Absorbable Tissue Spacer Market
The absorbable tissue spacer market is segmented by Technology (Hydrogel-Based Spacer, Biodegradable Hyaluronic Acid Spacer, Biodegradable Balloon Spacer) and End User (Hospitals, Ambulatory Surgical Centers, Specialized Clinics). Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Absorbable Tissue Spacer Market Size, Market Forecast and Outlook By FMI
The absorbable tissue spacer market is expected to expand from USD 542.0 million in 2026 to USD 1,027.0 million by 2036. Market revenue is anticipated to register a 6.6% CAGR during the forecast period. Hydrogel-based spacers are likely to lead with 70.2% share, and hospitals are projected to account for 69.0% of end user demand in 2026.
Summary of the Absorbable Tissue Spacer Market
- Demand and Growth Drivers
- Precision radiotherapy adoption is expected to raise spacer use across oncology procedures needing healthy tissue protection.
- Cancer care teams are likely to prefer absorbable spacers as treatment planning places stronger focus on toxicity reduction.
- Image-guided procedures are anticipated to improve clinical confidence in spacer placement across advanced radiation therapy settings.
- Product and Segment View
- Hydrogel-based spacers are expected to lead the technology segment as clinicians value stable separation during treatment windows.
- Biodegradable hyaluronic acid spacers are likely to gain attention as bioresorbable materials receive higher procedural interest.
- Hospitals are anticipated to lead end user demand as complex oncology care needs trained teams and radiotherapy infrastructure.
- Geography and Competitive Outlook
- India is expected to lead country-level expansion as cancer treatment access improves across advanced hospital networks.
- China is likely to record steady adoption as radiotherapy capacity increases across large urban medical systems.
- Companies with stronger biomaterial design and easier placement systems are anticipated to improve their standing in oncology care.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant for Healthcare at FMI, suggests, “The absorbable tissue spacer market is likely to advance as oncology teams place stronger emphasis on patient safety and radiation precision. Hydrogel-based spacer suppliers are expected to gain advantage as hospitals favor products with predictable absorption and easier clinical placement.”
- Absorbable Tissue Spacer Market Value Analysis
- The absorbable tissue spacer market is shifting from selective oncology use toward broader radiotherapy workflow adoption.
- Demand is likely to rise as hospitals seek tools reducing radiation exposure to healthy surrounding tissues.
- Adoption is expected to increase as hydrogel-based spacers offer predictable placement and reliable temporary tissue separation.
- Spending is anticipated to be shaped by cancer procedure volumes and hospital investment in precision radiotherapy systems.
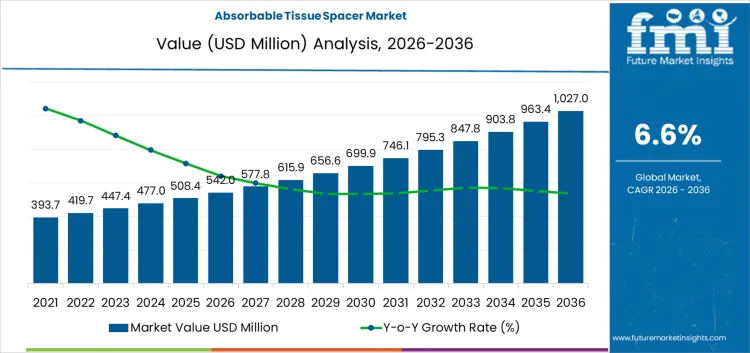
Absorbable Tissue Spacer Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 542 Million |
| Forecast Value (2036) | USD 1,027.0 Million |
| Forecast CAGR 2026 to 2036 | 6.60% |
How is the Global Absorbable Tissue Spacer Market Advancing?
The global absorbable tissue spacer market is experiencing significant growth, driven by the increasing use of absorbable tissue spacers in cancer treatments, particularly in radiation therapy for prostate cancer, breast cancer, and other malignancies. Absorbable tissue spacers are used to create a protective barrier between healthy tissue and radiation fields, reducing the risk of side effects from radiation therapy. As more cancer patients undergo radiation therapy, the demand for effective, safe, and minimally invasive treatment solutions is driving the adoption of absorbable tissue spacers.
The market is primarily driven by the growing prevalence of cancer, advancements in cancer treatment technologies, and an increasing focus on improving patient outcomes. Absorbable tissue spacers, made from biocompatible materials, offer several advantages, including reduced need for surgical intervention, fewer complications, and quicker recovery times compared to traditional spacer techniques. As more hospitals and cancer treatment centers embrace these innovative solutions to enhance precision in radiation therapy, the market is poised for continued growth.
Technological advancements in absorbable tissue spacer materials, such as improved bioabsorbable polymers and enhanced safety profiles, are expected to contribute to the market’s expansion. As the healthcare industry continues to prioritize patient safety, comfort, and the reduction of radiation-related side effects, the absorbable tissue spacer market is set to grow steadily, offering advanced therapeutic options for cancer patients.
What is the Segment-Wise Analysis for the Absorbable Tissue Spacer Market?
The absorbable tissue spacer market is segmented by technology and end user. Hydrogel-based spacers lead the market with a 70.20% share, followed by biodegradable hyaluronic acid spacers and biodegradable balloon spacers. These technologies are used to create temporary spacing between tissues during procedures such as radiation therapy, helping to protect healthy tissues from damage. In terms of end user, hospitals dominate the market with 69% of the share, followed by ambulatory surgical centers and specialized clinics.
Why Do Hydrogel-Based Spacers Lead the Absorbable Tissue Spacer Market?
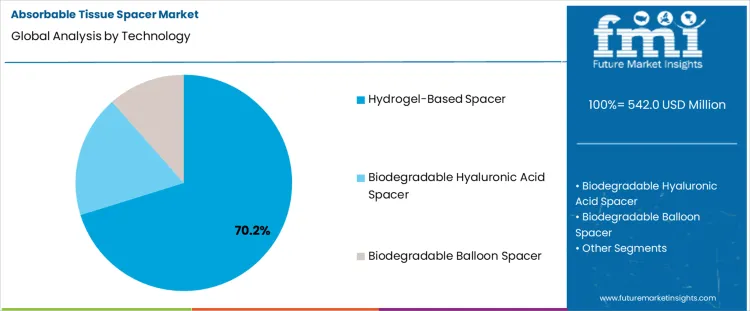
Hydrogel-based spacers account for 70.20% of the absorbable tissue spacer market share due to their superior biocompatibility, ease of application, and ability to maintain their structure during the therapeutic process. Hydrogel-based spacers are preferred for their ability to create temporary separation between tissues, especially in radiation therapy for cancer treatment. The hydrogel technology provides a soft, flexible, and highly absorbable material that reduces the risk of surrounding tissue damage while maintaining its function throughout the treatment. As the demand for less invasive, safer treatment options continues to rise, hydrogel-based spacers remain the leading choice for absorbable tissue spacers in medical procedures.
How Do Hospitals Contribute to the Absorbable Tissue Spacer Market?
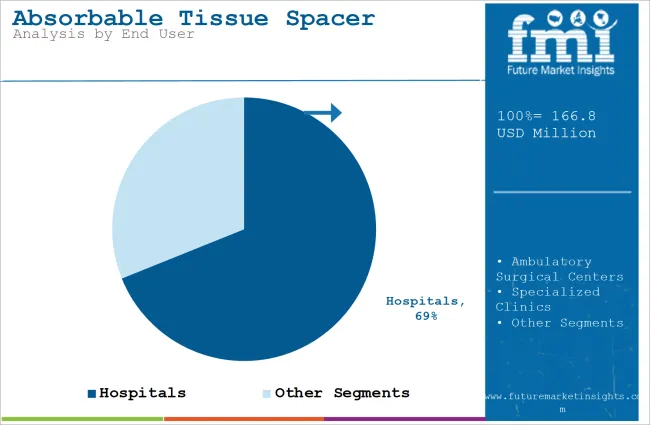
Hospitals represent 69% of the end user segment in the absorbable tissue spacer market. Hospitals are the primary settings where these spacers are used, especially for procedures such as radiation therapy, where protecting healthy tissues from radiation damage is critical. The demand for absorbable tissue spacers is growing in hospitals due to the increasing number of cancer treatments and the adoption of advanced therapeutic techniques. As hospitals focus on improving patient outcomes with less invasive procedures, the use of hydrogel-based and other types of absorbable tissue spacers continues to rise, solidifying hospitals as the dominant contributor to the market's growth.
What Are the Key Dynamics for the Absorbable Tissue Spacer Market?
Absorbable tissue spacers are used to physically separate tumors from adjacent critical organs, minimizing radiation dose to normal tissues and reducing complications. Increasing cancer incidence worldwide, particularly prostate, rectal, and gynecological cancers treated with radiotherapy, fuels demand. Advances in biomaterials and minimally invasive delivery techniques improve clinical adoption. Rising healthcare infrastructure investments and expanding radiotherapy capabilities in emerging countries support market growth as clinicians seek tools that enhance therapeutic outcomes while reducing long‑term side effects.
How Are the Key Drivers Shaping the Absorbable Tissue Spacer Market?
Growth in the Absorbable Tissue Spacer Market is shaped by the rising focus on precision oncology and patient safety in radiation treatment. Clinicians increasingly adopt absorbable spacers to protect healthy organs, such as the rectum in prostate cancer patients, thereby reducing toxicity and improving quality of life. Technological advancements in bioresorbable polymers and delivery systems make spacers easier to implant with predictable resorption profiles. Expansion of radiotherapy indications and higher utilization of dose‑escalated techniques also increase the need for spacers. Supportive reimbursement policies and clinical evidence demonstrating reduced adverse events contribute to broader acceptance across oncology centers globally.
What Are the Key Trends Shaping the Absorbable Tissue Spacer Market?
Key trends in the absorbable tissue spacer market include the development of next‑generation bioresorbable materials that offer tailored resorption rates and enhanced biocompatibility. There is increasing research into spacers with integrated imaging markers that improve placement accuracy and treatment planning. Use of minimally invasive and image‑guided delivery techniques enhances procedural safety and patient comfort. Growing evidence from clinical studies demonstrating reduced radiation‑induced toxicities strengthens adoption across a wider range of cancer types. Collaborations between biomaterial developers, oncology centers, and radiotherapy equipment manufacturers are accelerating innovation and expanding applications in both curative and palliative care settings.
What is the Growth Outlook for the Absorbable Tissue Spacer Market by Country?
| Country | CAGR (%) |
|---|---|
| India | 6.8% |
| China | 6.2% |
| South Korea | 5.9% |
| France | 5.3% |
| Germany | 5.0% |
The absorbable tissue spacer market is growing steadily across key regions, with India leading at 6.8%, driven by the country’s expanding healthcare infrastructure and increasing adoption of advanced cancer treatment technologies. The rising prevalence of cancers, particularly in the prostate and breast, has significantly contributed to the demand for absorbable tissue spacers in radiation therapy. China’s market grows at 6.2%, supported by its large population, growing awareness of cancer treatments, and improved healthcare access. South Korea’s market grows at 5.9%, fueled by the adoption of modern radiotherapy techniques and the country’s focus on advancing cancer care. France’s market grows at 5.3%, driven by increased adoption of absorbable tissue spacers in radiotherapy for localized cancers. Germany’s market grows at 5.0%, supported by strong healthcare infrastructure and a rising number of cancer diagnoses.
What is Driving the Growth of the Absorbable Tissue Spacer Market in India?
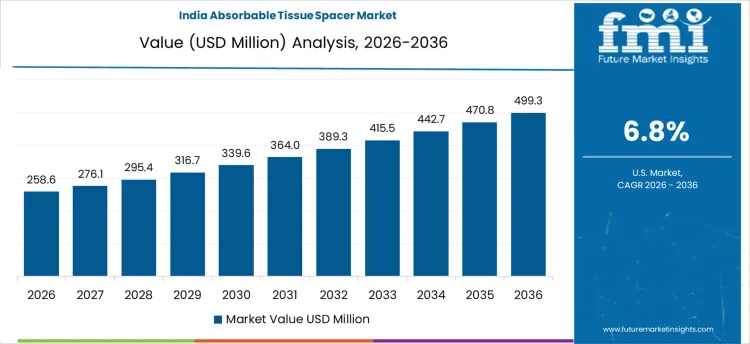
India’s absorbable tissue spacer market is growing at 6.8%, driven by the country’s expanding healthcare infrastructure and rising awareness of cancer treatments. As cancer incidence increases, particularly prostate and breast cancers, there is an increasing need for absorbable tissue spacers to improve the effectiveness of radiation therapy. India’s healthcare system is evolving, with greater access to advanced medical technologies, leading to a higher adoption rate of modern treatment methods, including the use of tissue spacers. India’s growing middle class and increasing medical tourism contribute to the demand for effective cancer care. As more hospitals and cancer centers incorporate these advanced technologies into their treatment protocols, India’s market for absorbable tissue spacers is expected to continue its growth trajectory.
How is the Absorbable Tissue Spacer Market Expanding in China?
China’s absorbable tissue spacer market is growing at 6.2%, supported by its vast population and the increasing prevalence of cancers such as prostate and breast cancer. As China’s healthcare infrastructure improves, the adoption of advanced cancer therapies, including the use of absorbable tissue spacers, is gaining traction. These spacers are crucial in radiation therapy to reduce damage to healthy tissues and enhance treatment precision. The Chinese government’s investment in healthcare, along with the increasing demand for high-quality cancer treatments, is contributing to market growth. With a growing awareness of the benefits of modern radiation therapy techniques, China’s market for absorbable tissue spacers is expected to expand as more medical institutions incorporate these technologies into their cancer treatment protocols.
What is Contributing to the Growth of the Absorbable Tissue Spacer Market in South Korea?
South Korea’s absorbable tissue spacer market is growing at 5.9%, driven by the country’s advanced healthcare system and rising demand for precision in cancer treatment. South Korea is known for its strong focus on research and development in medical technologies, and the adoption of absorbable tissue spacers is becoming increasingly common in radiotherapy for prostate and breast cancer. The growing number of cancer cases, particularly among the aging population, is fueling demand for effective and minimally invasive treatment options. The rise of medical tourism and South Korea’s reputation for high-quality medical care are contributing to the market’s growth. As the demand for more effective and personalized cancer treatments rises, South Korea’s market for absorbable tissue spacers is expected to grow steadily.
How is the Absorbable Tissue Spacer Market Expanding in France?
France’s absorbable tissue spacer market is growing at 5.3%, driven by an increasing focus on cancer treatment and innovations in radiation therapy. The country’s healthcare system is highly advanced, with a strong emphasis on providing high-quality cancer care. As the incidence of cancers like prostate and breast cancer rises, the adoption of absorbable tissue spacers in radiotherapy is becoming more prevalent. These spacers help minimize damage to healthy tissue during radiation therapy, improving patient outcomes. The French government’s efforts to enhance healthcare access and the rising awareness of modern cancer treatments further contribute to market growth. As French healthcare providers continue to embrace these advanced therapeutic techniques, the market for absorbable tissue spacers is expected to continue expanding.
What is Driving the Growth of the Absorbable Tissue Spacer Market in Germany?
Germany’s absorbable tissue spacer market is growing at 5.0%, supported by the country’s advanced healthcare infrastructure and a strong focus on cancer care. Germany is a leader in the adoption of cutting-edge medical technologies, and absorbable tissue spacers are increasingly being used in radiotherapy for prostate and breast cancer. The rising prevalence of cancer, along with Germany’s aging population, is driving the demand for precise, effective, and minimally invasive treatment options. The country’s well-established research and development sector, combined with growing awareness of radiation therapy advancements, is further boosting the market. As German healthcare institutions continue to adopt these innovative solutions, the demand for absorbable tissue spacers is expected to grow steadily in the coming years.
How Are Companies Competing in the Absorbable Tissue Spacer Market?
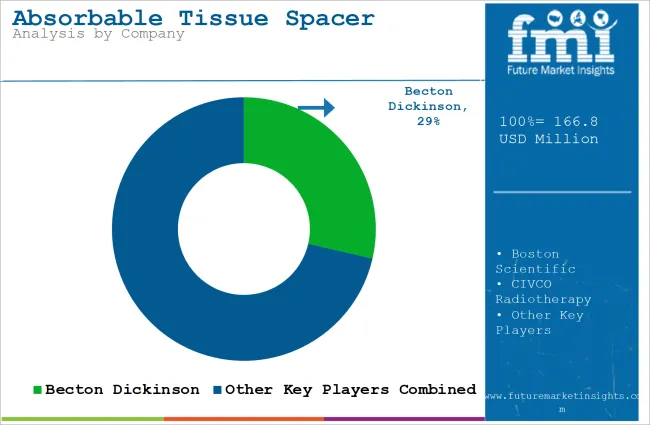
Competition in the absorbable tissue spacer market is driven by biocompatibility, ease of placement, predictable absorption profiles, and integration with radiation therapy workflows. Becton Dickinson positions its spacers with materials engineered for consistent separation and reliable bioresorption, and its product literature highlights ease of use in clinical procedures and compatibility with imaging guidance. Boston Scientific competes by offering precisely engineered spacer systems that emphasize controlled deployment and stable geometry during critical treatment windows.
CIVCO Radiotherapy and Elekta differentiate with image guided placement platforms and spacers designed to work seamlessly with radiation oncology workflows, with collateral stressing placement accuracy and minimal procedural complexity. IZI Medical Products focuses on user friendly spacer designs that support reproducible separation and integration with existing interventional techniques. Medtronic brings a broad medical device portfolio to the market, and its product materials underscore device reliability and clinical support infrastructure.
Qfix and Smith & Nephew compete with customized spacer solutions tailored for specific anatomical sites, marketed for optimal spacing and reduced patient discomfort. Stryker adds value with precision engineered systems backed by procedural training resources that help clinicians adopt spacer technologies with confidence. Varian Medical Systems rounds out the landscape with spacer offerings embedded within comprehensive radiotherapy solutions, and its documentation highlights alignment with treatment planning systems and imaging modalities. Across all players, product literature emphasizes predictable bioresorption, clinical effectiveness, ease of placement, and integration into therapeutic workflows as key differentiators that help clinicians improve treatment precision and patient outcomes.
Key Players in Absorbable Tissue Spacer Market
- Becton Dickinson
- Boston Scientific
- CIVCO Radiotherapy
- Elekta
- IZI Medical Products
- Medtronic
- Qfix
- Smith & Nephew
- Stryker
- Varian Medical Systems
Scope of Report
| Attributes | Description |
|---|---|
| Quantitative Unit (2026) | USD Million |
| Technology | Hydrogel Based Spacer, Biodegradable Hyaluronic Acid Spacer, Biodegradable Balloon Spacer |
| End Users | Hospitals, Ambulatory Surgical Centers, Specialized Clinics |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Germany, UK, France, Italy, Spain, Nordic, BENELUX, USA, Canada, Mexico, Brazil, Chile, Turkey, South Africa, Other GCC Countries, Other African Union |
| Key Companies Profiled | Becton Dickinson, Boston Scientific, CIVCO Radiotherapy, Elekta, IZI Medical Products, Medtronic, Qfix, Smith & Nephew, Stryker, Varian Medical Systems |
| Additional Attributes | Dollar sales by technology, end-user, and region; regional CAGR and growth outlook for absorbable tissue spacers. |
Absorbable Tissue Spacer Market by Key Segments
Technology
- Hydrogel‑Based Spacer
- Biodegradable Hyaluronic Acid Spacer
- Biodegradable Balloon Spacer
End User
- Hospitals
- Ambulatory Surgical Centers
- Specialized Clinics
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the absorbable tissue spacer market in 2026?
The global absorbable tissue spacer market is estimated to be valued at USD 542.0 million in 2026.
What will be the size of absorbable tissue spacer market in 2036?
The market size for the absorbable tissue spacer market is projected to reach USD 1,027.0 million by 2036.
How much will be the absorbable tissue spacer market growth between 2026 and 2036?
The absorbable tissue spacer market is expected to grow at a 6.6% CAGR between 2026 and 2036.
What are the key product types in the absorbable tissue spacer market?
The key product types in absorbable tissue spacer market are hydrogel‑based spacer, biodegradable hyaluronic acid spacer and biodegradable balloon spacer.
Which end user segment to contribute significant share in the absorbable tissue spacer market in 2026?
In terms of end user, hospitals segment to command 69.0% share in the absorbable tissue spacer market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- M
- arket Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology , 2026 to 2036
- Hydrogel‑Based Spacer
- Biodegradable Hyaluronic Acid Spacer
- Biodegradable Balloon Spacer
- Hydrogel‑Based Spacer
- Y to o to Y Growth Trend Analysis By Technology , 2021 to 2025
- Absolute $ Opportunity Analysis By Technology , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Ambulatory Surgical Centers
- Specialized Clinics
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Technology
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Technology
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Technology
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Technology
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Technology
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Technology
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Technology
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Technology
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Technology
- By End User
- Competition Analysis
- Competition Deep Dive
- Becton Dickinson
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Boston Scientific
- CIVCO Radiotherapy
- Elekta
- IZI Medical Products
- Medtronic
- Qfix
- Smith & Nephew
- Stryker
- Varian Medical Systems
- Becton Dickinson
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Technology , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Technology , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Technology , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Technology , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Technology , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Technology , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Technology , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Technology , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Technology , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Technology , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Technology
- Figure 6: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End User
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Technology , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Technology , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Technology
- Figure 23: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by End User
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Technology , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Technology , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Technology
- Figure 30: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by End User
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Technology , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Technology , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Technology
- Figure 37: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by End User
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Technology , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Technology , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End User
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Technology , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Technology , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Technology
- Figure 51: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by End User
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Technology , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Technology , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Technology , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

