Drug of Abuse Testing Market
Drug of Abuse Testing Market Growth - Trends & Forecast 2026 to 2036
Drug of Abuse Testing Market Forecast and Outlook 2026 to 2036
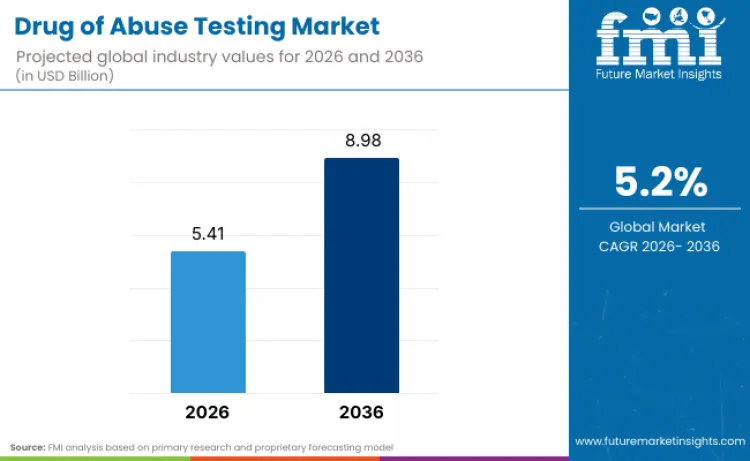
The global drug of abuse testing market is projected to reach USD 5.41 billion in 2026 and expand to USD 8.98 billion by 2036, registering a CAGR of 5.2% over the forecast period. According to FMI, the growth of the market is associated with the prevalence of substance use, regulation, and the adoption of routine screening in hospitals, diagnostic labs, forensic centers, and the workplace. The practice of immunoassay-based screening for high-volume testing, along with confirmatory chromatography methods such as GC-MS and LC-MS, is driving the demand for multi-modal testing platforms. Breakthroughs in rapid testing technology, analyzer automation, and assay specificity are enabling the possibility of rapid decision-making with analytical accuracy.
“The PharmChek Sweat Patch has a long track record of reliability and has been embraced by probation authorities, treatment programs, drug courts, and human services agencies across the country.” said Chris Linthwaite, CEO of SCRAM Systems.
The major players in the diagnostic market are also enhancing their market position with their never-ending innovations in the development of assays, analyzers, and integrated toxicology testing solutions. Abbott Laboratories is also enhancing their immunoassay-based drug screening solutions for the clinical and laboratory environments. F. Hoffmann-La Roche Ltd is also concentrating on laboratory automation and high specificity reagent systems that simplify toxicology confirmation solutions. Siemens Healthineers is also offering integrated clinical chemistry and immunoassay solutions that are employed in routine and emergency toxicology analyses. On the other hand, Thermo Fisher Scientific and Shimadzu Corporation are also playing an important role in confirmatory testing solutions through their chromatography and mass spectrometry systems.
Market development is also aided by the increasing application of drug testing in emergency medicine, rehabilitation programs, forensic analysis, and occupational health screening. The requirement for confirmatory testing, chain-of-custody analysis, and result defensibility is also propelling the demand for validated tests and high-quality analytical instruments. The end-user market includes hospitals, clinical laboratories, forensic labs, and other institutional settings, which have varying demands for speed, sensitivity, and specificity. The market trend is also moving towards an integrated testing process that includes screening, confirmation, data integrity, and regulatory compliance.
Summary of the Drug of Abuse Testing Market
What Is Growth Outlook for Drug of Abuse Testing Market as per Future Market Insights Projection?
Future Market Insights projects the drug of abuse testing market to expand from USD 5.41 billion in 2026 to USD 8.98 billion by 2036, registering a forecast CAGR of 5.2% over the period.
FMI Research Approach: Historical testing volumes, screening mandate coverage, drug-class demand proxies, and bottom-up revenue mapping by drug type, technique, and end user.
How Do FMI Analysts Perceive Drug of Abuse Testing Market to Evolve?
Future Market Insights analysts note that tiered workflows are being reinforced, with rapid screening being paired with targeted confirmatory testing to maintain compliance, speed, and chain-of-custody integrity.
FMI Research Approach: Evaluated immunoassay screening penetration, confirmatory chromatography attach rates, multi-panel expansion, and interoperability requirements across decentralized and lab settings.
Which Country Holds Largest Share in Global Drug of Abuse Testing Market?
Future Market Insights notes that the United States is being supported by large institutional screening volumes across healthcare, workplace, forensic, and public safety programs.
FMI Research Approach: Gathered country-level inputs on enforcement intensity, routine screening cadence, infrastructure availability, and accredited confirmatory capacity.
How Large Will Drug of Abuse Testing Market Be by 2036?
The global drug of abuse testing market is projected to reach USD 8.98 billion by 2036.
FMI Research Approach: Derived demand using weighted test frequency, panel mix, and confirmatory utilization factors across major end-user environments.
How are Regulatory Tailwinds Fueling Drug of Abuse Testing Adoption?
According to Future Market Insights analysts, sustained compliance expectations and documentation requirements are supporting routine, program-driven testing demand across jurisdictions.
FMI Research Approach: Incorporated mandate continuity, chain-of-custody standards, and turnaround-time expectations into adoption curves for screening and confirmatory workflows.
Drug of Abuse Testing Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 5.41 billion |
| Market Forecast Value (2036) | USD 8.98 billion |
| Forecast CAGR (2026 to 2036) | 5.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research.
How Do Regulatory Enforcement and Public Safety Frameworks Drive Demand for Drug of Abuse Testing?
The enforcement of regulations in the healthcare, occupational safety, and forensic sectors is instrumental in grounding the demand for drug of abuse testing. The need for drug of abuse testing is being driven by the requirement for legally defensible confirmation, which is reducing the tolerance for inconsistent practices. The testing of drugs of abuse is, therefore, being viewed as a compliance-critical diagnostic procedure rather than a discretionary process. In the regulated hospital, laboratory, and forensic environments, rapid immunoassay screening is being developed for high throughput, while chromatography-based confirmation is being used for evidentiary purposes. As the frameworks for accountability remain embedded, the baseline demand is expected to remain stable.
How is the Drug of Abuse Testing Market Segmented?
The drug of abuse testing market has been segmented based on drug type, technique type, end user, and geography. By drug type, the market is divided into psychostimulants, narcotics, cannabinoids, sedatives, and other abused substances. Based on technique type, the market is categorized into immunoassay screening, chromatography-based confirmatory testing, and rapid testing formats. By end user, the market is segmented into hospitals, diagnostic laboratories, forensic laboratories, and other institutional settings. Regionally, the market spans developed and emerging healthcare systems with established laboratory infrastructure and regulatory testing mandates.
Why Does Immunoassay-Based Testing Remain the Primary Technique?
Immunoassay-based testing remains at the forefront of technology adoption as it is being implemented as the front-line screen in hospitals, reference labs, emergency rooms, and workplace testing programs. Its utility is being established based on rapid turnaround, automated high-throughput processing, and the ability to handle large volumes of samples without a corresponding increase in technical labor time. Simultaneous multi-analyte panels can be tested in a single run, allowing rapid triage before confirmatory testing is even started. This has helped in reducing congestion in the laboratory while still facilitating timely clinical and administrative decisions. From an economic perspective, immunoassays are still the preferred choice in high-volume testing because of their lower cost per test compared with chromatography. Although confirmatory testing has its role in establishing evidentiary standards, immunoassays are likely to remain the “front door” of the testing algorithm workflow.
How Do End-User Requirements Influence Testing Modality Selection?
Diagnostic labs are the most influential group of end-users in determining the adoption of testing modalities in the drug of abuse testing market. These labs handle large sample volumes from hospitals, law enforcement agencies, and occupational health services, requiring flexible and standardized testing modalities. In this context, immunoassays are commonly used for screening, followed by chromatography-based methods for confirmatory testing in cases where legal or medical certainty is required. Forensic labs use chromatography for analytical specificity and defensibility of test results, while hospitals require rapid testing for acute clinical decision-making. Rapid testing formats are selectively used in decentralized or point-of-care applications where timeliness has greater priority over analytical complexity. Centralized labs, on the other hand, are the driving force behind volume-driven demand, establishing a stratified testing system where the choice of testing modalities is directly affected by turnaround time requirements.
How Are Integrated Testing Workflows Replacing Single-Method Approaches?
The market for drug of abuse testing is experiencing a transition from stand-alone testing procedures to integrated workflows that will facilitate the combination of rapid testing with analytical confirmation. The stand-alone testing procedure is increasingly being viewed as insufficient in a setting where the demand is for both speed and accuracy of results. With the volume of testing rising, laboratories are adopting multi-layered testing strategies that improve efficiency without compromising accuracy. This has resulted in the testing platform being viewed as part of a larger diagnostic environment.
How Is Automation and Throughput Efficiency Redefining Competitive Advantage?
There is an increasing trend in laboratories towards the implementation of automated systems that are compatible with continuous laboratory operations, standardized reagent use, and minimal hands-on time. High-throughput immunoassay analyzers are preferred in centralized laboratories, and modular chromatography systems are used for confirmatory testing without affecting regular activities. The trend in competitive differentiation among manufacturers is shifting from innovative tests to reliable systems, consistent reagents, and support services. Companies that can offer integrated laboratory solutions are preferred over companies that can offer testing solutions.
What Structural Shift Will Define Long-Term Market Leadership?
From 2026 to 2036, the future of the drug of abuse testing market will be shaped by the alignment of testing technologies with regulatory requirements, scalability in laboratories, and end-user requirements. The consistency of performance in hospital networks, diagnostic laboratories, and forensic facilities will remain the key to success. The drug of abuse testing market is undergoing a transition from a discretionary lab service to a compliance-critical diagnostic service. Suppliers who are able to align screening with confirmatory testing and scalability are well-positioned to unlock sustained value as the demand for testing continues to grow in the healthcare and non-healthcare sectors.
How Is the Drug of Abuse Testing Market Evolving Globally?
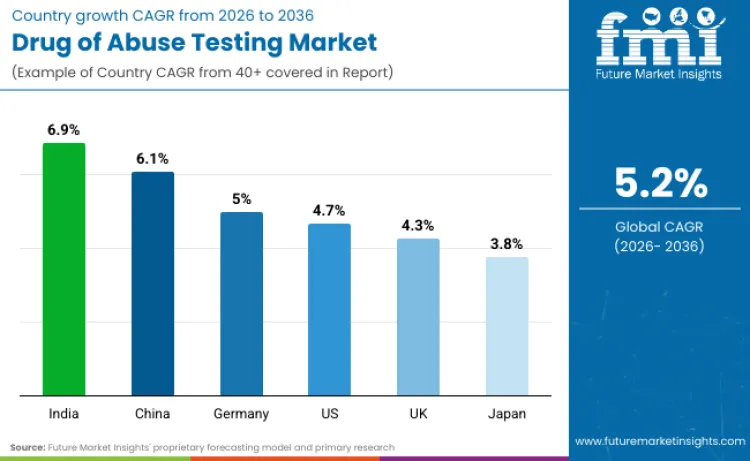
Future Market Insights has picked out the United States (CAGR of 4.7%), China (6.1%), India (6.9%), Germany (5.0%), the United Kingdom (4.3%), and Japan (3.8%) as the six key demand drivers until 2036. The United States is a leader in established workplace and clinical screening volumes, with strong forensic and public safety testing continuity. China and India are being driven by growing hospital capacity and institutional screening reach as testing infrastructure grows. Germany and the United Kingdom are being sustained by protocol-driven, compliance-led testing in healthcare and occupational settings. Japan’s market is being held back by validation-first adoption and conservative introduction of new testing modalities. FMI highlights that these markets balance enforcement toughness with institutional testing scale, maintaining steady demand.
| Country | CAGR (2026 to 2036) |
|---|---|
| United States | 4.7% |
| Germany | 5.0% |
| United Kingdom | 4.3% |
| China | 6.1% |
| Japan | 3.8% |
| India | 6.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
How is expanding public safety and healthcare surveillance in China driving testing adoption?
In China, the demand for drug of abuse testing solutions is being fueled by the growing public safety programs, the need for workplace compliance, and the rising integration of toxicology testing in hospital and emergency settings. As the urban population rises and the monitoring framework for regulation becomes more stringent, the testing processes are being normalized for use in law enforcement, transportation safety, and medical diagnostics. Immunoassays and confirmatory testing procedures are being adopted at an increasing rate to ensure accuracy and traceability. In this positioning, the China drug of abuse testing market is expected to register a CAGR of 6.1% over the forecast period. In the next five years, there will be a growing adoption at the secondary hospitals and testing centers, fueled by automation enhancements and rising accessibility to rapid testing solutions.
Is regulatory expansion in India strengthening demand for structured testing?
The market trends in India are being fueled by the rising government regulation of the market, the rising number of workplace drug testing programs, and the rising awareness about the abuse of drugs as a public health concern. The drug of abuse testing is being increasingly used in the transportation industry, industrial employment, and toxicology laboratories. The hospitals and laboratories are focusing on cost-effective solutions that can provide a balance between accuracy and efficiency. This is also seen in the India drug of abuse testing market, which is expected to grow at a CAGR of 6.9% during the forecast period. In the coming years, there will be increased penetration in the occupational health screening and government-supported testing programs.
Is compliance-driven healthcare in Germany supporting market stability?
In the German market, the demand is being driven by robust regulatory compliance needs and organized healthcare procedures for the detection of substance abuse. Drug of abuse testing solutions are being implemented in hospital labs, rehab facilities, and forensic organizations for organized diagnostics and documentation. The importance of test validity, accuracy, and quality assurance is driving adoption in the clinical and forensic segments. In this scenario, the Germany drug of abuse testing market is expected to register a CAGR of 5.0% during the forecast period. Over the next five years, incremental growth will be driven by improvements in lab automation, expanded test menus, and the adoption of organized confirmatory procedures.
How is public health policy in the United Kingdom influencing testing utilization?
In the United Kingdom, market development is being shaped by public health strategies addressing substance misuse, alongside workplace and criminal justice testing requirements. Drug of abuse testing is being utilized to support harm reduction programs, rehabilitation monitoring, and employment screening, with increasing emphasis on rapid turnaround and consistent result interpretation. Adoption is being guided by alignment with national healthcare protocols and budgetary discipline. As a result, the UK drug of abuse testing market is expected to grow at a CAGR of 4.3% during the forecast period. Future expansion is likely to be driven by selective deployment of rapid testing solutions in community healthcare and enforcement settings, supported by centralized laboratory confirmation workflows.
How is an aging society and clinical precision in Japan shaping testing demand?
In Japan, demand is being supported by the need for precise toxicology screening within a highly regulated healthcare system, where diagnostic accuracy and procedural consistency are strongly emphasized. Drug of abuse testing is being primarily adopted in hospital laboratories, forensic medicine departments, and controlled clinical environments. Conservative capital investment patterns are resulting in gradual, methodical adoption rather than rapid expansion. Under this framework, the Japan drug of abuse testing market is expected to grow at a CAGR of 3.8% during the study period. Over the next five years, growth is expected to be driven by system upgrades, enhanced assay sensitivity, and improved workflow integration rather than increased testing volumes.
How is the USA Market Advancing Through Regulatory and Workplace Maturity?
In the United States, the market development is being influenced by the presence of mandates for workplace drug testing, transportation safety laws, and well-established forensic and clinical toxicology infrastructure. The drug of abuse testing solutions are being standardized at an increasing rate among employers, healthcare organizations, and law enforcement agencies. Preference is being given to validated testing kits and automated laboratory platforms that are less prone to variability and facilitate high-throughput testing. Under such a mature regulatory and operational framework, the USA drug of abuse testing market is expected to register a CAGR of 4.7% during the forecast period. During the next five years, further adoption is expected to occur through panel expansion, enhanced accuracy of point-of-care testing, and increased use of digital reporting systems.
Who are the Major Players Active in the Drug of Abuse Testing Market?
The competitive environment in the drug of abuse testing market is fueled by the increasing demand for accuracy in rapid screening, regulatory needs, and scalability in the institutional testing environment. The market is being fueled by the need to support multiple drug panels, high-throughput analytical needs, and consistency in the hospital, laboratory, and forensic testing environment. The market is currently served by a mix of immunoassay-based screening instruments and confirmatory chromatography instruments, and market dynamics are being fueled by the end-user environment and testing volumes.
The major players are positioning themselves in the market through comprehensive assay offerings and instrumentation systems that provide capabilities for screening and confirmatory testing. Abbott Laboratories (Alere, Inc.) has expanded its presence in the market through immunoassay-based rapid testing solutions for decentralized hospital and point-of-care settings. F. Hoffmann La-Roche Ltd has concentrated its efforts on chromatography-based confirmatory solutions, providing high analytical specificity for regulated laboratory testing. Siemens AG (Siemens Healthineers) is continuing to utilize its infrastructure in the diagnostics market to provide comprehensive immunoassay testing capabilities in hospital and diagnostic laboratory settings.
Concurrently, Thermo Fisher Scientific, Inc. and Shimadzu Corporation have also enhanced their offerings in chromatography and analytical systems to better serve the needs of forensic labs and confirmatory testing. In terms of competitive differentiation in the major markets such as the USA, Germany, China, India, and Japan, the test menu, automation, and regulatory requirements are becoming more important.
Key Industry Developments:
- On August 28, 2025, SCRAM Systems, a leading manufacturer of alcohol and location monitoring devices and software, announced its acquisition of PharmChem, the industry leader in sweat-based drug testing and maker of the FDA-cleared PharmChek Drugs of Abuse Sweat Patch.
- In June, 2024, Align Capital Partners, toxicology testing solutions provider Premier Biotech has acquired Desert Tox, LLC, a provider of Substance Abuse and Mental Health Services Administration (SAMHSA) certified laboratory testing services.
Key Players in the Drug of Abuse Testing Market
- Abbott Laboratories (Alere, Inc.)
- F. Hoffmann La-Roche Ltd
- Siemens AG (Siemens Healthineers)
- Thermo Fisher Scientific, Inc.
- Drägerwerk AG & Co. KGaA
- Shimadzu Corporation
- Laboratory Corporation of America Holdings
Market Definition
The market for the drug of abuse test is defined as the global use of analytical and rapid screening instruments for the detection and monitoring of illicit and controlled substances in biological samples. The size of the market is measured in USD billion and projected from 2026 to 2036.
The market encompasses the revenue generated from immunoassay screening tests, chromatography confirmatory tests, and rapid tests. The application of these instruments is fueled by the demand for regulatory testing, workplace screening, and the demand for rapid and accurate substance detection.
The drug of abuse testing market is segmented into the following products, which come under the market scope and are segmented on the basis of drug type, technique type, and end user, as discussed in the market study report. The market scope includes the testing solutions for psychostimulants, narcotics, cannabinoids, sedatives, and others, based on immunoassay, chromatography, and rapid testing. The end users studied in the report are hospitals, diagnostics laboratories, forensic laboratories, and others. Excluded from the market for drug of abuse testing are non-analytical wellness screening devices, drug treatment services, rehabilitation facilities, and law enforcement activities that are not directly related to analytical testing platforms. Also excluded from the market are products that are not intended for drug detection, such as general clinical chemistry tests, molecular diagnostics that are not related to drug abuse testing, and non-regulated screening devices that do not meet certain analytical standards.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD 5.41 billion |
| Drug Type | Psychostimulants, Narcotics, Cannabinoids, Sedatives, Others |
| Technique Type | Immunoassay, Chromatography, Rapid Testing |
| End User | Hospitals, Diagnostics Laboratories, Forensic Laboratories, Others |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | USA, Germany, China, India, Japan, and 40+ countries |
| Key Companies Profiled | Abbott Laboratories (Alere, Inc.), F. Hoffmann La-Roche Ltd, Siemens AG (Siemens Healthineers), Thermo Fisher Scientific, Inc., Shimadzu Corporation |
| Additional Attributes | Dollar sales by drug type, technique type, and end user, regulatory compliance impact, workflow automation influence, test menu expansion and throughput optimization trends |
Source: FMI historical analysis and forecast data.
Drug of Abuse Testing Market by Segment
Drug Type:
- Psychostimulants
- Narcotics
- Cannabinoids
- Sedatives
- Others
Technique Type:
- Immunoassay
- Chromatography
- Rapid Testing
End User:
- Hospitals
- Diagnostics Laboratories
- Forensic Laboratories
- Others
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- Thailand
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- Netherlands
- Rest of Europe
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Colombia
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- Substance Abuse and Mental Health Services Administration. (2024). Mandatory Guidelines for Federal Workplace Drug Testing Programs: Urine, Oral Fluid, and Hair. SAMHSA.
- World Health Organization. (2023). Guidance on Screening and Laboratory Testing for Substance Use Disorders in Clinical and Public Health Settings. WHO.
- Clinical and Forensic Toxicology Society. (2023). Best Practices for Analytical Validation and Interpretation in Drugs of Abuse Testing. CFTS.
- Anderson, R. L., et al. (2023). Advances in immunoassay- and chromatography-based drug of abuse testing: Analytical performance, detection windows, and clinical relevance. Journal of Analytical Toxicology, 47, 512-526.
- Nguyen, P. T., et al. (2024). Clinical utility and regulatory considerations of point-of-care versus laboratory-based drug screening platforms. Clinical Chemistry, 70, 231-244.
Frequently Asked Questions
How big is the drug of abuse testing market in 2026?
The global drug of abuse testing market is estimated to be valued at USD 5.41 billion in 2026.
What will be the size of the drug of abuse testing market in 2036?
The market size for the drug of abuse testing market is projected to reach USD 8.98 billion by 2036.
How much will the drug of abuse testing market grow between 2026 and 2036?
Growth is expected to be registered at a 5.2% CAGR between 2026 and 2036.
What are the key technique types in the drug of abuse testing market?
Key technique types are Immunoassay, Chromatography and Rapid Testing in the drug of abuse testing market.
Which end user will contribute a significant share in the drug of abuse testing market in 2026?
A leading contribution is expected to be generated by Forensic Laboratories, accounting for an estimated 35.6% % share in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Primary Modes
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Desk Research Programme (Secondary Evidence)
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Drug Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Drug Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Drug Type , 2026 to 2036
- Psychostimulants
- Narcotics
- Cannabinoids
- Sedatives
- Others
- Psychostimulants
- Y to o to Y Growth Trend Analysis By Drug Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Drug Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technique Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technique Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technique Type, 2026 to 2036
- Immunoassay
- Chromatography
- Rapid Testing
- Immunoassay
- Y to o to Y Growth Trend Analysis By Technique Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Technique Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Diagnostics Laboratories
- Forensic Laboratories
- Others
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Drug Type
- By Technique Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Technique Type
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Drug Type
- By Technique Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Technique Type
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Drug Type
- By Technique Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Technique Type
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Drug Type
- By Technique Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Technique Type
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Drug Type
- By Technique Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Technique Type
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Drug Type
- By Technique Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Technique Type
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Drug Type
- By Technique Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Technique Type
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Technique Type
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Drug Type
- By Technique Type
- By End User
- Competition Analysis
- Competition Deep Dive
- Abbott Laboratories
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- F. Hoffmann La-Roche Ltd
- Siemens Healthineers
- Thermo Fisher Scientific, Inc.
- Drägerwerk AG & Co. KGaA
- Shimadzu Corporation
- Laboratory Corporation of America Holdings
- Others
- Abbott Laboratories
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Drug Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Technique Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Drug Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Technique Type, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Drug Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Technique Type, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Drug Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Technique Type, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Drug Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Technique Type, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Drug Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Technique Type, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Drug Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Technique Type, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Drug Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Technique Type, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Drug Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Drug Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Drug Type
- Figure 6: Global Market Value Share and BPS Analysis by Technique Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Technique Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Technique Type
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Drug Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Drug Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Drug Type
- Figure 26: North America Market Value Share and BPS Analysis by Technique Type, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Technique Type, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Technique Type
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Drug Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Drug Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Drug Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Technique Type, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Technique Type, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Technique Type
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Drug Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Drug Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Drug Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Technique Type, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Technique Type, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Technique Type
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Drug Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Drug Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Drug Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Technique Type, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Technique Type, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Technique Type
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Drug Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Drug Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Drug Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Technique Type, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Technique Type, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Technique Type
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Drug Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Drug Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Drug Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Technique Type, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Technique Type, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Technique Type
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Drug Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Drug Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Drug Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Technique Type, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Technique Type, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Technique Type
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

