Real-time Bioprocess Raman Analyzer Market
Real-time Bioprocess Raman Analyzer Market Analysis Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Real-time Bioprocess Raman Analyzer Market Forecast and Outlook 2026 to 2036
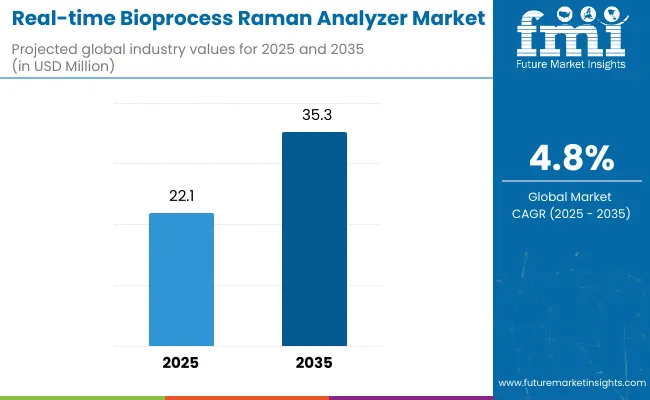
The real-time bioprocess Raman analyzer market is valued at USD 23.2 million in 2026 and is projected to reach USD 36.9 million by 2036, expanding at a CAGR of 4.8% during the forecast period. Demand growth is driven by the increasing need for real-time monitoring and control in biopharmaceutical manufacturing, where Raman spectroscopy is used for in-process analysis of critical quality attributes (CQAs), process parameters, and product consistency. The technology allows for direct, non-destructive measurements in complex processes such as cell culture, fermentation, and downstream processing.
Quick Stats for Real-time Bioprocess Raman Analyzer Market
- Market Value (2026): USD 23.2 million
- Market Forecast Value (2036): USD 36.9 million
- Forecast CAGR (2026 to 2036): 4.8%
- Leading Segment in Global Demand: Instruments 75% share
- Key Growth Regions in Global Demand: North America, Europe, Asia Pacific
- Top Key Players in Global Demand: Kaiser Optical Systems Inc., Resolution Spectra Systems Inc., Tornado Spectral Systems, Sartorius AG, Thermo Fisher Scientific Inc., MarqMetrix Inc., Wasatch Photonics Inc., Real Time Analyzer Inc.
Biopharmaceutical companies are increasingly adopting Raman analyzers for their ability to provide instant data on process changes, ensuring higher yield, quality, and reduced batch failures. Real-time monitoring of bioprocesses enhances decision-making, accelerates the product development cycle, and improves manufacturing efficiency. The demand for Raman analyzers is particularly strong in the production of biologics, vaccines, and cell-based therapies, where stringent quality control and process optimization are crucial.
Key benefits driving market growth include reduced dependency on traditional offline sampling techniques, faster turnaround times, and more accurate results. Raman spectroscopy also aligns with regulatory requirements for quality by design (QbD) and process analytical technology (PAT). Technological advancements focus on improving system integration, portability, data handling capabilities, and reducing operational costs.
Market dynamics are shaped by the increasing trend of automation in pharmaceutical production, the need for advanced process monitoring solutions, and the growing shift towards personalized medicine and biologic therapies. Suppliers focus on enhancing product features, expanding customer support services, and securing partnerships with major pharmaceutical manufacturers. Demand outlook reflects steady adoption across biopharma and biotechnology companies, alongside continued regulatory emphasis on advanced process monitoring, through 2036.
Real-time Bioprocess Raman Analyzer Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 23.2 million |
| Market Forecast Value (2036) | USD 36.9 million |
| Forecast CAGR (2026 to 2036) | 4.8% |
How Is the Real-Time Bioprocess Raman Analyzer Market Segmented?
The real-time bioprocess Raman analyzer market is segmented by product type, application, end-user, and region, reflecting differences in monitoring requirements and bioprocess complexity. By product type, the industry is divided into instruments and software, where instruments include Raman analyzers and Raman probes used for direct spectroscopic measurement. Application segmentation covers lab to process analysis and bioprocess analysis, addressing both development-stage monitoring and commercial manufacturing control. End-user segmentation includes biopharmaceutical companies, contract manufacturing organizations, and research organizations, each with distinct scale, compliance, and throughput needs. Regional segmentation spans North America, Europe, Asia Pacific, Latin America, and the Middle East and Africa. This structure highlights adoption patterns driven by biologics production intensity, regulatory frameworks, and investment in process analytical technologies across regions.
Why Do Instruments Dominate the Industry by Product Type?
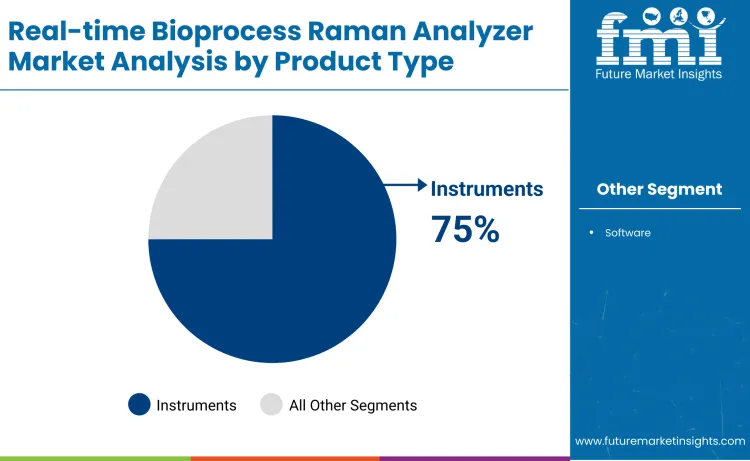
The instruments segment accounts for about 75% of total market share due to the essential role of hardware in real-time bioprocess monitoring. Raman analyzers and probes form the foundation of in-line and at-line spectroscopic measurement systems used in biomanufacturing. These instruments enable continuous monitoring of critical quality attributes and process parameters without interrupting production. Biopharmaceutical manufacturers depend on robust hardware for accuracy, stability, and repeatability under demanding process conditions. Compatibility with existing bioreactors and control systems supports wide deployment. Instrument reliability directly influences data quality and regulatory acceptance. Ongoing investment in miniaturization, probe durability, and system integration improves usability across development and commercial scales. These factors position instruments as the dominant product type in the real-time bioprocess Raman analyzer market.
Why Does Bioprocess Analysis Lead by Application?
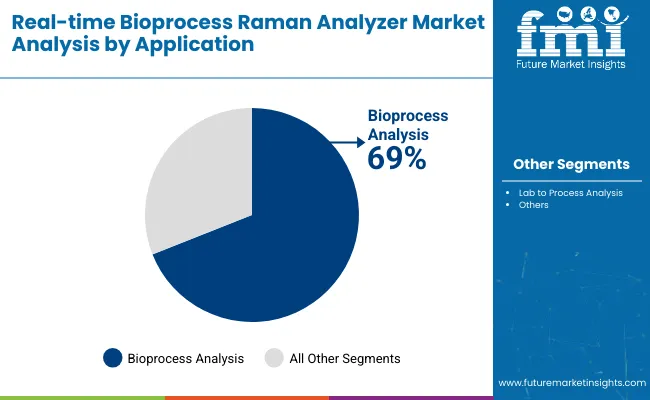
Bioprocess analysis represents about 69% of application-based demand due to its central role in commercial biologics manufacturing. This application focuses on real-time monitoring of cell culture performance, nutrient consumption, metabolite formation, and product expression. Continuous insight into these parameters supports tight process control and consistent product quality. Bioprocess analysis tools assist manufacturers in maintaining compliance with regulatory expectations for process understanding and control. Integration with automation platforms supports data-driven decision-making during production runs. As biologic therapies grow in complexity, real-time insight into process dynamics becomes increasingly valuable. Expansion of large-scale biologics production strengthens demand for bioprocess analysis solutions, reinforcing its leadership within the real-time bioprocess Raman analyzer market.
What Are the Key Drivers of the Real-Time Bioprocess Raman Analyzer Market?
Market growth is driven by rising demand for continuous monitoring in biologics manufacturing and increasing adoption of process analytical technology frameworks. Biopharmaceutical producers seek tools that improve process consistency and reduce batch variability. Real-time Raman analysis supports faster detection of deviations and improved yield management. Regulatory focus on process understanding encourages adoption of advanced analytical methods. Growth in cell and gene therapies increases need for precise monitoring of complex processes. Contract manufacturing expansion drives investment in flexible analytical platforms. Technological progress improves measurement accuracy and system integration. These drivers collectively support sustained demand for real-time bioprocess Raman analyzers across development and commercial manufacturing environments.
What Restraints Affect Market Adoption?
Market expansion faces challenges related to high upfront investment and technical complexity. Advanced Raman analyzer systems require significant capital expenditure, affecting purchasing decisions for smaller organizations. Integration with existing control systems demands specialized expertise and validation effort. Calibration and maintenance requirements add operational cost. Skilled personnel shortages limit efficient system operation in some facilities. Implementation timelines extend due to compliance documentation and process qualification needs. Budget constraints in research organizations influence adoption pace. These factors affect market penetration and slow broader deployment across cost-sensitive segments.
What Trends Are Shaping the Real-Time Bioprocess Raman Analyzer Market?
Key trends include deeper integration of Raman analyzers with automation and data management platforms. Manufacturers prioritize systems capable of seamless communication with control software. Demand grows for compact probes suitable for single-use bioreactors. Multivariate data analysis capabilities gain attention for improved process insight. Adoption increases in continuous manufacturing workflows. Expansion of biologics production in Asia Pacific supports regional growth. Vendors focus on usability improvements to reduce training burden. These trends guide product development and competitive positioning within the real-time bioprocess Raman analyzer market.
How Is the Real-time Bioprocess Raman Analyzer Market Developing from 2026 to 2036?
The real-time bioprocess Raman analyzer market from 2026 to 2036 continues to advance as biopharmaceutical manufacturers place stronger emphasis on continuous process insight and operational reliability. These analyzers enable inline and online measurement of critical variables such as metabolite concentration, nutrient consumption, and product formation without interrupting production. Adoption is closely linked to the global shift toward data-rich manufacturing models that reduce manual sampling and improve process transparency. Increasing production of biologics, vaccines, and cell based therapies drives demand for tools that support reproducibility across batches and sites.
Expansion of large scale biologics facilities and rising activity among contract development and manufacturing organizations further strengthen market momentum. Manufacturers seek integrated analytical platforms that align with digital manufacturing architectures and real-time decision workflows. Regulatory focus on process understanding and quality assurance reinforces long term deployment. While emerging markets prioritize new installations, mature regions focus on system modernization, software integration, and broader analytical coverage across upstream and downstream operations.
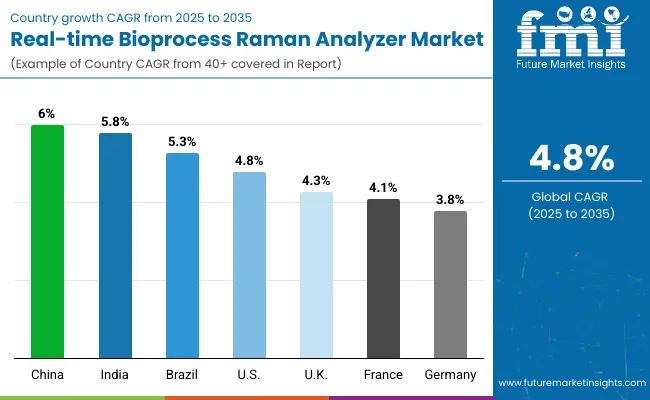
| Country | CAGR |
|---|---|
| China | 6.4% |
| India | 6.1% |
| Brazil | 5.6% |
| United States | 5.0% |
| United Kingdom | 4.6% |
| France | 4.4% |
| Germany | 4.0% |
Why Is China Leading Adoption of Real-time Bioprocess Raman Analyzers?
China is projected to grow at a CAGR of 6.4% from 2026 to 2036, supported by rapid expansion of domestic biopharmaceutical manufacturing capacity. Large scale investments in biologics production facilities increase demand for advanced analytical tools capable of supporting continuous operations. Raman analyzers are widely deployed to monitor cell culture health, nutrient utilization, and product concentration in real time, helping manufacturers maintain consistency across high volume production runs.
The growth of local biologics pipelines, biosimilar development, and innovative therapeutics accelerates adoption across both upstream and downstream processes. Industrial clusters emphasize process visibility to reduce variability and improve output reliability. Expansion of domestic instrument suppliers and technical service providers improves system accessibility and lowers deployment barriers. Strong regulatory oversight further encourages use of real-time monitoring technologies to support quality driven manufacturing across production hubs.
How Is India Strengthening Its Real-time Bioprocess Raman Analyzer Market?
India is expected to record a CAGR of 6.1% during 2026 to 2036 as its biopharmaceutical manufacturing base continues to diversify and scale. Growth of contract manufacturing and development services increases the need for robust process monitoring tools that support multiple client requirements. Raman analyzers are increasingly integrated into fermentation and cell culture workflows to enhance control over critical parameters and reduce production variability.
Strong focus on biosimilars, vaccines, and biologics exports drives investments in modern analytical infrastructure. Manufacturing hubs in western and southern regions adopt real-time analytics to improve efficiency and support regulatory compliance across global markets. Collaboration with international technology providers accelerates knowledge transfer, system validation, and workforce training. These factors collectively strengthen adoption across mid scale and large bioprocessing facilities.
What Is Driving Growth of Real-time Bioprocess Raman Analyzers in Brazil?
Brazil is projected to grow at a CAGR of 5.6% from 2026 to 2036, supported by expansion of domestic biopharmaceutical production capabilities. Increasing focus on local manufacturing of vaccines and therapeutic biologics creates demand for advanced monitoring solutions that improve process reliability. Raman analyzers help producers enhance efficiency by providing continuous insight into critical process variables without disrupting operations.
Public and private investments strengthen analytical capacity across new and existing facilities. Producers adopt real-time monitoring tools to align with evolving quality expectations and improve batch consistency. International collaborations and technology partnerships improve access to advanced instrumentation and analytical expertise. These developments support gradual but steady expansion of Raman based bioprocess monitoring across the country.
Why Does the United States Maintain Strong Demand for Raman-based Bioprocess Monitoring?
The United States is expected to expand at a CAGR of 5.0% during 2026 to 2036, driven by its well established biopharmaceutical manufacturing ecosystem. Leading biologics producers rely on Raman analyzers to gain real-time insight into complex production processes and support high standards of quality assurance. These systems are commonly integrated within advanced manufacturing and continuous bioprocessing frameworks.
Strong regulatory expectations reinforce adoption across both clinical and commercial manufacturing environments. Companies emphasize process optimization, digital transformation, and data driven decision models to improve efficiency and reduce risk. High concentration of biologics developers, coupled with ongoing facility upgrades and capacity expansion, sustains consistent demand for new installations and system enhancements.
How Is the United Kingdom Supporting Adoption of Real-time Bioprocess Raman Analyzers?
The United Kingdom is projected to grow at a CAGR of 4.6% from 2026 to 2036, supported by strong integration between research institutions and industrial manufacturing. Bioprocessing facilities adopt Raman analyzers to facilitate smooth transition from laboratory scale development to commercial production. Real-time monitoring improves process understanding during scale up and supports reproducibility across production stages.
Academic and industry collaboration accelerates deployment of advanced analytical technologies within manufacturing environments. Producers prioritize real-time data to improve process robustness and support innovation in biologics and advanced therapies. Growth of cell and gene therapy manufacturing further contributes to adoption, particularly in specialized and high value production sites.
What Sustains Steady Growth of Real-time Bioprocess Raman Analyzers in Germany?
Germany is expected to record a CAGR of 4.0% from 2026 to 2036, reflecting stable demand within its highly developed pharmaceutical and biopharmaceutical manufacturing environment. German manufacturers place strong emphasis on precision, process reliability, and regulatory alignment, which supports continued use of real-time Raman analyzers across biologics production lines. These systems enable continuous monitoring of critical parameters, helping facilities maintain tight control over complex upstream and downstream processes.
Growth in Germany is largely driven by replacement cycles, incremental capacity expansion, and modernization of existing analytical infrastructure rather than large scale greenfield projects. Facilities integrate Raman analyzers into established quality and automation frameworks to enhance process transparency and reduce manual intervention. Ongoing production of monoclonal antibodies, recombinant proteins, and advanced biologics sustains long-term demand for reliable real-time monitoring solutions across both commercial and clinical manufacturing sites.
What Drives Continued Adoption of Real-time Bioprocess Raman Analyzers in France?
France is projected to grow at a CAGR of 4.4% during 2026 to 2036, supported by steady expansion of biologics manufacturing and strong emphasis on quality driven production. Biopharmaceutical producers deploy Raman analyzers to support continuous monitoring, process verification, and compliance with stringent manufacturing expectations. These tools improve process understanding and help reduce variability across batches and production campaigns.
Growth in the French market is reinforced by investments in biologics, vaccines, and emerging advanced therapies. Facilities increasingly adopt real-time analytics to support scale-up activities and improve operational efficiency. Integration of Raman systems within digital manufacturing environments strengthens data visibility and decision making. Demand remains consistent as manufacturers upgrade analytical capabilities and expand biologics production capacity across both public and private sector facilities.
Competitive Landscape of the Real-Time Bioprocess Raman Analyzer Market
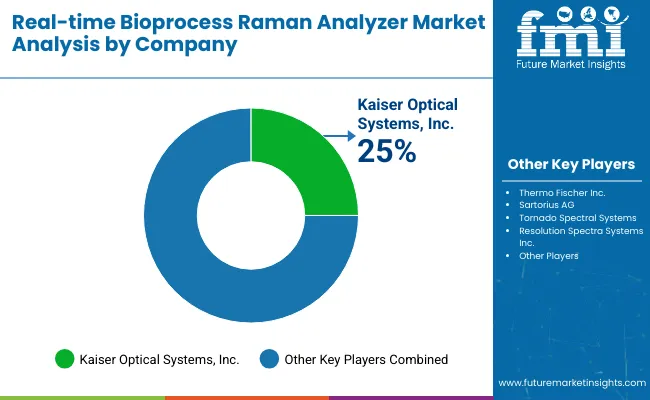
The real-time bioprocess Raman analyzer market is shaped by a focused group of analytical instrumentation providers and bioprocess technology companies supporting advanced process monitoring in biopharmaceutical manufacturing. Competition centers on measurement accuracy, system robustness, software integration, and the ability to deliver real-time, in-line analysis under GMP-compliant production environments. Adoption is driven by the growing use of process analytical technology in upstream bioprocessing, where continuous monitoring of critical quality attributes and process parameters is essential for process control and regulatory alignment.
Kaiser Optical Systems, Resolution Spectra Systems, and Tornado Spectral Systems lead the competitive landscape through high-performance Raman analyzers designed for real-time monitoring of fermentation and cell culture operations. These companies are recognized for probe durability, high signal stability, and advanced chemometric modeling that supports accurate tracking of metabolites, nutrients, and product concentration during bioprocess runs.
Large life science technology providers such as Thermo Fisher Scientific and Sartorius AG strengthen competition by embedding Raman spectroscopy into broader bioprocess automation and control platforms. Their advantage lies in end-to-end integration with bioreactors, data management systems, and manufacturing execution environments, supporting closed-loop process control.
Emerging and specialized players including MarqMetrix, Wasatch Photonics, and Real Time Analyzer focus on compact, fiber-coupled, and modular Raman systems suited for continuous biomanufacturing and pilot-scale operations. Competitive positioning increasingly depends on analytical reliability, ease of validation, and compatibility with modern digital bioprocess workflows.
Key Players in the Real-Time Bioprocess Raman Analyzer Market
- Kaiser Optical Systems Inc.
- Resolution Spectra Systems Inc.
- Tornado Spectral Systems
- Thermo Fisher Scientific Inc.
- Sartorius AG
- MarqMetrix Inc.
- Wasatch Photonics Inc.
- Real Time Analyzer Inc.
Scope of Report
| Attributes | Values |
|---|---|
| Market Value 2026 | USD 23.2 million |
| Projected Market Value 2036 | USD 36.9 million |
| CAGR 2026 to 2036 | 4.8% |
| Quantitative Units | USD million |
| Product Types Analyzed | Instruments (Raman Analyzers, Raman Probes), Software |
| Applications Analyzed | Lab to Process Analysis, Bioprocess Analysis |
| End-Users Analyzed | Biopharmaceutical Companies, Contract Manufacturing Organizations, Research Organizations |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, United Kingdom, Germany, France, China, Japan, South Korea, Brazil, Australia, India, and 40+ additional countries |
| Key Companies Profiled | Kaiser Optical Systems Inc., Resolution Spectra Systems Inc., Tornado Spectral Systems, Sartorius AG, Thermo Fisher Scientific Inc., MarqMetrix Inc., Wasatch Photonics Inc., Real Time Analyzer Inc. |
| Additional Attributes | Dollar sales by application and product type; regional adoption trends; competitive landscape analysis; preferences for in-line versus at-line analysis; integration with bioprocess monitoring systems; innovations in Raman spectroscopy, data analytics, and quality standardization |
Key Segmentation
By Product Type:
- Instruments
- Raman Analyzers
- Raman Probe
- Software
By Application:
- Lab to Process Analysis
- Bioprocess Analysis
By End-user:
- Biopharmaceutical Companies
- Contract Manufacturing Organizations
- Research Organizations
By Region:
- North America
- Europe
- Asia Pacific
- Rest of World
Bibliography
- U.S. Food and Drug Administration (FDA). (2022). Process Analytical Technology (PAT) Guidance for Industry. U.S. Department of Health and Human Services.
- U.S. National Institutes of Health (NIH). (2021). Biomanufacturing and Bioprocessing: Tools and Technologies. National Institute of Biomedical Imaging and Bioengineering.
- World Health Organization (WHO). (2023). Good Manufacturing Practices for Biopharmaceuticals. WHO Technical Report Series.
- International Society for Pharmaceutical Engineering (ISPE). (2022). Advances in Bioprocess Monitoring: The Role of Real-Time Data in Biomanufacturing. ISPE.
- European Medicines Agency (EMA). (2023). Guideline on the Manufacture of Biopharmaceutical Products. European Union.
Frequently Asked Questions
How big is the real-time bioprocess Raman analyzer market in 2026?
The industry is valued at USD 23.2 million in 2026.
What will be the size of the real-time bioprocess Raman analyzer market in 2036?
The industry is projected to reach USD 36.9 million by 2036.
How much will the real-time bioprocess Raman analyzer market grow between 2026 and 2036?
The industry is expected to grow at a 4.8% CAGR.
Which product type is projected to lead in the real-time bioprocess Raman analyzer market in 2026?
The instruments segment is projected to lead with a 75% market share.
Which application segment will contribute a significant share in the real-time bioprocess Raman analyzer market in 2026?
The bioprocess analysis segment is expected to command 69% share.
Table of Content
- Real-time Bioprocess Raman Analyzer Market Analysis Size And Share Forecast Outlook 2026 To 2036
- Real-time Bioprocess Raman Analyzer Market Forecast And Outlook (2026-2036)
- Market Definition And Scope
- Market Value (2026), Forecast Value (2036), And Forecast CAGR (2026-2036)
- Adoption Context In Biopharmaceutical Manufacturing
- Use Of Raman Spectroscopy For In-Process Monitoring
- Demand Across Cell Culture, Fermentation, And Downstream Operations
- Role Within PAT And Quality By Design Programs
- Deployment Models: In-Line, On-Line, And At-Line Use
- Data Handling, Chemometrics, And System Integration Requirements
- Outlook Through 2036
- Quick Stats For Real-time Bioprocess Raman Analyzer Market
- Real-time Bioprocess Raman Analyzer Market Key Takeaways
- How Is The Real-time Bioprocess Raman Analyzer Market Segmented?
- Segmentation By Product Type
- Segmentation By Application
- Segmentation By End User
- Segmentation By Region
- Why Do Instruments Dominate The Market By Product Type?
- Why Does Bioprocess Analysis Lead By Application?
- What Are The Key Drivers Of The Real-time Bioprocess Raman Analyzer Market?
- What Restraints Affect Market Adoption?
- What Trends Are Shaping The Real-time Bioprocess Raman Analyzer Market?
- How Is The Real-time Bioprocess Raman Analyzer Market Developing From 2026 To 2036?
- Country CAGR Comparison
- China
- India
- Brazil
- United States
- United Kingdom
- France
- Germany
- Competitive Landscape Of The Real-time Bioprocess Raman Analyzer Market
- Key Players In The Real-time Bioprocess Raman Analyzer Market
- Scope Of Report
- Real-time Bioprocess Raman Analyzer Market By Segments
- Frequently Asked Questions
- Bibliography
List of Tables
- Table 1: Real-time Bioprocess Raman Analyzer Market Value (USD Million), 2026 And 2036
- Table 2: Real-time Bioprocess Raman Analyzer Market Forecast CAGR (%), 2026-2036
- Table 3: Real-time Bioprocess Raman Analyzer Market Key Takeaways
- Table 4: Quick Stats For Real-time Bioprocess Raman Analyzer Market
- Table 5: Real-time Bioprocess Raman Analyzer Market Segmentation Framework
- Table 6: Market Share By Product Type (%)
- Table 7: Instruments Segment Share And Procurement Drivers (%)
- Table 8: Software Segment Role And Functional Coverage Mapping
- Table 9: Market Share By Application (%)
- Table 10: Bioprocess Analysis Share And Usage Mapping (%)
- Table 11: Lab To Process Analysis Role Across Development Stages
- Table 12: Market Demand By End User Category
- Table 13: Biopharmaceutical Companies: Deployment Drivers And Constraints
- Table 14: Contract Manufacturing Organizations: Deployment Drivers And Constraints
- Table 15: Research Organizations: Adoption Drivers And Budget Constraints
- Table 16: Regional Outlook Summary, 2026-2036
- Table 17: Country CAGR Comparison (%), 2026-2036
- Table 18: Competitive Landscape Summary
- Table 19: Vendor Capability Mapping Across Probes, Chemometrics, Integration, And Validation Support
- Table 20: Scope Of Report
- Table 21: Real-time Bioprocess Raman Analyzer Market By Segments Summary
List of Figures
- Figure 1: Real-time Bioprocess Raman Analyzer Market Overview And Value Snapshot (2026)
- Figure 2: Real-time Bioprocess Raman Analyzer Market Forecast Value Trend (USD Million), 2026-2036
- Figure 3: Real-time Bioprocess Raman Analyzer Market CAGR Representation (%), 2026-2036
- Figure 4: Real-time Bioprocess Raman Analyzer Market Ecosystem And Value Chain Flow
- Figure 5: Role Of Raman Spectroscopy Within Bioprocess Monitoring Workflows
- Figure 6: Placement Of Raman Analyzers Across In-Line, On-Line, And At-Line Monitoring
- Figure 7: Market Share By Product Type (%): Instruments Versus Software
- Figure 8: Instruments Segment Contribution Within Total Market Revenue (%)
- Figure 9: Raman Analyzer And Raman Probe Functional Architecture Diagram
- Figure 10: Software Role In Data Processing, Chemometrics, And Process Control
- Figure 11: Market Share By Application (%): Lab To Process Analysis Versus Bioprocess Analysis
- Figure 12: Bioprocess Analysis Share Highlight And Operational Scope (%)
- Figure 13: Raman Analyzer Usage Across Cell Culture, Fermentation, And Downstream Processing
- Figure 14: End User Distribution Of Real-time Bioprocess Raman Analyzer Market (%)
- Figure 15: Adoption Patterns Among Biopharmaceutical Companies
- Figure 16: Raman Analyzer Deployment In Contract Manufacturing Organizations
- Figure 17: Research Organization Usage Across Process Development And Scale-Up
- Figure 18: Integration Of Raman Analyzers With Bioreactors And Automation Systems
- Figure 19: Raman Analyzer Alignment With Process Analytical Technology Frameworks
- Figure 20: Data Flow From Spectral Acquisition To Real-time Decision Support
- Figure 21: Key Drivers Influencing Adoption Of Real-time Bioprocess Raman Analyzers
- Figure 22: Restraint Impact Mapping On Market Adoption And System Deployment
- Figure 23: Technology And Workflow Trends Shaping Market Evolution
- Figure 24: Global Regional Demand Distribution For Real-time Bioprocess Raman Analyzer Market (%)
- Figure 25: Country CAGR Comparison (%), 2026-2036
- Figure 26: China Real-time Bioprocess Raman Analyzer Market Growth Trajectory
- Figure 27: India Real-time Bioprocess Raman Analyzer Market Adoption Pattern
- Figure 28: Brazil Real-time Bioprocess Raman Analyzer Market Expansion Overview
- Figure 29: United States Real-time Bioprocess Raman Analyzer Market Maturity Curve
- Figure 30: United Kingdom Real-time Bioprocess Raman Analyzer Market Development Path
- Figure 31: Germany Real-time Bioprocess Raman Analyzer Market Stability And Upgrade Cycle
- Figure 32: France Real-time Bioprocess Raman Analyzer Market Adoption Profile
- Figure 33: Competitive Landscape Structure Of The Real-time Bioprocess Raman Analyzer Market
- Figure 34: Market Positioning Of Leading Raman Analyzer Manufacturers
- Figure 35: Competitive Differentiation Based On Hardware Performance And Integration Depth
- Figure 36: Vendor Capability Mapping Across Probes, Chemometrics, And Validation Support
- Figure 37: Real-time Bioprocess Raman Analyzer Market Segmentation Summary Diagram
- Figure 38: Scope Of Report Coverage Visualization

