China's infant formula market is experiencing unprecedented demand contraction driven by collapsing fertility. The National Bureau of Statistics recorded 9.02 million births in 2023, down from a 2016 peak of 17.86 million. This 50 percent decline reflects multiple reinforcing trends: rising marriage ages, urban cost pressures, and limited effectiveness of the three-child policy introduced in 2021. The shrinking birth cohort directly reduces formula consumption, as approximately 60 to 70 percent of Chinese infants consume some formula by six months of age.
Simultaneously, the market is bifurcating by price tier. Urban consumers, particularly in tier-one cities, continue purchasing premium formulas priced above RMB 300 per 900-gram tin, seeking advanced nutritional claims. However, volume growth has stagnated even in premium segments due to the absolute decline in buyers. Mass-market formulas serving tier-three and lower cities face acute pressure, with price competition intensifying as domestic brands compete for a shrinking pool of cost-conscious parents. This demographic reality is forcing manufacturers to shift strategy from volume growth to value extraction through premiumisation, ingredient innovation and brand loyalty programs.
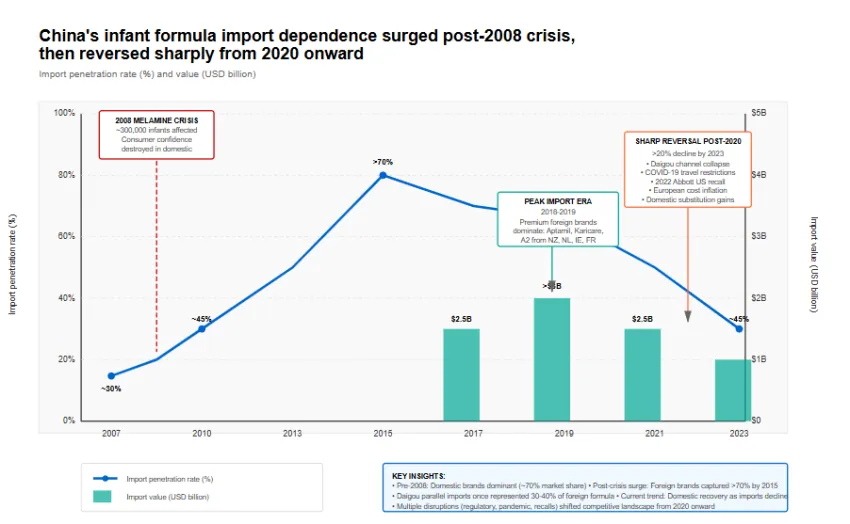
The 2008 melamine crisis, which caused kidney damage in approximately 300,000 infants, destroyed consumer confidence in Chinese formula. Import penetration surged from under 30 percent pre-crisis to peaks exceeding 70 percent by 2015. New Zealand, the Netherlands, Ireland and France became dominant suppliers, with brands like Aptamil, Karicare and A2 commanding premium pricing. UN Comtrade data under HS code 190110 shows China's infant formula imports peaked in value around 2018 to 2019, exceeding USD 3 billion annually.
Since 2020, import volumes have declined sharply. China Customs data indicates a contraction exceeding 20 percent in import value by 2023 compared to peak years. Multiple factors drove this reversal. The daigou parallel import channel, which once accounted for 30 to 40 percent of foreign formula entering China, collapsed under cross-border e-commerce regulation and COVID-19 travel restrictions. The 2022 Abbott recall in the United States, which shut down a major Michigan facility producing Similac, further disrupted supply chains and eroded confidence in foreign quality assurance. European suppliers faced their own challenges, including dairy input cost inflation and reduced China-specific export volumes as domestic substitutes gained traction.
China's State Administration for Market Regulation implemented a formula registration system in 2018 that fundamentally reshaped the competitive landscape. The system capped each domestic factory at three brands with nine SKUs total, consolidating a previously fragmented market and raising quality standards. Simultaneously, Beijing invested heavily in domestic dairy infrastructure, particularly in Heilongjiang and Inner Mongolia provinces, to secure raw milk supply chains with traceability systems comparable to New Zealand standards.
Feihe, China's largest domestic infant formula producer, achieved over RMB 22 billion in revenue in 2022, capturing approximately 18 percent domestic market share. The company positioned itself as a premium domestic alternative, pricing competitively with foreign brands while emphasizing formulas adapted to Chinese infant nutritional needs. Yili and Mengniu, traditionally focused on liquid dairy, expanded infant formula capacity through acquisitions and greenfield investments, collectively adding over 100,000 tons of annual production capacity since 2019. These three producers now supply over 60 percent of domestically manufactured formula.
Product innovation has focused on functional ingredients. Domestic brands have introduced formulas with human milk oligosaccharides, lactoferrin, probiotics and organic certifications to match or exceed foreign product claims. Manufacturing automation and quality control systems have been upgraded to international standards, with major facilities achieving certifications recognising dairy processing excellence. This capacity build-out has reduced dependence on imported finished goods while increasing imports of raw materials like whey protein concentrate and lactose from established dairy exporters.
The SAMR registration system represents China's most significant intervention in formula safety since 2008. As of 2023, SAMR had approved approximately 150 domestic infant formula brands and 80 foreign brands, down from over 2,000 unregistered products circulating pre-2018. Each registered formula undergoes ingredient verification, clinical testing and ongoing surveillance, creating enforceable quality standards that apply equally to domestic and imported products.
Ingredient regulations have tightened substantially. SAMR now mandates disclosure of all functional ingredients, restricts health claims to scientifically validated benefits, and requires traceability documentation from raw material sourcing through final packaging. The introduction of mandatory testing for contaminants, heavy metals and pathogenic bacteria at frequencies exceeding many international standards has narrowed the perceived quality gap between domestic and foreign formulas.
Consumer trust metrics reflect these regulatory gains. Market research indicates that parental willingness to purchase domestic formula has increased from approximately 40 percent in 2018 to over 55 percent by 2023, driven by transparency initiatives and high-profile safety certifications. However, foreign brands retain advantage in absolute trust rankings, particularly among affluent urban consumers who view imported products as insurance against residual safety risk. The trust gap, while narrowing, remains a structural feature favouring established foreign brands in premium segments.
China's infant formula imports have undergone compositional as well as volumetric changes. New Zealand's share of imports, historically above 50 percent, has declined as domestic production expands and European suppliers capture niche segments. Specific value data for 2023 indicates imports remain above USD 2 billion annually, but growth has reversed after years of double-digit expansion. Exports from China, while still modest, have increased modestly as domestic producers seek growth in Southeast Asian markets facing similar demographic transitions.
Pricing pressure has intensified across all tiers. Mass-market formulas priced below RMB 200 per tin face margin compression as domestic brands compete for volume in a shrinking market. Premium segment pricing has held relatively stable above RMB 300, but promotional activity has increased, with brands offering bundled purchases and loyalty discounts to retain customers. Foreign brands historically commanding 30 to 40 percent price premiums now compete within 10 to 20 percent of domestic equivalents in similar nutritional profiles.
Market restructuring is evident in channel dynamics. Traditional mother-and-baby stores, which once favoured foreign brands due to higher margins, now allocate comparable shelf space to premium domestic options as consumer acceptance grows. E-commerce platforms, which account for over 40 percent of formula sales, have accelerated domestic brand visibility through algorithm-driven recommendations and targeted promotions. The cross-border e-commerce channel, which once facilitated daigou imports, now operates under stricter customs and tax enforcement, reducing its role in circumventing formal import channels.
Sources
Annual births fell from 17.9 million in 2016 to approximately 9 million in 2023, cutting the addressable market by nearly half within seven years.
No. China's infant formula imports have declined sharply since 2020, with volumes falling over 20 percent by value between peak years and 2023 as domestic substitution accelerates.
Feihe, Yili and Mengniu have collectively gained over 15 percentage points of market share since 2019, driven by regulatory approvals, expanded manufacturing and premiumisation strategies.
China's 2018 formula registration system capped foreign SKUs, mandated stricter traceability and ingredient controls, and created quality parity conditions that narrowed the perceived safety gap.
Evidence indicates structural rather than cyclical change, with declining daigou channels, reduced reliance on New Zealand and European suppliers, and domestic capacity now meeting over 60 percent of total demand.
Infant Formula DHA Algae Oil Market Size and Share Forecast Outlook 2025 to 2035
Infant Formula Industry Analysis in United States Analysis - Size, Share, and Forecast Outlook 2025 to 2035
Infant Formula Ingredients Market Analysis - Size, Share & Forecast 2025 to 2035
This report covers the a2 infant formula market through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, a2 protein nutrition trends, infant feeding innovation, supply chain developments, and strategic growth opportunities.
Probiotic Infant Formula Market – Growth & Infant Nutrition Trends