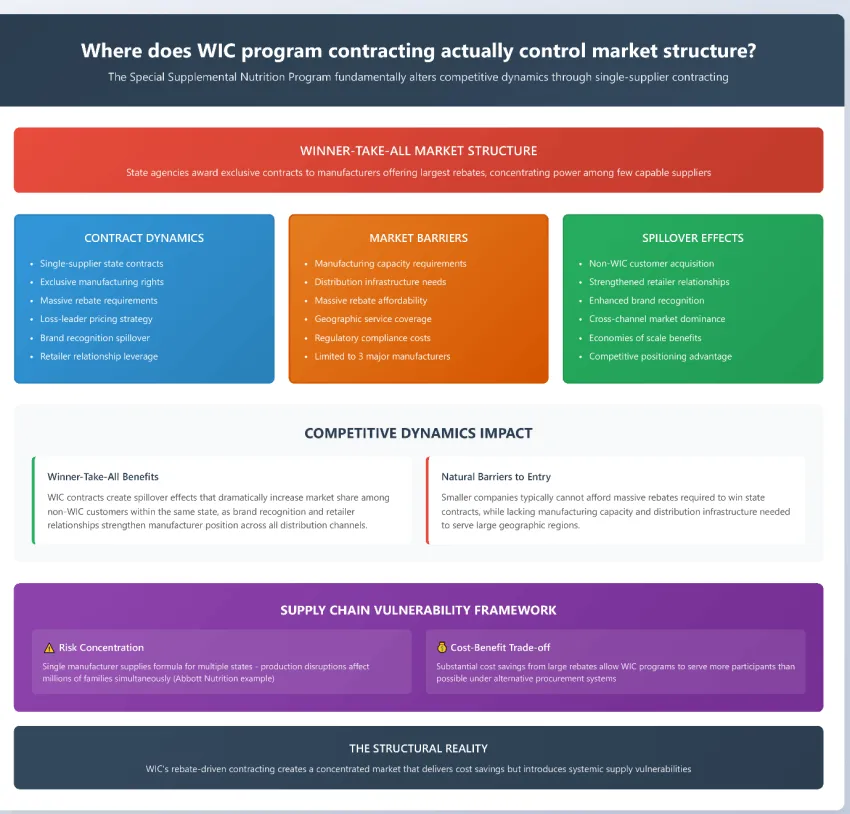
The Special Supplemental Nutrition Program for Women, Infants, and Children fundamentally alters competitive dynamics in the infant formula industry through its single-supplier contracting system. State agencies award exclusive contracts to manufacturers who offer the largest rebates, creating a winner-take-all market structure that concentrates power among the few companies capable of serving entire state populations.
WIC rebates have become so substantial that manufacturers often operate at minimal margins or even losses on direct program sales, viewing these contracts as loss leaders that generate broader market advantages. Winning a WIC contract creates spillover effects that dramatically increase market share among non-WIC customers within the same state, as brand recognition and retailer relationships strengthen the manufacturer's position across all distribution channels.
The contract bidding process creates natural barriers to entry that protect established manufacturers from new competition. Smaller companies typically cannot afford the massive rebates required to win state contracts, while lacking the manufacturing capacity and distribution infrastructure needed to serve large geographic regions. This dynamic has limited WIC contracts to just three major manufacturers over the past decade.
The system also creates supply chain vulnerabilities that became apparent during the Abbott Nutrition plant shutdown. When a single manufacturer supplies formula for multiple states, production disruptions can affect millions of families simultaneously. However, the substantial cost savings generated by large rebates allow WIC programs to serve more participants than would be possible under alternative procurement systems.
The infant formula industry exhibits extreme economies of scale due to the substantial fixed costs associated with regulatory compliance, specialized equipment, and quality assurance systems. Large manufacturers can spread these overhead expenses across massive production volumes while smaller players face disproportionately high per-unit costs for the same regulatory and quality requirements.
FDA regulations require extensive documentation, testing, and approval processes for any formula modifications or new product introductions. Maintaining compliance with current good manufacturing practices demands dedicated quality assurance personnel, sophisticated analytical laboratories, and comprehensive record-keeping systems. These regulatory overhead costs remain relatively fixed regardless of production volume, creating structural advantages for high-volume manufacturers.
Manufacturing equipment for infant formula production requires substantial capital investments in spray drying systems, specialized mixing equipment, and sterile packaging lines. The precision required for consistent nutritional composition and microbiological safety demands equipment capabilities that far exceed those needed for conventional food processing. These capital requirements create natural barriers to entry while favoring manufacturers with sufficient volume to justify the investments.
Research and development capabilities represent another area where scale provides competitive advantages. Developing new formulations requires extensive clinical testing, regulatory submissions, and validation studies that can extend over multiple years. Large manufacturers can maintain dedicated research facilities and clinical trial programs that smaller companies cannot economically support.
European formula manufacturers have introduced alternative approaches to infant nutrition that challenge the ingredient and processing strategies of established American brands. Rather than competing purely on nutritional equivalence, these companies emphasize different regulatory standards, organic ingredients, and manufacturing methods that appeal to health-conscious consumers willing to pay premium prices.
Organic formula production requires certified organic milk sources that command substantially higher prices than conventional dairy ingredients. The organic certification process adds complexity to supply chain management while requiring extensive documentation and testing to maintain compliance. However, the premium pricing achievable for organic products often justifies these additional costs for manufacturers serving the health-focused market segment.
European regulatory standards create different competitive dynamics by restricting certain ingredients commonly used in American formulas while requiring others that American manufacturers often omit. These regulatory differences create opportunities for European brands to differentiate their products based on ingredient purity and processing methods rather than competing solely on nutritional outcomes.
Direct-to-consumer distribution models allow some European and specialty brands to bypass traditional retail markups while building closer customer relationships. Online sales eliminate distributor margins and reduce inventory carrying costs, enabling more competitive pricing despite higher ingredient and manufacturing costs. This approach particularly benefits smaller brands that cannot achieve the retail distribution scale of established manufacturers.
Infant formula supply chains face unique vulnerabilities due to the specialized ingredients, regulatory constraints, and single-supplier dependencies that characterize the industry. Unlike other food categories where ingredient substitution and alternative sourcing provide flexibility, formula manufacturers must maintain precise nutritional specifications while meeting strict regulatory requirements.
The concentration of manufacturing capacity among a few large facilities creates systemic risks that became apparent during the Abbott Nutrition plant closure. When a significant portion of national production capacity is located at a single facility, any disruption can create widespread shortages that affect millions of families. This concentration reflects the substantial capital investments required for formula manufacturing that make geographic diversification economically challenging.
Specialized ingredient sourcing creates additional supply chain vulnerabilities. Many premium ingredients like probiotics, specialized oils, and vitamins come from limited suppliers with long lead times and strict quality requirements. When these suppliers face disruptions, formula manufacturers cannot easily substitute alternative sources without extensive testing and regulatory approvals.
The regulatory approval process for formula modifications creates inflexibility in responding to supply chain disruptions. Unlike other food products where recipe adjustments can be made quickly, any significant formula changes require FDA notification and potential testing that can take months to complete. This regulatory lag time makes it difficult for manufacturers to adapt quickly to changing ingredient availability.
Sources
Hypoallergenic formulas require complex protein hydrolysis processes that break down milk proteins into smaller, less allergenic fragments. This processing requires specialized enzymes, controlled reaction conditions, and extensive purification steps that significantly increase manufacturing costs compared to standard formula production.
WIC contracts create spillover effects where the contracted brand gains market share among non-WIC customers through increased brand recognition and retailer relationships. This market expansion helps manufacturers justify the large rebates offered to WIC programs by spreading fixed costs across larger total volumes.
Smaller manufacturers face significant disadvantages in regulatory compliance costs, manufacturing scale requirements, and distribution access that make direct competition extremely challenging. Success typically requires focus on specialized niches or premium segments rather than broad market competition.
The combination of substantial capital requirements, complex regulatory approvals, specialized manufacturing expertise, and established competitor advantages creates formidable barriers to entry that discourage new market participants despite attractive pricing levels.
Premium formulas command higher prices through organic certification, alternative ingredient sourcing, different processing methods, and marketing focused on ingredient purity rather than basic nutritional equivalence. These differentiators appeal to health-conscious consumers willing to pay for perceived quality advantages.
Infant Formula DHA Algae Oil Market Size and Share Forecast Outlook 2025 to 2035
Infant Formula Industry Analysis in United States Analysis - Size, Share, and Forecast Outlook 2025 to 2035
Infant Formula Ingredients Market Analysis - Size, Share & Forecast 2025 to 2035
This report covers the a2 infant formula market through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, a2 protein nutrition trends, infant feeding innovation, supply chain developments, and strategic growth opportunities.
Probiotic Infant Formula Market – Growth & Infant Nutrition Trends