Drug Discovery Services Market
Drug Discovery Services Market Forecast and Outlook 2026 to 2036
Drug Discovery Services Market Size and Share Forecast Outlook 2026 to 2036
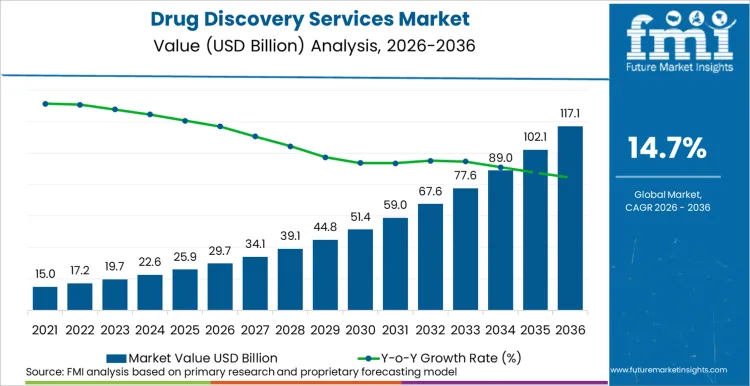
The global drug discovery services sector is on track to achieve a valuation of USD 117,163.1 Million by 2036, accelerating from USD 29,727.4 Million in 2026 at a CAGR of 14.7%. As per Future Market Insights, expansion is structurally underpinned by the pharmaceutical industry's accelerating shift toward externalized R&D models as internal discovery productivity declines despite rising expenditure. The USA Food and Drug Administration approved 55 novel drugs in 2024, and an increasing proportion of these approvals originated from programs that utilized outsourced discovery services, validating the CRO model's contribution to pipeline success. This approval momentum compels both large pharmaceutical companies and emerging biotech firms to deepen their outsourcing relationships with specialized discovery service providers who can offer integrated platforms spanning target identification, hit-to-lead optimization, and IND-enabling studies. Simultaneously the technology stack is shifting from traditional wet-lab-only workflows to hybrid AI-augmented discovery pipelines which forces service providers to invest in computational chemistry, machine learning, and high-dimensional experimentation capabilities.
Dr. Rob Howes, Senior Director at Charles River Laboratories, stated: 'Synthace allows us to conduct more technically demanding experiments with greater scientific rigour, reducing the amount of time we need to develop assays.' This statement, made in February 2026 during the announcement of a partnership between Charles River and Synthace for automated assay development through High Dimensional Experimentation, confirms that leading CROs are integrating software automation into their core service delivery.
FMI is of the opinion that the market is entering a consolidation and capability-stacking phase. IQVIA and Flagship Pioneering launched a strategic collaboration in August 2025 to accelerate the development of breakthrough biopharma companies within the Flagship ecosystem, granting portfolio companies access to IQVIA's AI-powered clinical research services. Separately, Pierre Fabre Laboratories and Iktos announced a strategic collaboration in January 2026 to utilize Iktos' generative AI and automated chemistry robotics for novel small-molecule oncology candidate discovery. FMI analysts note that these partnerships confirm the transition from transactional fee-for-service CRO relationships toward integrated, milestone-linked discovery alliances where technology providers share risk and reward.
Key Takeaways
Power in the drug discovery services value chain is concentrated among large integrated CROs such as Charles River Laboratories, WuXi AppTec, Eurofins Discovery, and Evotec. These firms control proprietary compound libraries, validated assay platforms, and regulatory filing expertise that create high switching costs for pharmaceutical clients. FMI analysts note that WuXi AppTec's passage of FDA on-site inspections at its Changzhou and Taixing sites with no observations in March 2025 demonstrates the compliance standards that entrench market leaders.
Pricing asymmetry in drug discovery services is driven by the gap between commoditized screening services, where competition has compressed margins, and high-value, integrated discovery platforms that combine AI target identification, medicinal chemistry optimization, and ADMET profiling. As per FMI, providers who offer end-to-end discovery solutions from target validation to IND filing command 30% to 50% higher contract values than providers offering isolated services.
Sourcing fragility in this market relates to the geographic concentration of key capabilities. China-based CROs, led by WuXi AppTec and Pharmaron, handle a substantial share of global medicinal chemistry and API synthesis. Geopolitical tensions, export controls, and proposed legislative restrictions (such as the U.S. BIOSECURE Act discussions) introduce supply chain risks for pharmaceutical companies that have centralized discovery outsourcing in China.
Brand and compliance risks are rising as regulatory authorities intensify scrutiny of data integrity, GLP compliance, and patient safety in outsourced discovery programs. The FDA's updated cybersecurity guidance for medical devices (February 2026) and the European Medicines Agency's data governance requirements create compliance burdens that underscore the importance of auditable, validated service delivery.
Geographic leverage in drug discovery services is anchored in three zones. North America is the largest market by value, driven by proximity to pharmaceutical headquarters and regulatory agencies. Europe retains strength in biologics discovery and academic translational partnerships. Asia Pacific, led by China and India, offers cost-competitive chemistry and manufacturing services, with Syngene International integrating its first U.S. site in Baltimore in December 2025 to serve as a bridge between Asian manufacturing and American regulatory requirements.
FMI opines that the executive-level reframing for this market is a transition from 'outsourcing to reduce cost' to 'outsourcing to access capability.' The USD 117,163.1 Million opportunity by 2036 is accessible to service providers who combine AI-augmented discovery platforms, regulatory-grade compliance infrastructure, and flexible commercial models that align incentives between CRO and pharmaceutical client.
Summary of Drug Discovery Services Market
What Is Growth Outlook for Drug Discovery Services Market as per Future Market Insights Projection?
Future Market Insights projects the drug discovery services market to expand at a CAGR of 14.7% from 2026 to 2036, increasing from USD 29,727.4 Million in 2026 to USD 117,163.1 Million by 2036.
FMI Research Approach: FMI proprietary forecasting model based on pharmaceutical R&D expenditure data, FDA novel drug approval rates, and CRO revenue disclosures.
How Do FMI Analysts Perceive Drug Discovery Services Market to Evolve?
FMI analysts perceive the market evolving toward integrated, AI-augmented discovery platforms where CROs function as technology partners rather than transactional service vendors, sharing milestone-linked risk and reward with pharmaceutical clients.
FMI Research Approach: Partnership tracking of IQVIA and Flagship Pioneering collaboration and Charles River and Synthace assay automation alliance.
Which Country Holds Largest Share in Global Drug Discovery Services Market?
The United States holds a significant share of the global drug discovery services market by value which is supported by the concentration of pharmaceutical company headquarters, FDA regulatory proximity, and venture-funded biotech firms.
FMI Research Approach: FMI country-level revenue modeling by CRO revenue disclosures and pharmaceutical outsourcing spend analysis.
How Large Will Drug Discovery Services Market Be by 2036?
The global drug discovery services market is projected to reach USD 117,163.1 Million by 2036.
FMI Research Approach: FMI long-term revenue forecast derived from pharmaceutical industry R&D spending projections and outsourcing penetration rate modeling.
What Is Definition of Drug Discovery Services Market?
The drug discovery services market comprises revenue generated from outsourced research services that support the identification, validation, optimization, and preclinical development of drug candidates on behalf of pharmaceutical, biotechnology, and academic clients.
FMI Research Approach: FMI market taxonomy and inclusion-exclusion framework.
What Are Globally Unique Trends Shaping Drug Discovery Services Market?
Globally unique trends include the deployment of generative AI for molecular design, the integration of automated high-dimensional experimentation into assay development, and the expansion of milestone-linked strategic alliances between CROs and biopharma innovators.
FMI Research Approach: Pierre Fabre and Iktos generative AI collaboration (January 2026) and Eurofins Discovery DiscoveryAI SAFIRE launch.
Drug Discovery Services Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 29,727.4 Million |
| Industry Value (2036) | USD 117,163.1 Million |
| CAGR (2026 to 2036) | 14.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Challenges and Opportunities
Challenge
High Costs and Lengthy Development Timelines
High cost and lengthy timeline of drug development is one of the significant challenges faced by the drug discovery services industry. With billions poured into the process of drug discovery from target identification to clinical trials, the high-cost, high-tech industry is rife with skilled individuals and just as many regulatory affairs.
In addition, the attrition rate is high in drug discovery, with many lead compounds failing in the later stages due to efficacy or safety issues. This leads to higher R&D costs, making it tough for small biotech companies to compete with large pharma companies.
Opportunity
Integration of AI and Machine Learning in Drug Discovery
Integration of AI and Machine Learning is a major opportunity for drug discovery services market. Data-driven approaches are evolving to expedite target identification, molecular modeling, and predictive analytics, and notably reduce the time and cost associated with traditional drug discovery techniques.
Drug repurposing, virtual screening and optimization of designs for clinical trials are increasingly carried out using AI-based platforms. Algorithms that break new pathways through the vast seas of edge-case data are razor-edge rock stars at drug development, driving efficiency, scaling success rates and buying time by scrubbing entire pipelines in silico.
Shifts in the Drug Discovery Services Market 2021 to 2025 and Future Trends 2026 to 2036
Between 2021 and 2025, the drug discovery services market experienced significant growth, fueled by increased pharmaceutical R&D spending, advancements in artificial intelligence (AI)-driven drug screening, and the rising demand for biologics and personalized medicine.
The rapid adoption of contract research organizations (CROs) to optimize drug development timelines and reduce costs further drove market expansion. Additionally, the surge in COVID-19-related drug and vaccine research accelerated investment in high-throughput screening (HTS) technologies and AI-based molecular modeling.
Between 2026 and 2036, the drug discovery services market will undergo a transformative shift, driven by AI-led automation, quantum-enhanced computational drug design, and precision medicine advancements. The adoption of lab-on-a-chip technologies, digital twin simulations, and organ-on-chip platforms will redefine preclinical drug testing, reducing dependency on animal models and accelerating human-relevant drug discovery.
Market Shifts: A Comparative Analysis 2021 to 2025 vs. 2026 to 2036
| Market Shift | 2021 to 2025 |
|---|---|
| Regulatory Landscape | Stricter clinical trial guidelines, emphasis on data integrity, and fast-track drug approval pathways. |
| Technological Advancements | AI-driven drug screening, CRISPR-based gene editing, and NGS-enabled precision drug discovery. |
| Industry Applications | Oncology, infectious disease drug discovery, biologics development, and rare disease therapeutics. |
| Adoption of Smart Equipment | Cloud-based drug discovery platforms, AI-assisted molecular docking, and big data-powered target identification. |
| Sustainability & Cost Efficiency | Cost-effective CRO outsourcing, AI-optimized preclinical research, and reduced trial timelines through predictive modeling. |
| Data Analytics & Predictive Modeling | AI-based hit-to-lead identification, cloud-integrated genomic data analysis, and in silico trials. |
| Production & Supply Chain Dynamics | COVID-19-driven supply chain disruptions, increased outsourcing to CROs, and AI-assisted compound library management. |
| Market Growth Drivers | Growth driven by AI-led drug discovery, increased biopharma R&D investments, and regulatory support for precision medicine. |
| Market Shift | 2026 to 2036 |
|---|---|
| Regulatory Landscape | AI-powered regulatory compliance frameworks, block chain-enabled data transparency, and decentralized clinical trial regulations. |
| Technological Advancements | Quantum-enhanced drug modeling, bio printed organoid-based testing, and federated AI-driven biomarker discovery. |
| Industry Applications | Expansion into neurodegenerative disorders, synthetic biology-driven drug engineering, and regenerative medicine discovery. |
| Adoption of Smart Equipment | Fully automated AI-driven robotic labs, digital twin simulations, and quantum-assisted compound screening. |
| Sustainability & Cost Efficiency | AI-driven cost reduction in drug R&D, sustainable biopharma production models, and autonomous AI-optimized clinical trial design. |
| Data Analytics & Predictive Modeling | Quantum-enhanced predictive toxicology, federated learning in AI-driven drug development, and block chain-secured clinical trial data. |
| Production & Supply Chain Dynamics | AI-optimized global drug discovery pipelines, decentralized manufacturing of drug compounds, and block chain-enabled pharmaceutical supply chain security. |
| Market Growth Drivers | Quantum-powered AI automation, organ-on-chip preclinical testing, and next-generation synthetic biology-driven therapeutic solutions. |
Country wise Outlook
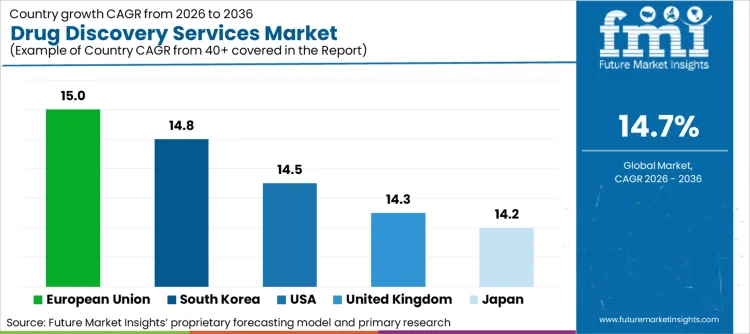
United States
The USA market for drug discovery services is experiencing quick growth, bolstered by the presence of key pharmaceutical & biotechnology companies, increasing R&D costs and government support to create medications.
The increasing demand for personalized medicine, adoption of porter AI-based drug discovery and the increasing demand for HTS technologies are also driving the growth of the market. Additionally, the explosion of biosimilar and biologics, coupled with new advances in gene and cell therapy, is accelerating the need for expert drug discovery services.
| Country | CAGR (2026 to 2036) |
|---|---|
| USA | 14.5% |
United Kingdom
Drug discovery services take root in the United Kingdom due to strong government support for life sciences research, the presence of blue-chip CROs (Contract Research Organizations), and the increasing number of academic-industry collaborators.
Pharma R&D expansion, investments in AI-driven drug discovery, and regulatory facilitation for faster approvals of drugs are ramping up market expansion. In addition, the UK presence of an early-stage drug development hub is increasing investment in drug discovery-outsource services.
| Country | CAGR (2026 to 2036) |
|---|---|
| UK | 14.3% |
European Union
The EU drug discovery services market is emerging ambitiously due to robust regulatory in place, increasing cash flows for research innovative drugs, and rising demand for biologics and small-molecule drugs.
Germany, France, and Switzerland lead in pharma R&D and are taking the lead in investing proactively in AI-based drug discovery, automation of screening technologies, and bioinformatics in drug development. In addition, the policies backed by the EU that promote cross-border cooperation in drug development and access to a large population database are further propelling the growth of the market.
| Region | CAGR (2026 to 2036) |
|---|---|
| European Union | 15.0% |
Japan
Japan's drug discovery services market is growing owing to the presence of developed pharmaceuticals industry, increased investment in the research and development of biopharmaceuticals, its governments' initiatives for precision medicines and regenerative therapies.
Increased focus on next generation drug screening methods and application of AI and machine learning During drug development is propelling the market growth. Positive presence of multi-national pharmaceutical majors and accelerating shift towards outsourcing of clinical trials is driving the growth of outsourced drug discovery services.
| Country | CAGR (2026 to 2036) |
|---|---|
| Japan | 14.2% |
South Korea
South Korea's drug discovery services market is experiencing strong growth through government support for biotech start-ups, increased foreign investment into pharma R&D and the expansion of the country's CRO industry.
The increasing usage of AI for drug discovery, soaring genomics studies and increasing academia partnerships with biotech firms are bolstering the market growth. Moreover, the rising demand for biologics and targeted therapies along with South Korea's rise as a center for clinical trials is strengthening the market growth.
| Country | CAGR (2026 to 2036) |
|---|---|
| South Korea | 14.8% |
Segmentation Outlook - Drug Discovery Services Market
Chemistry Services Lead Market Expansion as Demand for Hit-to-Lead Optimization Grows
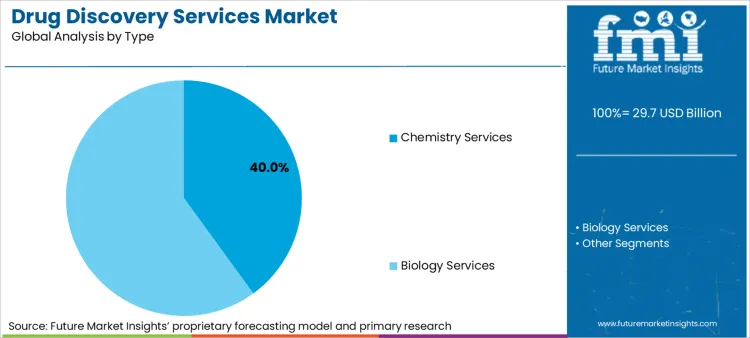
Chemistry services have emerged as one of the most effective segments in the drug discovery services marketplace, enabling pharmaceutical companies to develop superior drug candidates through structure-activity relationship (SAR) studies, medicinal chemistry optimizations, and route development.
Unlike traditional in-house research, outsourced chemistry services provide specialist knowledge and state-of-the-art technological platforms for faster acceleration of drug discovery operations. The growing emphasis on hit-to-lead optimization has significantly increased the uptake of chemistry services, as researchers seek to enhance potency, selectivity, and pharmacokinetic profiles by optimizing molecules.
Advancements in computational chemistry solutions, such as AI-powered molecular modeling, quantum mechanics simulations, and deep learning-driven predictive analytics, have driven market demand by ensuring reliable lead compound identification and optimization.
The integration of high-throughput screening (HTS) technologies, such as automated compound libraries, microfluidic platforms, and bioassay-guided fractionation, has also driven adoption, enabling efficient compound selection and hit validation. The formation of chemistry service partnerships, with CROs, pharmaceutical firms, and universities, has optimized market expansion through sharing knowledge, innovation, and cost-effective drug discovery mechanisms.
The practice of green chemistry approaches, with environmentally friendly synthetic pathways, biocatalysts-based processes, and green reagents, has fortified market growth through the union of drug discovery with environmentally safe research techniques.
While enjoying the advantages of efficiency, cost savings, and molecular specificity, the chemistry services market is faced with challenges such as compound attrition rates, regulatory issues regarding synthetic processes, and data reproducibility.
However, emerging innovations in AI-driven retrosynthetic analysis, block chain-based safe chemical data storage, and machine learning-driven lead optimization are driving efficiency, precision, and success rates to spur long-term growth for drug discovery services by chemistry globally.
Biology Services Drive Market Growth as Target Validation and Lead Identification Gain Popularity
Biology services have seen strong market adoption, particularly by biotech start-ups and pharmaceutical firms, as they increasingly outsource target validation expertise, mechanism-of-action research, and biomarker identification. Compared to in-house research, biology services provide rich preclinical data through functional genomics, high-content screening, and proteomics-based drug target assessment.
Growing need for target validation, ranging from CRISPR-based gene editing to RNAi screening, and ligand-receptor interactions has driven biology services adoption as scientists seek to validate therapeutic value before proceeding toward clinical development.
The expansion of biologics-based drug discovery, including monoclonal antibody screening, recombinant protein expression, and cell-based assays, has fueled market demand by broadening the scope of therapeutic modalities in preclinical research.
The use of AI-driven bioinformatics platforms, such as deep-learning models of protein-ligand interaction, next-generation sequencing (NGS) data analysis, and computerized pathway mapping, has also boosted adoption through increased predictive accuracy and productivity in target validation.
The advent of advanced cellular and organoid models, such as 3D cell cultures, PDX, and micro physiological systems, has accelerated market growth through the assurance of clinically relevant preclinical testing.
The adoption of ethical and regulatory-compliant biological testing platforms, such as GLP compliance, incorporation of biosafety measures, and standardization of in vitro and in vivo models, has further mandated market growth by aligning drug discovery with regulations and ethical research standards.
Though therapeutic validation, translational research, and preclinical precision are its strengths making it a go-to option, the biology services segment has a weakness in the guise of experimental variability, very high cost of sophisticated assay development, and un-scalability for niche therapeutic domains. But emerging innovation in AI-boosted biological data interpretation, cloud-integrated bioinformatics, and CRISPR-Cas9 gene function analysis is improving reproducibility, cost-effectiveness, and scalability and creating space for continued expansion for biology-led drug discovery services across the world.
Hit-to-Lead Identification Gains Market Demand as AI-Driven Compound Screening and Predictive Analytics Evolve
Hit-to-lead identification has emerged as one of the most widely adopted stages in the drug discovery process, enabling researchers to transition from initial screening hits to optimized lead compounds through iterative modifications and bioactivity assessments. Unlike traditional screening approaches, modern hit-to-lead strategies integrate AI-powered analytics, predictive modeling, and structure-based drug design to accelerate hit progression.
The rising demand for AI-driven compound screening, featuring machine learning algorithms for molecular docking, deep-learning models for toxicity prediction, and cheminformatics-based virtual screening, has fueled adoption of hit-to-lead services, as pharmaceutical firms prioritize computational efficiency in lead identification.
Despite its advantages in automation, cost efficiency, and computational accuracy, the hit-to-lead identification segment faces challenges such as compound attrition risks, algorithm biases in AI-driven screening, and regulatory concerns over AI-generated molecular predictions.
However, emerging innovations in quantum computing for molecular interactions, block chain-secured lead candidate verification, and AI-driven de novo drug design are improving accuracy, security, and lead success rates, ensuring continued market growth for hit-to-lead identification services worldwide.
Lead Optimization Expands as Molecular Refinement and Preclinical Testing Enhance Drug Viability
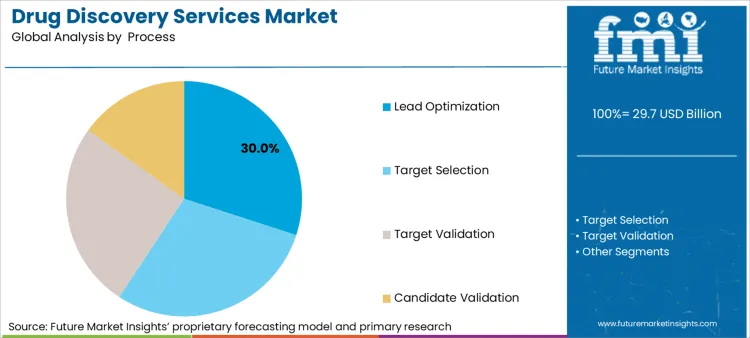
Lead optimization has gained strong market adoption, particularly among pharmaceutical developers and CROs, as they prioritize molecular refinement, pharmacokinetics optimization, and toxicity mitigation before advancing drug candidates to clinical trials.
Unlike early-stage discovery, lead optimization ensures that preclinical candidates meet safety, efficacy, and manufacturability criteria. The rising demand for structure-guided lead refinement, featuring AI-driven SAR analysis, fragment-based drug design, and dynamic molecular docking simulations, has driven adoption of lead optimization services, as researchers seek to enhance therapeutic precision and drug-like properties.
Despite its advantages in pharmacological refinement, regulatory compliance, and clinical trial readiness, the lead optimization segment faces challenges such as high attrition rates during late-stage preclinical testing, complex physicochemical property modifications, and scalability issues for biologics.
However, emerging innovations in AI-powered formulation design, digital twin simulations for pharmacokinetics modeling, and block chain-secured clinical data management are improving efficiency, accuracy, and regulatory compliance, ensuring continued expansion for lead optimization services worldwide.
Competitive Outlook
The Drug Discovery Services Market is expanding rapidly due to the increasing complexity of drug development and the growing demand for outsourced research solutions. Pharmaceutical and biotech companies are turning to contract research organizations (CROs) to streamline the discovery of novel therapeutics, improve efficiency, and reduce costs.
Advances in computational drug discovery, AI-driven screening, and high-throughput technologies are revolutionizing the market. The rising focus on biologics, gene therapies, and personalized medicine is further driving demand for specialized drug discovery services. Leading companies in the sector emphasize integrated research capabilities, AI-driven platforms, and global partnerships to maintain a competitive edge.
Market Share Analysis by Company
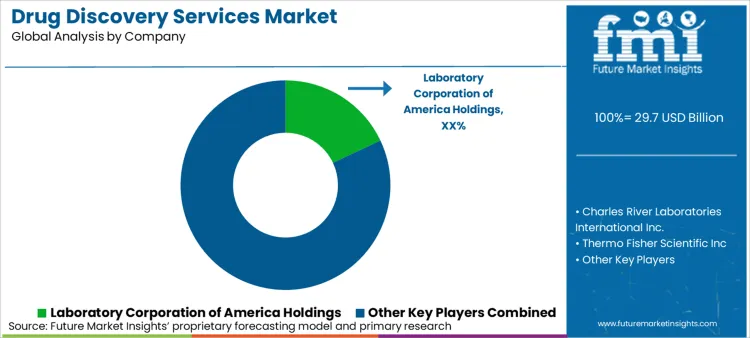
| Company Name | Estimated Market Share (%) |
|---|---|
| Laboratory Corporation of America Holdings | 18-22% |
| Charles River Laboratories International Inc. | 14-18% |
| Thermo Fisher Scientific Inc. | 12-16% |
| Evotec SE | 10-14% |
| Pharmaron Beijing Co., Ltd. | 8-12% |
| Genscript Biotech Corporatio | 6-10% |
| Other Companies (combined) | 30-40% |
Recent Developments
- In January 2026, Pierre Fabre Laboratories and Iktos announced a strategic collaboration to utilize Iktos' generative AI and automated chemistry robotics to identify and develop novel small-molecule drug candidates in oncology.
- In August 2025, IQVIA and Flagship Pioneering launched a strategic collaboration to accelerate the development of breakthrough biopharma companies within the Flagship ecosystem. The collaboration grants Flagship portfolio companies access to IQVIA's AI-powered clinical research services.
- In September 2024, Orion and Aitia entered into an agreement to create 'Gemini Digital Twins' for oncology drug discovery. The partnership uses Aitia's Causal AI technology and digital twin models alongside Orion's clinical data to discover and validate novel drug targets, with milestone payments exceeding USD 10 Million per target.
Market Definition
The drug discovery services market represents revenue generated from outsourced research activities spanning target identification and validation, hit finding and screening, lead optimization, ADMET profiling, and preclinical candidate development services.
Inclusions cover target-based and phenotypic screening, medicinal chemistry, in vitro and in vivo pharmacology, biomarker development, formulation support, and IND-enabling toxicology studies performed by contract research organizations and specialized discovery service providers.
Exclusions include clinical trial management services (Phase I to IV), commercial-stage manufacturing (CDMO revenue for marketed drugs), and in-house pharmaceutical company R&D spending. Academic grant-funded basic research not contracted to external service providers is also outside the scope.
Research Scope
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 29,727.4 Million |
| Service Type | Target Identification, Hit-to-Lead, Lead Optimization, ADMET, IND-Enabling Studies |
| Technology Platform | AI-Driven Design, High-Throughput Screening, Automated Chemistry, Digital Twins |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Key Companies Profiled | Charles River, WuXi AppTec, Eurofins Discovery, Evotec, IQVIA, Syngene International |
Drug Discovery Services Market Segmentation
By Process:
- Target Selection
- Target Validation
- Hit-to-lead Identification
- Lead Optimization
- Candidate Validation
By Type:
- Chemistry Services
- Biology Services
By Drug Type:
- Small Molecule Drugs
- Biologics Drugs
By Therapeutic Area:
- Neurology
- Infectious and Immune Systems Diseases
- Digestive System Diseases
- Oncology
- Other Therapeutic Areas
By End User:
- Pharmaceutical & Biotechnology Companies
- Academic Institutes
- Manufacturing
- Other End Users
By Region:
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia Pacific
- Middle East and Africa
Bibliography
- USA Food and Drug Administration. (2024). Novel Drug Approvals for 2024. FDA.
- Charles River Laboratories. (2026, February). Partnership with Synthace for Assay Development. Drug Target Review.
- WuXi AppTec. (2025, March). FDA Inspection Results at Changzhou and Taixing Sites. WuXi AppTec Newsroom.
- Evotec SE. (2025, November). Milestone Payment from Bristol Myers Squibb. Evotec Investor Relations.
Frequently Asked Questions
What was the overall size of the drug discovery services market in 2026?
The overall market size for drug discovery services market was USD 29,727.4 Million in 2026.
How big is the drug discovery services market expected in 2036?
The drug discovery services market is expected to reach USD 117,163.1 Million in 2036.
What will drive the demand for drug discovery services market during the forecast period?
The increasing demand for outsourcing drug discovery processes by pharmaceutical and biotechnology company’s fuels Drug discovery services Market during the forecast period.
List the top 5 countries contributing in drug discovery services market?
The top 5 countries which drives the development of Drug discovery services Market are USA, UK, Europe Union, Japan and South Korea.
Which is the leading segment in the drug discovery services market?
On the basis of type, chemistry services to command significant share over the forecast period.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Process
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Process , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Process , 2026 to 2036
- Lead Optimization
- Target Selection
- Target Validation
- Candidate Validation
- Lead Optimization
- Y to o to Y Growth Trend Analysis By Process , 2021 to 2025
- Absolute $ Opportunity Analysis By Process , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type, 2026 to 2036
- Chemistry Services
- Biology Services
- Chemistry Services
- Y to o to Y Growth Trend Analysis By Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Process
- By Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Process
- By Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Process
- By Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Process
- By Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Process
- By Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Process
- By Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Process
- By Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Process
- By Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Process
- By Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Process
- By Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Process
- By Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Process
- By Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Process
- By Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Process
- By Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Process
- By Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Process
- By Type
- Competition Analysis
- Competition Deep Dive
- Laboratory Corporation of America Holdings
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Charles River Laboratories International Inc.
- Thermo Fisher Scientific Inc
- Evotec SE
- Pharmaron Beijing Co., Ltd.
- Genscript Biotech Corporatio
- Laboratory Corporation of America Holdings
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Process , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Process , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Process , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Process , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Process , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Process , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Process , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Process , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Type, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Process , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Process , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Process
- Figure 6: Global Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Type
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Process , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Process , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Process
- Figure 23: North America Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Type, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Type
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Process , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Process , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Process
- Figure 30: Latin America Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Type, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Type
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Process , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Process , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Process
- Figure 37: Western Europe Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Type, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Type
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Process , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Process , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Process
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Type, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Type
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Process , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Process , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Process
- Figure 51: East Asia Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Type, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Type
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Process , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Process , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Process
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Type, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Type
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Process , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Process , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Process
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Type, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Type
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

