Bioprocessing Systems Market
The bioprocessing systems market is segmented by process, application, end user and region. Process includes single-use systems, upstream, downstream and integrated systems. Applications include vaccine development, cell therapy and plant cell cultivation, filters, tissue engineering, biosensors, bioreactors and mixers, plasma fractionation, chromatography, bio-therapeutic molecule production and environmental management aid. End users include biopharmaceutical companies, food and beverages, nutraceuticals, specialty products and industrial chemicals. Forecast for 2026 to 2036.
Bioprocessing Systems Market Size, Market Forecast and Outlook By FMI
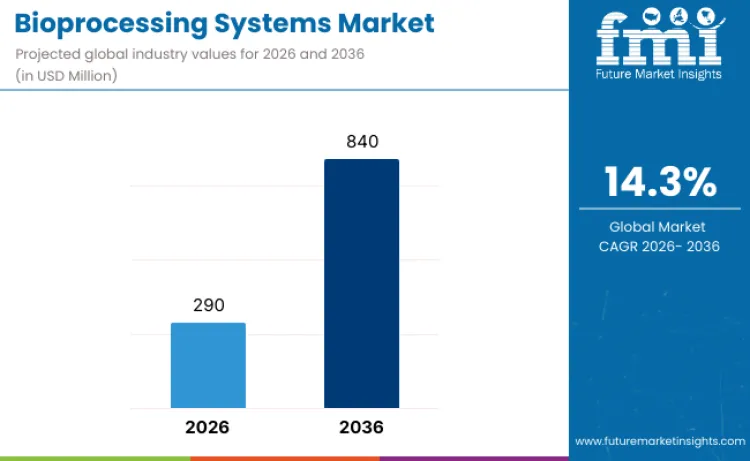
The bioprocessing systems market is estimated at USD 290.0 million in 2026. FMI forecasts the market at USD 840.0 million by 2036, with 14.3% CAGR from 2026 to 2036. Single-use systems remain the leading process with 44.3% share in 2026, while biopharmaceutical companies account for 48.0% of end-user demand.
Summary of the Bioprocessing Systems Market
- Demand and Growth Drivers
- Biologics pipeline expansion is raising the need for flexible production systems.
- Vaccine and cell therapy programs require faster setup and sterile handling.
- Automated systems help manufacturers improve batch control and reduce contamination risk.
- Product and Segment View
- Single-use systems lead because they remove cleaning validation from many workflows.
- Vaccine development holds strong demand due to dose volume and safety needs.
- Biopharmaceutical companies remain the core users because biologics pipelines require repeated process development.
- Geography and Competitive Outlook
- China expands quickly as domestic biotech capacity and biosimilar production rise.
- India gains from vaccine manufacturing scale and export-oriented facilities.
- The USA, Germany and Japan focus on automation, quality control and process intensification.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant at FMI, suggests, “Bioprocessing systems demand will depend on flexibility, sterility and data integrity. Vendors with single-use platforms, automated monitoring and strong validation support are likely to gain deeper relevance as biologics and cell therapy pipelines widen.”
- Bioprocessing Systems Market Value Analysis
- Bioprocessing systems are shifting from fixed plant hardware toward modular production platforms.
- Value improves when suppliers combine equipment, consumables and process analytics.
- Single-use formats reduce setup time and make multi-product facilities easier to manage.
- Continuous processing creates higher yield potential for biosimilars and complex biologics.
What Role Does Operating Margin Play in Technology Adoption?
Improving operating margin is a primary objective for Contract Development and Manufacturing Organizations (CDMOs). According to FMI's analysis, integrating automated perfusion bioreactors increases cell density yields by 40%, directly lowering the cost of goods sold (COGS). Higher volumetric productivity allows facilities to produce more doses per batch, diluting fixed overheads across a larger output. This efficiency gain translates to a 15% improvement in operating margins, providing the necessary capital to upgrade to fully closed, automated processing lines that minimize contamination risks.
How Does EBITDA Influence Capital Expenditure Strategies?
Strong EBITDA margins are critical for biotech startups seeking to invest in scalable infrastructure. FMI analysts opine that modular bioprocessing systems, which require 50% less upfront capital than traditional facilities, protect EBITDA by reducing depreciation expenses. Accelerating the timeline to clinical trials through flexible manufacturing improves valuation multiples for early-stage companies. Maintaining healthy earnings before interest and taxes ensures access to cheaper financing options for facility expansion, which is essential for capturing market share in the competitive biosimilar landscape.
Bioprocessing Systems Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 290.0 Million |
| Industry Value (2036) | USD 840.0 Million |
| CAGR (2026-2036) | 14.3% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Leading manufacturers are are acquiring filtration specialists to enhance their bioprocess technology portfolios, ensuring end-to-end compatibility. Large conglomerates are also diversifying into bioprocessing supplies to capture recurring revenue streams from consumables. Investment in next generation biomanufacturing allows brands to offer continuous processing solutions. Such strategic moves ensure resilience against raw material shortages while meeting evolving regulatory expectations for process analytical technology (PAT) integration.
What Drivers Are Accelerating Biologics Production?
Catalysts for production acceleration include the unprecedented demand for vaccines and gene therapies, driving sales of scalable upstream bioprocessing equipment. Despite high initial validation costs, manufacturers justify spending on automated systems to ensure batch consistency and regulatory compliance. Government grants for domestic vaccine security amplify this behavior. FMI analysts opine that this demand drives innovation in bioprocess containers and fluid transfer solutions, ensuring sterile integrity during liquid handling. Manufacturers are responding by launching pre-sterilized, ready-to-use assemblies that reduce setup times from days to hours. Decoupling production from rigid stainless steel piping allows brands to maintain relevance even as product pipelines diversify. This shift towards agile manufacturing is reshaping facility design strategies across the global bio-cluster network.
How Is the Bioprocessing Systems Market Segmented?
Global landscapes are intricately divided across process, application, and end-user, allowing stakeholders to identify specific value pools. Segmentation strategies enable manufacturers to tailor production capabilities, dedicating cleanrooms for single use bioreactors while optimizing assembly lines for filtration skids. Understanding distinctions between clinical-scale requirements and commercial-scale needs is critical for system design. Dominant segments dictate procurement of key components, with vaccine production requiring heavy investment in bioprocess validation services. Such granular analysis helps companies allocate R&D budgets effectively towards high-growth categories.
Why Do Single-Use Systems Maintain Dominance?
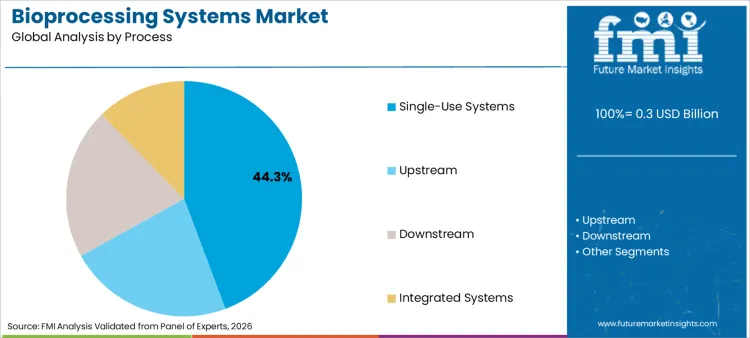
Single-use systems account for 44.3% of share in 2026, driven by their ability to eliminate cleaning validation and reduce cross-contamination risks. Operational flexibility provided by disposable bags and tubing allows for rapid product changeovers in multi-product facilities. Dominance is reinforced by continuous innovation in single use bioprocessing probes and sensors, ensuring real-time monitoring without sterilization downtime. Perceived safety of single-use technologies allows them to meet stringent regulatory standards better than reused equipment. Contract manufacturers remain loyal to these flexible formats, ensuring steady demand across diverse therapeutic modalities.
Why Does Vaccine Development Lead Applications?
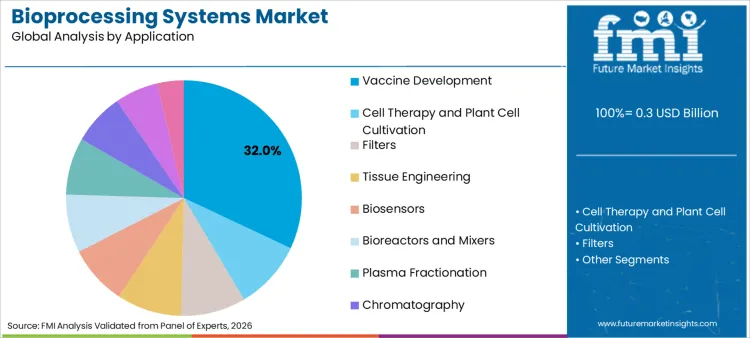
Vaccine development accounts for 32.0% of share in 2026, driven by the ongoing need for immunization programs and pandemic preparedness. The sheer volume of doses required necessitates high-throughput bioprocessing analytics equipment to monitor critical quality attributes. FMI is of the opinion that the segment benefits from the adoption of cellbag bioreactor chambers for rapid seed train expansion. Integration with real time bioprocess raman analyzer technologies allows for in-line quality control, reducing batch failures. This focus on speed and safety appeals to governments and NGOs, sustaining high volumes in the vaccine manufacturing sector.
Why Do Biopharmaceutical Companies Lead End Users?
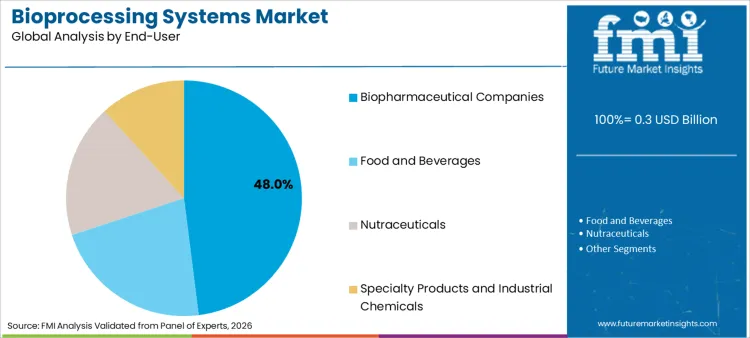
Biopharmaceutical companies account for 48.0% of share in 2026, driven by the massive pipeline of proprietary biologics entering clinical phases. Large pharma entities procure comprehensive suites of small scale bioreactors for process development and pilot runs. As per FMI's projection, the centralization of manufacturing excellence centers necessitates robust fleets of integrated upstream and downstream units. The sheer scale of operations and capital availability creates a continuous replacement cycle for outdated technology. Corporate procurement consolidations favor vendors who can supply end-to-end pharmaceutical filtration platforms.
What Drivers Are Fueling Continuous Processing?
A primary driver is the industry's push towards intensification to reduce facility footprints and operating costs. Biomanufacturers seek higher yields per liter, driving the adoption of liquid processing filter systems capable of extended operation. Integrating perfusion technology offers a distinct competitive edge in producing unstable proteins. Brands marketing continuous systems capture new consumption occasions among cost-conscious biosimilar producers. This "efficiency-first" trend allows facilities to improve throughput without expanding physical infrastructure.
What Restraints Are Hampering Market Expansion?
Intensifying supply chain constraints for plastic consumables, manifested through long lead times, act as a significant restraint. Policies restricting the disposal of single-use plastics create environmental compliance hurdles for large users. According to FMI's estimates, this pressure forces brands to invest in recycling programs for single use filtration assemblies, often increasing operational complexity. High costs of proprietary bioprocess integrity testing systems create unpredictable budget requirements for smaller labs. Manufacturers struggle to maintain premium pricing without demonstrating clear sustainability pathways.
Which Trends Are Reshaping Analytical Innovation?
"Process Analytical Technology" (PAT) movements, where real-time data informs process control, are reshaping product development. This aligns with regulatory shifts towards quality-by-design (QbD) frameworks. Rise of normal flow filtration products awareness pushes factories to implement automated integrity testing. Incorporating unique digital twins helps brands differentiate by predicting batch outcomes. Such innovations cater to a risk-averse industry seeking tools that minimize deviations and ensure batch release speed.
How Will the Bioprocessing Systems Industry Evolve in Major Nations?
Global landscapes are characterized by rapid capacity expansion in Asian nations, driving volume, while Western economies focus on process intensification and digitalization. Each region presents unique regulatory drivers, from NMPA standards in China to FDA modernization acts in the USA. Successful global players must operate decentralized support models, tailoring validation packages to local pharmacopeias while leveraging global supply chains for core hardware.
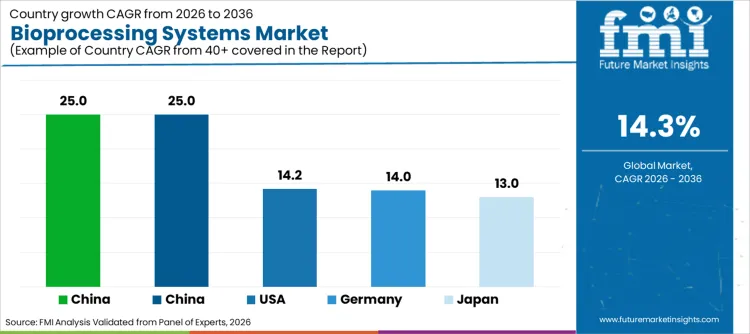
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 25.0% |
| India | 25.0% |
| USA | 14.2% |
| Germany | 14.0% |
| Japan | 13.0% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Why Is the Market in China Driving Hyper-Growth?
Demand for bioprocessing systems in China is set to grow at 25.0% CAGR. Growth is fueled by the government's "Made in China 2025" initiative targeting biotechnology independence. Chinese CDMOs are aggressively expanding capacity to serve global clients, driving demand for cost-effective stainless steel and hybrid systems. FMI analysts opine that the expansion of biosimilar approvals creates massive volume requirements for purification skids. The shift from generic small molecules to complex large molecules is a massive cultural driver, prompting startups to deploy aggressive infrastructure strategies.
Why Is the Market in India Matching Rapid Expansion?
Bioprocessing systems sales in India are projected to rise at 25.0% CAGR. Growth is underpinned by the country's status as the "vaccine capital of the world," requiring massive scale-up technologies. Manufacturers utilize cost-efficient hybrid systems to balance capital expenditure with production flexibility. As per FMI's analysis, India acts as a global hub for vaccine exports, necessitating compliance with WHO pre-qualification standards. The integration with domestic equipment suppliers allows for lower maintenance costs, creating a resilient ecosystem for high-volume manufacturing.
How Is the Domain in the USA Prioritizing Innovation?
The bioprocessing sector in the USA is poised to expand at 14.2% CAGR. Expansion is driven by the density of biotech startups in hubs like Boston and San Francisco focusing on cell and gene therapies. Manufacturers deploy automated, closed systems to reduce labor reliance and contamination risks. FMI is of the opinion that the U.S. market is also seeing a rise in personalized medicine manufacturing, requiring small-batch, highly flexible units. The focus is on systems that offer complete data integrity and audit trails, justifying the automation investment.
How Is the Landscape in Germany Focusing on Engineering?
Germany's bioprocessing domain is projected to rise at 14.0% CAGR. Characterized by engineering excellence, products often tie to precision fermentation and downstream processing efficiency. Manufacturers excel in creating robust, long-lasting hardware that integrates seamlessly with automation layers. "Industry 4.0" culture ensures steady demand for smart, connected bioprocessing units. Factories maintain highly automated production lines capable of supporting complex multi-product facilities.
Why Is the Industry in Japan Targeting Quality?
Bioprocessing demand in Japan is set to grow at 13.0% CAGR. Growth concentrates around regenerative medicine and high-quality monoclonal antibody production. Manufacturers focus on systems that guarantee sterility and particulate control, aligning with strict PMDA regulations. Innovation in single-use sensors enhances process control for sensitive cell lines. Quality-first culture drives the adoption of premium filtration and chromatography systems to ensure patient safety.
How Is the Competitive Landscape Evolving?
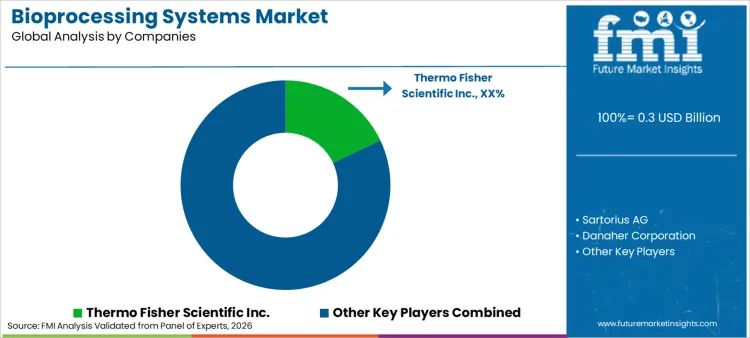
Market participants are increasingly focusing on strategic mergers and acquisitions to consolidate technological capabilities. Thermo Fisher Scientific Inc. leads the market with a dominant 18% share, leveraging its massive portfolio of end-to-end bioproduction solutions. FMI is of the opinion that Thermo Fisher's scale advantage allows it to offer bundled contracts including equipment, consumables, and analytics, creating high switching costs for customers. Leading players are also engaging in M&A activities to acquire niche automation firms, integrating digital control layers directly into hardware to offer "smart factories" out of the box.
Competition is further intensified by the entry of laboratory equipment manufacturers scaling up to industrial processing. Tech giants are partnering with software providers to launch cloud-native bioprocessing control systems that optimize yield across global sites. This trend forces incumbents to open their data architectures to third-party analytics to remain relevant. Strategic partnerships with academic institutions ensure early adoption of novel bioreactor designs. Service reliability and validation support are becoming key battlegrounds, with companies vying to offer the fastest facility startup times to CDMOs racing to market.
Key Players in Bioprocessing Systems Market
- Thermo Fisher Scientific Inc.
- Sartorius AG
- Danaher Corporation
- Pall Corporation
- Merck KGaA
- Lonza Group AG
- Eppendorf SE
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Process | Single-Use Systems, Upstream, Downstream, Integrated Systems |
| Application | Vaccine Development, Cell Therapy and Plant Cell Cultivation, Filters, Tissue Engineering, Biosensors, Bioreactors and Mixers, Plasma Fractionation, Chromatography, Bio-therapeutic Molecule Production, Environmental Management Aid |
| End-User | Biopharmaceutical Companies, Food and Beverages, Nutraceuticals, Specialty Products and Industrial Chemicals |
| Regions | North America, Europe, East Asia, South Asia, Latin America, MEA |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Bioprocessing Systems Market by Segments
Process
- Single-Use Systems
- Upstream
- Downstream
- Integrated Systems
Application
- Vaccine Development
- Cell Therapy and Plant Cell Cultivation
- Filters
- Tissue Engineering
- Biosensors
- Bioreactors and Mixers
- Plasma Fractionation
- Chromatography
- Bio-therapeutic Molecule Production
- Environmental Management Aid
End-User
- Biopharmaceutical Companies
- Food and Beverages
- Nutraceuticals
- Specialty Products and Industrial Chemicals
Bibliography
- Bio‑Process Systems Alliance. (2024). Single‑use technology guidance and best practices for biopharmaceutical manufacturing. BPSA, Washington, DC, United States.
- International Society for Pharmaceutical Engineering. (2023). GAMP 5® Good Practice Guide: Computerized Systems and Good Automated Manufacturing Practice. ISPE, Tampa, FL, United States.
- U.S. Food and Drug Administration. (2024). Emerging Technology Program (ETP). U.S. Department of Health & Human Services, Silver Spring, MD, United States.
- European Medicines Agency. (2025). Quality by Design (QbD) principles in pharmaceutical development. EMA, Amsterdam, Netherlands.
Frequently Asked Questions
How big is the global bioprocessing systems market?
The global bioprocessing systems market is estimated to be valued at USD 290.0 million in 2026.
What is the growth outlook for the bioprocessing systems market over the next 10 years?
The bioprocessing systems market is expected to grow at a CAGR of 14.3% from 2026 to 2036, reaching a valuation of USD 840.0 million.
Which Process drives demand for Bioprocessing Systems?
Single-Use Systems are the primary driver of demand, poised to capture approximately 44.3% of the global market share in 2026.
Who are the leading players in the bioprocessing systems market?
Leading players in the market include Thermo Fisher Scientific Inc., Sartorius AG, Danaher Corporation, and Pall Corporation, among others.
What are the key regional differences in the bioprocessing systems market?
Regional differences include hyper-growth in China and India due to vaccine and biosimilar expansion, while the USA and Germany focus on innovation and engineering excellence.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Process
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Process , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Process , 2026 to 2036
- Single-Use Systems
- Upstream
- Downstream
- Integrated Systems
- Single-Use Systems
- Y to o to Y Growth Trend Analysis By Process , 2021 to 2025
- Absolute $ Opportunity Analysis By Process , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Vaccine Development
- Cell Therapy and Plant Cell Cultivation
- Filters
- Tissue Engineering
- Biosensors
- Bioreactors and Mixers
- Plasma Fractionation
- Chromatography
- Bio-therapeutic Molecule Production
- Environmental Management Aid
- Vaccine Development
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End-User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End-User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End-User, 2026 to 2036
- Biopharmaceutical Companies
- Food and Beverages
- Nutraceuticals
- Specialty Products and Industrial Chemicals
- Biopharmaceutical Companies
- Y to o to Y Growth Trend Analysis By End-User, 2021 to 2025
- Absolute $ Opportunity Analysis By End-User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Process
- By Application
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Process
- By Application
- By End-User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Process
- By Application
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Process
- By Application
- By End-User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Process
- By Application
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Process
- By Application
- By End-User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Process
- By Application
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Process
- By Application
- By End-User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Process
- By Application
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Process
- By Application
- By End-User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Process
- By Application
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Process
- By Application
- By End-User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Process
- By Application
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Process
- By Application
- By End-User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Application
- By End-User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Process
- By Application
- By End-User
- Competition Analysis
- Competition Deep Dive
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Process , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Process , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Process , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Process , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Process , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Process , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Process , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Process , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End-User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Process , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Process , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Process
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End-User, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End-User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Process , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Process , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Process
- Figure 26: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Application
- Figure 29: North America Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End-User, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End-User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Process , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Process , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Process
- Figure 36: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Application
- Figure 39: Latin America Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End-User, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End-User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Process , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Process , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Process
- Figure 46: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Application
- Figure 49: Western Europe Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End-User, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by End-User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Process , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Process , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Process
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Application
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End-User, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End-User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Process , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Process , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Process
- Figure 66: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Application
- Figure 69: East Asia Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End-User, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by End-User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Process , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Process , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Process
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End-User, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End-User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Process , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Process , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Process
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End-User, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End-User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

