Healthcare Regulatory Affairs Outsourcing Market
The healthcare regulatory affairs outsourcing market is segmented by Service (Regulatory Consulting, Legal Representation, Regulatory Writing and Publishing, Product Registration and Clinical Trial Applications, Others), by End-User (Pharmaceutical and Biotechnology Companies, Medical Device Companies, Food and Beverage Companies), and by Region. Forecast for 2026 to 2036.
Healthcare Regulatory Affairs Outsourcing Market Size, Market Forecast and Outlook By FMI
In 2025, the healthcare regulatory affairs outsourcing market was valued at USD 2.3 billion. Based on Future Market Insights' analysis, demand for healthcare regulatory affairs outsourcing is estimated to grow to USD 2.45 billion in 2026 and USD 4.56 billion by 2036. FMI projects a CAGR of 6.7% during the forecast period.
An absolute dollar growth of USD 2.11 billion between 2026 and 2036 reflects regulatory intensification in pharmaceutical development pathways rather than volume-only clinical trial expansion. Growth depends on increased conversion from in-house regulatory functions to outsourced models that deliver specialized expertise in orphan drug designations, breakthrough therapy filings, and expedited approval pathways while maintaining compliance with evolving global standards. As per FMI, regulatory service providers are capturing value not from incremental full-time equivalent staffing alone but from strategic consulting complexity embedded in pediatric study planning, post-marketing commitment management, and real-world evidence integration that defines modern drug development programs.
Summary of the Healthcare Regulatory Affairs Outsourcing Market
- Market Definition
- The market comprises specialized services supporting regulatory compliance, submission preparation, and health authority interactions for pharmaceutical, biotechnology, and medical device companies.
- Demand Drivers
- Increasing complexity of global regulatory frameworks requiring specialized expertise unavailable in-house.
- Growing pharmaceutical and biotechnology pipelines with specialized therapeutic modalities demanding nuanced regulatory strategies.
- Expansion of expedited approval pathways requiring strategic regulatory planning and proactive agency engagement.
- Key Segments Analyzed
- Service: Regulatory Writing and Publishing leads, accounting for 27.4% share in 2025 due to persistent demand for compliant clinical study reports and electronic common technical document dossiers.
- End-User: Pharmaceutical companies dominate, driven by large pipelines and multi-region filing requirements.
- Region: North America leads value, while Asia-Pacific drives volume through clinical trial activity expansion.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare at Future Market Insights, comments: "This updated edition of the Healthcare Regulatory Affairs Outsourcing Market report captures a strategic inflection where service value is shifting from transactional document preparation toward strategic regulatory advisory integrated with advanced technology enablement. Vendors combining deep therapeutic expertise in cell and gene therapies with AI-powered regulatory intelligence platforms that monitor global health authority guidance changes in real time are securing preferred provider positions.”
- Strategic Implications/Executive Takeaways
- Invest in therapeutic area specialization, particularly in oncology, rare diseases, and advanced therapy medicinal products where regulatory precedent is limited.
- Develop AI-enabled regulatory intelligence platforms providing real-time updates on global guidance changes and competitor approval tracking.
- Build integrated service models spanning pre-IND consulting through post-marketing commitment management to capture full product lifecycle value.
- Methodology
- Built on primary interviews with regulatory service providers and pharmaceutical outsourcing executives.
- Benchmarked against drug development pipeline data and submission volume analysis.
- Validated using regulatory agency databases and internal peer review as per FMI modeling standards.

This performance reflects accelerating demand for specialized regulatory expertise across pharmaceutical, biotechnology, and medical device segments. North America leads global expansion supported by FDA's expedited program proliferation and complex post-marketing requirements. Europe follows driven by Medical Device Regulation implementation and pharmaceutical vigilance obligations. Asia-Pacific maintains steady growth through clinical trial activity expansion and emerging market regulatory harmonization.
Healthcare Regulatory Affairs Outsourcing Market Definition
The healthcare regulatory affairs outsourcing market encompasses the global provision of specialized services supporting pharmaceutical, biotechnology, and medical device companies in navigating regulatory requirements for product development, approval, and lifecycle management. Services include regulatory strategy formulation, dossier preparation and electronic common technical document publishing, clinical trial application management, product registration, labeling compliance, post-marketing surveillance, and regulatory intelligence. These services help sponsors achieve and maintain compliance with health authority requirements across global markets while optimizing development timelines and resource allocation.
Market Inclusions
The report includes global and regional market size estimates in value terms, with a forecast period from 2026 to 2036. It provides segmentation by Service, End-User, and Region. The analysis incorporates service delivery trends, technology adoption patterns, regulatory alignment strategies, and performance benchmarking across outsourcing engagement models. Regional demand dynamics, country-level growth rates, and competitive positioning are also evaluated as per FMI methodology.
Market Exclusions
The scope excludes clinical trial conduct services, contract research organization activities beyond regulatory support, and manufacturing process development services. It also excludes revenues from in-house regulatory functions and focuses strictly on the outsourced service layer. Consulting engagements for non-healthcare regulated industries are not considered due to market boundary definitions.
Research Methodology
- Primary Research: Interviews were conducted with regulatory affairs outsourcing providers, pharmaceutical regulatory executives, biotechnology compliance officers, medical device regulatory managers, and health authority relations consultants.
- Desk Research: Public company filings, regulatory agency guidance documents, industry association publications, and clinical trial registries were reviewed.
- Market-Sizing and Forecasting: A hybrid top-down and bottom-up model was constructed, reconstructing demand from drug development pipeline activity, regulatory submission volumes, and outsourcing penetration rates across end-user segments.
- Data Validation and Update Cycle: Outputs were subjected to variance testing against industry association data, vendor revenue reports, and structured internal review prior to publication.
Healthcare Regulatory Affairs Outsourcing Market Drivers, Restraints, and Opportunities
Future Market Insights analysis indicates the market has historically existed as a tactical support function tied to submission preparation cycles. Its baseline scale is anchored in global pharmaceutical research and development activity, with outsourcing penetration increasing as regulatory complexity outpaces in-house capacity expansion. As per FMI, the market's valuation is increasingly decoupled from submission volumes alone, driven by strategic advisory engagement across development pathways and lifecycle management requirements.
FMI analysts observe a clear stratification between conventional regulatory support optimized for standard drug applications and next-generation services engineered for breakthrough therapies, orphan indications, and personalized medicine approaches. Standard service offerings face pressure where automation reduces manual document preparation requirements, while growth concentrates in strategic engagements where specialized expertise justifies premium pricing through accelerated approval timelines and optimized regulatory pathways.
- Increasing Complexity of Global Regulatory Frameworks: The FDA issued multiple guidance documents addressing real-world evidence integration, AI-enabled drug development tools, and complex clinical trial designs that demand specialized expertise for compliant implementation [1]. The European Union Medical Device Regulation implementation expanded clinical evidence requirements and post-market obligations across twenty-seven member states, creating sustained demand for regulatory support from device manufacturers lacking in-house European expertise [2]. China's National Medical Products Administration overhauled electronic submission formats and tightened data integrity requirements, forcing international sponsors to engage local regulatory specialists for market access [3].
- Proliferation of Expedited Approval Pathways: The FDA granted accelerated approval to fifteen oncology drugs in 2024, each requiring post-marketing confirmatory studies that generate ongoing regulatory obligations [4]. Breakthrough therapy designations and regenerative medicine advanced therapy designations require proactive agency engagement and strategic planning that sponsors increasingly outsource to specialized consultants. The European Medicines Agency's PRIority MEdicines scheme and adaptive pathway pilots create additional complexity in development planning that favors external regulatory expertise [5].
- Expansion of Specialized Therapeutic Modalities: Cell and gene therapies present unique regulatory challenges including comparability, potency assay validation, and long-term safety monitoring that demand specialized expertise unavailable within most sponsor organizations [6]. Oligonucleotide therapies and antibody-drug conjugates require nuanced CMC regulatory strategies that outsourcing providers have developed through repeated engagement. Rare disease programs accounting for three hundred one orphan drug designations in 2024 require regulatory consultants familiar with natural history study integration and surrogate endpoint acceptance strategies [7].
- Regulatory Information Management System Adoption: The FDA's eCTD 4.0 mandate effective January 2025 intensified outsourcing among firms lacking necessary systems or trained staff for compliant electronic submissions [8]. Cloud-based regulatory information management platforms combined with AI-powered document assembly reduce submission cycle times while requiring specialized implementation expertise. Vendors embedding natural language processing for auto-compilation of eCTD modules achieve submission cycle reductions while maintaining compliance with evolving technical specifications [9].
- Post-Marketing Commitment Management Requirements: The FDA Sentinel Initiative and EMA EudraVigilance systems mandate continuous safety monitoring requiring expedited periodic safety update report generation [10]. Post-approval commitments for accelerated approval products generate sustained regulatory obligations that sponsors outsource to maintain compliance without diverting internal resources. Real-world evidence generation for label expansions creates additional regulatory complexity favoring external expertise in observational study design and health authority engagement [11].
Segmental Analysis
Healthcare Regulatory Affairs Outsourcing Market Analysis by Service
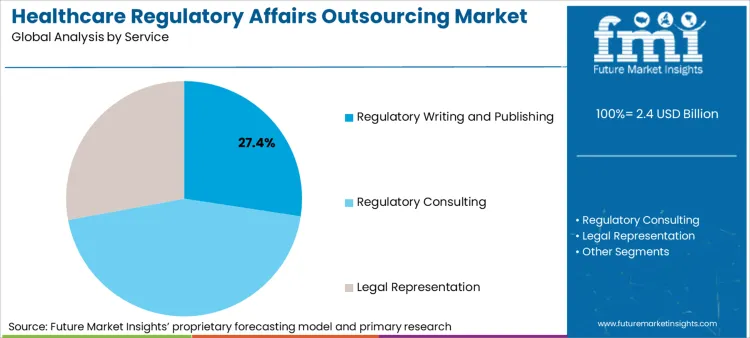
Based on FMI's healthcare regulatory affairs outsourcing market report, demand for regulatory writing and publishing is estimated to lead the service segment, capturing 27.4% of the market share in 2025 and maintaining dominance through 2035. Regulatory writing and publishing services encompass clinical study report preparation, investigator brochure development, summary document creation, and electronic common technical document compilation.
The segment's leadership reflects persistent demand for compliant documentation meeting ICH formatting requirements across global submissions. Growth is supported by increasing clinical trial activity and expanding requirements for detailed safety narratives in integrated summaries. Publishing services benefit from eCTD mandate adoption across emerging markets requiring specialized software expertise and validation capabilities.
Product registration and clinical trial application services represent the second-largest segment, driven by parallel filing strategies across multiple regions that compress timelines and require coordinated agency engagement. Regulatory consulting services command premium pricing for strategic assignments tied to breakthrough therapy designations, orphan drug applications, and novel modality programs where agency precedent is limited.
Legal representation services maintain niche but high-margin activity for patent term extensions, exclusivity negotiations, and citizen petition responses. Post-marketing and lifecycle management services grow at accelerated rates driven by FDA Sentinel Initiative requirements and periodic safety update report obligations.
Healthcare Regulatory Affairs Outsourcing Market Analysis by End-User
Pharmaceutical companies generate maximum demand for healthcare regulatory affairs outsourcing, accounting for the largest share driven by extensive pipelines, multi-region filing requirements, and complex post-marketing obligations. Large pharmaceutical companies maintain core regulatory strategy in-house while outsourcing high-volume activities including regional dossier adaptation, labeling revisions, and periodic safety update report generation.
Mid-size pharmaceutical companies outsource broader regulatory functions due to resource constraints while maintaining strategic oversight of core programs. Virtual and emerging pharmaceutical companies outsource substantially all regulatory activities from pre-IND consulting through post-approval commitments.
Biotechnology companies represent the fastest-growing end-user segment, driven by specialized therapeutic modalities requiring nuanced regulatory strategies and limited internal regulatory capacity. Cell and gene therapy developers engage outsourcing partners for CMC regulatory strategy, clinical trial application preparation, and breakthrough therapy designation requests. Medical device manufacturers show accelerated outsourcing growth following EU Medical Device Regulation implementation, requiring clinical evaluation report preparation, technical documentation updates, and notified body engagement support. Food and beverage companies maintain smaller but steady demand for regulatory support in novel food ingredient approvals and health claim substantiation.
Regional Analysis
The healthcare regulatory affairs outsourcing market is segmented geographically across North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa. Growth dynamics differ based on pharmaceutical development activity, regulatory framework maturity, and outsourcing adoption rates.
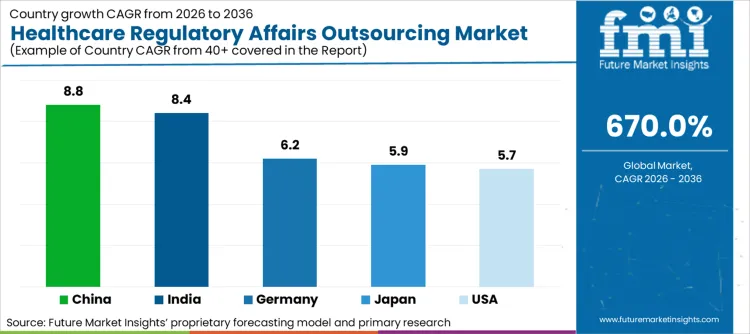
| Country | CAGR (2026-2036) |
|---|---|
| China | 8.8% |
| India | 8.4% |
| Germany | 6.2% |
| Japan | 5.9% |
| United States | 5.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Market Analysis
North America represents the largest value market for healthcare regulatory affairs outsourcing, driven by FDA's extensive guidance framework, complex post-marketing requirements, and concentration of pharmaceutical development activity.
The region's regulatory environment features multiple expedited pathways, real-world evidence integration, and evolving digital health guidance that demand specialized expertise. The FDA's Project Optimus requiring oncology programs to include dose-optimization studies adds documentation layers that strain sponsor resources, favoring outsourcing engagement.
- United States: Demand for healthcare regulatory affairs outsourcing in the United States is projected to rise at 5.7 % CAGR through 2036. The USA market benefits from the world's largest pharmaceutical research and development expenditure and FDA's complex regulatory framework requiring specialized expertise for efficient navigation. The agency's real-world evidence program authorizes observational data for label expansions while offering limited design guidance, driving sponsors to consultants who can craft defensible methodologies.
The FDA granted accelerated approval to multiple oncology drugs in 2024, each requiring post-marketing confirmatory studies generating sustained regulatory obligations. Virtual biotech companies operating with lean staff outsource substantially all compliance tasks from pre-IND through post-market surveillance. The eCTD 4.0 mandate effective January 2025 intensified outsourcing among firms lacking necessary systems or trained staff for compliant submissions.
FMI's analysis of the healthcare regulatory affairs outsourcing market in North America consists of country-wise assessment including the United States and Canada. Readers can find regulatory guidance tracking, outsourcing penetration analysis, and competitive intelligence.
Europe Market Analysis
Europe represents a mature market characterized by harmonized regulatory frameworks across member states layered with country-specific requirements. The European Medicines Agency oversees centralized procedures while national competent authorities manage national procedures and maintain local language requirements.
The EU Medical Device Regulation fully implemented in 2024 expanded clinical evidence requirements and post-market obligations across twenty-seven member states, creating sustained demand for regulatory support from device manufacturers lacking in-house European expertise.
- Germany: Demand for healthcare regulatory affairs outsourcing in Germany is projected to rise at 6.2 % CAGR through 2036. Germany's strong pharmaceutical industry and position as Europe's largest medical device market create sustained demand for regulatory support services. The country's Federal Institute for Drugs and Medical Devices maintains rigorous assessment standards requiring comprehensive documentation and proactive agency engagement.
German pharmaceutical companies expanding into emerging markets require support for local registration requirements and regulatory intelligence. The European Health Data Space implementation adds novel regulatory considerations for real-world evidence generation and secondary data use that drive demand for specialized consulting expertise.
FMI's analysis of the healthcare regulatory affairs outsourcing market in Europe consists of country-wise assessment including Germany, United Kingdom, France, Italy, and Spain. Readers can find regulatory framework tracking, outsourcing adoption analysis, and competitive landscape insights.
- Asia-Pacific Market Analysis: Asia-Pacific represents the fastest-growing regional market for healthcare regulatory affairs outsourcing, driven by clinical trial activity expansion, emerging market regulatory evolution, and increasing pharmaceutical development investment. The region's diverse regulatory frameworks require specialized expertise for market access across countries with varying requirements and approval timelines.
- China: Demand for healthcare regulatory affairs outsourcing in China is projected to rise at 8.8 % CAGR through 2036. China leads regional growth as the world's second-largest pharmaceutical market with rapidly evolving regulatory framework under the National Medical Products Administration. The agency overhauled electronic submission formats and tightened data integrity requirements, forcing international sponsors to engage local regulatory specialists for market access.
China's acceptance of ICH guidelines and implementation of expedited approval pathways create opportunities for outsourcing providers familiar with both domestic requirements and international standards. Domestic pharmaceutical companies expanding globally require regulatory support for FDA and EMA submissions, driving demand for bilingual consultants familiar with Western regulatory expectations. WuXi AppTec has built extensive regulatory service capabilities supporting both international sponsors entering China and domestic companies expanding globally, creating a comprehensive outsourcing ecosystem.
FMI's analysis of the healthcare regulatory affairs outsourcing market in Asia-Pacific consists of country-wise assessment including China, India, Japan, and South Korea. Readers can find regulatory evolution tracking, service provider mapping, and market access strategy insights.
- India: Demand for healthcare regulatory affairs outsourcing in India is projected to rise at 8.4 % CAGR through 2036. India's position as a major generic pharmaceutical supplier to global markets creates sustained demand for regulatory support in preparing abbreviated new drug applications, maintaining drug master files, and responding to FDA deficiency letters.
The country's clinical trial activity expansion generates requirements for ethics committee submissions, clinical trial applications, and regulatory correspondence management. Indian regulatory service providers have developed cost-effective delivery models combining deep generic drug expertise with English language capabilities that appeal to international sponsors. The government's Production Linked Incentive scheme for pharmaceuticals encourages manufacturing expansion requiring regulatory support for facility registrations and product approvals across global markets.
- Japan: Demand for healthcare regulatory affairs outsourcing in Japan is projected to rise at 5.9 % CAGR through 2036. Japan's Pharmaceuticals and Medical Devices Agency maintains rigorous review standards requiring comprehensive documentation and proactive engagement strategies. The country's accelerated approval pathways for regenerative medicines introduced conditional approval frameworks requiring long-term follow-up studies that expand regulatory service requirements.
Japanese pharmaceutical companies expanding internationally require regulatory support for FDA and EMA submissions, driving demand for bilingual consultants familiar with Western regulatory expectations. The aging population and high healthcare utilization rates sustain pharmaceutical development investment requiring ongoing regulatory support services.
Competitive Landscape for Market Players

The healthcare regulatory affairs outsourcing market is characterized by a mix of global contract research organizations with regulatory service divisions, specialized regulatory consulting firms, and professional services companies with life sciences practices. Leading firms are differentiating through therapeutic area specialization, technology-enabled service delivery, and integrated offerings spanning development through lifecycle management.
IQVIA combines regulatory expertise with extensive clinical trial data assets, enabling evidence-based regulatory strategy development and accelerated response to health authority information requests. The company's orchestrated clinical trials platform flags compliance gaps during protocol creation, shifting quality control upstream and reducing submission cycle times.
Charles River Laboratories leverages deep preclinical expertise to support regulatory strategy development from early development through IND submission. Syneos Health integrates regulatory consulting with clinical development and commercial services, offering comprehensive support for emerging pharmaceutical companies. ICON maintains extensive regulatory writing and publishing capabilities supporting global submissions across therapeutic areas.
Specialized regulatory consulting firms including PAREXEL and Promedica International focus exclusively on regulatory affairs, developing deep expertise in breakthrough therapy designations, orphan drug applications, and novel modality programs. These firms command premium pricing for strategic assignments where agency precedent is limited and regulatory pathway optimization can meaningfully compress approval timelines.
WuXi AppTec has built comprehensive regulatory service capabilities supporting both international sponsors entering China and domestic companies expanding globally, creating a unique position in the Asia-Pacific market.
Buyer behavior in this market reflects dual sourcing strategies to mitigate dependency on single providers and maintain pricing discipline. Framework agreements with pharmaceutical companies often include multi-year volume commitments with rate structures tied to service complexity and therapeutic area. Pricing power for suppliers depends less on labor rates alone and more on demonstrated regulatory success rates, therapeutic expertise depth, and ability to support rapid response to health authority information requests.
Recent Developments
- IQVIA - Technology Platform Expansion (2025): IQVIA expanded its regulatory intelligence platform incorporating AI-powered monitoring of global health authority guidance changes with real-time alerts for pharmaceutical clients.
- Charles River Laboratories - Service Integration (2025): Charles River integrated preclinical consulting with regulatory strategy services offering seamless transition from non-clinical development through IND submission.
- WuXi AppTec - Capability Expansion (2025): WuXi AppTec expanded its regulatory service capabilities supporting international clients navigating evolving NMPA requirements and domestic clients preparing FDA and EMA submissions.
Key Players in the Healthcare Regulatory Affairs Outsourcing Market
- IQVIA
- Accell Clinical Research, LLC
- Charles River Laboratories
- Syneos Health
- Laboratory Corporation of America Holdings
- ICON PLC
- Medpace, Inc.
- PAREXEL International Corporation
- Thermo Fisher Scientific Inc. (PPD)
- Promedica International
- WuXi AppTec
Scope of Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 2.45 billion (2026) to USD 4.56 billion (2036), at a CAGR of 6.7% |
| Market Definition | The healthcare regulatory affairs outsourcing market comprises specialized services supporting regulatory compliance, submission preparation, and health authority interactions for life sciences companies. |
| Service Segmentation | Regulatory Consulting, Legal Representation, Regulatory Writing and Publishing, Product Registration and Clinical Trial Applications, Others |
| End-User Segmentation | Pharmaceutical and Biotechnology Companies, Medical Device Companies, Food and Beverage Companies |
| Region Segmentation | North America, Latin America, Europe, Asia-Pacific, Middle East & Africa |
| Key Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, China, India, Japan, South Korea, and 40+ countries |
| Key Companies Profiled | IQVIA, Accell Clinical Research, LLC, Charles River Laboratories, Syneos Health, Laboratory Corporation of America Holdings, ICON PLC, Medpace, Inc., PAREXEL International Corporation, Thermo Fisher Scientific Inc. (PPD), Promedica International, WuXi AppTec |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling validated through primary interviews with regulatory service providers and pharmaceutical outsourcing executives, supported by drug development pipeline analysis and submission volume tracking |
Healthcare Regulatory Affairs Outsourcing Market Analysis by Segments
Service:
- Regulatory Consulting
- Legal Representation
- Regulatory Writing and Publishing
- Product Registration and Clinical Trial Applications
- Others
End-User:
- Pharmaceutical and Biotechnology Companies
- Medical Device Companies
- Food and Beverage Companies
Region:
- North America
- Latin America
- Europe
- Asia-Pacific
- Middle East & Africa
Bibliography
- USA Food and Drug Administration. (2025). Guidance Document: Considerations for the Use of Real-World Data and Real-World Evidence to Support Regulatory Decision-Making. FDA Center for Drug Evaluation and Research.
- European Commission. (2025). Medical Devices Regulation (EU) 2017/745 Implementation Report 2025. Publications Office of the European Union.
- National Medical Products Administration. (2025). NMPA Annual Report on Drug Regulation 2024. China Food and Drug Administration.
- USA Food and Drug Administration. (2025). Accelerated Approval Program Annual Report 2024. FDA Center for Drug Evaluation and Research.
- European Medicines Agency. (2025). PRIME: Priority Medicines Scheme Annual Report 2024. EMA.
- International Society for Cell & Gene Therapy. (2025). Regulatory Considerations for Cell and Gene Therapy Development. ISCT.
- USA Food and Drug Administration. (2025). Orphan Drug Designations and Approvals Report 2024. FDA Office of Orphan Products Development.
- USA Food and Drug Administration. (2024). Electronic Common Technical Document (eCTD) Compliance and Implementation Guidance. FDA.
- International Council for Harmonisation. (2025). ICH M8: Electronic Common Technical Document Implementation Guide. ICH.
- European Medicines Agency. (2025). EudraVigilance Annual Report on Pharmacovigilance 2024. EMA.
- USA Food and Drug Administration. (2025). Real-World Evidence Program Two-Year Progress Report. FDA.
- Pharmaceutical Research and Manufacturers of America. (2025). Pharmaceutical Industry Profile 2025. PhRMA.
- European Federation of Pharmaceutical Industries and Associations. (2025). EFPIA Annual Report 2025. EFPIA.
- Japan Pharmaceutical Manufacturers Association. (2025). JPMA Annual Report 2025. JPMA.
- International Federation of Pharmaceutical Manufacturers and Associations. (2025). IFPMA Regulatory Harmonization Progress Report. IFPMA.a
This Report Addresses
- Market intelligence to enable structured strategic decision-making across regulatory service providers, pharmaceutical outsourcing executives, and healthcare investors.
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, including validated CAGR and absolute dollar opportunity sizing.
- Growth opportunity mapping across Service, End-User, and regional demand pockets, with emphasis on regulatory writing specialization and strategic consulting expansion.
- Segment and regional forecasting covering regulatory consulting, writing and publishing, and product registration services, alongside end-user demand trajectories across pharmaceutical, biotechnology, and medical device companies.
- Competition strategy assessment, including therapeutic area specialization investment, technology platform development, and buyer leverage dynamics shaping service agreement structures.
- Service and technology tracking focused on AI-enabled regulatory intelligence, electronic submission platforms, and integrated lifecycle management offerings.
- Regulatory policy analysis covering global health authority guidance evolution, expedited pathway proliferation, and post-marketing requirement expansion influencing outsourcing demand.
- Report delivery in PDF, Excel, and presentation-ready formats to support executive planning, service line prioritization, and competitive benchmarking.
Frequently Asked Questions
How large is the demand for healthcare regulatory affairs outsourcing in the global market in 2026?
Demand for healthcare regulatory affairs outsourcing is estimated to be valued at USD 2.45 billion in 2026.
What will be the market size of healthcare regulatory affairs outsourcing in the global market by 2036?
Market size for healthcare regulatory affairs outsourcing is projected to reach USD 4.56 billion by 2036.
What is the expected demand growth for healthcare regulatory affairs outsourcing between 2026 and 2036?
Demand for healthcare regulatory affairs outsourcing is expected to grow at a 6.7% CAGR between 2026 and 2036.
Which service segment is poised to lead global sales?
Regulatory writing and publishing services are estimated to lead the service segment, holding 27.4% share in 2025, reflecting persistent demand for compliant clinical study reports and electronic common technical document dossiers.
How significant is the role of pharmaceutical companies in driving outsourcing demand?
Pharmaceutical companies represent the dominant end-user segment, driven by extensive pipelines, multi-region filing requirements, and complex post-marketing obligations requiring specialized regulatory support.
What is driving demand in North America?
FDA's extensive guidance framework, complex post-marketing requirements, concentration of pharmaceutical development activity, and eCTD 4.0 mandate implementation are supporting North American demand growth.
What is the United States growth outlook in this report?
The United States is projected to expand at a 5.7% CAGR during 2026 to 2036.
What is driving demand in Europe?
EU Medical Device Regulation implementation, pharmaceutical vigilance obligations, and the European Health Data Space framework are supporting European market growth.
What is the Germany growth outlook in this report?
Germany is projected to expand at a 6.2% CAGR during 2026 to 2036, supported by its position as Europe's largest pharmaceutical and medical device market.
Does the report cover China in its regional analysis?
Yes, China is included within Asia-Pacific and is a high-growth country market covered in the report.
What is the China growth outlook in this report?
China is projected to expand at an 8.8% CAGR during 2026 to 2036, supported by NMPA regulatory evolution, international sponsor market access requirements, and domestic company global expansion.
Does the report cover India in its regional analysis?
Yes, India is included within Asia-Pacific and is covered in the country-wise assessment.
What is the India growth outlook in this report?
India is projected to expand at an 8.4% CAGR during 2026 to 2036, supported by generic pharmaceutical export activity and clinical trial expansion.
Does the report cover Japan in its regional analysis?
Yes, Japan is included within Asia-Pacific under the regional scope of analysis.
What is the Japan growth outlook in this report?
Japan is projected to expand at a 5.9% CAGR during 2026 to 2036, supported by PMDA regulatory requirements and domestic company international expansion.
What is driving demand in Asia-Pacific?
Clinical trial activity expansion, emerging market regulatory evolution, and increasing pharmaceutical development investment are supporting Asia-Pacific demand growth.
What defines healthcare regulatory affairs outsourcing as part of this market?
Services are included if they support regulatory compliance, submission preparation, or health authority interactions for pharmaceutical, biotechnology, or medical device companies on an outsourced basis.
What is included in the scope of this healthcare regulatory affairs outsourcing market report?
Scope includes market sizing and forecasting for 2026 to 2036, segmentation by service, end-user, and region, along with competitive assessment and country-wise outlook.
What is excluded from the scope of this report?
The scope excludes clinical trial conduct services, contract research organization activities beyond regulatory support, manufacturing process development services, and in-house regulatory functions.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Service
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Service , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Service , 2026 to 2036
- Regulatory Writing and Publishing
- Regulatory Consulting
- Legal Representation
- Regulatory Writing and Publishing
- Y to o to Y Growth Trend Analysis By Service , 2021 to 2025
- Absolute $ Opportunity Analysis By Service , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Service
- By Country
- Market Attractiveness Analysis
- By Country
- By Service
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Service
- By Country
- Market Attractiveness Analysis
- By Country
- By Service
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Service
- By Country
- Market Attractiveness Analysis
- By Country
- By Service
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Service
- By Country
- Market Attractiveness Analysis
- By Country
- By Service
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Service
- By Country
- Market Attractiveness Analysis
- By Country
- By Service
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Service
- By Country
- Market Attractiveness Analysis
- By Country
- By Service
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Service
- By Country
- Market Attractiveness Analysis
- By Country
- By Service
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Service
- Competition Analysis
- Competition Deep Dive
- IQVIA
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Accell Clinical Research, LLC
- Charles River Laboratories
- Syneos Health
- Laboratory Corporation of America Holdings
- ICON PLC
- Medpace, Inc.
- PAREXEL International Corporation
- IQVIA
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Service , 2021 to 2036
- Table 3: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Service , 2021 to 2036
- Table 5: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: Latin America Market Value (USD Million) Forecast by Service , 2021 to 2036
- Table 7: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Western Europe Market Value (USD Million) Forecast by Service , 2021 to 2036
- Table 9: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Eastern Europe Market Value (USD Million) Forecast by Service , 2021 to 2036
- Table 11: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: East Asia Market Value (USD Million) Forecast by Service , 2021 to 2036
- Table 13: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: South Asia and Pacific Market Value (USD Million) Forecast by Service , 2021 to 2036
- Table 15: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Middle East & Africa Market Value (USD Million) Forecast by Service , 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Service , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Service , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Service
- Figure 6: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Region
- Figure 9: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 10: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 11: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 12: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 17: North America Market Value Share and BPS Analysis by Service , 2026 and 2036
- Figure 18: North America Market Y-o-Y Growth Comparison by Service , 2026-2036
- Figure 19: North America Market Attractiveness Analysis by Service
- Figure 20: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 21: Latin America Market Value Share and BPS Analysis by Service , 2026 and 2036
- Figure 22: Latin America Market Y-o-Y Growth Comparison by Service , 2026-2036
- Figure 23: Latin America Market Attractiveness Analysis by Service
- Figure 24: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 25: Western Europe Market Value Share and BPS Analysis by Service , 2026 and 2036
- Figure 26: Western Europe Market Y-o-Y Growth Comparison by Service , 2026-2036
- Figure 27: Western Europe Market Attractiveness Analysis by Service
- Figure 28: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: Eastern Europe Market Value Share and BPS Analysis by Service , 2026 and 2036
- Figure 30: Eastern Europe Market Y-o-Y Growth Comparison by Service , 2026-2036
- Figure 31: Eastern Europe Market Attractiveness Analysis by Service
- Figure 32: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: East Asia Market Value Share and BPS Analysis by Service , 2026 and 2036
- Figure 34: East Asia Market Y-o-Y Growth Comparison by Service , 2026-2036
- Figure 35: East Asia Market Attractiveness Analysis by Service
- Figure 36: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 37: South Asia and Pacific Market Value Share and BPS Analysis by Service , 2026 and 2036
- Figure 38: South Asia and Pacific Market Y-o-Y Growth Comparison by Service , 2026-2036
- Figure 39: South Asia and Pacific Market Attractiveness Analysis by Service
- Figure 40: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Middle East & Africa Market Value Share and BPS Analysis by Service , 2026 and 2036
- Figure 42: Middle East & Africa Market Y-o-Y Growth Comparison by Service , 2026-2036
- Figure 43: Middle East & Africa Market Attractiveness Analysis by Service
- Figure 44: Global Market - Tier Structure Analysis
- Figure 45: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

