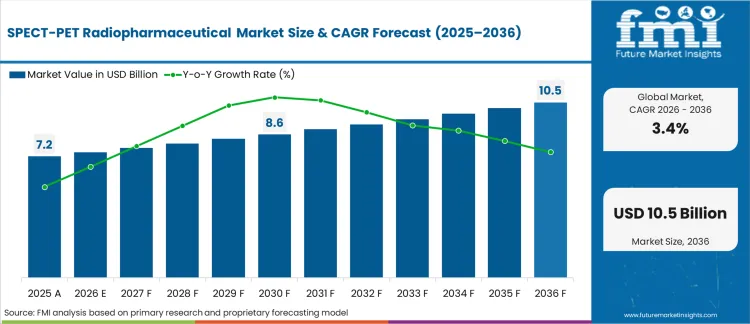
In 2026, the SPECT/PET radiopharmaceutical market was valued at USD 7,485.0 million. Based on Future Market Insights analysis, demand for SPECT/PET radiopharmaceuticals is estimated to grow to USD 10,456.8 million by 2036. FMI projects a CAGR of 3.4% during the forecast period.
Absolute dollar growth of USD 2,971.7 million over the decade signals steady expansion rather than a step-change cycle. As per FMI, growth is expected to be supported by broader utilisation of nuclear imaging and radiotheranostic pathways, while manufacturing lead times, regulated transport controls, and site qualification requirements are expected to limit rapid capacity switching.
“The FDA’s acceptance of our NDA is an important regulatory milestone in advancing this new radiopharmaceutical treatment option for patients with GEP-NETs, In the Phase 3 COMPETE trial, 177Lu-edotreotide demonstrated extended PFS, a straightforward dosing regimen, and a favorable safety profile, supporting its potential to improve the current treatment paradigm. We look forward to working closely with the FDA toward potential approval.’ said Dr. Celine Wilke, Chief Medical Officer of ITM.5]
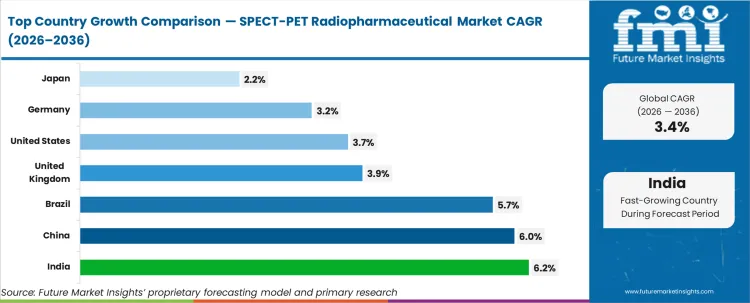
India (6.2% CAGR, supported by expanding nuclear medicine capacity and higher diagnostic throughput), China (6.0% CAGR, driven by installed-base expansion and wider oncology imaging use), and Brazil (5.7% CAGR, linked to rising access to advanced imaging) are expected to lead growth. The UK (3.9% CAGR) and the U.S. (3.7% CAGR) are expected to grow through repeat imaging demand. Germany (3.2% CAGR) and Japan (2.2% CAGR) are expected to contribute more through replacement demand, constrained by tight scheduling governance and budget-led reimbursement pressure.
The market includes radiopharmaceuticals for SPECT and PET procedures for imaging and targeted therapy. The products are used in controlled clinical environments and are sold to hospitals, imaging centers, and cancer therapy centers based on protocol and tracer availability. The market includes radioisotopes used for imaging and therapeutic purposes and are presented as oral solution and IV solution formats. The market is driven by procedure volumes, oncology and cardiology case mix, and regulatory requirements for handling and transporting the products. The revenue stream is driven by dose sales and repeat business based on imaging and therapy schedules.
The report provides global and regional market sizes and a 10-year forecast from 2026 to 2036. The market is sized by radioisotope, application, dosage form, and end user, with country-level CAGR comparisons across key markets. The report also examines the role of supply chain and site readiness factors in purchasing, continuity of scheduling, and vendor selection, through primary research and desk research from authorized public sources.
The scope excludes PET and SPECT scanners, cyclotrons and generators sold as capital equipment, dose calibrators, injectors, shielding consumables, and imaging software. It also omits radiology service revenue and procedure fees, focusing strictly on radiopharmaceutical product value. Non-nuclear contrast media and radiotherapy devices are excluded. Research-only isotopes not supplied for clinical imaging or therapeutic use are excluded unless sold under compliant clinical supply pathways.
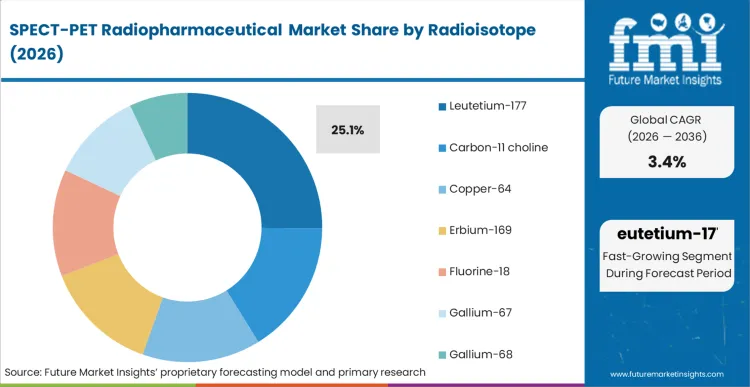
Based on FMI’s report, lutetium-177 is estimated to hold 25.1% share in 2026, followed by fluorine-18 at 17.6% share. Lu-177 leadership is supported by its role in targeted radionuclide therapy workflows, where treatment courses create repeat dosing schedules and higher value per patient cycle. F-18 remains the second largest share due to its broad PET imaging footprint and routine ordering patterns that track daily scan throughput.
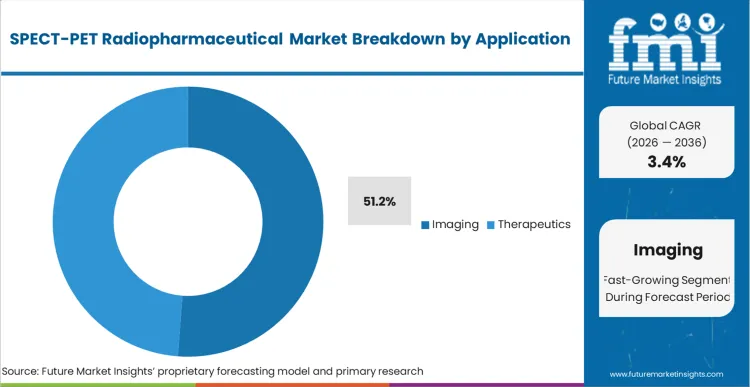
Imaging accounts for 51.2% share in 2026, followed by therapeutics at 48.8%, based on FMI’s report. Imaging remains marginally higher as scan volumes are spread across oncology staging, cardiology perfusion, and neurological assessments, keeping repeat tracer procurement steady. Therapeutics is close behind as theranostic pathways expand, where higher value per administration supports revenue growth even when treated patient counts are smaller than scan volumes.
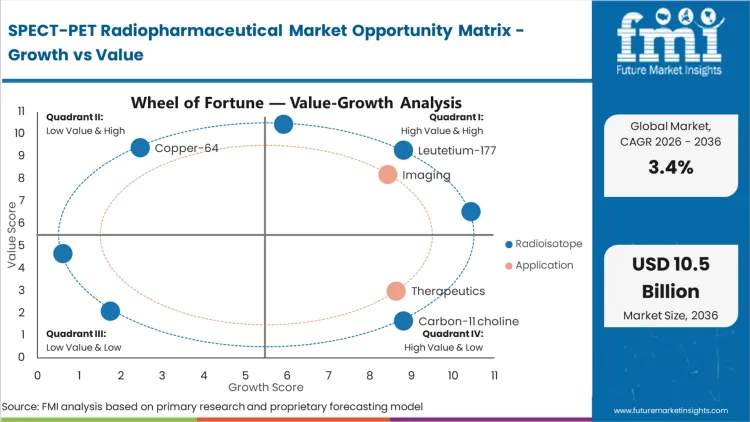
Future Market Insights analysis that historical patterns point at a capacity- and logistics-governed category where demand is anchored in repeat nuclear medicine imaging throughput and scheduled radionuclide therapy cycles. Procurement is being shaped by short half-life handling requirements, dose-day delivery discipline, and site scheduling realities that convert any disruption into cancelled scans or deferred therapy sessions, as per FMI.
While demand expansion is being paced by isotope availability limits, licensed site readiness, and strict cold-chain execution windows that increase cancellation risk, value growth is being supported by rising theranostic utilization and continued dependence on routine PET imaging tracers. Based on FMI’s report, the forecast reflects a new operating balance where supplier qualification is being tightened around release reliability, contingency planning, and documented handling consistency across hospitals and imaging networks.
.webp)
Based on the regional analysis, SPECT/PET radiopharmaceutical market is segmented into North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia, and Middle East & Africa across 40+ countries. Regional performance is assessed using country-level demand signals linked to cyclotron and reactor access, radiopharmacy network density, reimbursement discipline, and hospital and imaging-center uptake of imaging and therapeutic isotopes, as per FMI. The full report also offers market attractiveness analysis based on regional trends.
| Country | CAGR |
|---|---|
| United States | 3.7% |
| India | 6.2% |
| Germany | 3.2% |
| United Kingdom | 3.9% |
| Japan | 2.2% |
| China | 6.0% |
| Brazil | 5.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America is shaped by theranostics scale-up and hospital-led nuclear medicine throughput, where product selection is governed by delivery reliability, site readiness for therapy, and payer coverage discipline. Novartis AG is the largest scale participant through broad radioligand therapy footprint and associated supply infrastructure. GE Healthcare holds strong positioning in PET imaging tracers and radiopharmacy reach aligned to hospital and imaging networks. Lantheus Holdings, Inc. competes through a focused nuclear medicine portfolio with strong USA channel penetration According to Future Market Insights analysis, the growth is still limited by the availability of isotopes and the risk of scheduling, although the value share linked to therapy continues to increase.
FMI’s report includes a detailed analysis of the growth in the North American region, along with a country-wise assessment that includes the United States. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the North America region.
The Asia Pacific continues to be the fastest-growing market, driven by the development of nuclear medicine infrastructure, the increasing need for oncology imaging, and the eventual development of local isotope production and radiopharmacy distribution infrastructure. Curium Pharma and local vendors engage through tracer product lines and radiopharmacy services, while hospital systems and cancer centers drive usage growth as new facilities come on line. FMI analysts point out that the growth trajectory of the region is being led by site licensing, radiopharmacy staffing, and the ability to maintain cold-chain delivery windows.
The full report provides an overview of the SPECT/PET Radiopharmaceuticals market in East Asia and South Asia from 2021 to 2036. The trends that dictate the market are discussed in the report, which covers countries like China, Japan, South Korea, India, Indonesia, Malaysia, and Thailand.
Latin America has a mixed public and private adoption pattern, where access to the cyclotron, import dependence for certain isotopes, and distributor reach impact adoption and pricing. Lantheus and Curium operate through tracer distribution channels, and radiopharmacies in the region help with distribution in large urban agglomerations. FMI believes that the market is being fueled by the expansion of PET imaging availability and oncology diagnostics, with challenges related to infrastructure concentration and reimbursement variability.
The report provides a complete analysis of the market in Brazil, Mexico, Chile, Argentina, Peru, and Rest of Latin America. The reader can gain insights into various aspects, including the pricing trends, which are currently influencing the Latin America market.
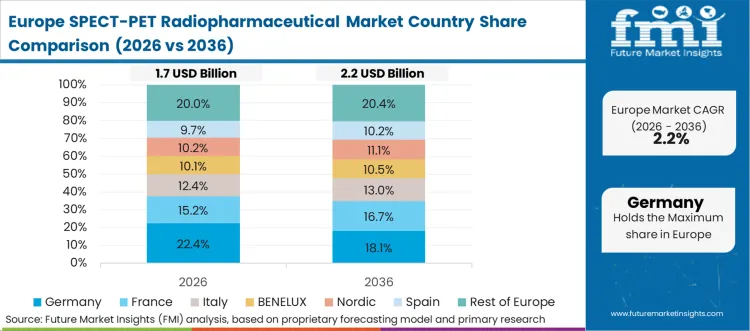
Europe is a compliance-driven and capacity-constrained market, where the demand for radiopharmaceuticals is driven by reactor and cyclotron capacity, transportation windows, and reimbursement control in the national healthcare systems. Novartis AG and Curium Pharma operate in the therapy and imaging franchises, which are linked to the regulated supply chains. GE Healthcare maintains engagement with the imaging tracer portfolios and distribution infrastructure. According to the FMI analysis, market growth is fueled by the demand for oncologic imaging, and therapy market growth is fueled by site readiness and isotope availability.
FMI’s analysis of SPECT/PET radiopharmaceutical market in Europe consists of country-wise assessment that includes the Germany, Italy, France, United Kingdom, Spain, Russia, Nordic, Benelux and Rest of Europe. Readers can know various regulations and latest trends in the regional market.
Market structure remains fragmented, yet practical competition is concentrated among a limited set of suppliers that can control radionuclide access, run GMP radiopharmaceutical manufacturing with repeatable batch release, and execute time bound distribution that matches imaging slots and therapy appointments. Competition is shaped by execution reliability more than price swings, since dose usability is constrained by isotope decay and shipping windows. Portfolio breadth across PET and SPECT tracers supports account stability, yet performance gates such as on time delivery, cancellation rates, and deviation handling tend to determine allocation decisions, as per FMI.
The suppliers with structural advantages are usually placed at various points in the chain, ranging from isotope production or secure long-term isotope supply, through formulation and aseptic fill finish, to regional radiopharmacy or distribution reach. Redundancy in manufacturing across facilities minimizes the risk of disruption due to cyclotron, reactor, or processing downtime. High-quality systems, quick release testing, and cold chain capability safeguard valuable time at the point of care. Those without redundancy usually operate on single geography networks or isotope reach, which heightens vulnerability to contract loss as service levels deteriorate, according to the report by FMI.
The buyer behavior also perpetuates leverage. Hospitals and large health systems usually prequalify multiple suppliers for routine tracers, maintain approved alternates active, and employ substitution clauses to manage product shortages without postponing procedures. Imaging center networks usually contract with radiopharmacy partners and maintain the ability to switch when delivery performance deteriorates. Cancer treatment centers can be more protocol bound for certain therapeutic agents, yet contingency arrangements are still maintained to protect scheduling continuity. This procurement approach limits pricing power for routine imaging products, while measured premiums are retained mainly where supply is tight or handling requirements raise switching friction, Future Market Insights analysis.
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
| Metric | Value |
|---|---|
| Quantitative Units | USD 7,485.0 million (2026) to USD 10,456.8 million (2036), at a CAGR of 3.4% |
| Market Definition | The SPECT and PET radiopharmaceutical market comprises the global production and trade of diagnostic and therapeutic radioisotope based agents supplied to nuclear medicine and molecular imaging services, where demand is linked to oncology and cardiology imaging volumes, radionuclide therapy expansion, and supply chain constraints tied to isotope production, distribution windows, and site licensing requirements. |
| Radioisotope Segmentation | Carbon-11 choline, Copper-64, Erbium-169, Fluorine-18, Gallium-67, Gallium-68, Indium-111, Iodine-123, Iodine-125, Leutetium-177, Nitrogen-13, Radium-223, Rhenium-186, Rubidium-82, Strontium-89, Technetium-99, Thallium-201, Yttrium-90 |
| Application Coverage | Imaging, Therapeutics |
| Dosage Form Coverage | Oral Solution, IV Solution |
| End User Coverage | Hospitals, Diagnostic Imaging Centers, Cancer Treatment Centers, Specialty Centers, Research and Academic Institutes, Others |
| Regions Covered | North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe, and Middle East & Africa. |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia and 40+ countries |
| Key Companies Profiled | Novartis AG, GE HealthCare Technologies Inc., Cardinal Health, Inc., Eli Lilly and Company, Lantheus Holdings, Inc., Bayer AG, Boston Scientific Corporation, Life Molecular Imaging GmbH, Curium Pharma, Q BioMed Inc., Jubilant DraxImage Inc., NorthStar Medical Radioisotopes, LLC |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with radiopharmaceutical suppliers, radiopharmacies, and nuclear medicine stakeholders, supported by isotope supply benchmarking and procedure volume triangulation, as per FMI. |
How large is the demand for SPECT/PET Radiopharmaceuticals in the global market in 2026?
Demand for SPECT/PET Radiopharmaceuticals in the global market is estimated to be valued at USD 7,485.0 million in 2026, as per FMI.
What will be the market size of SPECT/PET Radiopharmaceuticals in the global market by 2036?
Market size for SPECT/PET radiopharmaceuticals is projected to reach USD 10,456.8 million by 2036.
What is the expected demand growth for SPECT/PET Radiopharmaceuticals in the global market between 2026 and 2036?
Demand for SPECT/PET Radiopharmaceuticals in the global market is expected to grow at a CAGR of 3.4% between 2026 and 2036.
Which application is poised to lead global demand by 2026?
Imaging is expected to be the dominant application, capturing 51.2% share in 2026.
Which dosage form is expected to account for the largest share in 2026?
IV Solution is expected to hold the highest share at 98.3% in 2026.
How significant is the role of hospitals in driving adoption in 2026?
Hospitals are projected to hold 49.4% share of end user demand in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.