The United States maintains structural advantages in antibiotic innovation through regulatory incentives and pharmaceutical industry concentration. The FDA operates dedicated pathways for antibacterial drug development, including the Qualified Infectious Disease Product designation, which grants five additional years of regulatory exclusivity for drugs targeting serious bacterial infections. This framework has accelerated approval of agents such as cefiderocol and plazomicin, both of which address multidrug resistant uropathogens.
Major pharmaceutical companies maintain US headquarters or primary regulatory relationships with the FDA, resulting in sequential approval strategies that prioritize the American jurisdiction. The agency has established streamlined clinical trial requirements for uncomplicated and complicated UTI indications, reducing development timelines compared to multi country regulatory submissions. The combination of regulatory clarity, intellectual property protection, and established clinical trial infrastructure positions the United States as the first approval territory for most novel anti infectives.
Beyond novel agents, the United States has seen reformulation and lifecycle management strategies that improve dosing convenience and resistance mitigation. Extended release formulations of nitrofurantoin and fosfomycin, as well as combination products pairing beta lactams with beta lactamase inhibitors, have received FDA approval ahead of other jurisdictions. This leadership in drug availability directly impacts prescribing options and resistance management strategies.
Hospital based antimicrobial stewardship programs have become mandatory in the United States through accreditation requirements established by The Joint Commission and the Centers for Medicare and Medicaid Services. These programs enforce prescribing audits, formulary restrictions, and prospective authorization requirements for broad spectrum antibiotics commonly used in complicated UTI treatment.
Stewardship committees review antibiotic utilization data, track resistance patterns within institutions, and implement interventions that direct prescribers toward narrow spectrum agents when clinically appropriate. For UTI treatment, this has translated into reduced use of fluoroquinolones in uncomplicated cases and tighter control over carbapenem use in complicated infections. Many institutions have implemented automatic stop orders for empiric therapy pending culture results, forcing reassessment based on microbiological data.
Outpatient stewardship has gained traction through electronic health record interventions, clinical decision support tools, and payer prior authorization requirements. These mechanisms reduce inappropriate antibiotic prescribing for asymptomatic bacteriuria and cystitis, conditions where overtreatment contributes significantly to resistance pressure. The Centers for Disease Control and Prevention has published national action plans reinforcing stewardship principles, creating a policy environment that prioritizes judicious antibiotic use over volume based prescribing.
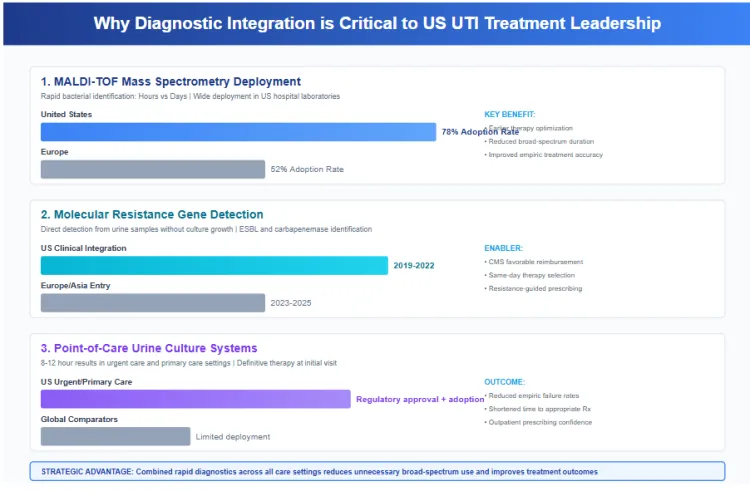
The United States has achieved higher adoption rates of rapid microbiological diagnostics compared to most other developed countries. Matrix assisted laser desorption ionization time of flight mass spectrometry, which identifies bacterial species within hours rather than days, has been deployed widely in US hospital laboratories. This technology enables earlier therapy optimization and reduces the duration of empiric broad spectrum coverage.
Molecular diagnostics capable of detecting resistance genes directly from urine samples have entered clinical use in the United States ahead of Europe and Asia. These platforms identify extended spectrum beta lactamase production and carbapenemase genes without requiring culture growth, allowing clinicians to select appropriate therapy at the time of diagnosis. The integration of these diagnostics into clinical workflows has been supported by favorable reimbursement decisions from the Centers for Medicare and Medicaid Services.
Point of care urine culture systems have also gained regulatory approval and commercial adoption in US urgent care and primary care settings. These devices provide preliminary identification and susceptibility data within eight to twelve hours, enabling outpatient providers to prescribe definitive therapy during the initial visit. The combination of rapid hospital diagnostics and expanding point of care capabilities has reduced empiric treatment failure rates and shortened time to appropriate therapy.
US healthcare reimbursement structures create financial incentives for adherence to evidence based treatment guidelines. Medicare and major private payers have implemented bundled payment models and quality metrics that penalize avoidable UTI related readmissions and complications from inappropriate antibiotic selection. These mechanisms pressure hospitals and physician groups to follow guideline concordant therapy.
Documentation requirements enforced by payers demand justification for broad spectrum antibiotic use, including evidence of prior treatment failure or documented resistance patterns. This administrative burden naturally directs prescribers toward narrow spectrum first line agents when clinical criteria permit. Prior authorization requirements for certain antibiotics create additional friction that reinforces stewardship principles.
Medical liability considerations also shape prescribing behavior in the United States more than in countries with different tort systems. Physicians face malpractice risk for both undertreatment and overtreatment of infections. This dual pressure has resulted in standardization around evidence based guidelines issued by the Infectious Diseases Society of America and other specialty organizations, as adherence to published standards provides legal defensibility. The combination of reimbursement constraints and liability exposure creates powerful incentives for protocol driven UTI treatment.
Clinical practice guidelines published by American professional societies serve as reference documents for international guideline developers. The Infectious Diseases Society of America UTI treatment guidelines are widely cited in European, Asian, and Latin American protocols, establishing de facto international standards for empiric therapy selection and duration.
Most multinational clinical trials for new antibiotics are led by US based investigators and coordinated through American academic medical centers. These trials establish the evidence base that regulatory agencies worldwide use for drug approvals. The FDA approval of a new antibiotic creates momentum for subsequent submissions to the European Medicines Agency and other regulators, with clinical data packages largely determined by US trial design.
Pharmaceutical companies structure global product launches around initial US approval and commercialization, followed by sequential international rollouts. This sequencing means that clinical experience, prescribing patterns, and resistance surveillance data accumulate first in the United States. International clinicians subsequently adopt treatment approaches validated in the US healthcare system, reinforcing American leadership in UTI therapeutic strategies.

Bibliography
Treatment protocols vary based on local resistance patterns, antibiotic availability, diagnostic infrastructure, and regulatory frameworks. The United States maintains broader access to novel agents and enforces stricter prescribing governance through stewardship programs, while other countries may prioritize cost containment or have different resistance epidemiology.
Resistance patterns determine which empiric antibiotics remain effective for uncomplicated UTI treatment. Rising fluoroquinolone resistance has shifted US guidelines toward nitrofurantoin and fosfomycin for uncomplicated cystitis. Hospitals with high extended spectrum beta lactamase prevalence require carbapenem use for complicated infections, driving stewardship interventions.
FDA regulatory pathways, intellectual property protection, commercial infrastructure, and reimbursement predictability make the United States the preferred initial launch territory. Companies prioritize US approval to establish commercial presence and generate clinical experience before pursuing international registrations.
Rapid identification and susceptibility testing enable early transition from empiric broad spectrum therapy to pathogen directed narrow spectrum agents. Molecular diagnostics that detect resistance genes prevent use of antibiotics predicted to fail, reducing treatment duration and inappropriate prescribing for colonization rather than infection.
Evidence demonstrates that antimicrobial stewardship reduces antibiotic related adverse events, healthcare associated infections, and institutional resistance rates without increasing treatment failures. Stewardship interventions that optimize antibiotic selection, dosing, and duration have been associated with improved clinical outcomes and reduced healthcare costs.
Uncomplicated Urinary Tract Infection Treatment Market Analysis by Penicillin, Quinolones, Sulfonamide, Nitrofuran, and Others Through 2035
Market Share Breakdown of Uncomplicated Urinary Tract Infection Treatment
Demand for Uncomplicated Urinary Tract Infection Treatment in Japan Size and Share Forecast Outlook 2025 to 2035
Urinary Tract Infection (UTI) Treatment Market (UTI) Analysis - Size, Share, and Forecast 2025 to 2035
Complicated Urinary Tract Infections Treatment Market is segmented by Drug Class (Cephalosporin, Penicillin and Combinations, Quinolones, Azoles and Amphotericin B, Nitro Furans), Distribution Channel (Hospital Pharmacies, Gynecology and Urology Clinics, Drug Stores, Retail Pharmacies), Mechanism Of Action (Broad-Spectrum Antibiotics, Beta-Lactamase Inhibitor Combinations, Carbapenems, Aminoglycosides), and Region. Forecast for 2026 to 2036.