Blister Packaging Market
Blister Packaging Market The blister packaging market is segmented by Product Type (Thermoformed, Cold-Form, Others), Application (Pharmaceuticals, Medical Devices, Consumer Healthcare, Food & Confectionery, Electronics, Others), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Blister Packaging Market Size, Market Forecast and Outlook By FMI
The blister packaging market is expected to expand from USD 30.3 billion in 2026 to USD 52.3 billion by 2036. The market is anticipated to register a 5.6% CAGR during the forecast period. Thermoformed formats are likely to lead product type with a 64.0% share, while pharmaceuticals are projected to account for 52.0% share of application revenue in 2026.
Summary of the Blister Packaging Market
- Demand and Growth Drivers
- Prescription-drug traceability requirements are expected to support blister packaging use across regulated pharmaceutical supply chains.
- Circularity rules are likely to move material redesign into routine packaging procurement decisions.
- Package-level coding needs are anticipated to increase investment in print quality and inspection-ready blister formats.
- Product and Segment View
- Thermoformed formats are expected to lead the product type segment due to high-speed line compatibility.
- Pharmaceuticals are likely to remain the main application area because patient-safety routines depend on traceable unit packs.
- Cold-form and specialty blister formats are anticipated to support categories where barrier performance remains a priority.
- Geography and Competitive Outlook
- India is expected to lead country-level growth due to QR-code compliance and plastic packaging enforcement.
- China is likely to record strong expansion as reimbursement-linked traceability increases packaging-line control requirements.
- Companies with stronger validation systems and recyclable material roadmaps are anticipated to improve their competitive position.
- Analyst Opinion
- Ismail Sutaria, Principal consultant for Packaging at FMI, suggests, “The blister packaging market is likely to shift toward suppliers that can combine compliance discipline with material redesign. Pharma-facing converters are expected to gain advantage when they offer validated print quality and audit-ready documentation.”
- Blister Packaging Market Value Analysis
- The blister packaging market is moving from a standard pack-format category into a compliance-linked packaging system.
- Demand is likely to rise as pharmaceutical traceability makes code readability a routine requirement.
- Use is expected to strengthen where thermoformed films support high-speed filling and stable inspection performance.
- Spending is anticipated to be backed by circularity rules and stronger documentation needs across regulated packaging lines.
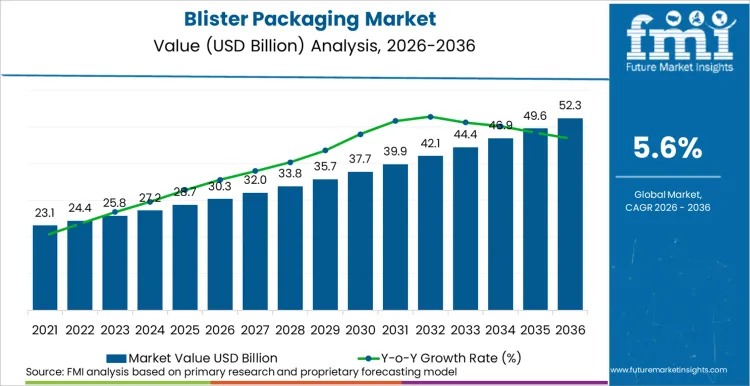
How are Packaging Policy and Compliance Frameworks Shaping Blister Packaging Scale Economics?
| Region/Program Cluster | Scale Drivers for 2026 |
|---|---|
| Asia Scale Programs |
|
| North America and Europe |
|
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Blister Packaging Market Key Metrics
| Metric | Value |
|---|---|
| Industry Size (2026E) | USD 30.3 billion |
| Industry Value (2036F) | USD 52.3 billion |
| CAGR (2026 to 2036) | 5.6% |
Investment logic also overlaps with procurement routines associated with the global pharmaceutical packaging market and the global unit dose packaging market.
Why is the Global Blister Packaging Expanding?
Prescription-drug traceability requirements are translating into packaging-line modernization that benefits unit-dose formats. FDA DSCSA compliance policies established a stabilization period through November 27, 2024, creating a bounded window for trading partners to harden processes and systems for interoperable, electronic tracing at package level. That timetable pulls demand toward blisters that accommodate consistent machine-readable identifiers, stable lidding print performance, and verification-friendly geometry that maintains readability through high-speed cartoning and distribution handling.
Sustainability programs now translate into measurable cost and compliance mechanics. EU PPWR entered into force in February 2025 and applies generally from August 2026, adding a single rulebook that strengthens incentives to reduce hard-to-recycle combinations across EU supply chains. In UK, Plastic Packaging Tax sets per-tonne rates that increase over time, reinforcing renewed scrutiny on recycled-content evidence, component declarations, and defensible exemptions where medical compliance limits options. Execution also mirrors modernization programs seen across healthcare packaging and child resistant packaging.
How is the Blister Packaging Market Segmented?
Why are High-Throughput Pharma Lines and Recyclable Film Roadmaps reinforcing Thermoformed leadership?
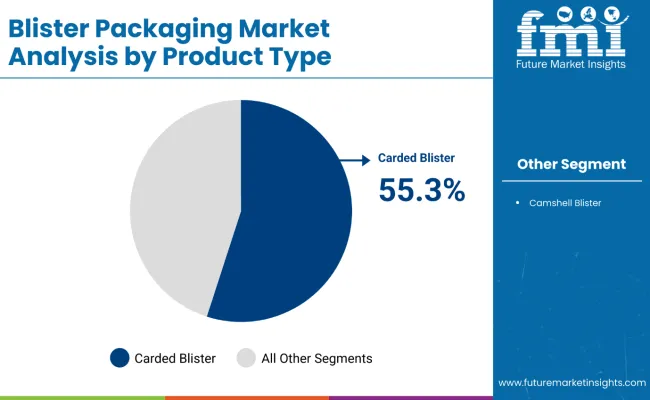
Thermoformed formats hold a 64.0% share because high-cavity, high-speed lines in solid oral dose production rely on predictable forming windows and wide material availability across regional converter bases. Supplier roadmaps increasingly position thermoform films as a pathway to lower-impact structures while protecting machinability and inspection performance.
Klöckner Pentaplast launched kpNext as a recyclable packaging film for medical device packaging, signalling continued investment in recyclability-aligned film platforms that can be qualified into regulated packaging workflows where documentation, change control, and performance evidence matter as much as run speed
Material choices also reflect a rising preference for traceable inputs and documented circularity claims. Tekni-Plex has highlighted circular polymers for pharma-grade packaging applications, reinforcing how resin and film suppliers are building post-use or advanced-recycled content pathways that can still support regulated packaging documentation expectations. This keeps thermoformed platforms structurally advantaged when converters need a scalable substrate family that can absorb specification changes without destabilizing line capability.
Why does Pharmaceuticals anchor a Substantial Application Share through Traceability and Safety Routines?
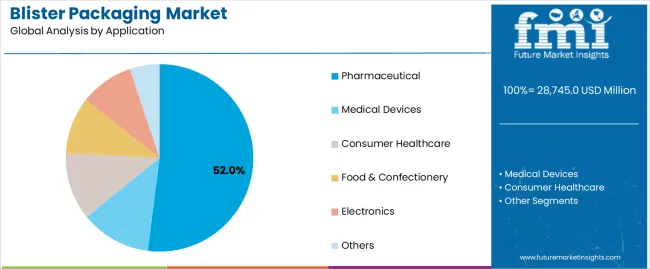
Pharmaceuticals account for a 52.0% share because traceability and patient-safety controls increasingly bind packaging choices to downstream scanning and verification workflows. In India, government updates describe implementation of barcode or QR code requirements for major drug brands, elevating importance of code placement, print permanence, and verification routines across packs used in retail and institutional dispensing. That policy stance changes value perception of blister lidding and carton surfaces, favouring structures that can deliver consistently scannable codes at industrial speeds.
China adds an additional operational lever through reimbursement-linked traceability. NHSA notices set staged collection of drug traceability codes with full collection from January 1, 2026, increasing demand for packaging and inspection systems that sustain high read rates under production and logistics stress. As traceability becomes embedded in transaction controls, packaging formats that simplify code placement and inspection throughput retain preference in pharmaceutical primary packs.
What are the Key Blister Packaging Market Dynamics?
Compliance digitization is turning print and inspection performance into procurement criteria. DSCSA timelines in USA and reimbursement-linked traceability code programs in China convert policy into measurable penalties for unreadable identifiers, missing codes, or weak transaction integrity, shifting vendor selection toward suppliers that validate print quality, maintain process capability, and document change control with regulator-ready discipline. This elevates value of packaging partners that can prove code quality under production variability and demonstrate disciplined supplier qualification across films, foils, inks, and coatings.
Circularity programs are simultaneously tightening material roadmaps. EU PPWR, India plastic waste amendments, and UK tax plus EPR reporting steps signal policy intent to reduce waste, improve recyclability, and strengthen documentation across packaging life cycles, elevating redesign programs that simplify structures and support verified declarations. Roadmaps often align with decision structures associated with sustainable packaging and flexible packaging, especially where brand owners need auditable claims discipline alongside mechanical and barrier performance.
How do Key Countries exhibit Promising Trends in the Blister Packaging Landscape?
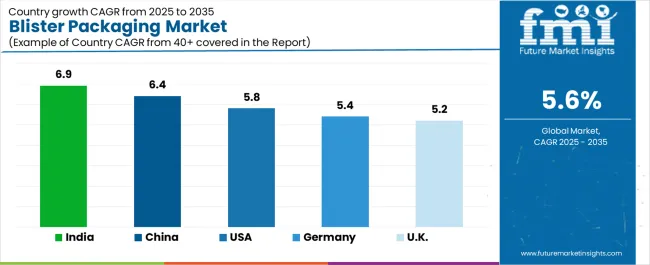
| Country | CAGR (2026-2036) |
|---|---|
| India | 7.1% |
| China | 6.6% |
| USA | 6.0% |
| Germany | 5.6% |
| UK | 5.4% |
How is India’s QR-code Compliance and Plastic Packaging Enforcement shaping Blister Pack Redesign Discipline?
Blister packaging expansion in India is projected at a 7.1% CAGR, supported by on-pack identification expectations for medicines and stronger governance of plastic packaging waste. Government updates describe requirements to print or affix barcodes or QR codes on packaging labels for leading drug brands, increasing need for robust code placement, print permanence, and verification steps on packs that travel through retail and institutional channels. This pushes brand owners toward packaging specifications that reduce code failure rates and support inspection workflows without lowering line productivity.
Plastic Waste Management (Amendment) Rules, 2024 add enforcement and reporting structure that supports EPR execution, including separate waste-stream models that can be used to fulfil EPR obligations. That governance strengthens pressure to document material choices, channelization routes, and compliance evidence for packaging waste. Suppliers that provide clear technical files and documented recyclability positioning gain advantage when procurement teams require compliance-ready packaging specifications, not only price-based quotes.
Will China’s Reimbursement-linked Traceability and GMP Reinforcement tighten Packaging Material Controls?
China is expected to advance at a 6.6% CAGR, reinforced by policy that makes traceability codes operationally central in reimbursement and distribution control. NHSA notices set staged collection of drug traceability codes with full collection from January 1, 2026, increasing demand for packaging lines that can print, verify, and sustain high read rates at speed, including blister lidding and secondary packaging. This shifts investment toward inspection, rejection handling, and data capture systems that protect code integrity across high-volume SKUs.
Regulatory tightening also reaches upstream packaging materials. NMPA issued a GMP annex for pharmaceutical packaging materials and containers in 2025, reinforcing process control and documentation expectations for packaging material manufacturers. That raises qualification thresholds for films and foils used in blisters and increases value of suppliers able to demonstrate controlled manufacturing, validated performance, and audit-ready documentation.
How is DSCSA readiness along with Recyclable Blister Innovation Shaping Procurement Criteria in USA?
Blister packaging expansion in USA is forecast at a 6.0% CAGR, driven by traceability execution and sustainability-linked redesign programs that remain compatible with regulated supply chains. FDA DSCSA compliance policies established a stabilization period through November 27, 2024, enabling trading partners to implement and mature interoperable electronic systems for package-level tracing of prescription drugs. This elevates demand for packaging solutions that support reliable identification and verification, including consistent print performance and disciplined change control.
Material roadmaps are also becoming more visible in commercially positioned offerings. Amcor reported a commercial order for AmSky blister system in 2025, positioning recyclable blister packaging for healthcare applications and signalling that qualification is moving beyond pilots into scaled procurement. Suppliers that can pair traceability-ready execution with defensible sustainability documentation gain leverage in sourcing cycles that increasingly ask for both compliance evidence and circularity narratives.
Why does Recyclability Governance shape Film Selection in Regulated Primary Packaging in Germany?
The blister packaging market in Germany is projected to grow at a 5.6% CAGR, shaped by design-for-recycling governance and EU harmonization that affects packaging design choices. Central Agency Packaging Register publishes a minimum standard that defines design-for-recycling criteria used by dual systems, and 2024 updates reinforce how material combinations are evaluated for recyclability. That governance creates commercial pressure to redesign blister components that underperform in recyclability assessments, especially within consumer healthcare portfolios where alternatives can be qualified.
EU PPWR entered into force in 2025 and applies generally from August 2026, adding a common compliance baseline that accelerates portfolio standardization across EU supply chains. Suppliers that provide documentation mapping to German recyclability evaluation frameworks can reduce qualification friction for brand owners balancing barrier needs against circularity constraints.
How will Cost Signalling through Packaging Taxes and EPR reporting reshape Blister Packaging Economics in the UK?
UK blister packaging is expected to expand at a 5.4% CAGR, influenced by cost-signalling mechanisms that convert sustainability into direct financial incentives. HMRC Plastic Packaging Tax sets per-tonne rates that increase over time, reinforcing evaluation of recycled-content feasibility and evidence requirements, especially for non-pharma blisters serving consumer healthcare, confectionery, and electronics where alternative substrates can be qualified.
Packaging EPR also adds reporting and fee foundations. UK government guidance requires obligated organisations to collect and report packaging data for 2024, with reporting deadlines in 2025, increasing attention to component-level material declarations and end-of-life classification. Blister programs therefore require tighter data governance around components, suppliers, and material identities to avoid reporting errors and fee leakage, particularly where multi-material combinations complicate classification.
Blister Packaging Market Competitive Landscape

Competitive differentiation is consolidating around compliance-ready manufacturing discipline and defensible sustainability transitions. DSCSA, reimbursement-linked traceability code programs in China, and EU PPWR timelines convert policy into operational requirements that affect artwork control, print verification, material qualification, and supplier documentation. Suppliers that provide validated change control, code readability assurance, and audit-ready technical files are increasingly preferred for regulated primary packaging and for adjacent consumer healthcare categories.
Innovation is becoming more visible in commercially positioned offerings that target barrier-led segments. Constantia Flexibles positioned REGULA CIRCULAR as a recyclable cold-form foil solution, addressing applications where barrier needs historically constrained redesign options. Amcor’s AmSky order in 2025 signals adoption movement for recyclable blister systems in healthcare. These roadmaps parallel pathways tracked in aluminum foil packaging, especially where foil performance and circularity requirements must be reconciled through documented, regulator-ready execution.
Blister Packaging Market Key Players
- Amcor plc
- Constantia Flexibles
- Klöckner Pentaplast
- Tekni-Plex Inc.
- Bemis Company Inc., now part of Amcor plc
- Bilcare Limited
- Honeywell International Inc.
- Sonoco Products Company
- Westrock Company
- Winpak Ltd.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Product Type | Thermoformed; Cold-Form; Others |
| Application | Pharmaceuticals; Medical Devices; Consumer Healthcare; Food & Confectionery; Electronics; Others |
| Region | North America; Latin America; Western Europe; Eastern Europe; East Asia; South Asia & Pacific; Middle East & Africa |
Blister Packaging Market Segmentation
By Product Type
- Thermoformed
- Cold-Form
- Others
By Application
- Pharmaceuticals
- Medical Devices
- Consumer Healthcare
- Food & Confectionery
- Electronics
- Others
By Region
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia & Pacific
- Middle East & Africa
Bibliography
- Central Agency Packaging Register. (2024). Minimum standard for packaging design for recycling.
- European Union. (2025). Regulation (EU) 2025/40 of the European Parliament and of the Council on packaging and packaging waste.
- Ministry of Environment, Forest and Climate Change, Government of India. (2024). Plastic Waste Management (Amendment) Rules, 2024.
- National Healthcare Security Administration, People’s Republic of China. (2025). Notice on the collection of drug traceability codes.
- National Medical Products Administration, People’s Republic of China. (2025). Annex: Good Manufacturing Practice for pharmaceutical packaging materials and containers.
- Press Information Bureau, Government of India. (2025). Steps taken to prevent sale of adulterated drugs.
- U.S. Food and Drug Administration. (2023). DSCSA compliance policies establish 1-year stabilization period for implementing electronic systems.
Frequently Asked Questions
How Opportunistic are Growth Prospects for Global Blister Packaging Market in 2026?
In 2026, blister packaging industry value is projected to attain USD 30.3 billion.
How much value will Global Blister Packaging Market generate by 2036?
Blister packaging industry is anticipated to total USD 52.3 billion by 2036.
How much will the Global Blister Packaging Market rise from 2026 to 2036?
The blister packaging market is likely to advance at a 5.6% CAGR from 2026 to 2036.
Which Product Category is expected to capture largest share of Global Blister Packaging industry?
Thermoformed blister packs dominate by product type, expected to capture 64.0% of total revenues in 2026.
Where will Blister Packaging Solutions find Maximum Deployment in terms of Application?
According to FMI, the blister packaging market shows incredible promise in the pharmaceutical industry, generating 52% revenue in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Roots Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended A8oach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Diagnostics
- Monitoring
- Therapeutic
- Consumer Products
- Diagnostics
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- General Health and Wellness
- Maternity Care
- Menstrual Health
- Pelvic and Urine Healthcare
- Cancer
- Chronic Disease
- General Health and Wellness
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Direct to Consumer
- Hospitals
- Surgical Centers
- Fertility Clinics
- Diagnostic Centers
- Direct to Consumer
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Flo Health Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Clue by BioWink GmbH
- Natural Cycles USA Corp.
- Elvie (Chiaro Technology Ltd.)
- Willow Innovations, Inc.
- Others
- Flo Health Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Product Type
- Figure 26: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Application
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Product Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Application
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Application
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Application
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Product Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Application
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

