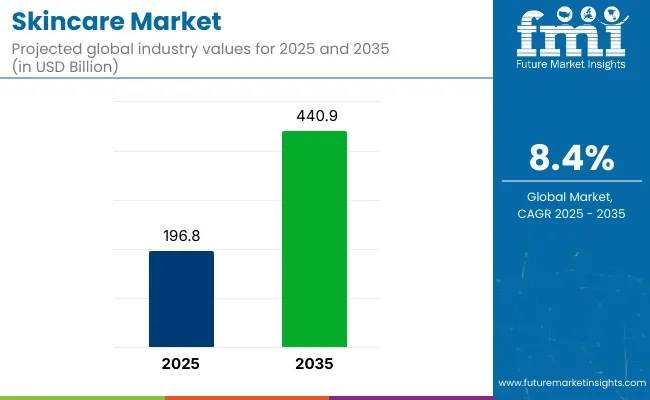
Skincare market valuation is expected to reach USD 215.4 billion in 2026, supported by stronger dermatology literacy, wider adoption of preventive routines, and rising demand for solutions linked to barrier repair, photoaging, pigmentation, and acne control. Sales are projected to expand at an 8.1% CAGR through 2036, taking the sector beyond USD 467.8 billion. Growth is being reinforced by aging demographics that prioritize corrective care, alongside younger consumers building early habits around daily moisturization, sun protection, and active based regimens.
As per FMI’s projection, product leaders are increasing investment in clinical and dermatology led substantiation to defend ingredient claims, improve tolerability profiles, and reduce sensitivity related drop off in repeat purchases. The competitive focus is moving beyond basic moisturizers toward targeted formats that deliver measurable outcomes for specific concerns, including retinoid driven renewal, peptide supported firmness, ceramide based barrier restoration, and hyaluronic acid hydration systems. As per FMI’s estimates, formulators are placing greater weight on concentration accuracy, delivery efficiency, and safety validation, which is accelerating demand for dermatologically tested portfolios that are easier for professionals to recommend and consumers to trust.
FMI is of the opinion that personalization is becoming a core conversion lever, with brands building routines by skin type, climate exposure, and concern severity rather than selling single hero products. Government and regulatory oversight on ingredient safety, labeling, and claim language is also tightening the link between evidence and marketing, pushing brands toward standardized testing, clearer usage guidance, and compliant communication. At the same time, advanced delivery approaches such as microencapsulation and penetration optimizing systems are being used to improve active stability and performance while maintaining skin comfort, helping premium and mass brands scale scientifically supported skincare without compromising accessibility.
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 215.4 Billion |
| Industry Value (2036) | USD 467.8 Billion |
| CAGR (2026-2036) | 8.1% |
Source: Future Market Insights analysis, supported by a proprietary forecasting model and primary research
Retinoid adoption is accelerating when brands can convert clinical credibility and improved tolerability into measurable funnel performance, repeat behavior, and sustained usage. Acquisition should track CAC and conversion rate, because education-led traffic only matters if shoppers move from awareness to a first purchase after understanding expected outcomes and irritation management. Engagement is reflected in DAU/MAU, session duration, and feature adoption, especially where routines, reminders, skin-assessment tools, or onboarding content help users introduce retinoids gradually and stick to protocols. Retention is captured through churn rate, retention rate, and stickiness, since many drop-offs are tied to early irritation, unrealistic timelines, or misuse; products and guidance that reduce friction drive longer continuity. Revenue is validated through LTV, ARPU, and MRR/ARR, as regimens often expand into complementary cleansers, moisturizers, and SPF bundles that increase basket depth. Customer satisfaction should follow NPS and CSAT, linking perceived efficacy and tolerability to advocacy. Usage patterns such as bounce rate and task completion rate indicate whether education and regimen steps are clear enough to keep users progressing rather than abandoning.
Global landscape is intricately segmented to address diverse requirements of the dermatological wellness industry, categorizing sector by product type, skin type, formulation, application, and distribution channel. Structural division allows stakeholders to identify specific therapeutic targets, such as rising demand for gel formulations in oily skin management or preference for cream textures in dry skin applications. By analyzing these segments, manufacturers can tailor production lines to meet distinct needs of dermatological clinics versus direct-to-consumer brands. Segmentation also highlights shift from generic skincare approaches to targeted dermatological interventions. As industry matures, granular segmentation becomes essential for understanding therapeutic pathways and regulatory compliance, ensuring product development aligns with evolving dermatological guidelines.
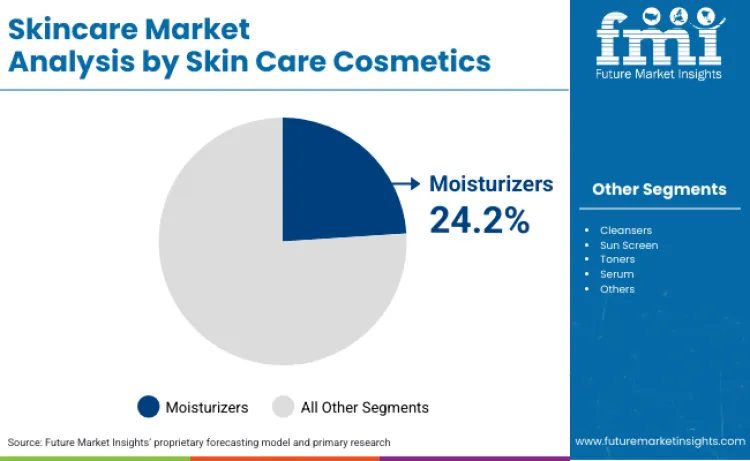
Moisturizers account for 24.2% of product share in 2026, driven by extensive clinical evidence supporting skin barrier function benefits. Leadership position is underpinned by dermatological approvals from healthcare authorities recognizing hydration maintenance therapeutic claims. Segment benefits from continuous formulation improvements, such as ceramide integration technology removing moisture loss concerns. Research focused on hyaluronic acid stability is renewing interest among cosmetic manufacturers. Critical role in managing skin barrier repair formulations further solidifies standing, as these products reduce transepidermal water loss. As dermatological guidelines prioritize hydration maintenance, demand for clinically-validated moisturizing concentrates is expected to remain robust.
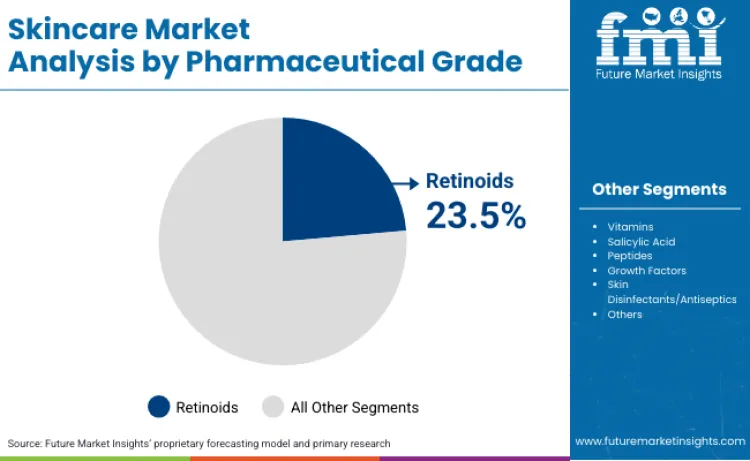
Retinoids command 23.5% share of pharmaceutical grade sector in 2026, reflecting dermatological preference for scientifically-proven anti-aging delivery systems. Dominance is linked to superior bioavailability of vitamin A derivatives compared to cosmetic alternatives. Dermatologists rely on specialized retinoid technology to ensure precise dosing and photostability protection. These formulations are essential for delivering concentrated tretinoin and adapalene, driving priority status among prescription skincare brands. Relentless pursuit of FDA approval ensures products designed for photodamage reversal remain primary choice. High dermatological acceptance necessitates dynamic manufacturing capacity, bolstering sustained revenue streams for pharmaceutical suppliers.
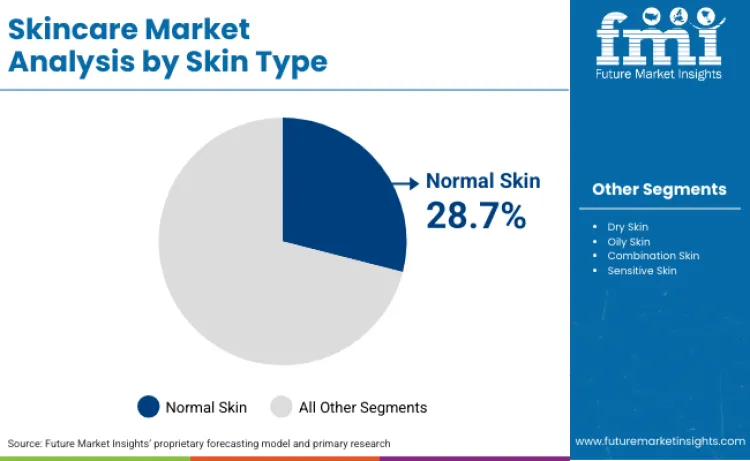
Normal skin applications capture 28.7% of market share in 2026, owing to widespread dermatological maintenance requirements demanding balanced skincare intervention. Dominance stems from clinical guidelines recommending preventive skincare alongside therapeutic treatments for optimal skin health management. Consumers prefer these products for their gentle benefits and minimal sensitivity risks. Usage in maintaining healthy skin barrier function drives continuous volume growth among preventive skincare populations. Compatibility with various dermatological formulation matrices allows for combination therapies across different skin concerns. Manufacturers focusing on pH-balanced and non-comedogenic formulations for these maintenance conditions secure significant loyalty among dermatology practices.
An older consumer base is shifting skincare demand toward products that can deliver visible results while respecting thinner, drier, and more reactive skin. This is pushing brands to develop age-specific regimens that prioritize barrier support, gentler dosing, and compatibility with common age-linked concerns such as persistent dryness, uneven tone, and sensitivity. Combination anti-aging is also gaining share because mature users often want one routine to address multiple endpoints, which increases demand for multi-active systems built around careful tolerability logic. Providers and clinics influence purchase decisions more strongly in this segment, so manufacturers that cannot align claims, instructions, and safety expectations with dermatology-led practice risk losing prescription pull-through and premium shelf space.
Clean beauty is affecting growth by changing how products are evaluated and purchased, not just what ingredients are used. The category is increasingly shaped by disclosure, traceability, and clearer risk signaling around allergens and irritants, which elevates compliance-ready documentation and transparent labeling as competitive advantages. Online channels amplify this shift because shoppers compare formulations quickly, filter for sensitivities, and expect brands to justify choices with plain-language explanations and credible standards. As a result, product teams are reformulating to reduce friction points that trigger returns or negative reviews, while marketing strategies emphasize ingredient provenance and safety positioning that can stand up to scrutiny from informed consumers and clinical advisors.
Formulation innovation is being driven by delivery systems that improve active performance without increasing irritation, especially as consumers demand stronger outcomes with less downtime. Encapsulation, advanced emulsions, and controlled-release designs are being used to protect unstable actives, manage release rates, and support consistent sensory feel across climates and storage conditions. At the same time, regulatory expectations for substantiated claims are raising the bar on testing strategy, pushing brands toward stronger validation of absorption, tolerability, and real-world efficacy. Companies that can pair innovation with defensible clinical support and scalable manufacturing are better positioned to win premium share in performance skincare.
Global landscape for dermatological products is characterized by diverse regulatory frameworks, influenced by healthcare systems and clinical practice standards. Established beauty markets prioritize evidence-based formulations and pharmaceutical-grade manufacturing, whereas emerging economies focus on accessible pricing and basic skin protection. Asia-Pacific is emerging as rapid growth hub due to increasing K-beauty influence and expanding skincare awareness. Conversely, North American sectors pivot towards precision dermatology and personalized formulation protocols. Government initiatives in developing nations supporting healthcare access ensure sustained demand for dermatological products across all demographics.
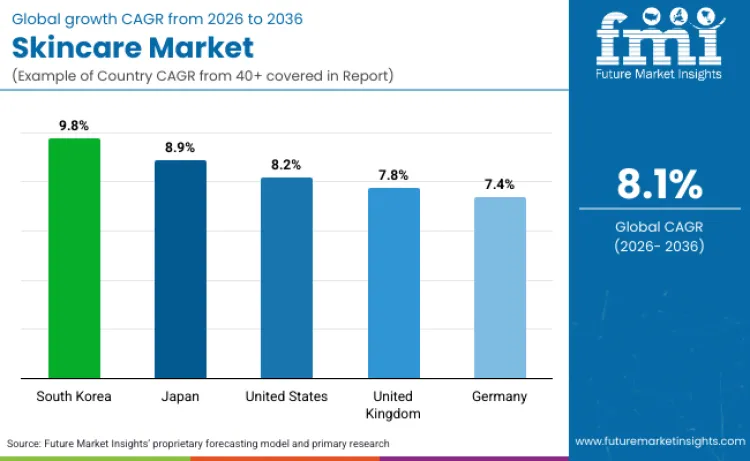
| Country | CAGR (2026 to 2036) |
|---|---|
| South Korea | 9.8% |
| Japan | 8.9% |
| United States | 8.2% |
| United Kingdom | 7.8% |
| Germany | 7.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Skincare growth in South Korea is being reinforced by export led scale and a regulatory structure that supports rapid commercialization of differentiated products. Korea’s official government channels have highlighted record cosmetics export performance and global ranking strength, which expands production volumes, new product launches, and cross border brand visibility for Korean skincare. On the compliance side, MFDS defines and manages “functional cosmetics” under a distinct framework, which supports faster development of performance positioned skincare when products fit recognized functional categories and evaluation pathways.
Japan’s skincare demand is rising because domestic consumption is strongly tied to healthy aging needs and a structured pathway for products positioned closer to functional benefit claims. Japan’s Cabinet Office documents a very high share of older population, which sustains routine demand for anti aging, barrier support, and prevention oriented skincare across long use cycles. For efficacy adjacent positioning, Japan uses a defined quasi drug pathway with formal manufacturing and marketing procedures, which encourages brands to invest in validated actives and disciplined claims, supporting premiumization and repeat purchase.
Skincare expansion in the United States is being shaped by tighter federal compliance expectations that raise the baseline for safety, traceability, and accountable product stewardship, which favors organized brands and clinically positioned product lines. FDA’s MoCRA framework expands oversight and sets requirements such as facility registration and product listing, pushing the category toward better documented portfolios and strengthening consumer and retailer confidence in regulated supply chains.
UK skincare growth is supported by a clear, enforced cosmetics compliance framework in Great Britain that requires responsible placement on the market and aligns product safety expectations with established European style rules. UK government guidance explains how Regulation (EC) No 1223/2009 applies in Great Britain through domestic enforcement regulations, which strengthens consistency in labeling, safety obligations, and market surveillance expectations. This clarity supports broader commercialization of evidence based skincare and helps scalable brands expand across retail and online channels with fewer compliance ambiguities.
Germany’s skincare momentum is strongly linked to a compliance and safety culture that supports therapeutic and sensitive skin positioning, alongside high consumer pull for natural and organic aligned products. German federal bodies contribute directly to cosmetics safety and surveillance, with BFR assessing risks of cosmetic ingredients and BVL referencing the EU wide legal framework used for cosmetics placed on the German market. In parallel, Germany has a significant organic farming footprint, which supports ingredient sourcing narratives and demand for natural positioned skincare lines that emphasize traceable, sustainably sourced inputs.
The skincare landscape is increasingly shaped by science-led portfolio building through acquisitions, as major groups look to lock in clinical credibility, faster innovation cycles, and loyal communities that already trust “active” routines. L’Oréal has used M&A to strengthen both ends of the premium spectrum, adding luxury skincare via Aesop and expanding its science-driven skincare push through a majority stake in Medik8.
Shiseido has moved in the same direction by acquiring Dr. Dennis Gross Skincare, leaning into dermatologist-led positioning and treatment-style regimens that sit closer to clinical skincare than traditional cosmetics. Estée Lauder’s completed acquisition of DECIEM signals how seriously incumbents are taking ingredient-forward brands that win on efficacy narratives and repeatable routines. Unilever is also buying into actives-led skincare momentum, its India business announced an agreement to acquire Minimalist, reflecting how mass players are using acquisitions to upgrade into higher-growth, “results-first” segments.
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Skin Care Cosmetics Segments | Cleansers; Sun Screen; Toners; Serum; Moisturizers; Facial Mask; Exfoliators; Hydrating & Anti-Aging Cream; Depilatories; Eye Creams; Skin Conditioners |
| Pharmaceutical Grade Categories | Vitamins; Retinoids; Salicylic Acid; Peptides; Growth Factors; Skin Disinfectants/Antiseptics; Solution; Spray; Skin Barrier Repair Products |
| Prescription Categories | Corticosteroids; Non-steroidal drugs; Antifungal; Antibiotics; Immunosuppressants; Biologics; Others |
| Skin Type Categories | Normal Skin; Dry Skin; Oily Skin; Combination Skin; Sensitive Skin |
| Distribution Channel Types | Dermatology Clinics; Beauty & Wellness Centers; Medical Spa; Retail Pharmacy Chains; Drug Stores; Hyper Market & Super Market; Mono Brand Stores; Specialty Stores; Online Sales Channels |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Key Countries | South Korea, Japan, United States, United Kingdom, Germany |
| Key Companies Profiled | L'Oréal; Unilever; Procter & Gamble; Johnson & Johnson; Bausch Health Companies; Estée Lauder; SkinCeuticals; CeraVe; Beiersdorf AG |
| Additional Attributes | Dollar sales measured for skincare products used in dermatological wellness and cosmetic applications, specified by active ingredient type (retinoids, peptides, ceramides, hyaluronic acid), product formulation, skin type preference, application focus (anti-aging, hydration, acne treatment), retail model (clinical vs. consumer), and compliance alignment with evolving safety standards and dermatological regulations. |
How big is the global skincare market?
The global skincare market is valued at USD 215.4 billion in 2026.
What is the growth outlook for the skincare market over the next 10 years?
The market is projected to grow at an 8.1% CAGR from 2026 to 2036, reaching USD 467.8 billion.
Which product segments or formats drive demand in this market?
Demand is driven primarily by moisturizers, pharmaceutical-grade actives such as retinoids, and clinically positioned anti-aging and hydration formulations.
How does consumer behavior differ by region?
Asia-Pacific markets emphasize innovation-led and routine-based skincare, while North America and Europe prioritize clinically validated, dermatologist-endorsed, and prescription-adjacent products.
What are the main risks and constraints affecting this market?
Key constraints include regulatory scrutiny on active ingredients, rising compliance costs, ingredient sensitivity concerns, and heightened consumer expectations for safety and efficacy claims.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.