About The Report
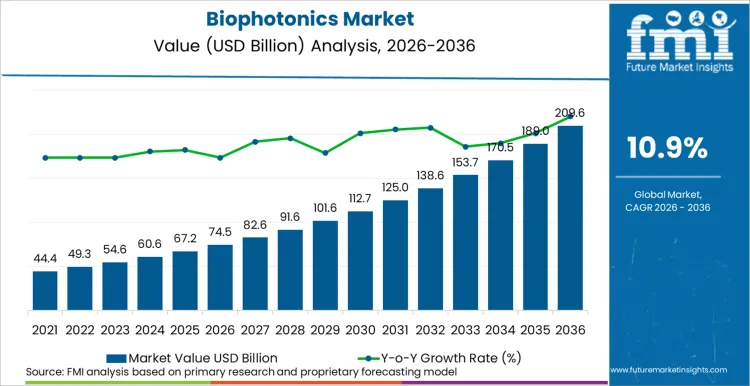
In 2025, the biophotonics market was valued at USD 67.2 billion. Based on Future Market Insights' analysis, demand for biophotonics is estimated to grow to USD 74.5 billion in 2026 and USD 209.9 billion by 2036. FMI projects a CAGR of 10.9% during the forecast period.
Absolute dollar growth of USD 135.4 billion over the decade signals transformational expansion driven by convergence of AI-enhanced imaging, regulatory approval acceleration for photonic diagnostics, and miniaturization of biosensing platforms. Unlike mature industrial adhesive markets, biophotonics sustains double-digit growth through continuous technological disruption in non-invasive diagnostics and point-of-care testing, though equipment capital intensity and reimbursement variability introduce cyclical pressure in hospital procurement.
As Akira Hiruma, CEO of Hamamatsu Photonics, noted regarding precision requirements in biosensing infrastructure, "Photon detection precision is fundamental to next-generation biosensing platforms, and our investment in hybrid PMT systems reflects that commitment." In March 2025, Hamamatsu reported widespread deployment of photomultiplier tubes in national cancer research centers across Japan and Germany, enabling single-molecule resolution in fluorescence-based cell diagnostics [1].
United States (10.2% CAGR) drives expansion through NIH-funded precision medicine initiatives and FDA approvals for optical imaging devices. European Union (10.5% CAGR) benefits from Horizon Europe bio-imaging allocations and point-of-care diagnostic integration. Japan (10.3% CAGR) and South Korea (10.4% CAGR) leverage semiconductor manufacturing capabilities for photonic chip development. United Kingdom (9.8% CAGR) gains from Biomedical Catalyst Programme funding supporting optical biosensor commercialization across regenerative medicine applications.
Biophotonics comprises global production and deployment of light-based technologies for biological analysis, encompassing optical imaging systems, laser therapeutics, and biosensing platforms. The market serves as the technical foundation for non-invasive diagnostics in oncology, ophthalmology, and neurology, utilizing photon interactions with biological tissues to enable molecular detection, structural visualization, and therapeutic intervention at cellular resolution.
The report includes comprehensive analysis of market dynamics, featuring Global and Regional Market Sizes (Volume and Value) and a 10-year Forecast (2026-2036). It covers segmental breakdowns by application (See-Through Imaging, Biosensors, Microscopy), technology (In-vivo, In-vitro), and end-users (Medical Therapeutics, Medical Diagnostics, Non-Medical Application). Analysis incorporates device pricing trends, regulatory approval timelines, and cross-border technology transfer patterns.
The scope excludes non-optical diagnostic modalities such as magnetic resonance imaging (MRI) or computed tomography (CT) unless integrated with photonic components. It also omits downstream clinical services like radiology interpretation fees or surgical procedure costs, focusing strictly on photonic instrumentation, laser systems, and optical component supply. Report does not account for basic research photonics with no commercial diagnostic or therapeutic pathway.
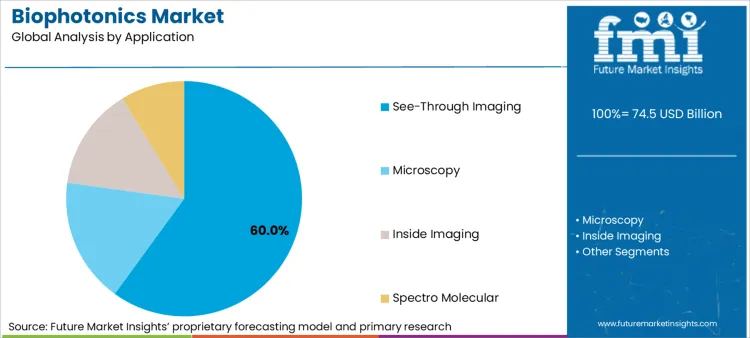
Based on FMI's biophotonics market report, consumption of see-through imaging and biosensors is estimated to hold 60% combined share in 2026. Growth in demand for these applications comes from clinical necessity for non-invasive, real-time diagnostic capabilities that reduce patient risk while maintaining molecular-level detection precision across oncology, cardiology, and neurology workflows.
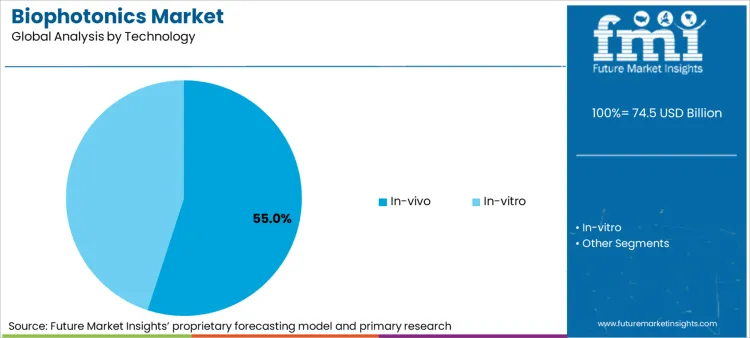
In-vivo biophotonics 55% market share in 2026 stems from its irreplaceable role in real-time surgical navigation and neuroscience research, yet this dominance creates dependency on hospital capital budgets and reimbursement approval cycles. The segment's reliance on high-cost imaging platforms transforms any delay in institutional funding or regulatory clearance into immediate revenue disruption, while technical barriers around tissue penetration depth and signal scattering require continuous R&D investment with uncertain commercial timelines.
Future Market Insights analysis indicates the biophotonics market operates in a growth acceleration phase where technological maturation intersects with regulatory modernization. The 2026 valuation reflects intensified hospital adoption following FDA's 2024 approval of four photonic imaging devices for clinical cancer screening, coupled with European Medicines Agency pathway harmonization. Unlike commodity chemical markets constrained by feedstock volatility, biophotonics growth derives from expanding clinical applications where photonic precision creates distinct diagnostic value unavailable through conventional imaging.
While equipment capital costs remain elevated, demand composition is shifting from research-grade instrumentation to clinical-ready, FDA-cleared diagnostic platforms commanding premium pricing through reimbursement pathway integration. The forecast accounts for accelerating replacement cycles as AI-enhanced imaging systems obsolete first-generation optical coherence tomography installations, with growth sustained through 2036 by point-of-care biosensor miniaturization expanding addressable markets beyond traditional hospital settings into home healthcare and remote monitoring applications.
Based on the regional analysis, biophotonics market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
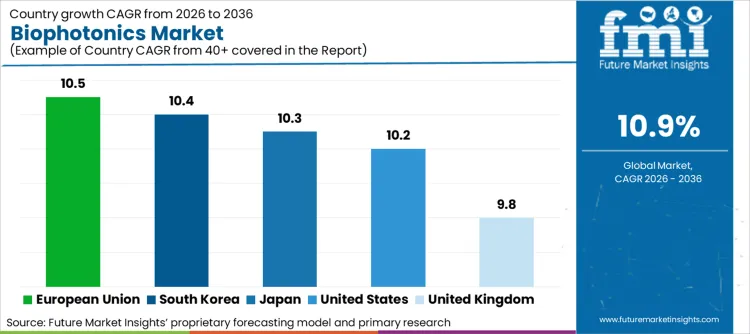
| Country | CAGR (2026 to 2036) |
|---|---|
| United States | 10.2% |
| European Union | 10.5% |
| Japan | 10.3% |
| South Korea | 10.4% |
| United Kingdom | 9.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
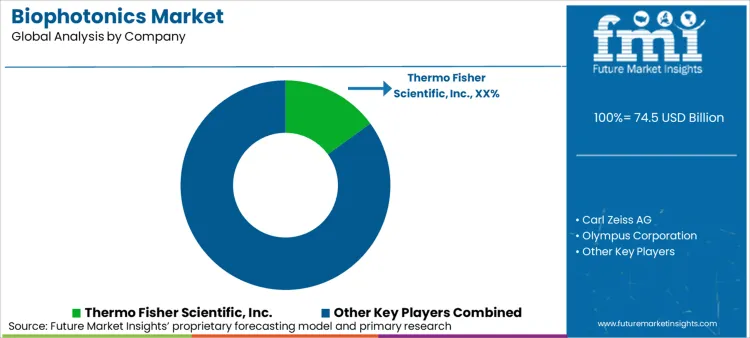
North America is characterized by research funding dominance and regulatory pathway maturity, where NIH-directed precision medicine investments and FDA's accelerated approval mechanisms for AI-integrated diagnostics establish the global benchmark for photonic technology commercialization. Key players in North America include Thermo Fisher Scientific, specializing in biosensors and fluorescence imaging for clinical diagnostics; Carl Zeiss and Olympus Corporation, leading in surgical visualization and confocal microscopy; and BD (Becton Dickinson), focusing on flow cytometry and molecular diagnostics.
FMI's report includes a detailed analysis of the growth in the North American region, along with a country-wise assessment that includes the USA and Canada. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the North America region.
Europe functions as the global center for publicly-funded photonics research infrastructure, where Horizon Europe programme allocations and national innovation initiatives drive bio-imaging technology development with emphasis on healthcare accessibility and environmental sustainability. Leading players in Europe include BASF and Zeiss maintaining R&D leadership in optical materials and precision imaging systems, Leica Microsystems advancing AI-driven biomedical optics, and Toptica Photonics specializing in high-resolution laser spectroscopy.
FMI's analysis of biophotonics market in Europe consists of country-wise assessment that includes Germany, Italy, France, UK, Spain, Russia, Nordic, Benelux and Rest of Europe. Readers can know various regulations and latest trends in the regional market.
Asia Pacific serves as the manufacturing powerhouse for photonic components and the fastest-growing market for clinical diagnostic adoption, leveraging semiconductor industry expertise and government healthcare modernization programmes to accelerate optical imaging deployment. ARCL Organics, Kanoria Chemicals and Huaqiang Chemical Group contribute to regional supply chains; however, Olympus Corporation, Hamamatsu Photonics, Sony, and Nikon dominate photonic technology development and biomedical imaging systems manufacturing.
The full report analyzes the biophotonics market across East and South Asia from 2026-2036, covering pricing, trends, and growth drivers in China, Japan, South Korea, India, Indonesia, Malaysia, and Thailand. The assessment highlights trends that dictate regional technology adoption and procurement strategies.
Market structure exhibits moderate concentration where top five photonic imaging and biosensing manufacturers command approximately 50 to 60 percent of global revenue, yet practical competition intensifies across specialized application segments. Roughly half of global capacity resides with vertically integrated optical component manufacturers possessing in-house laser development and AI algorithm capabilities, while remaining capacity operates through component assembly models with limited differentiation beyond distribution reach. Primary competitive variable is technology integration depth, specifically the ability to deliver AI-enhanced image reconstruction and regulatory-cleared diagnostic workflows, not standalone hardware performance.
Companies with captive photonic component fabrication and proprietary AI imaging algorithms sustain pricing power through platform lock-in effects, as clinical validation datasets and trained neural networks create switching costs for hospital systems. Suppliers without vertical integration depend on third-party optical components and generic image processing software, increasing exposure to commoditization pressure and procurement cycle volatility. Manufacturers with established FDA and EMA approval track records for photonic diagnostics compress time-to-market for next-generation platforms through regulatory pathway familiarity and existing quality management system infrastructure.
Customer concentration reinforces buyer leverage across hospital networks and centralized procurement authorities, yet willingness to pay premium pricing for AI-integrated, clinically validated systems sustains manufacturer margins. Large healthcare systems multi-source equipment to prevent vendor dependency where interoperability standards permit, limiting price pass-through, though proprietary imaging algorithms and clinical decision support integration create partial switching barriers that preserve installed base revenue through software licensing and maintenance contracts.
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
| Metric | Value |
|---|---|
| Quantitative Units | USD 74.5 billion (2026) to USD 209.9 billion (2036), at a CAGR of 10.9% |
| Market Definition | The biophotonics market comprises global production and deployment of light-based technologies for biological analysis, encompassing optical imaging systems, laser therapeutics, and biosensing platforms utilized for non-invasive diagnostics in oncology, ophthalmology, and neurology through photon interactions with biological tissues enabling molecular detection, structural visualization, and therapeutic intervention at cellular resolution. |
| Application Segmentation | See-Through Imaging, Microscopy, Inside Imaging, Spectro Molecular, Analytics Sensing, Light Therapy, Surface Imaging, Biosensors |
| Technology Segmentation | In-vivo, In-vitro |
| End User Segmentation | Tests and Components, Medical Therapeutics, Medical Diagnostics, Non-Medical Application |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East and Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia and 40 plus countries |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Carl Zeiss AG, Olympus Corporation, Hamamatsu Photonics K.K., BD (Becton Dickinson and Company), PerkinElmer Inc., Leica Microsystems, Toptica Photonics AG, Zecotek Photonics Inc., Lumenis Ltd., Bruker Corporation, BASF SE |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling validated through primary interviews with optical imaging system manufacturers and clinical diagnostics laboratories, supported by FDA approval databases and hospital procurement records |
Demand for Biophotonics in the global market is estimated to be valued at USD 74.5 billion in 2026.
Market size for Biophotonics is projected to reach USD 209.9 billion by 2036.
Demand for Biophotonics in the global market is expected to grow at a CAGR of 10.9% between 2026 and 2036.
See-through imaging and biosensors combined are expected to be the dominant applications, capturing approximately 60.0% of global market share in 2026 due to clinical diagnostic necessity and non-invasive detection capabilities.
In-vivo technology represents a critical segment, projected to hold approximately 55.0% share of the total market in 2026 as real-time surgical navigation and neuroscience research prioritize live-organism imaging capabilities.
Hospital adoption acceleration following FDA approval of photonic imaging devices, NIH precision medicine funding exceeding USD 2.8 billion annually, and defense sector biosensing investments are driving growth.
FDA medical device clearance pathways and NIH biomedical research grant requirements are referenced as key regulatory benchmarks.
The United States is projected to grow at a CAGR of 10.2% during 2026 to 2036.
Growth is supported by Horizon Europe programme allocations exceeding EUR 340 million for bio-imaging and national innovation initiatives across member states.
Publicly-funded research institution procurement and hospital adoption of optical diagnostics for personalized medicine dominate regional consumption.
European Union is projected to expand at a CAGR of 10.5% during 2026 to 2036.
Japan is included within East Asia under the regional scope of analysis.
Ministry of Health, Labour and Welfare healthcare technology assessment programmes and budget documentation covering precision medicine investments are cited as primary reference sources.
Asia Pacific demand is associated with semiconductor industry capabilities enabling photonic chip development and government healthcare modernization programmes driving clinical diagnostic adoption.
South Korea is included within East Asia under the regional coverage framework.
Electronics manufacturing capabilities supporting biosensing technology development and personalized medicine emphasis driving AI-powered optical imaging adoption are emphasized.
In-vivo imaging technologies are prioritized due to real-time surgical navigation capabilities and neuroscience research requirements across hospital settings.
Biophotonics encompasses light-based technologies primarily used for non-invasive biological analysis, molecular detection, and precision medical imaging in oncology, ophthalmology, and neurology applications.
Biophotonics market refers to global production, deployment, and clinical adoption of optical imaging systems, laser therapeutics, and biosensing platforms in medical and non-medical applications.
Scope covers biophotonics technologies by application, key platforms such as see-through imaging and biosensors, and end-use industries including medical therapeutics and diagnostics.
Non-optical diagnostic modalities, downstream clinical services, and basic research photonics without commercial diagnostic pathways are excluded unless part of integrated photonic systems.
Market forecast represents a model-based projection built on defined assumptions for strategic planning purposes.
Forecast is developed using hybrid top-down and bottom-up modeling validated through hospital procurement records, FDA approval databases, and manufacturer capacity disclosures.
Primary interviews with clinical laboratories and verifiable regulatory databases are used instead of unverified syndicated market estimates.
Our Research Products

The "Full Research Suite" delivers actionable market intel, deep dives on markets or technologies, so clients act faster, cut risk, and unlock growth.

The Leaderboard benchmarks and ranks top vendors, classifying them as Established Leaders, Leading Challengers, or Disruptors & Challengers.

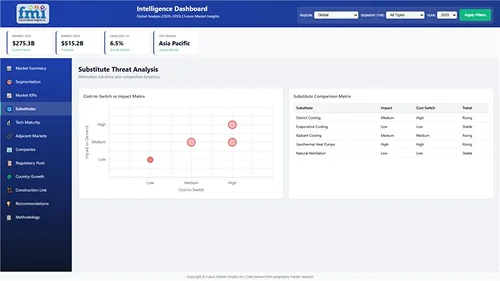
Locates where complements amplify value and substitutes erode it, forecasting net impact by horizon

We deliver granular, decision-grade intel: market sizing, 5-year forecasts, pricing, adoption, usage, revenue, and operational KPIs—plus competitor tracking, regulation, and value chains—across 60 countries broadly.

Spot the shifts before they hit your P&L. We track inflection points, adoption curves, pricing moves, and ecosystem plays to show where demand is heading, why it is changing, and what to do next across high-growth markets and disruptive tech

Real-time reads of user behavior. We track shifting priorities, perceptions of today’s and next-gen services, and provider experience, then pace how fast tech moves from trial to adoption, blending buyer, consumer, and channel inputs with social signals (#WhySwitch, #UX).
Partner with our analyst team to build a custom report designed around your business priorities. From analysing market trends to assessing competitors or crafting bespoke datasets, we tailor insights to your needs.
Supplier Intelligence
Discovery & Profiling
Capacity & Footprint
Performance & Risk
Compliance & Governance
Commercial Readiness
Who Supplies Whom
Scorecards & Shortlists
Playbooks & Docs
Category Intelligence
Definition & Scope
Demand & Use Cases
Cost Drivers
Market Structure
Supply Chain Map
Trade & Policy
Operating Norms
Deliverables
Buyer Intelligence
Account Basics
Spend & Scope
Procurement Model
Vendor Requirements
Terms & Policies
Entry Strategy
Pain Points & Triggers
Outputs
Pricing Analysis
Benchmarks
Trends
Should-Cost
Indexation
Landed Cost
Commercial Terms
Deliverables
Brand Analysis
Positioning & Value Prop
Share & Presence
Customer Evidence
Go-to-Market
Digital & Reputation
Compliance & Trust
KPIs & Gaps
Outputs
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.