The flow cytometry market is projected to reach USD 7.3 billion by 2026 and expand to USD 15.9 billion by 2036, reflecting an 8.1% CAGR. Growth formation is increasingly tied to whether laboratories can translate higher-dimensional assays into controlled, decision-grade workflows. Demand is clustering around environments where assay complexity is matched with reproducibility controls, rather than simply higher parameter counts.
FMI opines that value creation is now driven by workflow completeness rather than instrument placement alone. Buyers are prioritizing vendors that can integrate panel design discipline, reagent consistency, instrument stability, and governed analysis pipelines into a single operating routine. In such settings, recurring consumption of panels, controls, and calibration materials expands faster than capital replacement cycles.
Two operational shifts are changing how flow cytometry demand converts into spend. First, high-parameter immunophenotyping is moving from expert-built panels to repeatable panel families that can be deployed across cohorts with lower failure risk.
This transition is raising the value of reagent lot discipline, compensation controls, and standardized QC routines because the penalty for drift increases sharply when programs track subtle population shifts over long timelines. End use customers are therefore selecting suppliers who can support panel continuity, consistent fluorochrome performance, stable antibody availability, and documented performance characteristics that preserve comparability across multi-quarter studies.
Second, analytics is becoming a throughput constraint rather than the instrument itself. Many sites now generate more events and channels than their analyst capacity can reliably gate with consistency, which pushes demand toward governed analysis templates, audit-ready reporting, and services that reduce operator-to-operator variance.
This is especially visible in multi-center studies and translational pipelines where cross-site comparability is a prerequisite for decision-making. Spending is widening beyond instruments into software, method standardization, and workflow services that keep interpretation stable as assay complexity increases.
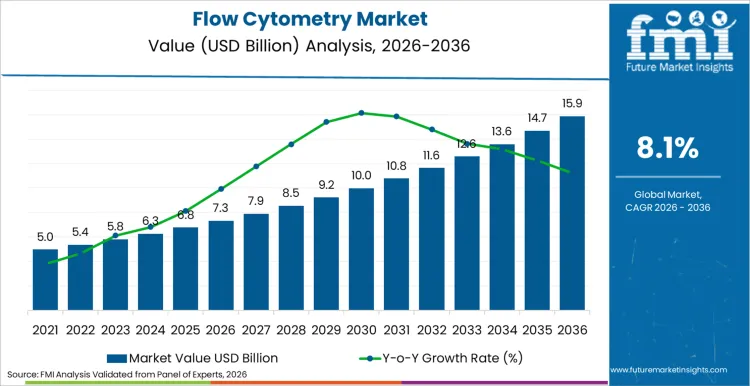
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 7.3 Billion |
| Industry Value (2036) | USD 15.9 Billion |
| CAGR (2026-2036) | 8.1% |
Source: Future Market Insights (FMI)
Expansion is being reinforced by the transition from single-readout assays to multi-parameter cellular decisioning, where laboratories require faster resolution on phenotype, function, and response signatures. This shift increases reliance on standardized reagents, stable panels, and repeatable analytical templates that preserve longitudinal comparability. Execution is often aligned with decision structures associated with single-cell analysis, particularly where high-dimensional profiling must be scaled across cohorts without inflating technical noise.
Portfolio planning is frequently evaluated alongside frameworks associated with immunology research, especially where panel families become reusable assets across discovery and translational pipelines. Renovation cycles are also being coordinated with laboratory strategies linked to clinical diagnostics, particularly where workflow governance determines whether cytometry outputs can support regulated or quasi-regulated decision paths.
Product type, application, usage, and end user segment the flow cytometry market. Product type includes reagents, consumables & accessories; flow cytometer systems; software; and services. Applications include immunophenotyping, cell sorting, cell cycle analysis, apoptosis, cell proliferation assays, intracellular calcium flux, detection & sorting of exosomes, and others. Usage is split between research use and clinical diagnostics. End users include academic and research institutes, diagnostic laboratories, hospitals, and biopharmaceutical companies.
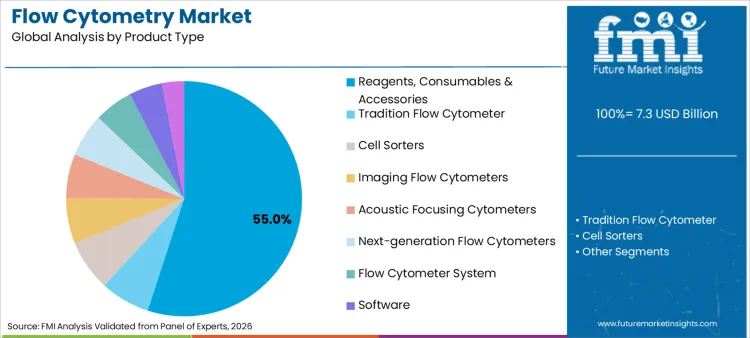
Reagents, consumables & accessories hold a 55.0% share because cytometry workflows are panel-driven and repeat-dependent. Every incremental cohort and timepoint multiplies recurring reagent demand. This dominance is reinforced where assay design is coordinated with development structures associated with biomarker discovery, particularly where validated panels are reused across multi-phase programs.
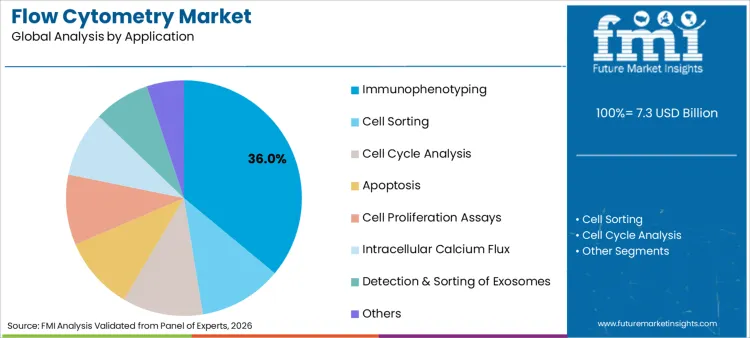
Immunophenotyping leads with a 36.0% share because it anchors both discovery and translational profiling workflows. Demand is sustained where program design is aligned with application stacks associated with oncology diagnostics, especially as immune profiling increasingly informs response stratification and study gating.
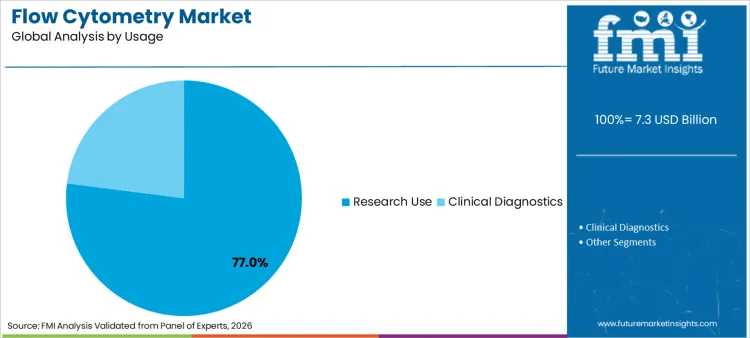
Research use leads with 77.0% share as the installed base of academic and translational labs continues to widen assay dimensionality. This is reinforced where throughput planning is coordinated with operating models associated with laboratory automation, particularly where standardized sample prep and template-based analysis reduce marginal labor per sample.
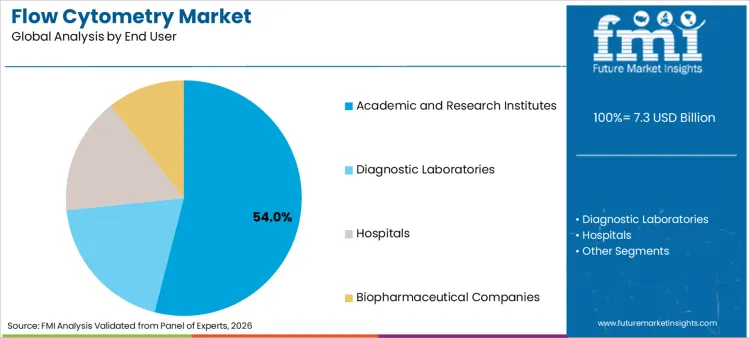
Academic and research institutes lead with 54.0% share due to continued expansion of shared core facilities and multi-parameter study designs. Utilization planning is increasingly aligned with governance structures associated with biobanking, particularly where sample scarcity makes high-information assays economically rational.
A primary driver is the rising standardization premium in high-parameter assays. As panels become more complex, laboratories increasingly pay for confidence: validated reagent lots, stability claims that reduce drift, and controls that preserve comparability across long studies. This elevates recurring demand for QC kits, validation services, and training, shifting purchasing behavior from ad-hoc replenishment to programmatic contracts. A second driver is the operationalization of sorting as upstream infrastructure for advanced pipelines.
Cell sorting is increasingly treated as a gating step that determines downstream success rates. This raises expectations around recovery efficiency, purity stability, and gentle handling of sensitive populations, creating a clear path for premiumization in instruments and service contracts. On the opportunity side, analysis governance is becoming decisive. Demand is moving toward rule-governed pipelines that reduce subjective gating drift and support cross-site reproducibility. New growth pockets are emerging around rare-event detection and exosome workflows, where background control, pre-analytical discipline, and application-specific kits become differentiators. (Approx. 200 words)
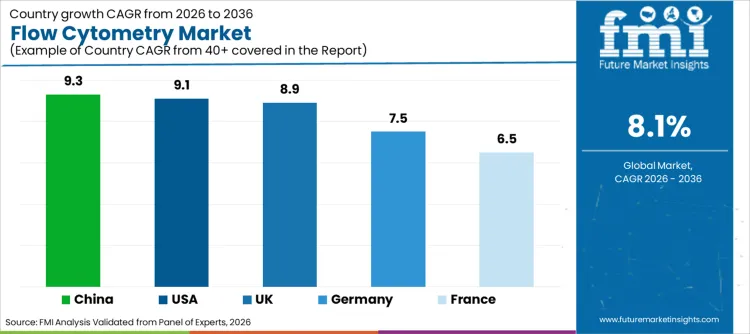
| Country | CAGR (2026-2036) |
|---|---|
| USA | 9.1% |
| Germany | 7.5% |
| UK | 8.9% |
| China | 9.3% |
| France | 6.5% |
Source: Future Market Insights (FMI)
The USA is projected to expand at a 9.1% CAGR, driven in part by NIH-funded multi-site translational programs that require harmonized flow cytometry protocols across academic medical centers. Initiatives under the NIH Human Immunology Project Consortium and Cancer Moonshot are enforcing standardized antibody panels and instrument performance tracking across networks, directly lifting demand for calibrated mid- to high-parameter analyzers and certified QC consumables. In parallel, leading core facilities are consolidating around a limited set of vendor platforms, notably BD and Thermo Fisher, to simplify cross-site data comparability. This is increasing long-term service contracts, shared reagent sourcing, and standardized software licensing, anchoring growth more in installed-base expansion and workflow compliance than in greenfield instrument purchases.
Germany is expected to grow at a 7.5% CAGR, strongly shaped by GMP enforcement under EU Annex 1 and PEI (Paul-Ehrlich-Institut) guidance for cell and gene therapy manufacturing. These frameworks are increasing the use of validated flow cytometry for in-process control and release testing, particularly for purity and viability assessment in sorting-intensive workflows. At the same time, Fraunhofer-linked translational centers are expanding high-speed cell sorting capacity for immunotherapy and regenerative medicine programs. This is driving selective demand for high-end sorters with documented performance tracking, while routine analyzers are procured with pre-qualified calibration and compliance packages.
The UK is projected to expand at an 8.9% CAGR, supported by the rapid scaling of cell therapy manufacturing under MHRA-regulated Advanced Therapy Medicinal Product (ATMP) frameworks. Flow cytometry is increasingly embedded in release-adjacent testing for CAR-T and stem cell products, tightening requirements around instrument qualification and controlled antibody sourcing. In parallel, NHS-linked translational labs are facing a shortage of experienced cytometrists, accelerating adoption of automated compensation, fixed gating templates, and vendor-supported analysis pipelines. This is shifting spending toward software upgrades and validated analysis modules rather than frequent hardware replacement.
The market for flow cytometry in China is projected to advance at a 9.3% CAGR, driven by national biopharma capacity programs under the 14th Five-Year Plan, which mandate expansion of immunology and oncology testing platforms in tertiary hospitals. Large-scale procurement programs are favoring standardized mid-range flow cytometers bundled with training and service, rapidly expanding the installed base. At the same time, domestic players such as Mindray and Accuri-aligned distributors are gaining share in public tenders by offering pre-configured immunophenotyping packages. This is anchoring growth in volume-driven analyzer deployments and recurring reagent pull-through rather than in high-end custom systems.
France is expected to grow at a 6.5% CAGR, shaped by the central role of flow cytometry in INSERM-led immunology networks and hospital immunophenotyping platforms. National reference laboratories are standardizing leukemia and lymphoma panels, reinforcing demand for fixed clinical configurations rather than exploratory multi-omics cytometry. Meanwhile, budget discipline within public hospitals is extending instrument replacement cycles, with upgrades prioritized only when optical stability or laser compatibility limits panel continuity. As a result, growth is structurally weighted toward antibodies, QC reagents, and software compliance updates rather than accelerated analyzer turnover.
Competition is increasingly defined by who can deliver an end-to-end cytometry operating system: stable instruments, validated reagents, reproducible software, and service architectures that preserve multi-parameter consistency over time. Instrument vendors are differentiating through usability and stability improvements that reduce operator-driven variance. Reagent portfolios are becoming programmatic, with panel families and controls designed to reduce failure risk as marker counts rise. Software is moving from optional to decisive as labs demand templates, traceability, and reproducibility safeguards.
| Strategy | What it changes in outreach and conversion |
|---|---|
| Build panel-first commercial bundles by application | Converts procurement into a workflow decision; increases recurring pull-through of reagents and controls. |
| Offer audit-ready analysis templates | Improves multi-site adoption and reduces buyer fear of analyst drift. |
| Create performance-stability service guarantees | Shifts justification from capex to avoided failures in high-value cohorts. |
| Standardize sorting assurance packages | Enables scale-up of purity-sensitive workflows without quality collapse. |
| Formalize application-specific onboarding | Reduces ramp time and sustains consistent utilization across sites. |
| Items | Values |
| Quantitative Units | USD Billion |
| Product Type | Reagents, Consumables & Accessories; Flow Cytometer System; Software; Services |
| Application | Immunophenotyping; Cell Sorting; Cell Cycle Analysis; Apoptosis; Cell Proliferation Assays; Intracellular Calcium Flux; Detection & Sorting of Exosomes; Others |
| Usage | Research Use; Clinical Diagnostics |
| End User | Academic and Research Institutes; Diagnostic Laboratories; Hospitals; Biopharmaceutical Companies |
| Regions | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East & Africa |
What is the Outlook for the Flow Cytometry Market in 2026?
The flow cytometry market is projected to total USD 7.3 billion in 2026.
What is the forecast for the Flow Cytometry Market in 2036?
By 2036, flow cytometry demand is expected to reach USD 15.9 billion.
Which Product Type is expected to lead in 2026 in the Global Flow Cytometry Market?
Reagents, consumables & accessories are expected to lead with a 55.0% share in 2026.
As of 2026, which Application Category is expected to Dominate in the Flow Cytometry Landscape?
Immunophenotyping applications are expected to lead with a 36.0% share in 2026 in the flow cytometry landscape.
By Usage Category, Which Segment shows Most Promising Prospects in the Market for Flow Cytometry?
Research use demonstrates significant prospects for flow cytometry, expected to capture a 77.0% share in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.