian pharmaceutical industries depend heavily on PVDC coated films for blister packaging meeting international export standards. Regulatory authorities in India, China, and ASEAN countries maintain approval frameworks aligned with ICH guidelines that recognize PVDC as an acceptable barrier material. The region produces significant volumes of generic pharmaceuticals for global distribution, necessitating packaging materials accepted across multiple regulatory jurisdictions.
Processed food categories experiencing rapid growth in Asia require extended shelf life capabilities that PVDC coatings reliably deliver. Coffee, snack foods, processed meats, and cheese products utilize PVDC barrier technology to maintain quality through distribution networks with variable temperature control. Regulatory systems prioritize food safety outcomes over packaging material composition, viewing barrier performance as a public health benefit that reduces foodborne illness risk.
Export oriented manufacturing creates additional incentives for PVDC retention, as Asian producers supply packaged goods to North American and other permissive regulatory zones. Maintaining PVDC coating capabilities allows flexible response to customer specifications across different destination requirements. Investment in coating technology remains economically justified by serving multiple geographic segments with differentiated regulatory tolerances.
Global brand owners increasingly operate parallel packaging specifications that deploy PVDC coated films in permissive regions while using alternative barrier technologies in Europe. This dual strategy requires maintaining relationships with multiple material suppliers, qualifying different film structures for equivalent products, and managing supply chain complexity across regional operations.
Pharmaceutical companies face particular challenges reconciling global product registration requirements with regional packaging preferences. A drug approved with PVDC blister packaging in one jurisdiction may require separate stability studies to support alternative packaging in another region. This creates strong incentives to maintain PVDC specifications globally despite European pressure, as the regulatory and validation costs of regional variation often exceed material cost differences.
Food manufacturers adopt more flexible regional approaches, reformulating package structures for European distribution while retaining PVDC platforms elsewhere. This involves parallel converter relationships, separate SKU management systems, and regional quality assurance protocols. The operational complexity imposes costs but remains preferable to either universal PVDC elimination or losing European distribution access.

Regulatory philosophies differ fundamentally between precautionary European approaches emphasizing recyclability and risk-based North American frameworks focusing on use-specific safety, creating divergent material acceptability standards.
Current trends suggest continued regional divergence rather than convergence toward universal prohibition, as performance requirements and infrastructure capabilities vary significantly across geographies.
Through parallel specifications, regional supplier relationships, and supply chain segmentation that increases operational complexity but maintains access to all geographic segments.
Pharmaceutical blister packaging and certain processed food categories where no regulatory-approved alternative delivers equivalent barrier performance at commercial scale.
Convergence appears unlikely in the near term given fundamental differences in environmental policy priorities, waste management infrastructure, and chemical risk assessment methodologies across major regulatory zones.
Aluminum Oxide Coated Films Market
Top Coated Direct Thermal Printing Films Market Size and Share Forecast Outlook 2025 to 2035
Competitive Breakdown of Top Coated Direct Thermal Printing Films Providers
Coated Fabrics Market is segmented by Material Type (Polymer-coated Fabrics, Rubber-coated Fabrics, Fabric-backed Vinyl Composites, Silicone-coated Fabrics, Fluoropolymer-coated Fabrics), Substrate (Polyester, Nylon, Cotton and Blends, Fiberglass, Aramid and High-modulus Fibers), Application (Transportation, Protective Clothing, Industrial, Commercial Tents and Structures, Furniture and Upholstery, Other Applications), Coating Chemistry (PU, PVC, TPU, Silicone, PTFE/PVDF Systems), Distribution Channel (Direct OEM Supply, Converters and Fabricators, Industrial Distributors, Project-based Procurement, Aftermarket and Replacement Sales), Performance Property (Abrasion Resistance, Chemical Resistance, Flame Retardancy, Weatherability, Low-emission Interiors), and Region. Forecast for 2026 to 2036.
Coated Label Market Size and Share Forecast Outlook 2025 to 2035